Abstract
Background
Digital health technologies have potential to address the challenges associated with traditional cardiac rehabilitation (CR). However, it is not complete enough for prescribed exercise guidance and remote monitoring.
Objective
We aimed to evaluate the feasibility of a smartphone app for prescribed exercise tutoring by exercise videos combined with wearable devices to monitor heart rate in patients with stable coronary heart disease (CHD).
Methods
The study is a quasi-experimental design study with a single group. A total of 31 patients were included with an average age of 56.2 years (SD 13.4). They participated in a 12-week remote digital CR program. We employed a wearable heart rate monitoring device connected with an app to monitor the patients’ exercise intensity. The app can display the videos corresponding to an exercise prescription to guide the exercise. Cardiorespiratory endurance, blood pressure, blood glucose, cholesterol, blood uric acid, left ventricular ejection fraction and quality of life (QoL) were assessed at the beginning and end of the intervention. Compliance and safety events were recorded as well.
Results
Completion rate reached 90.3%. Average daily effective exercise time was 39.4 min (SD 17.8), and 92.9% of the patients could exercise in the prescribed intensity for at least 20 min per day. Average effective exercise days per week were 4.6 days (SD 2.2), and 67.9% of the patients could exercise in the prescribed intensity for at least 3 days per week. Patients’ peak VO2 (P = 0.041) and peak metabolic equivalents (P = 0.018) were significantly increased, low-density lipoprotein (P = 0.036) and diastolic blood pressure at rest (P = 0.044) were significantly decreased, and depression (GAD-7, P = 0.014) and anxiety (PHQ-9, P = 0.013) were significantly improved.
Conclusions
It is feasible, safe, and helpful for stable CHD patients to use the app for prescribed exercise tutoring with videos combined with wearable devices to monitor heart rate.
Trial Registration
ChiCTR1800019144.
Keywords
Introduction
Coronary heart disease (CHD) is the most common cause of death worldwide. “World health statistics 2023: Monitoring health for the SDGs, sustainable development goals” mentioned that CHD caused 17.9 million deaths worldwide in 2019. 1 Exercise-based cardiac rehabilitation (CR) has been shown to reduce all-cause mortality, myocardial infarction rates, all-cause hospitalizations, and associated healthcare costs in patients with CHD and improve health-related quality of life. 2 Therefore, CR has become an important part of the long-term treatment of patients with CHD and is recommended by the European Society of Cardiology, American Heart Association, and American College of Cardiology Class I, and exercise intervention has been identified as a central element. 3 Even so, patient participation in CR remains low, with an overall participation rate of approximately 40% in recent decades. 4 CR started late in China, and due to insufficient attention, family economic and transportation difficulties, etc., the participation rate of patients is also low. 5
Studies have shown that patients who receive interventions with digital health technologies (i.e. the use of the Internet, wearables, and apps to provide medical services) appear to have higher adherence than those who use traditional CR. 6 Early remote digital CR cannot monitor physiological parameters of patients during exercise, and there are disadvantages such as complicated operation and limited to restricting participants to a fixed position. 7 In recent years, increasingly powerful smartphone and wearable sensor technology have realized the automatic collection and real-time upload of data, so supervisors can monitor the exercise safety of participants at almost any location and at any time. A recent systematic review demonstrated the high availability, utility, acceptability, and acceptance of digital CR for these smartphones combined with wearable sensors. 8 But at present, doctors/coaches still need to spend more time on exercise instruction to patients through pre-use training, video conferencing, and phone calls, 6 or patients can only perform simple exercises such as walking and jogging. 9 In order to try to explore a new remote digital CR model, we assessed the feasibility of a smartphone app for prescribed exercise tutoring with exercise demonstration videos combined with wearable devices to monitor heart rate (HR) in patients with stable CHD. Furthermore, we evaluated its clinical efficacy as exploratory secondary result. The clinical efficacy of this app in patients with type 2 diabetes 10 and COVID-19 11 has been confirmed before, but it has not been used in patients with CHD.
