Abstract
Objective
To facilitate replication and future intervention design of web-based multibehavior lifestyle interventions, we describe the rationale, development, and content of the AiM, Plan, and act on LIFestYles (AMPLIFY) Survivor Health intervention which provides healthy eating and exercise behavior change support for older cancer survivors. The intervention promotes weight loss, improvements in diet quality, and meeting exercise recommendations.
Methods
The Template for Intervention Description and Replication (TIDieR) checklist was used to provide a comprehensive description of the AMPLIFY intervention, consistent with CONSORT recommendations.
Results
A social cognitive theory web-based intervention founded on the core components of efficacious print and in-person interventions was conceptualized and developed through an iterative collaboration involving cancer survivors, web design experts, and a multidisciplinary investigative team. The intervention includes the AMPLIFY website, text and/or email messaging, and a private Facebook group. The website consists of: (1) Sessions (weekly interactive e-learning tutorials); (2) My Progress (logging current behavior, receiving feedback, setting goals); (3) Tools (additional information and resources); (4) Support (social support resources, frequently asked questions); and (5) Home page. Algorithms were used to generate fresh content daily and weekly, tailor information, and personalize goal recommendations. An a priori rubric was used to facilitate intervention delivery as healthy eating only (24 weeks), exercise only (24 weeks), or both behaviors concurrently over 48 weeks.
Conclusions
Our TIDieR-guided AMPLIFY description provides pragmatic information helpful for researchers designing multibehavior web-based interventions and enhances potential opportunities to improve such interventions.
Introduction
Nearly 17 million Americans are living with a history of cancer, a number estimated to exceed 22 million by 2030. 1 The vast majority (89%) of these individuals are 50 years of age or older and are at increased risk of secondary primary cancers, medical comorbidities and disabilities, and accelerated ageing.1–3 These risks can be prevented, attenuated, and possibly reversed with a healthy diet, regular exercise, and beneficial weight management.4,5 Hence, it is concerning that national surveys indicate that 73% of primarily middle-aged and older cancer survivors do not meet the recommendations for vegetable and fruit intake, 53% do not meet aerobic exercise recommendations, over 90% do not meet resistance exercise recommendations, and 67% are overweight or obese.6,7
Healthy lifestyle promotion interventions are critical to address this important public health need and doing so with a web-based approach could enhance reach and impact. Although the number of eHealth interventions for promoting healthy eating, regular exercise, and/or weight loss is growing among cancer survivors, most have not been tested in large randomized controlled trials with long-term follow up.8–10 Moreover, their effects are inconsistent with benefits modest, at best, and few have explicitly targeted older cancer survivors.9–12 Hence, there is a need to leverage preferences from the target population to enhance outcomes. To do so, we identified features of web-based healthy lifestyle interventions preferred by a racially and geographically diverse sample of middle-aged and older cancer survivors from the region where the intervention was developed (i.e. 51% women, 40% Black, 56% ≥ 65 years old, 33% ≤ 12 years of education, and 33% rural with varied cancer types (30% breast, 32% prostate, 38% mixed)). 13 These features (e.g. reliable information from content experts, interactive/tailoring, flexibility regarding time and frequency of participation, privacy) informed the design of the AiM, Plan, and act on LIFestYles (AMPLIFY) Survivor Health web-based intervention.13,14
As previously described, AMPLIFY efficacy testing includes three inter-related randomized controlled trials testing the effects of a: (1) diet only module, (2) exercise only module, and (3) diet and exercise modules delivered in sequence (diet then exercise or vice versa), or concurrently (combined module) on body weight, diet quality, physical activity, and related outcomes in cancer survivors 50 years of age or older. 14 The purpose of this article is to describe the rationale, development, and content of the web-based intervention modules for AMPLIFY and, in so doing, complement our prior randomized trial description. Reporting intervention details offers an important opportunity for future researchers and intervention developers to replicate and improve upon current designs. 15
Methods
We use the Template for Intervention Description and Replication (TIDieR) checklist 16 to guide our comprehensive description of the AMPLIFY intervention. This intervention is currently being tested in three inter-related randomized controlled trials led by the University of Alabama at Birmingham and is recruiting nationwide across the United States. 14 We provide, in this Educational Piece, new methods related to development and integration of multiple behavior web-based interventions for older cancer survivors. By providing a comprehensive description of the AMPLIFY intervention design, processes, modifications, and interface with nonintervention research activities, this report complements our prior randomized study design publication. 14
The TIDieR checklist was chosen because of its rigorous development using literature review and Delphi survey with international experts. 16 Further, TIDieR has been successfully used to describe digital health interventions in populations other than cancer survivors and is highly recommended by CONSORT for intervention descriptions.17–19 Our results are organized according to each of the 12 TIDieR criteria which are the intervention name, why (rationale, theory, or goal), what (material and procedures), who (intervention providers), how (delivery mode), where (intervention setting), when and how much (intervention time period, timing, and frequency of intervention activities), tailoring (personalization), modifications (intervention changes during the course of the study), and intervention delivery (adherence and fidelity). A completed TIDieR checklist is provided as Supplemental TIDieR Checklist File.
Results
Overview of development process
In practical terms, the development approach was a pragmatic and iterative process of brainstorming, idea selection based on feasibility, and refinement based on user input. This collaborative process involved a multidisciplinary investigative team, web design experts, and cancer survivors. The investigators and the web designers had an established relationship based on prior collaborations. The relationship with the cancer survivors was developed using an Institutional Review Board approved protocol (UAB IRB-300002068) recruiting cancer survivors for the formative focus groups, beta testing, and the ongoing process evaluation recruitment. 13
The multidisciplinary team of investigators and the web design team met biweekly with interim action items completed and intervention element reviews performed between meetings. Two years were required to develop a sufficient nidus of intervention material to proceed with the efficacy launch after which regular meetings continued to complete the final elements in time for participants to receive them on schedule and address programming or content issues not identified during beta testing. We continue to meet regularly to address the infrequent refinement required during efficacy testing. A regular agenda item for these meetings has been cancer survivor input facilitated by rapid cycle analyses of our focus groups then beta testing feedback and now ongoing process evaluation based on best practices for mixed methods approach. 20 Because of our foundation of efficacious interventions (criterion 2), intervention development was based on the Cultural Adaptation Process model, an accepted adaptation science approach to translating efficacious interventions to new contexts and delivery channels.21,22 An overview of the intervention description by TIDieR criteria is provided in Table 1 with additional relevant details reported in the following sections.
