Abstract
Objective
To investigate if in-clinic measures of physical function and real-world measures of physical behavior and mobility effort are associated with one another and to determine if they predict future hospitalization in participants with chronic kidney disease (CKD).
Methods
In this secondary analysis, novel real-world measures of physical behavior and mobility effort, including the best 6-minute step count (B6SC), were derived from passively collected data from a thigh worn actigraphy sensor and compared to traditional in-clinic measures of physical function (e.g. 6-minute walk test (6MWT). Hospitalization status during 2 years of follow-up was determined from electronic health records. Correlation analyses were used to compare measures and Cox Regression analysis was used to compare measures with hospitalization.
Results
One hundred and six participants were studied (69 ± 13 years, 43% women). Mean ± SD baseline measures for 6MWT was 386 ± 66 m and B6SC was 524 ± 125 steps. Forty-four hospitalization events over 224 years of total follow-up occurred. Good separation was achieved for tertiles of 6MWT, B6SC and steps/day for hospitalization events. This pattern persisted in models adjusted for demographics (6MWT: HR = 0.63 95% CI 0.43–0.93, B6SC: HR = 0.75, 95% CI 0.56–1.02 and steps/day: HR = 0.75, 95% CI 0.50–1.13) and further adjusted for morbidities (6MWT: HR = 0.54, 95% CI 0.35–0.84, B6SC: HR = 0.70, 95% CI 0.49–1.00 and steps/day: HR = 0.69, 95% CI 0.43–1.09).
Conclusion
Digital health technologies can be deployed remotely, passively, and continuously to collect real-world measures of physical behavior and mobility effort that differentiate risk of hospitalization in patients with CKD.
Keywords
Introduction
Patient focused medicine is a shift to systematically putting patient perspectives at the center of drug development and clinical care. At a fundamental level, this requires developing and validating clinical outcome measures that reflect aspects of health that are meaningful to patients. The 21st Century Cures Act, passed in 2016, spurred this paradigm shift and opened the door for real-world data (RWD) collected outside of a clinic setting to be used to develop a new class of digital clinical measures that are objective, sensitive, clinically relevant, and meaningful to patients. 1 The current COVID-19 pandemic has further accelerated this shift and forced previously reluctant drug developers to embrace pragmatic trials that place less emphasis on traditional in-clinic assessments and leverage RWD to ensure continued participant oversight.2–5.
Measures of real-world physical behavior and mobility effort derived from wearable actigraphy sensors are one of the most promising examples of how RWD can provide a more comprehensive and ecologically valid representation of disease manifestation and health status. 6 Importantly, such measures can be captured remotely, passively, and continuously to generate large amounts of objective RWD that directly or indirectly represent a patient's physical functioning in their own real-world environment.7,8 This is a departure from the status quo where physical function outcome assessments have historically relied on subjective report (from the patient, caregiver, or clinician) or standardized in-clinic performance tests. Subjective assessments have the benefit of directly surveying physical function within a range of relevant contexts, but their reliability compared to objective measures can be limited by reporting bias. Conversely, objective performance tests assess physical function within a single structured and standardized environment that does not reflect patients’ real-world environments.
Novel, real-world assessments of physical behavior and mobility effort passively measured as patients go about their daily lives can complement existing physical function assessments and/or be used when existing methods are inappropriate or not feasible.9–12. For example, existing in-clinic performance-based tests are designed to reflect or mimic movements (e.g. walking, sit-to-stand transitions, etc.) needed to complete common activities of daily living. Wearable actigraphy sensors combined with advanced analytics can now be used to assess if and how these same movements are being performed in a patient's own real-world environment. While such measures provide a peak into a patient's lived experience, there is a lack of consensus on their true meaning and clinical relevancy across therapeutic areas. Thus, significant work remains to develop and define fit-for-purpose13–16 measures that accurately capture concepts of interest and are clinically relevant.
The purpose of this investigation was to gain insight into the clinical utility of real-world measures of physical behavior and mobility effort passively collected as participants go about their daily lives. In this secondary analysis, we leveraged an existing dataset from a sample of participants with chronic kidney disease (CKD), a condition that known to induce severe and prevalent physical dysfunction. 17 Specifically, we investigated how real-world measures of physical behavior and mobility effort correlated with accepted in-clinic physical function performance tests and whether real-world or in-clinic assessments predicted hospitalization.