Methods
Study design
We conducted a quasi-experimental design study with a single group registered with the Chinese Clinical Trials Registry (ChiCTR1800019144). We planned to recruit 30 participants intentionally from the Department of Cardiovascular Medicine, Peking University People's Hospital, China, from January 2018 to April 2018, then all subjects participated in a 12-week remote digital CR program. Participants aged ≥18 years old, diagnosed with stable CHD (including stable angina pectoris, stable period after myocardial infarction treatment, stable period after percutaneous coronary intervention (PCI) or coronary artery bypass grafting, etc.), own and use a smartphone, would be included in the study. In addition, individuals with any of the following conditions were excluded as follows:
Resting angina pectoris, unstable angina pectoris, acute myocardial infarction, previous history of myocardial infarction without surgical treatment, left main coronary artery stenosis or three-vessel disease without surgical intervention. Combined with one or more of the following cardiovascular diseases: heart failure, frequent premature ventricular contractions, ventricular tachycardia, uncontrolled atrial tachycardia (including atrial tachycardia, atrial flutter, atrial fibrillation, etc.), severe sinus bradycardia (HR < 50 bpm); acute myocarditis, pericarditis, ruptured aneurysm, acute pulmonary embolism or pulmonary infarction; uncontrolled hypertension (resting systolic blood pressure > 160 mmHg, diastolic blood pressure > 100 mmHg), orthostatic hypotension. Combined with acute non-cardiac diseases, such as infection, renal failure, hyperthyroidism, etc. Uncontrolled diabetes or recurrent hypoglycemia. Nerve, muscle, skeletal muscle, and rheumatic diseases that may be aggravated by exercise. Mental illnesses that are unable to live independently or cooperate with treatment.
This study was approved by the Research Ethics Committee of Peking University People's Hospital. All patients completed and obtained written informed consent.
Procedure
After the recruitment of subjects, the demographic data as well as clinical data such as current symptoms, medication status, and past history were collected first. All subjects underwent baseline assessments, including height, weight, laboratory tests, quality of life, mental health assessments, and cardiopulmonary exercise testing (CPET).
The intervention was a 12-week remote digital CR program. According to the consensus of Chinese experts on exercise therapy for patients with CHD, all subjects were prescribed 30–60 min of exercise therapy every day. 12 An exercise prescription included aerobic exercise 3–5 days per week (preferably 7 days per week), resistance exercise 2–3 days per week (at least 1 day apart), and flexibility exercise 3–5 times per week. Patients need to achieve the defined exercise time according to the requirements, but we do not specify a fixed exercise time, patients could schedule exercise on their own time.
Aerobic exercise intensity was calculated using the anaerobic threshold (AT) method (measured by CPET): target HR = anaerobic threshold HR ± 5–10 bpm, and Borg score as a secondary measure of exercise intensity was required to be 11–16 points. For patients with myocardial ischemia during exercise, target HR = HR of induced myocardial ischemia—10 bpm. Based on the results of the CPET, the doctor developed a personalized exercise prescription for the subjects and explained to them prior to the initiation of exercise therapy. All subjects also received training on exercise safety precautions.
Subjects downloaded the R Plus Health App V1.0 patient terminal (Recovery Plus Inc), which wirelessly connects to a chest-worn HR belt (Recovery Plus Inc) and can measure exercise frequency, intensity, time, volume, and progress. The app has a movements library with more than 2000 movements, and doctors can select and combine movements on the R Plus health App V1.0 doctor terminal (Recovery Plus Inc) according to the exercise prescription and set the target HR, safe HR, exercise frequency, intensity, and time of the exercise prescription. Then, the app will send different exercise prescriptions suitable for patients according to the assessment results, elderly patients and young patients have different exercise prescriptions, and for patients who cannot stand, it will also provide bedridden exercise prescriptions to ensure the maximum benefit of all patients. During exercise, the app provides demonstration videos of each exercise in the personalized exercise prescription, and participants were asked to follow the videos and wear a HR belt. If the target HR is not reached, the app will give a voice reminder to speed up the frequency of movement. Similarly, when the patient's HR is fast, the app alerts the patient to slow down. When the patient's HR exceeds the safe HR range, the app will directly ask the patient to stop exercising and automatically send a message to the doctor terminal to remind that the existing exercise prescription may be too intense and needs to be adjusted. Doctors can also remotely monitor patient performance and view patient information and symptom alerts by the doctor terminal. In addition, the app can automatically calculate the cumulative exercise time to reach the target HR, showing the amount of exercise remaining for a week to reach the target. The user interface of the R Plus Health APP is shown in Figure 1. In addition, researchers conducted monthly follow-up phone calls with all subjects to understand their progress and answer their questions.