Overview of the

FAQ: frequently asked questions.
TIDieR criterion 1: Brief name
When funded by the National Institutes of Health (NIH), the title of this project was AMPLIFI (Adapting Multiple behavior
TIDieR criterion 2: Why (rationale, theory, or goal of the intervention core components)
Rationale for the theoretical underpinning
The intervention is based on the social cognitive theory of health behavior change which has frequently been used to promote a healthy diet and regular exercise in cancer survivors.23–27 Self-efficacy and other important constructs such as perceived barriers, outcome expectations, behavioral capability, and role models were targeted in our intervention content, messaging, and structure. 28 Participants are encouraged to achieve recommendations for body weight, diet, and exercise for cancer survivors and older adults (Table 2).29–33
AMPLIFY diet and exercise goals based on current healthy eating and exercise recommendations for cancer survivors and older adults

To avoid the risk of underweight, messaging encourages weight maintenance and discourages further weight loss at a BMI of 21.75 kg/m2. BMI: body mass index.
Efficacious elements adapted during intervention design
The AMPLIFY intervention adapts two efficacious social cognitive theory-based behavior change interventions. First, Reach-out to ENhancE Wellness in Older Cancer Survivors (RENEW) employed tailored print materials and telephone counseling to increase exercise, improve diet, and reduce body weight in order to enhance quality of life and physical functioning. 34 Secondly, the Better Exercise Adherence after Treatment for Cancer (BEAT Cancer) exercise intervention used print, in-person, group, and home-based activities to increase the odds of meeting aerobic physical activity recommendations for cancer survivors. 35
The adaptation effort focused on retaining the core components, which for RENEW and BEAT Cancer interventions included beneficial program elements from the participants’ perspective (individualization (personalized), accountability, encouragement, etc.) and social cognitive theory mediators of intervention effect (self-efficacy, barriers, etc.).25,36–42 The adaptation to a web-based platform was also facilitated by the development of an early prototype called Survivorshine (adapted from RENEW), 43 successful adaptation of BEAT Cancer core components to other contexts (e.g. rural cancer survivors),36,44–46 and formative focus groups with cancer survivors. 13 The components were translated by using the iterative process of brainstorming ideas, selecting ideas based on feasibility followed by creating and refining the element. Throughout this process we mapped core components to specific intervention elements and content (Table 3). The core component integration permeated all discussions while ensuring a user-friendly interface was a priority when making decisions regarding final core component application.
Elements of the AMPLIFY website

Core components refer to “active ingredients” retained during adaptation as described in the text, TIDieR item 2 (rationale for intervention core components). These included both social cognitive theory constructs and intended products of intervention activities.
Diet supplies sent to participants receiving the diet only or combined module. Exercise supplies sent to participants receiving the exercise only or combined module.
Private Facebook group membership was based on module received by participant.
FAQ: frequently asked questions.
After development and before launching efficacy testing, beta testing was performed with 15 cancer survivors (53% women and 41% African American; mean age of 64 ± 10 years). Institutional Review Board approval was obtained for beta testing activities and all beta testers provided informed consent before initiating any beta testing activities. Each beta tester was asked to complete a brief survey after viewing each e-learning tutorial and three think aloud interviews regarding website usability, etc. The resultant intervention elements mapped to the core components of the RENEW and BEAT Cancer interventions adapted for each feature are provided in Table 3.
TIDieR criterion 3: Physical or informational materials used in the intervention, including those provided to participants or used in intervention delivery or in training of intervention providers
Intervention overview
Elements of the AMPLIFY intervention include an interactive website, messaging (email and/or Short Messaging Service (SMS)), diet-related and exercise-related supplies shipped to the participants’ home address, and private Facebook groups. Figure 1 provides an overview of the inter-relationships among these elements. The AMPLIFY website is used to deliver the individual behavior, sequenced, or concurrent interventions using three foundational modules: (1) diet module (24 weeks); (2) exercise module (24 weeks); and (3) combined module used for concurrent delivery of diet and exercise (48 weeks). 18 The modules were developed in concert (see criterion 8) to promote design efficiency and consistency. The individual behavior modules were used for the sequenced intervention.
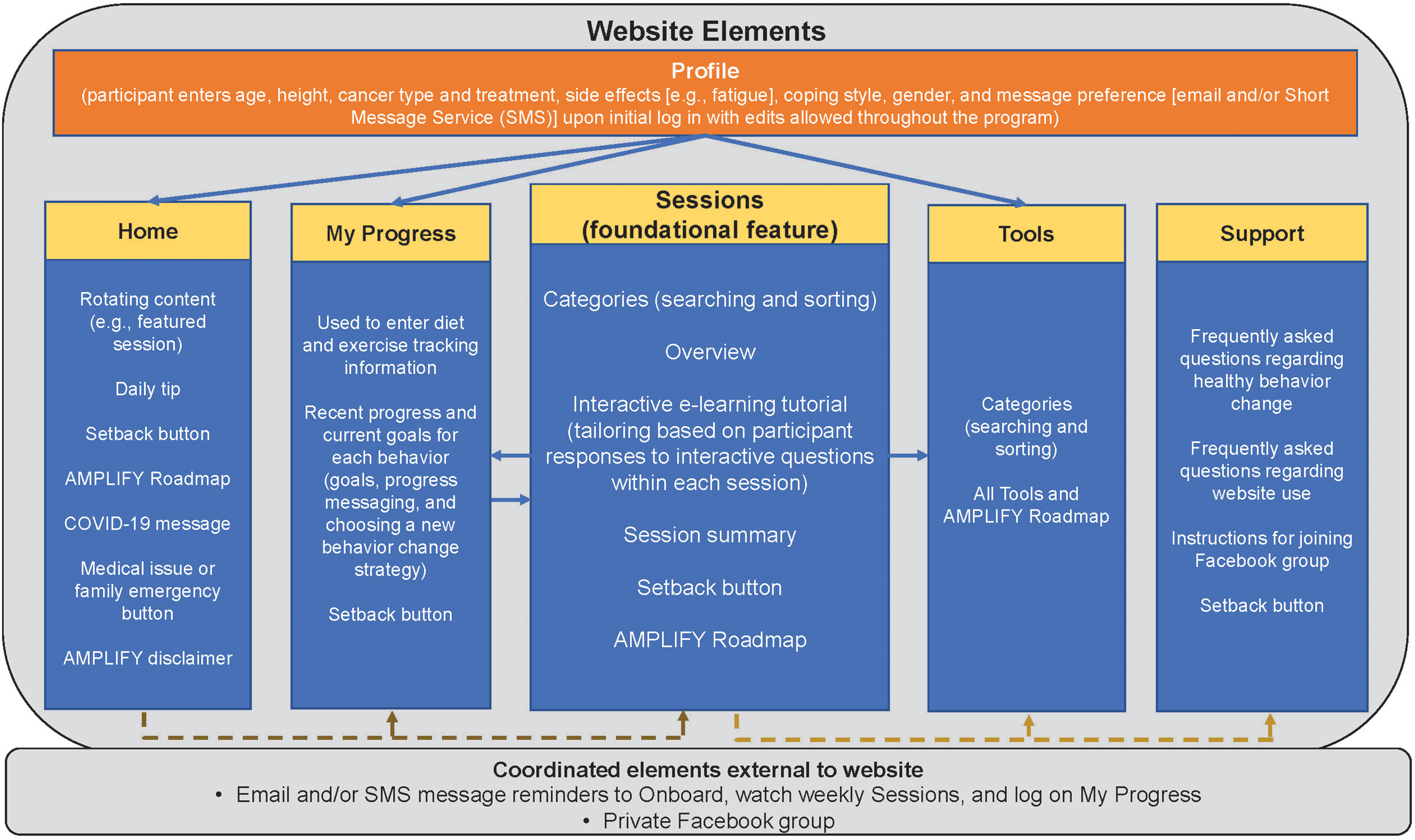