Methods
This is a secondary analysis of baseline and follow-up data from the Sit Less, Interact, Move More (SLIMM) pilot study (NCT02970123). For a detailed description of the SLIMM study see Lyden et al. (2021). 18 The SLIMM study was a 24-week, single center, pilot, open-labeled, randomized controlled trial conducted at The University of Utah and sponsored by National Institute of Diabetes and Digestive and Kidney Diseases. The study was approved by the University of Utah Institutional Review Board. All participants provided written informed consent.
Study population
Trained study staff at The University of Utah recruited participants from March 23, 2017, through December 15, 2018. Potential participants were approached during clinic visits or through an opt-in letter mailed to a list of potential participants identified through the University of Utah electronic medical records as approved by the Institutional Review Board. Participants were required to meet the following inclusion criteria: (1) CKD (stage 2 CKD with eGFR 60 to <90 ml/min/1.73 m2 with urine albumin/creatinine ratio > 30 mg/g or stage 3 or 4 CKD with eGFR 15 to <60 ml/min/1.73 m2 or on maintenance hemodialysis or kidney transplant), (2) BMI 25.0 to 39.9 kg/m2 and (3) able to perform the SLIMM intervention (gait speed ≥ 0.7 m/s and able to walk ≥ 250 m in 6 minute walk test). Pregnancy, incarceration, life expectancy <1 year in the opinion of the primary provider, unlikely to comply with research protocol, inability to obtain accelerometer data before randomization, enrolled in interventional trials using drugs or devices, and inability to ambulate were exclusion criteria.
Accelerometry
Prior to randomization, study participants wore the activPAL™ (PAL Technologies, Glasgow, Scotland) activity monitor for 24 hours/day and for 7 consecutive days in their free-living environments. Previous studies validated the use of activPAL™ in measuring sedentary behavior, physical activity and stepping behavior.19–23. Although not specifically validated in patients with CKD, the activPAL™ has been validated in clinical populations with low physical function and mobility and has been extensively used to study real-world behavior in these populations.18,24– 26. The small (23.5 × 43 × 5 mm) and lightweight (9.5 g) accelerometer was attached directly to the skin on the midline of the thigh halfway between the hip and the knee, using a non-allergenic adhesive pad. Data were sampled at 20 Hz across a range of −2 to +2 g. The thigh wearing location allowed the assessment of detailed metrics of posture (lying, sitting, standing, stepping), physical activity intensity and transitions from sitting to standing. Following best practice, a valid day of measurement was defined as ≥10 waking hours of valid data. 27 To be included in analyses, at least 4 valid days, with at least 1 day being a weekend day, were required.
Randomization and SLIMM intervention
Details of SLIMM randomization and intervention procedures have been published 28 and more details are provided in the supplemental material. Briefly, eligible participants were block randomized to either standard of care (SOC) or SLIMM intervention. SOC participants were encouraged to achieve federally recommended physical activity guidelines. 29 Participants assigned to the SLIMM intervention were instructed to get up from sitting or lying postures while awake at least once per hour and to engage in light intensity activities. SLIMM participants underwent extensive education and were provided individualized strategies based on their own real-world behavior on how to replace sedentary time with light intensity walking.
Physical function
The exact definition of physical function has been debated for years and many different terminologies have been used to describe the same concept. 30 – 33. For the purposes of this manuscript, physical function is defined as the ability to perform both basic and instrumental activities of daily living 34 and is assessed using objective, in-clinic performance tests.
In-clinic physical function was assessed using the 6-minute walk test (6MWT), 4-m gait speed test and shuttle test. Tests were performed following standardized procedure 28 and more details are provided in the supplemental material.
