Abstract
During recruitment for a large, decentralized clinical trial for high-risk individuals with COVID-19, respondents were either transferred in real-time to a clinical research coordinator (i.e. warm transfer), or a callback time was arranged. A retrospective analysis was conducted on 2341 respondents comparing the rate of enrollment among those who were warm-transferred and those for whom a callback was arranged. A respondent who warm-transferred was significantly more likely to enroll in the clinical trial.
Introduction
The difficulty with participant recruitment in clinical trials is well established.1,2,3 Despite great costs in both time and money, many clinical trials fail due to poor recruitment. In a study of 894 randomized clinical trials (RCTs), it was demonstrated that the most common reason for study discontinuation was poor recruitment. 1 If an RCT is not ended prematurely, other consequences of poor recruitment include increased cost of research, diminished validity, and, perhaps worst of all, a delay in the availability of new treatments and diagnostics for patients in clinical settings. Clinical trial investigators and study sponsors opt for various recruitment strategies, which may involve some combination of advertisement, word of mouth, and referral. In order to understand the research efforts to enhance recruitment in clinical trials, previous literature has identified successful strategies that can be built upon and improved. These strategies include various interventions such as implementing phone call follow-ups and making alterations to study design, aiming to optimize recruitment processes and address potential barriers to improve overall recruitment rates.3,4 Because of the COVID-19 pandemic and the infeasibility of traditional clinical trial procedures (e.g. during the pandemic, people were advised not to leave their homes, likely limiting their ability to participate in a clinical trial that involves in-person visits), clinical trial stakeholders have been introduced to a variety of decentralization methods, some of which might be expected to improve enrollment outcomes and prevent study discontinuation. 5 Given the aforementioned consequences of poor recruitment, we propose that innovative recruitment strategies that utilize digital technologies are warranted and may be implemented to save time, and money, and ultimately bring new treatments to patients in a faster and more efficient way.
A recruitment strategy utilizing warm transfers (WTs) was developed at a decentralized clinical research site in the context of a large COVID-19 clinical trial. Here, the term “warm transfer” refers to the immediate transfer, via phone, of a pre-screened potential clinical study participant by a patient experience coordinator (PEC) to an available clinical research coordinator (CRC) in real-time (Figures 1 and 2).

Warm transfer workflow.
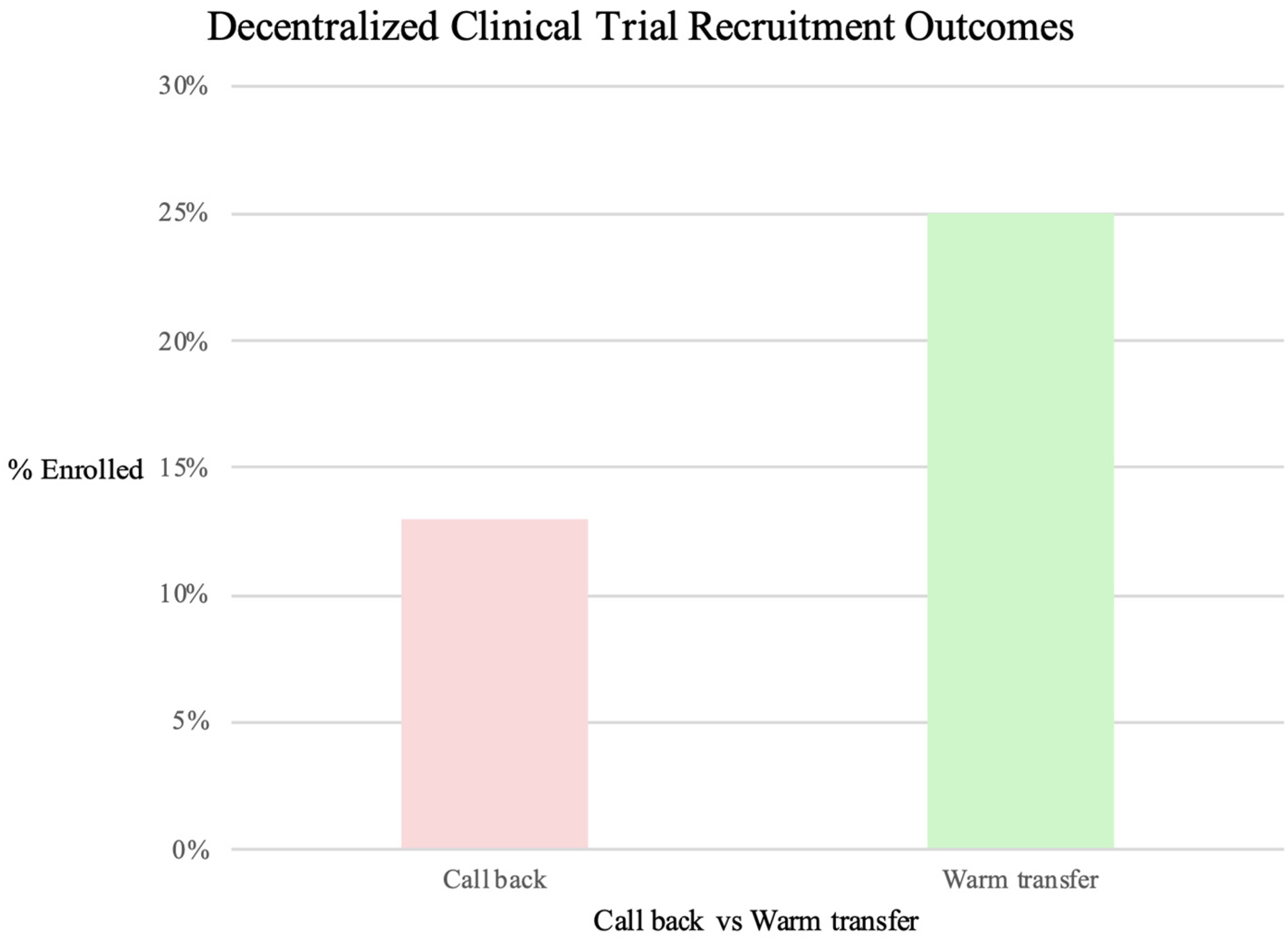
Rate of enrollment, warm transfer versus call back.