The user interface of the R Plus Health APP.
Outcomes
The primary outcome was the feasibility of the project. Feasibility was defined as the extent to which subjects actively participated in and maintained the program. Participants were considered active if their daily effective exercise (HR during exercise reached the target HR) was ≥10 min or ≥3 effective exercise days per week as automatically recorded by the app. Safety events and medication adjustments will also be recorded.
All subjects were assessed before (T0) and after the intervention (T1). To measure cardiorespiratory endurance, all participants underwent symptom-limited CPET using the PFT4 ergo CPET system (COSMED QUARK, ITALIA) on a treadmill according to the Bruce protocol. The 12-lead electrocardiogram (ECG) and HR were recorded continuously before, during, and 10 min after the test until HR, ECG, and oxygen uptake(VO2) returned to baseline values. Blood pressure (BP) was measured manually every 2 min with a sphygmomanometer. Subjects were encouraged to exercise. When they have reached their own defined limits of functional capacity (feeling strained or breathless), or have angina-like chest pain, extreme fatigue, elevated arterial BP greater than 230/120 mmHg, ST segment depression of at least 2 mm on ECG, or severe arrhythmia, the test stops. During and at the end of CPET, the patient's subjective level of perceived exertion was quantified every minute using the Borg (6–20) scale. All CPET procedures were performed by a cardiologist and an assistant. The following parameters were collected as follows: peak metabolic equivalents, peak VO2, oxygen consumption at AT (AT VO2), peak cardiac output (CO), resting and peak HR, resting and peak BP.
Fasting blood glucose, total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, blood uric acid, serum creatinine, white blood cell, red blood cells, and hemoglobin were all measured by drawing venous blood. In addition, the left ventricular ejection fraction (LVEF) was measured by echocardiography.
The Patient Health Questionnaire-9 (PHQ-9) is a self-report measure of depression consisting of nine items matching the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria of major depression. Patients are asked to rate each of the items on a scale of 0 to 3 on the basis of how much a symptom has bothered them over the last 2 weeks (0 = not at all, 1 = several days, 2 = more than half the days, 3 = nearly every day). The summed-item score simply adds up the scores from each of the items to give a total score ranging from 0 to 27. A cutoff score of 10 or above on the summed-item score has been recommended as a method of screening for major depressive disorder. 13
A 12-Item Short Form Survey (SF-12) was used to measure the quality of life of patients. SF-12 contains 12 items with a total score ranging from 0 to 100, with higher scores indicating a better quality of life. 14
We used the ENRICHD Social Support Inventory (ESSI) to assess social support among participants. The full-length ESSI is a seven-item self-report survey that was developed to screen post-myocardial infarction patients for low levels of perceived support as part of the inclusion criterion for the Enhancing Recovery in Coronary Heart Disease (ENRICHD) trial. It measured the four defining attributes of social support as follows: the emotional, instrumental, informational, and appraisal by using the questions. The response options of each item ranged from 1 (none of the time) to 5 (all of the time) except the question of “Are you currently married or living with a partner?” which is a Yes/No type of question. According to the criteria of the ENRICHD protocol (Version 7.0), low perceived social support was defined as a total score of ≤18 and a score ≤3 in terms of at least 2 items, excluding items 4 and 7. 15
Pittsburgh sleep quality index (PSQI) is the most widely used sleep health assessment tool. The PSQI consists of 24 questions to be rated, 19 of which are self-reported and 5 of which require secondary feedback from a room or bed partner. Only the self-reported items (15 rated as 0–3 while 4 open-ended) are used for quantitative evaluation of sleep quality as perceived by the patient. The open-ended items are also finally scored as structured categorical values (rated at 0–3) as per the range of values reported. These 19 self-reported items are used to generate categorical scores representing the PSQI's seven components. Finally, the scores for each component are summed to get a total score (range: 0–21). This score provides an efficient summary of the patient's sleep experience and quality for the past month. 16
General Anxiety Disorder-7 (GAD-7) consists of seven items measuring worry and anxiety symptoms. Each item is scored on a four-point Likert scale (0–3) with total scores ranging from 0 to 21 with higher scores reflecting greater anxiety severity. Scores above 10 are considered to be in the clinical range. 17
Statistical analysis
Use PASS 2021 software to calculate confidence intervals for one mean. Taking the peak VO2 as the reference index, the distance from mean to limits was set to 0.5, the standard deviation was set to 1, and the confidence level was set to 0.95.