Schematic representation of the AMPLIFY website describing contents of each website section; dashed arrows indicate section connections that improve website navigation; solid arrows indicate data connections that tailor (or individualize) information seen by the participant (e.g. profile information used to tailor Home page cancer survivor story to cancer coping type, My Progress red meat and alcohol recommendations tailored to gender, and Tools to cancer type and side effects; My Progress information is used to tailor e-learning tutorial messaging to tracking behavior; Sessions section is used to tailor My Progress options (e.g. hide request to track red meat if a vegetarian) and Tools to most appropriate weekly session); email/SMS reminder messages and Facebook groups are nonwebsite elements that coordinate and track with intervention progress (e.g. messages are consistent with the weekly session topic).
Website overview
The AMPLIFY website (https://amplifymyhealth.org) is accessible via participants’ personal computers and portable devices (e.g. tablets, smart phones). Participants log onto the website with a username and password. In addition to a Home page, the website has the following sections or tabs: Sessions, My Progress, Tools, and Support (Table 1).
Sessions
Each module includes weekly educational diet (24 sessions), exercise (24 sessions), or combined sessions (48 sessions) located in the Sessions section of the website (Table 1). These educational sessions are the central aspect of the website and provide a foundation for the remaining intervention elements. 14 The Sessions section includes an overview page with categories for easier weekly session searching. Diet-related categories include managing weight, eating to reduce cancer risk, shopping and food preparation, and making healthy behavior changes. Exercise-related categories include motivation, social support, restarting exercise, getting stronger, and overcoming behavior change barriers.
The weekly sessions are based on a “Spiral Curriculum” 47 so that each session builds on what was presented in earlier sessions. This is especially important for resistance exercise requiring multiple sessions to introduce recommendations, basic information about safety and how to do the exercises, gradually adding new exercises over time and helping participant develop a personal plan, and progression. However, each session is designed to “stand alone” if the participant chooses to skip earlier sessions. Details regarding session topics are provided in Supplemental Table S1.
Session 1 for each module is a self-assessment that includes comparison of current behavior with recommendations for cancer survivors and older adults (i.e. long-term goals; Table 2).29–33 This initial self-assessment is required to progress to and view further intervention content. Self-assessments for diet ask for the participant's weight, dietary/health practices (e.g. snacking after dinner, proportion of whole to refined grains, and supplement use) and food frequencies (e.g. intakes of red meat, vegetables, fruits, alcohol). Self-assessments for exercise ask about aerobic, resistance, flexibility, and balance exercise behavior. Exercise self-assessment also asks about walking self-efficacy 48 and physical function-related abilities (e.g. can you get up off the floor by yourself, can you walk a mile, how many flights of stairs can you climb) to inform messaging that directs the individual's focus toward physical functioning and endurance benefits they are achieving (or can achieve) from increased physical activity and exercise. Self-assessments are then repeated at weeks 12 and 24 for a total of three within the Diet and Exercise modules. Initial and follow-up self-assessments in the combined module require two weekly sessions (one for diet and one for exercise) and occur at weeks 1, 2, 23, 24, 45, and 46. Follow-up self-assessments are encouraged but are not required to view additional content.
Sessions other than self-assessments include interactive, educational e-learning tutorials on healthy eating, weight management, exercise, and other health promoting strategies (e.g. managing stress, social support, preventing setbacks; Supplemental Table S1). These tutorials were created using the Articulate Storyline© software (https://articulate.com/360) which uses branching and feedback to tailor to participant responses and increase engagement. Where appropriate, tutorials interactively ask participants to respond to questions and responses are used to personalize content within the e-learning tutorial. Each tutorial ends with “take-home messages” and a behavioral challenge (“weekly challenge”) for the upcoming week (Supplemental Table S1). The subsequent week's tutorial begins by asking the participant how they did with last week's challenge and provides personally tailored reinforcement. The weekly session webpage provides a summary of the tutorial and links to relevant website resources (e.g. Tools) below the tutorial. Because content from diet and exercise tutorials were integrated for the combined module, careful attention and iterative reviews of the combined module were implemented to remove redundant content (see criterion 8).
My Progress
The My Progress section (Table 1; Figures 2 and 3) encourages self-monitoring, goal-setting, self-efficacy, use of behavior modification strategies, accountability, and motivation. The behaviors available for tracking are based on the session content (Supplemental Table S1). To enhance personalization and reduce participant burden, all tracking except for weight, aerobic exercise, and resistance exercise can be turned off 3 weeks after introduction on the website. Participants who do not eat meat or who never use alcohol are not asked to track intakes of these constituents. Daily calorie intake is only tracked during the first two weeks of the diet only and combined modules to further reduce participant burden.