Real-world mobility effort
Two real-world measures of mobility effort were derived from daily accelerometer data. The motivation for investigating the two measures selected was that they (1) have parallels to commonly used in-clinic performance tests of walking capacity (e.g. 6MWT, gait speed) and sit-to-stand body transfers (e.g. timed-up-and-go), both of which have been shown to differentiate patients with CKD who have difficulties with activities of daily living and those at increased risk for mortality,17,35–41 and (2) they are easily measured using a single wearable actigraphy sensor that is acceptable to patients with CKD and can accurately capture other meaningful physical behaviors.
28
Best 6-minute step count (B6SC) was defined as the number of steps during the most active continuous 6-minute period that the activPAL™ monitor was worn. B6SC was derived using a proprietary sliding window algorithm included in the activPAL™ analysis software.
42
A priori, we used B6SC as the primary measure of real-world mobility effort as this domain conceptually corresponds to the in-clinic 6-minute walk distance. It was hypothesized that higher B6SC in the real-world corresponds to higher physical function. Rise time was defined as the average time to rise from a seated or lying position to a standing one. Rise time was derived using a proprietary algorithm included in the activPAL™ analysis software.
42
It was hypothesized that longer rise time corresponds to lower physical function.
Real-world physical behavior
Three common measures of physical behavior were derived from daily accelerometer data. To account for confounding effects of non-wear and sleep time, we normalized physical behavior duration for each time domain variable to 16 waking hours per day by the following formula: (total activity duration recorded per day in minutes divided by total waking wear time per day in minutes) × 60 × 16.43,44
Step count—the median number of steps accumulated per day. Sedentary minutes per day—the average number of minutes per day spent performing sedentary behaviors during waking hours. Sedentary behavior was defined as time spent in a lying or seated position with energy expenditure less than 1.5 METs.
29
It was hypothesized that more sedentary minutes per day corresponds to decreased physical function. Moderate-to-vigorous physical activity (MVPA) minutes per day—the average number of minutes per day spent performing MVPA during waking hours. MVPA was defined as time spent in activity of at least 3 METs.
29
It was hypothesized that more MVPA minutes per day corresponds to increased physical function.
Hospitalizations
University of Utah Electronic Medical Records systems were used to track hospitalizations between randomization and an administrative censoring date of June 30th, 2021. Our main outcome was time from randomization until first hospitalization. Participants were censored at the administrative censor date of June 30th, 2021, last date of EMR encounter, or death.
Statistical methods
Baseline characteristics were compared between tertiles of B6SC using one-way ANOVA for continuous measures and Pearson's Chi-squared test for categorical variables. Pairwise correlations were calculated between baseline measures of physical function. Histograms were checked to ensure appropriate normal distribution of baseline characteristics.
Kaplan–Meier failure for hospitalization was plotted by tertiles of each of the baseline physical function characteristics. We next investigated if each of the physical function characteristics predicted hospitalizations using multivariate Cox proportional hazard regressions. The first model was adjusted for age, gender, race, and the SLIMM intervention group. The second model additionally adjusted for smoking, alcohol use, CKD, diabetes, coronary artery disease, hypertension, congestive heart failure, peripheral vascular disease, and BMI. Schoenfeld residuals were tested for proportionality and no abnormalities were detected. Analysis was performed using Stata/MP 15.1.
Results
Participant characteristics
Baseline assessments and follow-up data were analyzed 106 participants who took part in the SLIMM pilot study. Mean age was 69 ± 13 years, 43% were women and 90% were Caucasian. The baseline distribution of CKD stages was 5% stage 2, 38% stage 3A, 37% stage 3B and 21% stage 4/5 or end stage kidney disease (ESKD). In this older adult population with CKD, baseline physical function was relatively low with a mean 6-minute walk distance of 386 ± 66 m, gait speed of 1.1 ± 0.2 m/s and shuttle walk distance of 251 ± 118 m. Study participants spent most of their awake time sedentary (648 ± 128 minutes/day) and were modestly active with MVPA duration of 18 ± 32 minutes/day and 5272 median (3943–7189) steps/day. The baseline B6SC was 524 ± 125 steps, and the average rise time was 0.96 ± 0.19 seconds.
Table 1 summarizes participant characteristics by tertiles of B6SC. While demographics and trial assignment were similar across baseline B6SC tertiles, BMI and prevalence of type 2 diabetes were lower and the MVPA duration and the number of steps/day were higher in the highest versus the lowest tertile of B6SC. Supplemental Table S1 summarizes baseline characteristics by whether they were subsequently hospitalized or not.