Statistical analysis was performed using SPSS 26.0. Categorical variables will be described as percentages (%). Normally distributed variables were described with mean (SD), and within-group analysis was performed using paired-samples t-test. Non-normally distributed variables were described as medians (IQR), and within-group analyses were performed using the Wilcoxon signed-rank test. Missing values are treated with replacement missing values. The level of significance was defined as P ≤ 0.05.
Results
The sample size was calculated to be 18. To account for patient withdrawals, we planned to recruit 30 participants. A total of 31 subjects with stable CHD registered and all met the inclusion criteria (8 cases after PCI, 23 cases of CHD), and all subjects successfully started the program. A total of 28 patients (90.3%) completed all interventions and follow-up. Three people (9.7%) withdrew; the reason for the withdrawal including one went abroad, one moved to another city, and one refused to use it for webless. The flow chart of study participants is shown in Figure 2.

Flow chart of study participants.
The demographic and clinical characteristics of the study population are shown in Table 1. The mean age was 56.2 years (SD 13.4), 96.8% (30/31) were male and 3.2% (1/31) were female. Mean BMI was 26.2 kg/m2 (SD 2.9). A total of 32.3% (10/31) had hypertension, 9.7% (3/31) had diabetes, and 54.8% (17/31) had dyslipidemia. A total of 67.7% (21/31) had a history of smoking.
Clinical and demographic characteristics of the study population.
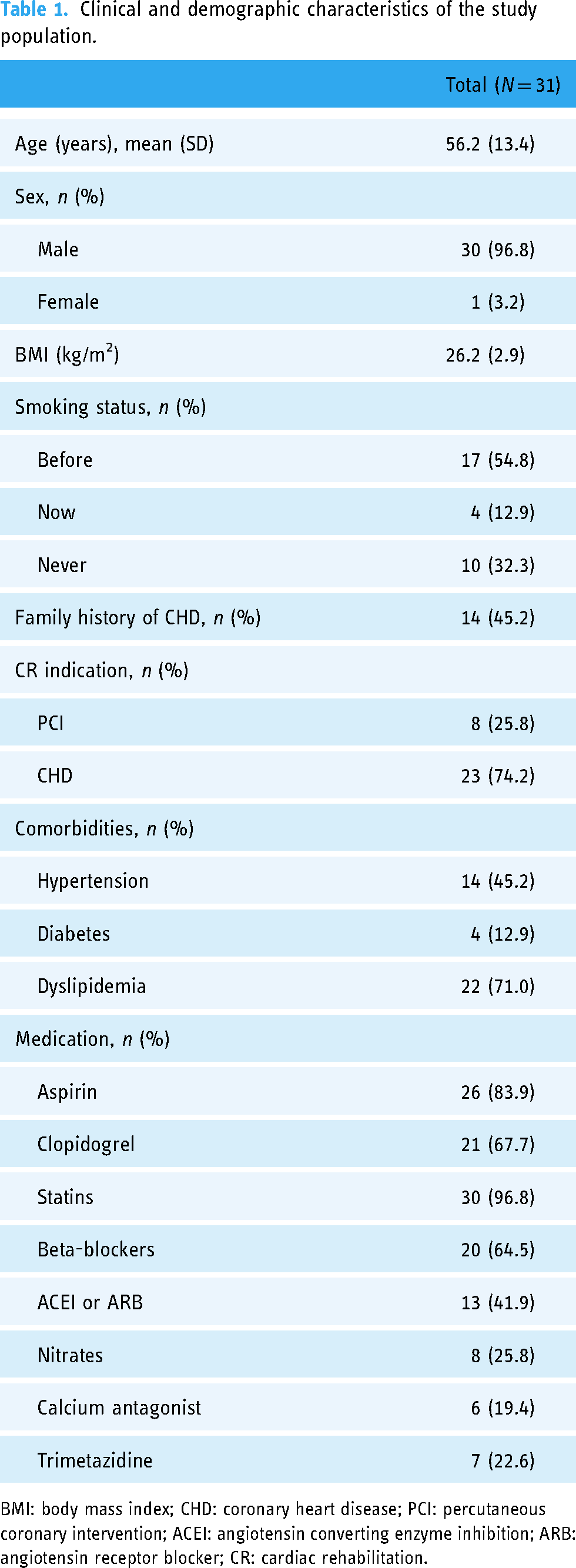
BMI: body mass index; CHD: coronary heart disease; PCI: percutaneous coronary intervention; ACEI: angiotensin converting enzyme inhibition; ARB: angiotensin receptor blocker; CR: cardiac rehabilitation.