My Progress tracking screen for the AMPLIFY diet module.
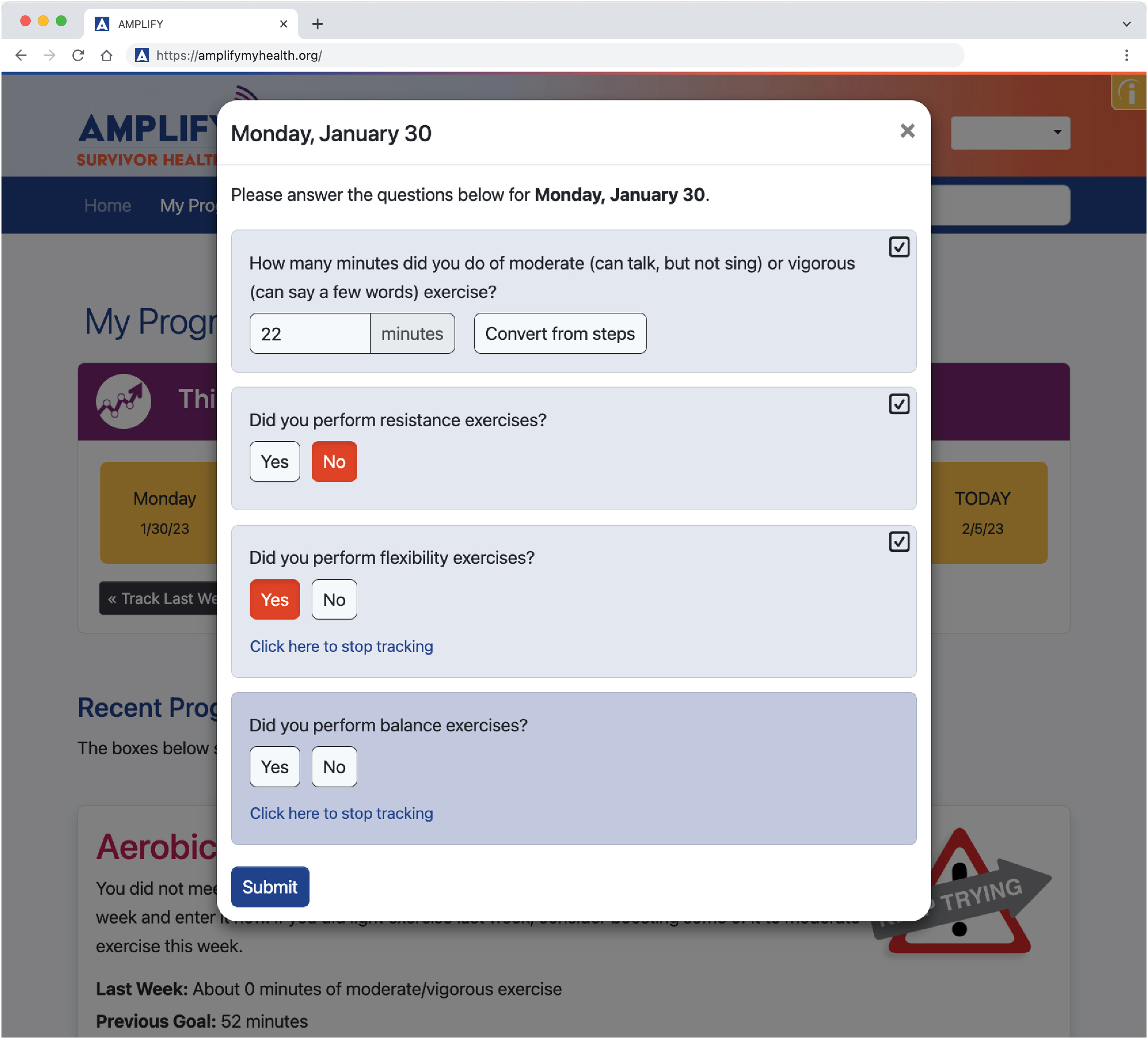
My Progress tracking screen for the AMPLIFY exercise module.
The My Progress messaging algorithms are based on setting weekly short-term behavior goals that contribute to meeting several long-term diet and exercise goals consistent with current recommendations for body weight, healthy eating, and exercise for cancer survivors and older adults (Table 2).29–33 The suggested diet goals are based on body weight change and intake frequency of food groups (e.g. servings of red meat) or eating behaviors (e.g. days per week participant snacked after dinner) tracked in the past week. The suggested short-term aerobic exercise goal is based on gradually increasing duration and intensity based on the self-assessment sessions and recent activity logged by the participant on My Progress. For simplicity, goals for resistance, flexibility, and balance exercise focus on weekly frequency with resources provided in the Sessions and Tools related to progressing intensity, repetitions, etc. To encourage progress toward meeting self-determined short and long-term goals, a menu of 5 to 10 modification strategies for each healthy eating and exercise behavior are provided using a pull-down menu which allows participants to choose one; strategies are rotated each week based on the week's topic, a predetermined order, or deterministic shuffling algorithm.
Tools
Additional education and application resources supporting healthy eating, weight management, and exercise are provided as Tools (Supplemental Table S1). There are 73 healthy eating/weight management tools and 48 exercise tools, all of which were integrated into the combined module. Among the Tools, the AMPLIFY Roadmap warrants highlighting because it was developed based on beta testing feedback (see criterion 10) requesting an overview of participant progress related to program content. The Roadmap, which is accessible from the Home page, Tools, and Sessions, lists the weekly focus (healthy eating or exercise) along with the week's session title, specific topic, and weekly behavioral challenge. The participant can review prior weeks to reinforce a sense of progress (enhances self-efficacy and offers encouragement) while future weeks are hidden to minimize potential cognitive overload and confusion. Sessions on the Roadmap are hyperlinked making it an easy navigation tool.