Participant characteristics by tertiles of best 6-minute step count (B6SC). a

Mean ± SD for continuous variables and N (%) for categorical variables are presented.
Excludes participants on dialysis or transplant recipients.
Correlations of in-clinic and real-world measures
B6SC had low positive correlations with in-clinic physical function measures; 6-minute walk distance (ρ = 0.26, p = 0.007), 4-m gait speed (ρ = 0.20, p = 0.04) and shuttle walk test (ρ = 0.29, p = 0.003) (Table 2). Rise time had low negative correlations with the in-clinic measures; 6-minute walk distance (ρ = -0.26, p = 0.010), 4-m gait speed (ρ = -0.23, p = 0.02) and shuttle walk test (ρ = -0.22, p = 0.03). Sedentary duration was not correlated with in-clinic physical function measurements, while MVPA and the number of steps/day had modest correlation with in-clinic 6-minute walk distance (Table 2).
Spearman rank correlation coefficients between baseline in-clinic and real-world measures.
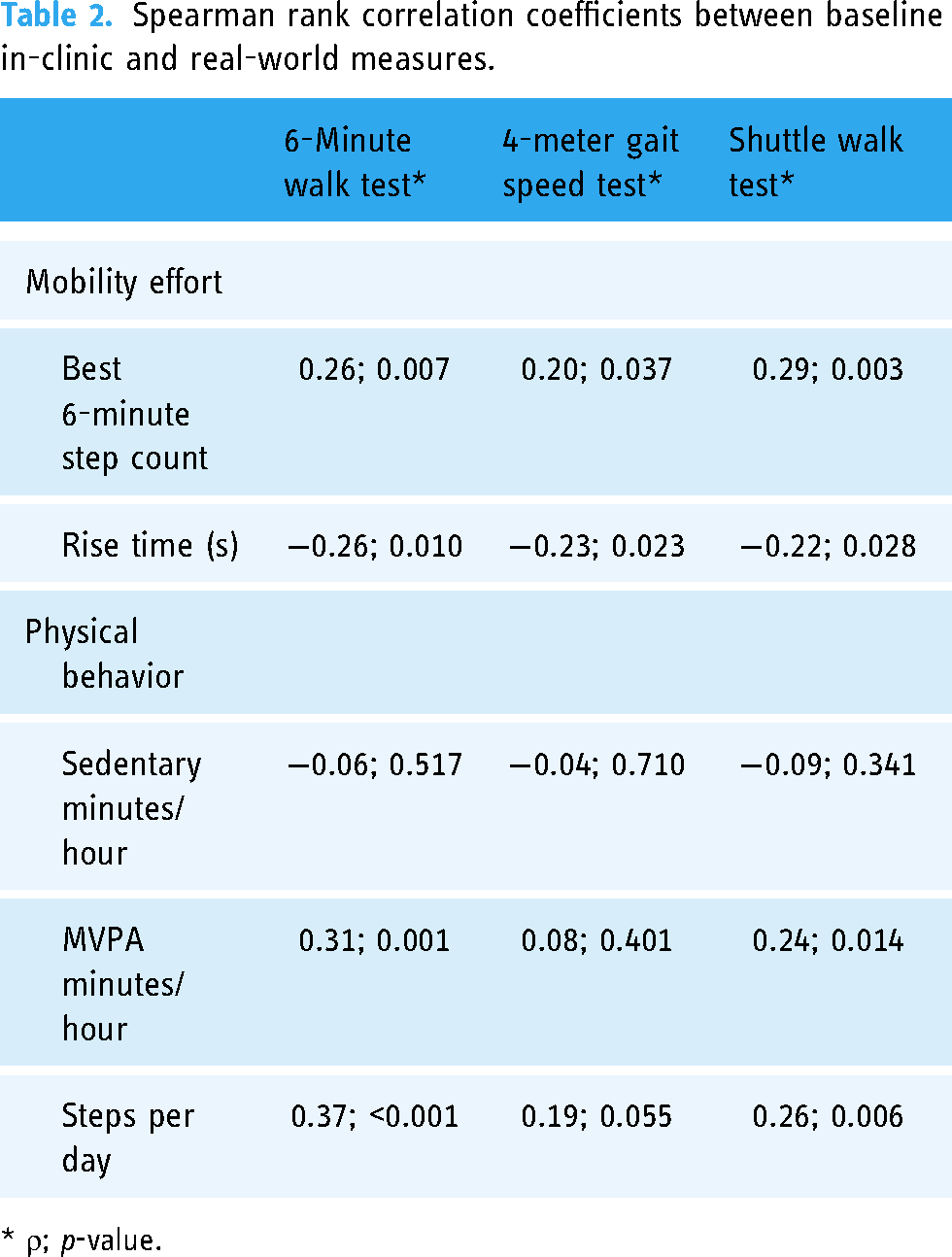
* ρ; p-value.