All 28 people who completed the program had a daily effective exercise time of more than 10 min, with an average of 39.4 min (SD 17.8), of which 53.6% (15/28) had a daily effective exercise time of ≥30 min, 39.3% (11/28) were in 20–30 min, and 7.1% (2/28) were less than 20 min. The average effective exercise days per week was 4.6 days (SD 2.2), of which 50.0% (14/28) were ≥5 days, 17.9% (5/28) were between 3 and 5 days, and 32.1% (9/28) were less than 3 days.
After 12-week of intervention, the patient's cardiorespiratory endurance significantly improved, peak VO2 increased from 22.43 to 23.66 ml/kg/min (MD 0.6, 95%CI 0.1 to 2.4, P = 0.041), peak metabolic equivalents increased from 6.35 to 7.04 METs (MD 0.3, 95%CI 0.1 to 1.2, P = 0.018). The mean DBP at rest decreased significantly, from 72.29 to 67.36 mmHg (MD 2.3, 95%CI −9.7 to −0.1, P = 0.044). LDL dropped from 2.35 to 1.91 mmol/l (MD 0.2, 95%CI 0.0 to 0.8, P = 0.036). In addition, GAD-7 (95%CI −3.5 to 0.0, P = 0.014) and PHQ-9 (95%CI −2.0 to −0.5, P = 0.013) scores were significantly lower than those before intervention. In addition, SBP at rest, AT VO2, total cholesterol, triglycerides, blood uric acid, serum creatinine, and PSQI score trended down, while peak CO, HDL, red blood cells, hemoglobin, LVEF and ESSI score increased, although the difference was not statistically significant. Differences in outcome measures after the intervention are shown in Table 2.
Exercise records and effectiveness between baseline and 12 weeks.

HR: heart rate; CO: cardiac output; LVEF: left ventricular ejection fraction.
Blood glucose, total cholesterol, HDL, LAL, and triglycerides data were missing at T1 from 18 participants.
Blood uric acid and serum creatinine data were missing at T1 from 19 participants.
White blood cell, red blood cells, and hemoglobin data were missing at T1 from 21 participants.
LVEF data were missing at T1 from 20 participants.
*Significant treatment effect, P < 0.05.
Regarding safety, no serious adverse events occurred during the 3-month course. The patient reported a total of three muscle weakness and three muscle pain.
Discussion
In this study, we evaluated the clinical efficacy and feasibility of a smartphone app for prescribed exercise tutoring with exercise demonstration videos combined with wearable devices to monitor HR in patients with stable CHD. Of the 31 subjects who successfully followed the app to start exercising, 28 (90.3%) eventually completed the 12-week program. After the intervention, we observed significant improvements in cardiorespiratory endurance (peak VO2 and metabolic equivalents), DBP at rest, LDL, PHQ-9, and GAD-7, without serious adverse events. Therefore, we initially believe that the app is valuable and can be used by patients with stable CHD, but only if the patient can use a smartphone. But, these findings should be considered exploratory.