Support
The Support section (Table 1) includes a link to the Social Support Tool, Setback button, and access to frequently asked questions (FAQs) related to making healthful changes and using the website. Clicking on the Setback button takes the participant to a Setback webpage that includes an inspirational video of how another cancer survivor dealt with diet and/or exercise behavior change setbacks and, once released, the Setback session with e-learning tutorial is also available. After participants complete session 1 (diet or exercise only module) or sessions 1 and 2 (combined module), instructions for creating a Facebook account and accessing the AMPLIFY Facebook group appear in this section. Facebook is an optional component to provide participants social support (Figure 1). The relevance of social support and encouragement to accessing the Facebook group is reinforced at week 6 of the diet or exercise only module, and at weeks 11 and 12 of the combined module. The module-specific Facebook groups are monitored regularly (≥2/week) by trained research staff. Scheduled weekly posts on the group newsfeed (written to target social cognitive theory constructs) are used to encourage engagement. Ad hoc posts are used in response to relevant news stories released by reputable groups such as the American Society of Clinical Oncology.
Home page
The Home page integrates connections to all sections of the website and includes rotating and static content (Table 1; Figures 1, 4, and 5). Because the sessions are the central element of the intervention, the Home page highlights the next weekly session when due in addition to displaying a static Sessions section shortcut (Table 1). The Home page also includes a button for users to notify the AMPLIFY staff if they have recently experienced a medical issue or family emergency. To reinforce that healthy behavior setbacks are common, the Setback button is accessible from the Home Page as well as My Progress and Support sections. Rotating content also includes daily tips and social cognitive theory-based cancer survivor testimonials tailored to cancer coping style.

Home page for the AMPLIFY diet module.

Home page for the AMPLIFY exercise module.
Email and/or SMS messaging
Messages encouraging website engagement (Table 1 and Supplemental Table S1) are sent by email and/or SMS. Participants can opt in or out of email and/or SMS. Messages are initiated after the participant has initiated website use (see onboarding under criterion 5) and are sent each Monday (highlighting the new weekly session) and Saturday (reminder to track using My Progress).
Supplies provided to participants
Participants who receive the diet-only and exercise-only modules in sequence receive the materials specific to the module they are currently assigned as described in Table 3. Those assigned to the combined module receive both healthy eating and exercise materials upon module initiation.
Materials used in intervention delivery or in training of intervention providers
Website administration
A secure server/network infrastructure is required to house the website; this is provided by the website development firm (www.peopledesigns.com).
General management
Institutional research staff are provided secure internet access, secure email client, and computers to register participants on the website, download the administrative data used to generate staff notifications, and receive/respond to participant emails. Due to the large amount of administrative user data that is generated, a data management system (i.e. StudyTRAX©) is used to calculate any lapses in password creation, onboarding, etc. and generate staff notifications (also known as “flags”—details regarding flags are described in the paragraphs that follow). For participant-initiated email communication, a single AMPLIFY email alias is employed with a protocol for the staff member responsible for responding or delegating emails for management.
Facebook moderation
A study investigator with social media expertise (DP) uses a written protocol to train staff members implementing the Facebook protocol. Staff moderating Facebook require institution-provided computers, email, and internet access. All staff protocols and training materials are stored on a cloud server for easy staff access (Box®; Redwood City, CA) or locally on an institutional shared drive.
TIDieR criterion 4: Procedures, activities, and/or processes used in the intervention
After randomization, the participant is shipped the box of supplies (see criterion 3; Table 2) and directed through a series of steps to session 1 as shown in Figure 6. The profile information is used to personalize the Home page, My Progress, and Tools sections. “Flags” notify staff if a participant has not: registered within 2 weeks of being sent website registration instructions by email; completed onboarding within 2 weeks of registering; or completed the initial self-assessment session(s) within 2 weeks (3 weeks for the combined module). The participant is unable to view material beyond the initial session(s) because the participant-provided information obtained during the session(s) is crucial to tailoring messages provided during later self-assessments based on the participant progress (see criterion 3 for details). Once the participant has completed the initial session(s), the website programming automatically releases sessions each week (see Supplemental Table S1 for content details) regardless of whether the participant has viewed the prior week's session. However, once the initial session is completed, flags are generated if the participant does not log in to the website for more than 4 weeks. Protocol that follows after a flag is generated is described under criterion 11.
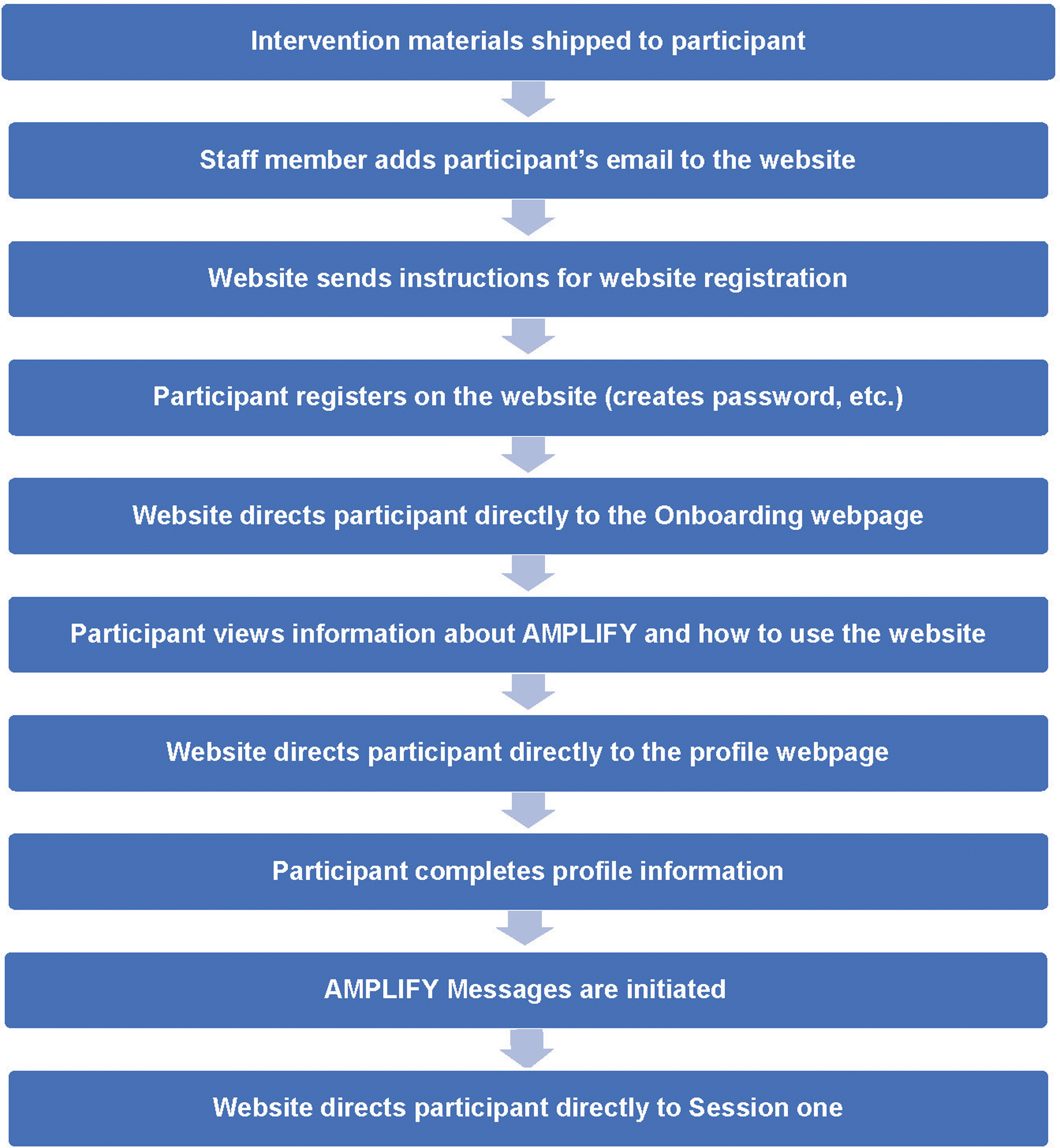
Participant flow from shipping of intervention materials to accessing session 1 of the AMPLIFY intervention (process begins after randomization).