Probability of hospitalization
The average follow-up duration was 2.1 ± 1.2 years with 8 deaths. There were 44 hospitalization events over 224 person-years of total follow-up. Unadjusted associations of tertiles of in-clinic and real-world measures with hospitalization are presented in Figure 1. Within each domain, tertiles of 6-minute walk distance (Figure 1(a)), B6SC (Figure 1(b)) and steps/day (Figure 3(c)) achieved the best separation for hospitalization events.

Kaplan–Meier curves. Probability of hospitalization by tertiles of the predictor variable for (a) in-clinic and real-world measures of (b) mobility effort and (c) physical behavior.
Cox regression models are illustrated in Figure 2(a) and 2(b). In a model adjusted for age, gender, race and SLIMM intervention group, each SD increase in 6-minute walk distance was significantly associated with lower risk of hospitalization (hazard ratio (HR) 0.68 (95% confidence interval (CI) 0.49–0.96). This remained significant with further adjustment for smoking, alcohol use, CKD stages, diabetes, hypertension, heart failure, peripheral vascular disease, and body mass index (HR 0.63, 95% CI 0.43–0.93). In corresponding models, hospitalization hazard ratios for each SD higher B6SC (HR 0.75, 95% CI 0.56–1.02 and HR 0.70, 95% CI 0.49–1.00) had similar magnitude of associations with hospitalization, however, statistical significance was marginal. Physical behavior measures (steps, sedentary duration, and MVPA duration per day) were non-significantly associated with lower risk of hospitalization (HR 0.75, 95% CI 0.50–1.13 and HR 0.69, 95% CI 0.43–1.09).

Hazard ratios per standard deviation of the predictor variable for unadjusted (a) and adjusted (b) Cox regression models.
Discussion
In this secondary, retrospective analysis of the SLIMM trial, the traditional in-clinic 6MWT, the novel real-world measure B6SC, and real-world step count achieved good separation for hospitalization events in patients with CKD. Although the small sample size and brief follow-up period limit this to a proof of principle study, the novel finding was that real-world measures of physical behavior (step count) and mobility effort (B6SC), which are passively measured from a body worn accelerometer as individuals go about their daily lives, provide additional information not captured by in-clinic assessments of physical function and warrant further investigation. Findings also confirm existing evidence to support the clinical utility of the 6MWT in patients with CKD.
To gain insight into how novel real-world measures compare to accepted performance test commonly used in patients with CKD, the associations between in-clinic and real-world measures were also tested. The correlation analyses demonstrated the expected directional relationships with low, but significant, strength. These results were expected as real-world measures do not replicate in-clinic tests, but they do similarly capture health concepts of interest.45,46 To our knowledge, no previous research has investigated the relationship between novel real-world measures and accepted clinical outcome assessments in patients with CKD. Note that negative correlations with rise time were expected as increased rise time corresponds to lower physical function while increased values for in-clinic measures correspond to higher physical function.
B6SC is defined as the number of steps taken within the continuous 6-minute window that contains the most steps for a given calendar day. It parallels the in-clinic 6MWT but has three key differences; (1) B6SC is not an active performance test, but rather a passive measure that captures a person's best natural effort as they go about their typical activities of daily living, (2) it is measured daily rather than at episodic timepoints and (3) it is measured in the participant's own real-world environment rather than in structured clinic setting. In this sense, B6SC is an ecologically valid measure of physical function that reflects an individual's lived experience. Others have proposed alternative measures to capture best natural effort, 47 such as peak 30-minute cadence, but those were not explored in the current study.