A recent systematic review showed that patient adherence in all types of digital CR was almost equal to or better than traditional center-based CR, 6 most reported program completion rates of over 80%,9,18 with only one exception. 19 The program completion rate of our study reached 90.3%, higher than other remote digital CR studies with the same duration of intervention.20,21 In addition, 92.9% (26/28) of the patients were able to exercise in the prescribed intensity for at least 20 min per day, and 67.9% (19/28) of the patients were able to exercise in the prescribed intensity for at least 3 days per week, meeting the requirements of the exercise prescription in guidelines for CR programs 22 and Chinese expert consensus. 12 This suggests that exercise demonstration videos may improve patients’ motivation to exercise at home, and professional exercise demonstration videos allow patients to schedule more exercise according to their own time rather than having to schedule appointments with a trainer/physical therapist. It is worth noting that in the only study with a low completion rate, the authors mention that part of the reason is that the patient's HR or BP exceeded the safety threshold built into the app during the warm-up phase, causing frequent interruptions and affecting patient motivation. 19 This reminds that subsequent similar studies may need to appropriately adjust the safe physiological parameter range or the intensity of exercise, and conduct extensive testing in advance.
In our study, patients’ cardiorespiratory endurance (peak VO2 and metabolic equivalent) was significantly improved after the 12-week intervention, which was consistent with the results of previous studies. However, it appears that the degree of improvement in cardiorespiratory endurance was lower compared to other studies with similar intervention. Patients in our study had an average increase in peak VO2 of 0.6 ml/kg/min and an average increase in metabolic equivalents of 0.3 METs. However, peak VO2 increased on average by about 4 ml/kg/min in studies of Frederix et al. 23 and Kraal et al., 21 metabolic equivalents increased by an average of 1 METs in the study of Harzand et al. 20 Compared with previous studies, our study is more effective in improving cardiorespiratory endurance in CHD patients than usual care intervention with only routine follow-up,24,25 but not as good as center-based CR.21,23 This result is different from the previous view that remote digital CR has almost the same improvement in cardiorespiratory endurance in CHD patients as center-based CR,18,26,27 or even better.9,19 The reason may be our consideration of exercise safety. Because the app is a new product, we do not know how the instructional video affects the user's motivation to exercise, and our target population is CHD patients. Therefore, for the sake of exercise safety, we maintained the patient's exercise prescription intensity during these 12 weeks without progressing. But in fact, almost all exercise-based CR interventions need to be advanced to achieve greater benefits. Based on the good completion rate of this study and no serious adverse events, we will adjust the intensity of the exercise prescription based on patient feedback in the follow-up study. It should be noted, however, that the cardiorespiratory endurance benefit of remote digital CR appears to be temporary. Smolis-Bak et al. 28 found that the peak VO2 of patients increased significantly after 3–4 months of intervention, but returned to the baseline level at the 12-month follow-up. More research is needed to explore the continuation of the effect of remote digital CR on cardiorespiratory endurance.
The intervention program in our study also demonstrated a BP management effect (DBP at rest dropped significantly), supporting the view that remote digital CR intervention can improve BP management in some studies.20,25,29 However, some studies have not found significant impact on BP management.9,23,30 The reason for the heterogeneity may lie in the adequacy of exercise intensity. Improvements in BP have been observed in almost all studies that set a target exercise intensity for patients based on the results of exercise function tests, including our study. However, if only the duration and frequency of exercise are recorded, BP does not seem to be improved. Therefore, monitoring and urging patients to achieve target exercise intensity seem to be the key to good benefits of remote CR. Most studies concluded that the impact of remote CR on BP management was similar9,23 or even better than that of center-based CR.25,29 Contrasting results were observed in two studies, one of which monitored exercise only by means of a patient diary, 31 while the other reported poor patient compliance due to technical reasons and did not implement exercise prescriptions as required. 19
In addition, we also found a significant decrease in LDL after the intervention, although some data were missing (18), but still reflected some trends. It is well known that LDL is one of the risk factors for CHD. 32 It has been found that digital CR can reduce triglycerides23,29 and increase HDL, 23 which is consistent with the effect of center-based CR.18,19,23 At present, there is no research report that remote digital CR can reduce LDL. Studies have shown that aerobic exercise can significantly reduce LDL and bring benefits to blood lipids in patients with CHD, but the exercise intensity may be at least 65% VO2. 33 In our study, we set the target exercise intensity through the AT method and monitored the HR to urge the patient to reach the exercise intensity. Therefore, sufficient exercise intensity may have caused the reduction of LDL, which once again emphasizes the importance of exercise intensity monitoring for remote CR.