If the participant indicates a health condition or other life event interfering with intervention participation by clicking the medical issue or family emergency button (Home page), the website sends an email to the AMPLIFY team. A study staff member contacts the participant (via telephone and/or email) to discuss the situation and, if necessary, pauses intervention messages (email or SMS); however, the website continues to release the weekly sessions.
TIDieR criterion 5: Description of the expertise, background, and specific training given to intervention providers
The coauthors brought multidisciplinary expertise to intervention development (Table 1) along with consultants, as needed, for e-learning tutorial design. For intervention implementation, study staff prepare, ship, and explain the intervention supplies (portion plate, pedometer, etc.) using checklists and scripts.
Intervention implementation also requires a designated study staff member responsible for facilitating participant website registration, website engagement, integration with the research data team and responding to inquiries from participants. This staff member is equipped with experience with healthy lifestyle clinical trials, electronic data capture platforms (e.g. REDCap and StudyTRAX©), digital technologies, and social media while also possessing critical thinking skills, excellent organizational skills, and familiarity with the intervention and research study protocol.
The intervention implementation also requires a registered dietitian and an exercise physiologist with expertise working with chronic disease and/or cancer populations. These expert staff oversee respective Facebook groups and also manage participant communications related to their expertise. If necessary, these communications are escalated to doctoral level research investigators who are registered dietitian, exercise physiologists, licensed physicians, and licensed clinical psychologists. This level of expertise optimizes safety during efficacy testing; issues requiring expert attention and clinical trial adverse events will be used to reevaluate the most appropriate level of support needed when AMPLIFY is used outside of a clinical trial.
Lastly, the website design company hosts the website, maintains a secure server and infrastructure, assists with technical support of study staff and end users, and refines programming, as necessary. The website design team also assists with engagement user data extraction and delivery of the messages (including Mailgun® for email delivery and a Vonage® messaging service for SMS delivery).
TIDieR criterion 6: Mode of delivery
The web-based intervention is delivered via the AMPLIFY website (htttps://amplifymyhealth.org) and accessible via participants’ computers and portable devices (e.g. tablets or smart phones). When needed, research staff assist participants in establishing a functional email address prior to website registration.
TIDieR criterion 7: Type(s) of location(s) where the intervention occurred, including any necessary infrastructure or relevant features
The intervention is housed in cloud-based infrastructure managed by People Designs, Inc. (Durham, NC) while the remaining infrastructure and materials are at the University of Alabama Birmingham (Birmingham, AL). Mailgun® and Vonage® support message delivery.
TIDieR criterion 8: Number of times the intervention was delivered and over what period of time including the number of sessions, their schedule, and their duration, intensity or dose
The AMPLIFY intervention is delivered across 48 weeks as a sequence (i.e. the 24-week diet module then the 24-week exercise module or vice versa) or concurrently (i.e. the 48-week combined module). Since rolling enrollment is used for the study, intervention implementation is ongoing. 14
As described in Supplemental Table S1, a rubric of weekly session topics served as the foundation for development of the diet only, exercise only, and combined modules. The sessions for the diet and exercise only modules were developed first and were based on the rubric which was crucial to balancing the behavioral content of each individual behavior module and facilitating the later combining process. The process of integrating these sessions for the combined module began as the individual behavior modules were completed allowing an iterative refinement process as the combined module identified necessary edits to the diet or exercise only modules. This structure also facilitated consistency among all three modules regarding content of e-learning tutorials, Tools, etc.
Several overarching approaches were used to combine the diet only and exercise only modules when creating the concurrent combined module. First, e-learning tutorials required editing to reduce redundancies and match the ordering of the weekly challenges within the combined module. Also, Tools with similar content were combined into a single Tool referring to both diet and exercise. Similarly, the AMPLIFY Roadmap content was streamlined to improve flow and avoid excess repetition when combined. My Progress content was linked to the weekly topic so it was not altered when creating the combined module however minor edits were made to the messaging to match the order of the combined module sessions.
TIDieR criterion 9: Tailoring of the intervention
Tailoring (based on participant's website profile entered during onboarding and self-assessment sessions) is done on the Home page, Sessions, My Progress, and Tools sections using website programming and/or interactive options within the e-learning tutorials as outlined in Table 1. Decisions regarding tailoring were based on our theoretical model (e.g. exercise self-assessment messaging was tailored to walking self-efficacy). Tailoring decisions were also made to improve intervention relevance (e.g. only participants who reported drinking alcohol were asked to track this; participants reporting fatigue as a symptom were presented Tools related to this symptom) and increase engagement (e.g. e-learning sessions asked participants about the prior weekly challenge with session messaging tailored based on the participant's success or lack thereof; Home page rotated new material based on the last session completed by the participant).