Real-world measures of physical behavior, including step count, sedentary time and MVPA, have previously been shown to be associated with morbidity and mortality in healthy and diseased populations.29,48–57 However, in the current study measures of real-world physical behavior were non-significantly associated with future hospitalizations, with steps/day having the strongest association. This was not entirely surprising given our small, inactive sample. Median steps/day and prevalence of MVPA were minimal (Table 1) and it is possible that in a different population which accumulates more activity, hospitalization would be significantly predicted. Sedentary time was high among participants, but there was little variability between participants (Table 1), making it difficult to reveal associations between sedentary duration and hospitalizations in this homogenous sample.
The 6MWT is arguably one of the most widely used in-clinic performance test. 58 Its origins stem from the 12-minute run test developed by Cooper et al. (1968) which was shown to have good correlation with maximum oxygen consumption measured during treadmill running. 59 A walking version of the test was first used by McGavin et al. (1976) in populations with respiratory disease 60 and later shortened to 6 minutes. 61 In its current form, the 6MWT is a simple, pragmatic assessment and has demonstrated value as a prognosticator of disease severity and predictor of morbidity and mortality in many clinical populations including kidney disease.17,31,35–41,62–64 Results from the current study contribute to the large body of evidence supporting the clinical utility of the 6MWT.
Despite the broad appeal of in-clinic performance tests, there is an opportunity for real-world measures that are captured continuously, for long periods of time, as individuals go about their daily lives to compliment in-clinic performance test like the 6MWT.9,10 First, unlike in-clinic assessments that are episodically captured at discreet timepoints, they capture day-to-day variation to provide a more granular picture of health. Second, they are not captured under standardized conditions not reflective of patients’ true environment, but rather capture a patient’s true lived experience in their own environment. Third, they are not influenced by administrator motivation or test-retest bias (i.e. the “practice effect”). 65 Although not investigated in the current study, the continuous and passive nature of real-world measures may be particularly useful for measuring disease progression with higher granularity and sensitivity, allowing clinicians to make better informed and more timely treatment decisions.
When using passively collected real-world measures as outcome assessments, it is important to consider that an individual's choice to participate in a given behavior is complex and multidimensional with factors other than disease severity and symptom expression interacting to influence behavioral choices. For example, lack of participation in a given behavior doesn’t necessarily reflect one's ability to perform that behavior. For many individuals, participation in active and inactive behaviors is unrelated to functional ability or health status, but rather individual, social, and environmental factors such as professional responsibilities, urban versus rural living and/or seasonal variability. 66 However, it is also important to consider that, as reported by Koufaki 2022, maintaining functional independence is a hallmark of clinical care for patients with CKD and accurate measures of physical function are key to putting this into practice. 17 Thus, if the ultimate goal is to maintain (and measure) independence, outcome measures that are influenced by multiple factors related to an individual's lived experience may, in certain contexts, be more appropriate for assessing the status, or change, of functional independence on an individual level than standardized functional performance tests. In fact, The World Health Organization (WHO) and the International Classification Framework of Functioning, Disability and Health (ICF) acknowledge that function is a result of the interactions between biological, environmental, and personal factors. In other words, the context in which the person exists cannot be ignored when assessing function. 30
The idea that real-world data from digital health technologies can transform drug development and healthcare has been touted for years by industry stakeholders, care providers and academic partners alike.9,10,12,67 And the proliferation of wearables and smart devices in the consumer health market has signaled that patients are not only open to, but ready for, this transformation; a notion further supported by substantial industry and academic research. A recent national survey of US adults found that 82% of respondents were willing to share health data from a wearable device with a clinician. 68 Rightfully so however, the transformation has been slow and cautious with multiple factors hindering mass adoption including lack of (1) know-how, (2) operational infrastructure, (3) reimbursement models, (4) robust security and privacy systems and, perhaps most importantly, (5) the evidence needed to support the validity and relevancy of digital clinical outcome measures.11,14,15
In the current study we investigated three real-world measures of physical behavior and two measures of mobility effort, however many additional measures can be derived from wearable actigraphy sensors. To fully understand the clinical relevancy and health concepts of interest reflected in real-world measures, population-specific natural history studies and interventional trials should investigate measure characteristics in conjunction with accepted clinical endpoints and outcomes.