PECs and CRCs were trained on the WT workflow, in which PECs administered a pre-screen questionnaire (PSQ) to interested respondents. PECs were trained to administer an Institutional Review Board (IRB)-approved script prior to the PSQ. The PSQ contained questions designed to elicit information relevant to the inclusion/exclusion criteria of the study (see the Methods section). Upon completion, PECs “handed off” potentially eligible respondents to CRCs for the next part of the study, which involved further screening and opportunities for participant education. To facilitate this step, a PEC either transferred the respondent to a CRC in real-time via phone or arranged for the respondent to be called back. Callbacks occurred when a CRC was not immediately available to speak with a respondent at the moment. To our knowledge, this is the first study investigating the utility of WT in a clinical research setting using a comparator. Quantifying the potential value added by WTs has implications for training, resource allocation, and the efficient planning of a recruitment strategy for clinical trials.
Digital communication tools were used to facilitate phone calls and real-time messaging among the study staff.
Methods
A retrospective analysis was conducted on 2341 respondents who expressed interest in participating in a clinical trial and completed a brief telephone screen (administered by a PEC). The respondents in this study were identified in a variety of ways including both traditional (e.g. referral and word of mouth) and digital (e.g. social media) methods. Respondents were individuals interested in participating in a clinical trial for COVID-19 (see https://clinicaltrials.gov/ct2/show/NCT04504032 for a full listing of the inclusion/exclusion criteria for the parent study as well as primary/secondary outcome measures). Respondents were identified using IRB-approved recruitment materials. Written informed consent was obtained for all participants in the parent study. Permission to retain data was obtained from the participants.
Respondents were classified by group: those who were warm-transferred and those for whom a callback was arranged and then by screening outcome. A chi-square test of independence was performed to determine if there was an association between group and screening outcome. The data were sorted according to group and values were tabulated using R language and environment for statistical computing (r-project.org). Here, the screening outcome is the result of what happened after a participant was screened: either screen failed or not screen failed. “Enrolled” refers to a participant who has signed a consent form.
Results
Twenty-five percent of respondents who were warm-transferred enrolled in the study (65 out of 260) while only 12.9% of those respondents for whom a callback was arranged ultimately enrolled in the study (270 out of 2081). A greater proportion of callbacks were arranged than WTs were made at approximately 8:1. A chi-square test of independence indicated that there was a significant association between group and screening outcome X2 (1, N = 2341) = 27.26, p < 0.00001.
A second chi-square test was conducted to determine if there was an association between the group and screen fail outcome. The chi-square test did not show a significant association between group and screen fail status X2 (1, N = 335) = 1.53, p = 0.217. Minor differences in the rate of screen failure after enrollment as WT respondents’ screens failed at a rate of 9.23% (6 out of 65) while callback respondents’ screens failed at 5.19% (14 out of 270). The comparative demographics data between the WT and callback participants, including information such as age, sex, and race can be found in Appendix 1.
Discussion
Our data indicate that there is a strong association between WTs and enrollment in a clinical trial, relative to callbacks. These results support the hypothesis that there is value in a recruitment strategy that utilizes WTs.
Several variables may explain this association. We suggest that competent staff who are prepared to educate prospective research participants in real-time may build trust and add credibility to the recruiters while decreasing the likelihood that a potential participant declines participation on the basis of confusion or misunderstanding. The “callback” can also set the stage for such helpful interactions, but there is an added barrier: the respondent must answer the phone call from the study staff at the predesignated time. For the WT, the participant is already on the line and the conversation simply continues uninterrupted as the participant moves through the recruitment and screening phases of the study.