TIDieR criterion 10: Modifications of the intervention during the study
A few modifications were made after efficacy launch. First, we reduced the frequency of messages sent for failure to log in because overlap with the regularly scheduled twice weekly messages caused confusion. Although the content of the My Progress section was not changed, the option to track progress in a separate window was added because some participants were experiencing issues with data not automatically updating. Lastly, due to low engagement with the optional Facebook groups early in efficacy testing, we made the following refinements: (1) participants may join Facebook after completing session 1 (diet or exercise) or sessions 1 and 2 (combined) and (2) a link to the Support page with Facebook information was added to Social Support Tool and weekly session summaries for week 6 for diet or exercise (weeks 11 and 12 for combined). Additionally, we now include more active Facebook engagement strategies and activities such as polls and posting pictures.
TIDieR criterion 11: Planned procedures for how adherence or fidelity was assessed, describe how and by whom, and if any strategies were used to maintain or improve fidelity
As described under criteria 3.3 and 4, website engagement is encouraged with messaging and, if necessary, staff contacts. When a staff contact is indicated through a flag, a call is placed to the participant until contact is made or contact is attempted on at least three different days (if no contact is made, at least two messages are left using all means necessary—voicemail, email, and/or SMS). Staff continues efforts to contact the participant by telephone as long as the participant is answering and/or calling back. If, however, there is no response after 6 weeks, the participant is considered a withdrawal. Staff contact attempts and outcomes are tracked in REDCap and StudyTRAX©. As previously described, 14 user engagement is tracked using website data.
Fidelity of website delivery (e.g. material delivered as intended by the investigative team) was ensured through extensive and iterative investigative and website development firm review of a staging website before the programs were released for beta testing. Once released, beta testing feedback was reviewed within 2 weeks to allow facile intervention refinements. An extensive investigative and development team review was repeated after beta testing and before releasing the final version for efficacy testing.
Fidelity of SMS messaging was ensured with pilot testing and use of a reputable company (Vonage®). The SMS system tracks whether messages are sent out. Given the peripheral nature of the SMS messaging with regard to intervention content and other budget priorities, messages received or read are not being tracked.
Fidelity of the Facebook group moderators is facilitated with a written moderator protocol, staff training, and a priori written weekly posts. Fidelity is confirmed by comparing moderator posts and responses to a checklist based on the written protocol.
TIDieR criterion 12: Actual adherence or fidelity
The randomized efficacy trials are ongoing (primary endpoints of body weight, diet quality, and accelerometer-measured physical activity). 14 This information will be reported in manuscripts published after trials are completed and on the ClinicalTrials.gov website (NCT 04000880).
Discussion
Conclusions
Given the rising number of middle-aged or older cancer survivors who are at increased risk for second cancers, comorbidities and functional decline, the design and testing of an integrated, interactive, and tailored web-based intervention that assists this patient population in addressing healthy eating, weight management, and exercise is of critical public health importance. Developing such an intervention required a reciprocal and iterative partnership among cancer survivors, a multidisciplinary team of content experts, and experienced web designers. Given the identified research gap related to web-based interventions addressing multiple behaviors, 12 it is notable that an a priori rubric was crucial to consistency across the intervention modules and, as such, is highly recommended for other investigators developing similar inter-related multibehavior interventions used for testing optimal methods for addressing multiple behaviors (e.g. sequential vs. combined). Also, our reporting here uses TIDieR to demonstrate how the weekly sessions (a critical piece of the intervention because of the curriculum content) are placed within the context of other intervention elements. These details are infrequently described in the scientific literature yet are critical to the scientific discourse surrounding the pragmatic process of web-based intervention design and testing. Moreover, the comprehensive details provided here will enhance the interpretation and replication related to our randomized efficacy trial results, once available. In so doing, this report holds significant potential for advancing the science of health behavior change with high reach, low touch web-based interventions.
Knowledge gap addressed
Only a few web-based healthy lifestyle interventions (e.g. diet and/or exercise) for cancer survivors have published a description of the intervention design with none providing in-depth details as done in this report.12,49–54 Of the eight design descriptions,49–56 all but one have used a behavior theory foundation (e.g. social cognitive theory, theory of planned behavior, transtheoretical model, self-determination theory, and rewards based). All but one incorporated tailoring with five describing specific tailoring factors (e.g. age, gender, cancer treatment, current diet or exercise behavior, health status). Five focused on both diet and exercise,49,50,53–55 four integrated SMS messaging,49,50,54,55 and three targeted mixed cancer types.49,52,53 Also, the intervention duration has often been 3 months or less and none have extended past 6 months. Similar to our report, these prior publications demonstrate the integration of web-based education with other strategies (e.g. SMS messaging, videoconference-based counseling) and the ability to provide a variety of additional resources (e.g. Tools) which participants can explore based on preference and need. In contrast, no description of a web-based diet and/or physical activity intervention for cancer survivors has used the TIDieR checklist, an oversight that is troubling in light of recent reviews indicating that descriptions of health behavior interventions for this population are often insufficient to support replicability.19,57,58
Our AMPLIFY description advances this literature in several ways. It is the first description of a web-based diet and/or exercise intervention for cancer survivors to illustrate how core components from nonweb-based efficacious interventions can be applied to web-based intervention development. It is also the first to discuss integrating a private Facebook group as an optional intervention element. Notably, we report here the modifications and pragmatic decisions as recommended by the TIDieR checklist. In a recent review, only 2% of technology-based health behavior interventions for chronic disease populations included information on intervention modifications. 19 By using the TIDieR checklist, we include our modifications which suggest lessons learned related to the digital intervention development process (e.g. proactively consider potential confusion caused when “as needed” messages overlap with regularly scheduled messages). Also, the unexpected low Facebook engagement prompting modifications suggests that future design teams should consider if and how Facebook can be a viable option for providing virtual support for older cancer survivors as adjunct to a web-based program. This type of information is useful for future intervention development yet is missing from prior intervention descriptions in cancer survivors. Further, our detailed AMPLIFY description is the first focused specifically on older adults with a history of mixed cancer types and the only 12-month description.