Studies have shown that patients have negative emotions when faced with a CHD diagnosis, anxiety and depression are predictors of poorer outcomes. 34 Several previous studies have reported improvements in psychological status of patients with remote digital CR intervention.27–29,31 In our study, patients’ GAD-7 (anxiety) and PHQ-9 (depression) scores were also significantly improved after the intervention. However, there are also studies pointing out that remote digital CR does not seem to improve patients’ psychological status.26,35,36 We found that too much pushing patients to focus on their disease may be the reason for their poor psychological improvement. For example, patients in the study of Brough et al. 35 needed to pass online exams on CHD and cardiac physiology to get the next stage of the CR program, which may cause additional psychological stress for patients. Dale et al. 37 also speculate that remote CR did not improve psychological status of patients as much as center-based CR did because of the anxiety caused by these text messages about their own disease. Educating patients about their disease is important, but how can we not to stress them out too much? Therefore, further and longer interventions are warranted to examine the effects of remote CR on the psychological status of patients with CHD.
We also observed improvements in multiple secondary outcomes, including decreases in total cholesterol, triglycerides, serum uric acid, serum creatinine, PSQI scores, and increases in HDL, red blood cells, hemoglobin, LVEF, and ESSI scores. Perhaps due to the small sample size, the changes in these outcomes were not statistically significant, but they still need attention because of their relevance to the prognosis of CHD patients. There are outcomes that have been confirmed by other studies, such as reduction of triglycerides, 23 increase of HDL 18 and hemoglobin, 25 but other outcomes such as serum uric acid and serum creatinine were absent.
One study provided exercise demonstration videos online for patients performing CR remotely, but did not use wearables to monitor exercise safety. 26 At present, the digital CR intervention of smart mobile devices combined with wearable sensors still requires doctors/coaches to spend a lot of time on exercise guidance for patients. 6 Our study is the first to evaluate the clinical efficacy and feasibility of a smartphone app for prescribed exercise tutoring with exercise demonstration videos combined with wearable devices in patients with stable CHD. However, our study was a single-arm feasibility study, so several limitations should be considered, including the absence of a control group, the short duration of the intervention (12 weeks), and the small sample size. The study included only stable CHD patients with a low risk of exercise safety and were mostly male (96.8%), so results cannot be extrapolated to other patients.
Conclusions
It is feasible and safe for patients with stable CHD to use the smartphone app for prescribed exercise tutoring with exercise videos combined with wearable devices to monitor HR, and it can improve the patient's cardiorespiratory endurance, BP, LDL, depression, and anxiety. Quantitative monitoring and tutoring on an exercise prescription may be the key to improving the effect of remote CR. Patients showed good adherence, so this smartphone app is a viable alternative when patients cannot participate in center-based CR for various reasons, but whether it can achieve the effect of central rehabilitation and ensure safety needs further long-term, controlled, and multicenter studies.
Footnotes
Acknowledgements
The authors would like to thank the past and present members of the continuing cardiac care team for support and assistance throughout this study and Recovery Plus for its products and technical support.
Contributorship
KX designed the protocol for this study, and CB drafted this article. KX and CB contributed equally to this work and should be considered co-first authors. YK, XF, YL, and BL were responsible for patient management and data collection. RD provided supervision for the entire project. CB and XC provided support for the pre- and post-test data analyses. All authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was registered with the Chinese Clinical Trials Registry (ChiCTR1800019144) and approved by the Research Ethics Committee of Peking University People's Hospital (2018PHB080-01).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number No.81772446).
Guarantor
Rongjing Ding