Additionally, disease progression or recovery can occur rapidly in COVID-19 and other populations in a way that affects eligibility for research studies. A participant who is eligible and interested in participation in a clinical trial may experience clinical deterioration within hours after first engaging with the study team, preventing enrollment into the trial because of the challenges of speaking on the phone when one is not feeling well. Since the WT occurs in real-time, the participant stands to benefit from a faster enrollment process.
Limitations of our study are noted. First, while we have identified a robust association, it is unclear if this result is generalizable. That is, it remains to be seen if this association persists across different study types and in different disease populations. Importantly, the parent study's inclusion criteria were time sensitive such that a participant would be deemed ineligible if the COVID-19 symptoms were greater than seven days old. This meant that participants had a narrow window of eligibility, likely bolstering our observed WT benefit. Other clinical trials with looser criteria and/or longer eligibility windows might not observe such a robust association. Our PSQ did not undergo validation. Also, though recruitment started in September 2020, the study team did not start tracking whether a respondent was a WT or callback until November 2020. Therefore, approximately 14% of the total respondents could not be included in this analysis. An additional 2% were not included due to incompleteness in the record. Finally, our results are agnostic of the referral source. Therefore, we are unable to determine if a particular recruitment channel (e.g. a digital channel) is more or less likely to result in a favorable recruitment outcome in the WT paradigm. It is possible that an individual who was ascertained via a digital method (e.g. social media advertising) is more likely to respond favorably or be advantaged in some way with respect to the WT. We believe that referral source and its relation to the WT is an interesting question that could be explored in further research. However, we do note that those who received a WT did not differ significantly in terms of their rate of screen failure (see the Results section) or demographics (see Appendix 1).
WTs are not without challenges. For a WT to occur, either a staff member must be dedicated to the cause of receiving WTs in real-time, or a staff member must hold other work to take a phone call at that moment. In this study, an approximate eight-fold greater proportion of callbacks were arranged than WTs were made. Despite this disparity, we noted a higher conversion rate from screening to enrollment for those participants that experienced a WT as part of their recruitment pathway. Given the potential upside, a dedicated staff member and/or team who handles 100% of the WTs could be warranted. However, as with any recruitment strategy, an analysis of the potential benefits weighed against the potential costs should be considered.
While WTs have been described in the clinical setting and clinical research setting, there appears to be no data on their effectiveness in improving recruitment outcomes. Herein, we have demonstrated that WTs are a promising option that could be used as part of a broader clinical trial recruitment strategy to improve recruitment efficiency. This appears to be especially relevant as the industry continues the trend toward decentralized clinical trial adoption, leveraging new technologies and reducing past inefficiencies. 6 Future research should be aimed at understanding the reproducibility and generalizability of this effect and what characteristics of clinical trials such as patient population, size, and therapeutic area might lend themselves to a recruitment approach that includes WTs.
Conclusion
In this study, we demonstrate an association between WTs and enrollment in a clinical trial. Therefore, WTs may be a useful strategy to improve recruitment outcomes in clinical research studies.
Footnotes
Acknowledgements
We thank the members of the Patient Engagement and Clinical Operations teams at Science 37 along with the volunteers who expressed interest in participating in the clinical trial. We thank the Bill & Melinda Gates Foundation for their collaboration with the clinical trial. We thank Binesh Kumar for his assistance with organizing the demographic data.
Contributorship
KP and JC conceived and designed the analysis. KP wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript. AG carried out the data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Respondents were identified using IRB approved recruitment materials. Permission to retain data was obtained from the participants. The Advarra IRB (Columbia, Maryland), which served as the central IRB, approved the parent study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
KP
Appendix 1

| Demographics comparison | Warm transfer enrolled participants (n=65) | Callback enrolled participants (n=270) |
|---|---|---|
| Median age (min–max), years | 51 (25–73) | 49 (20–74) |
| <65 | 56 (86.2%) | 232 (85.9%) |
| ≥65 | 9 (13.8%) | 38 (14.1%) |
| Male | 24 (36.9%) | 110 (40.7%) |
| Female | 41 (63.1%) | 160 (59.3%) |
|
|
||
| American Indian or Alaska Native | 0 (0.0%) | 1 (0.37%) |
| Asian | 0 (0.0%) | 2 (0.74%) |
| Black or African American | 9 (13.8%) | 16 (5.9%) |
| White | 55 (84.6%) | 240 (88.9%) |
| Mixed race | 0 (0.0%) | 0 (0.0%) |
| Other | 1 (1.5%) | 8 (2.9%) |
| Unknown | 0 (0.0%) | 3 (1.1%) |
Note. The table presents comparative demographic characteristics of the warm transfer participants enrolled in the study versus the callback participants enrolled in the study. The values represent frequencies and percentages within each category.