Strengths and limitations
Very few eHealth interventions in any chronic disease population have been thoroughly described using the complete TIDier checklist as done in this report. 19 Thus AMPLIFY represents the first to use this taxonomy in cancer survivors and, in so doing, provides a foundation for other researchers developing web-based interventions for this understudied and at-risk population. Notably, web-based interventions for cancer survivors have largely been developed without input from racial minorities while in contrast, African American cancer survivors made up 41% of AMPLIFY's beta testers. Moreover, several novel aspects of our description are not routinely reported in the literature (e.g. mapping of core components, rubric facilitating multiple behavior integration). Of particular significance for future research, using the TIDieR checklist to describe AMPLIFY advances the science of web-based interventions in several ways, as it: (1) provides an example that encourages the use of TIDieR for standardizing the description of other similar interventions in cancer survivors; (2) identifies ways future researchers could build upon our intervention (e.g. narration by voice actors, additional tailoring); and (3) provides information needed to determine if and how to adapt the intervention for implementation with other populations and contexts.
However, we acknowledge several limitations to our intervention design and description. Describing complex and integrated interventions such as AMPLIFY (e.g. multiple elements, opportunities for tailoring) can be challenging. We address this complexity with figures, table summaries and screenshots because multimedia URLs are not currently available for public access. Further, tailoring was constrained by cost and study timeline, but we offset this by identifying opportunities for more extensive tailoring in future intervention designs. Also, the use of a medical issue or family emergency button and staff contact for initial engagement (important for efficacy testing) may not be feasible when released into broader use, thus potentially reducing eventual intervention effectiveness. Although translation to a web-based delivery was acceptable and website engagement was high during beta testing, final evaluation of these aspects is pending completion of the ongoing efficacy testing. Combining these data, once available, with evaluating cost-effectiveness based on intervention implementation cost outside a clinical trial context is critical to understanding AMPLIFY's potential for broader dissemination and implementation success.
Implications and future directions
We found that the TIDieR checklist was a useful framework for ensuring that the web-based intervention description was comprehensive. Application of certain criterion was more difficult for our automated digital (vs. human delivered) intervention. For example, staff training was less appropriate for our “low touch” intervention so the Criterion interpretation was expanded to include describing what role staff played in the delivery (e.g. housing of the intervention, programming, debugging, etc.). Further, TIDieR application to a web-based intervention could potentially be improved by specifying that the inter-relationships and integration among the various components or adjunct technologies used should be described.
The efficacy of the AMPLIFY modules are currently being tested in three inter-related randomized controlled trials. 14 As part of these trials, we will use engagement data to identify “active ingredients” or “key features” of the interventions along with testing of social cognitive theory mediators of the anticipated behavior change. While waiting on the results of these trials, researchers in the field can use the description provided in this report to improve other web-based interventions being developed for cancer survivors (e.g. prioritizing resources to optimize tailoring opportunities and use of actor narration or high-quality computer-generated voice). Also, research is needed using Multiphase Optimization Strategy (MOST) or Sequential Multiple Assignment Randomized Trial (SMART) study designs to determine which intervention elements are the most important for behavior change and for whom. Lastly, the use of TIDieR checklist for future web-based intervention descriptions will continue to advance the field of improving the health and wellbeing of cancer survivors through healthy behavior change and accelerate implementation of programs into community and clinical practice.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231182805 - Supplemental material for Using the TIDieR checklist to describe development and integration of a web-based intervention promoting healthy eating and regular exercise among older cancer survivors
Supplemental material, sj-docx-1-dhj-10.1177_20552076231182805 for Using the TIDieR checklist to describe development and integration of a web-based intervention promoting healthy eating and regular exercise among older cancer survivors by Laura Q. Rogers, Dori Pekmezi, Yu-Mei Schoenberger-Godwin, Kevin R. Fontaine, Nataliya V. Ivankova, Amber W. Kinsey, Teri Hoenemeyer, Michelle Y. Martin, Maria Pisu, David Farrell, Jonathan Wall, Kaitlyn Waugaman, Robert A. Oster, Kelly Kenzik, Kerri Winters-Stone and Wendy Demark-Wahnefried in DIGITAL HEALTH
Supplemental Material
sj-docx-2-dhj-10.1177_20552076231182805 - Supplemental material for Using the TIDieR checklist to describe development and integration of a web-based intervention promoting healthy eating and regular exercise among older cancer survivors
Supplemental material, sj-docx-2-dhj-10.1177_20552076231182805 for Using the TIDieR checklist to describe development and integration of a web-based intervention promoting healthy eating and regular exercise among older cancer survivors by Laura Q. Rogers, Dori Pekmezi, Yu-Mei Schoenberger-Godwin, Kevin R. Fontaine, Nataliya V. Ivankova, Amber W. Kinsey, Teri Hoenemeyer, Michelle Y. Martin, Maria Pisu, David Farrell, Jonathan Wall, Kaitlyn Waugaman, Robert A. Oster, Kelly Kenzik, Kerri Winters-Stone and Wendy Demark-Wahnefried in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors thank our intervention beta testers and the many University of Alabama at Birmingham research staff and trainees who have supported the AMPLIFY intervention initiative. We also dedicate this publication in memory of our colleague and friend, Dr Karen Meneses, whose passion for improving the health and well-being of cancer survivors remains an inspiration for the research team.
Contributorship
LQR, DP, YS-G, NVI, MYM, MP, DF, RAO, KK, and WD-W contributed to conceptualization, funding acquisition, investigation, methodology, project administration, supervision, and writing original draft. KRF contributed to methodology, project administration, and supervision. AWK, TH, and KW-S contributed to methodology and project administration. JW and KW-S contributed to methodology. AWK initiated the first draft which was finished by LQR. All authors reviewed and edited the manuscript draft and approved the final draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The research study using the AMPLIFY intervention along with the amendment approving beta testing was approved by the University of Alabama at Birmingham (UAB) Institutional Review Board (UAB IRB-300002068), which served as the single IRB for this multisite study including UAB and the University of Tennessee Health Science Center. Beta testers provided informed consent prior to participation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is supported by the National Cancer Institute (P01 CA229997, P30 CA013148, R25 CA076023, T32 CA047888) and the American Cancer Society (CRP-19-175-06-COUN).
Guarantor
LQR and WD-W.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