Strengths of this secondary analysis of the SLIMM trial includes (1) inclusion of a range of participants at various stages of CKD, (2) use of a rigorously validated wearable activity monitor, (3) inclusion of multiple in-clinic measures of physical function and (4) inclusion of multiples objective, real-world measures that were passively collected as participant went about their daily lives.
There are also several limitations: (1) a small sample size with low participation in active behaviors, (2) a relatively short follow-up time, (3) a single center study in a predominantly Caucasian population, (4) hospitalization data obtained only within the University of Utah Health system, (5) measurement of real-world behavior for only 7 consecutive days and (6) additional patient centered outcomes such as self-reported physical function and quality of life were not evaluated in relation to real-world measures of physical behavior and mobility effort. Recent improvements in device miniaturization, computational power, and extended memory now allow data to be collected for weeks to months at a time. Future work should leverage these technological advancements to investigate more complex measure of real-world physical behavior and mobility collected over time.
The current study provided valuable proof-of-principle evidence that real-world measures of physical behavior and mobility effort are clinically relevant in patients with CKD. By simultaneously measuring novel real-world measures with accepted in-clinic assessments and hard clinical outcome data (hospitalization) we demonstrated that RWD passively captured from a wearable digital health technology, provides an ecologic look into health status. Importantly, these data do not duplicate commonly used in-clinic tests and thus future studies should examine how RWD can complement existing performance tests. Additionally, future work should examine how digital health technologies that are deployed remotely, passively, and continuously can be used to measure disease progression with accuracy and sensitivity.
Conclusion
Digital health technologies can be deployed remotely, passively, and continuously to collect real-world measures of physical behavior and mobility effort in patients with CKD. In this proof of principle study, physical behavior (step count) and mobility effort (B6SC), which are passively measured as individuals go about their daily lives, provide additional information not captured by in-clinic assessments of physical function and warrant further investigation. Longitudinal investigations in larger samples will provide additional insight into the clinical relevancy of real-world digital clinical measures.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231181234 - Supplemental material for Predicting hospitalization from real-world measures in patients with chronic kidney disease: A proof-of-principle study
Supplemental material, sj-docx-1-dhj-10.1177_20552076231181234 for Predicting hospitalization from real-world measures in patients with chronic kidney disease: A proof-of-principle study by Kate Lyden, Nikita Abraham, Robert Boucher, Guo Wei, Victoria Gonce, Judy Carle, Sydney E. Hartsell, Jesse Christensen and Srinivasan Beddhu in DIGITAL HEALTH
Footnotes
Acknowledgements
We would like to thank all participants who volunteered to take part in this study.
Authors' Note
Kate Lyden ia also affiliated at VivoSense, Inc Newport Coast, CA, USA.
Clinicaltrials.gov registration
Sit Less, Interact, Move More (SLIMM) Intervention for Sedentary Behavior in Chronic Kidney Disease (CKD) (SLIMM). NCT02970123 (https://clinicaltrials.gov/ct2/show/NCT02970123?term=beddhu&draw=2&rank=3).
Contributorship
KL and SB researched literature and conceived the study. All authors were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. KL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
Kate Lyden is employed by VivoSense, Inc and previously consulted for wearable device manufacturers. The authors declared no other potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the University of Utah Institutional Review Board.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases (R21DK106574). Statistical analyses and preparation of this manuscript were also supported by grants from National Institute of Diabetes and Digestive and Kidney Diseases (R01 DK091437) and the National Heart, Lung and Blood Institute (R21HL145494) and the University of Utah Study Design and Biostatistics Center (funded in part from the Public Health Services research grant numbers UL1-RR025764 and C06-RR11234 from the National Center for Research Resources).
Guarantor
SB.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
