Abstract
Background
According to the World Health Organization (WHO), pneumonia is the leading infectious cause of death in children below 5 years old. Hence, the early detection of pediatric pneumonia is crucial to reduce its morbidity and mortality rates. Even though chest radiography is the most commonly employed modality for pneumonia detection, recent studies highlight the existence of poor interobserver agreement in the chest X-ray interpretation of healthcare practitioners when it comes to diagnosing pediatric pneumonia. Thus, there is a significant need for automating the detection process to minimize the potential human error. Since Artificial Intelligence tools such as Deep Learning (DL) and Machine Learning (ML) have the potential to automate disease detection, many researchers explored how such tools can be implemented to detect pneumonia in chest X-rays. Notably, the majority of efforts tackled this problem from a DL point of view. However, ML has shown a higher potential for medical interpretability while being less computationally demanding than DL.
Objective
The aim of this paper is to automate the early detection process of pediatric pneumonia using ML as it is less computationally demanding than DL.
Methods
The proposed approach entails performing data augmentation to balance the classes of the utilized dataset, optimizing the feature extraction scheme, and evaluating the performance of several ML models. Moreover, the performance of this approach is compared to a TL benchmark to evaluate its candidacy.
Results
Using the proposed approach, the Quadratic SVM model yielded an accuracy of 97.58%, surpassing the accuracies reported in the current ML literature. In addition, this model classification time was significantly smaller than that of the TL benchmark.
Conclusion
The results strongly support the candidacy of the proposed approach in reliably detecting pediatric pneumonia.
Keywords
Introduction
Recent technological advancements in the healthcare industry have contributed to the digitization and storage of medical health records in large databases.1–3 Consequently, numerous efforts have been utilized to investigate the potential of using AI tools in the era of big data to assist medical practitioners in making more informed diagnostic decisions using available medical databases to ensure high patient care quality.4,5 The use of AI in the healthcare industry is promising because it can aid in preventing, treating, and diagnosing illnesses with expert-level accuracy while decreasing human error. 6 To date, AI tools have been shown to be effective in detecting several diseases such as skin cancer, 7 lung cancer, 8 breast cancer, 9 heart diseases,10,11 eye diseases, 12 tuberculosis, 13 covid-19, 14 and pneumonia.15–19 Within the scope of pneumonia detection, the implementation of AI has been investigated using multiple medical modalities including clinical data, computed tomography, ultrasounds, and chest X-rays. 16
Notably, pneumonia is a critical respiratory disease that limits a person's necessary oxygen intake by filling the lung's air-sacs, also known as the alveoli, with fluid. This disease is caused by bacteria, viruses, or fungi and may be fatal if left untreated. According to the World Health Organization (WHO), 20 despite being curable, pneumonia is the “single largest infectious cause of death in children worldwide” where it accounted for 15% of the fatalities of children under the age of five in 2017, making its early detection essential to reduce the mortality rate. Currently, chest radiography is the most commonly utilized imaging modality for pediatric pneumonia detection.21,22 However, the work of Voigt et al. 23 proves that there is a poor interobserver agreement between radiologists when it comes to diagnosing pediatric pneumonia using the same chest X-ray images. As such, the authors recommend standardizing the pneumonia detection process and setting up compulsory training programs to reduce the high interobserver variability. Furthermore, in several low- to middle-income countries, pediatric pneumonia is diagnosed in chest X-rays by non-radiologist clinicians, 21 and Fawole et al. 24 confirm the existence of variability in such clinicians’ diagnosis. In addition, the authors suggest that even though training interventions have the potential to reduce the diagnosis variability of pediatric pneumonia in chest X-rays in the short run, further studies must be conducted to monitor whether such progress can be retained in the long run.
Thus, it can be deduced that there is a significant need for an automated approach that can enable medical practitioners to increase the reliability and accuracy of their pediatric pneumonia diagnosis in chest X-ray images, especially in under-developed countries such as those present in sub-Saharan Africa and South Asia where pneumonia is most widespread. 20 In response to this, numerous efforts have been utilized by researchers to evaluate the candidacy of Artificial Intelligence (AI) tools, such as Deep Learning (DL) and Machine Learning (ML), when it comes to automating the pneumonia diagnosis process. By analyzing the existing literature, it is observed that the majority of the efforts explored the potential of DL to detect pneumonia in chest X-ray images while little work has been conducted to explore that of ML.
Despite the high resulting accuracies of DL models and their popularity amongst researchers, several challenges have been raised to evaluate their clinical applicability. Firstly, DL models employ architectures that extract features automatically from the data, 15 and they behave like a black-box problem, which lowers the medical interpretability of the model output and undermines the clinical effectiveness of utilizing DL in healthcare. Secondly, DL models require a large volume of data to produce acceptable results. 15 As a result, such models require computationally expensive systems over large training periods, which can be impractical in healthcare. Consequently, evaluating ML has the potential to overcome such challenges, and this is the main subject of interest for this research.
In contrast to DL models, ML models possess a higher potential for clinical interpretability as they permit choosing the feature extraction method, enabling the model to focus on features directly related to the symptoms of the disease in question. In addition, ML models possess the ability to provide comparable accuracies to DL models with significantly lower computational time and effort when fine-tuned. Furthermore, Bhardwaj et al. 1 suggest that ML has the potential to enhance the patient-doctor relationship while reducing the growing cost of healthcare. As such, the purpose of this paper is to propose an ML approach that can accurately and reliably detect pediatric pneumonia in chest X-rays with significantly reduced training time. It is demonstrated that using a Quadratic SVM model delivers a 97.58% accuracy surpassing the current ML accuracies reported in the literature with significantly smaller classification time than that of the used Transfer Learning (TL) benchmark. This strongly promotes ML for further development in pediatric pneumonia detection in the future.
Literature review
Automated pneumonia detection
In the field of automated pneumonia detection in chest X-ray images, the majority of the utilized efforts explored the potential of DL and its subset, TL, as opposed to ML. To illustrate, Kundu et al. 17 developed an automated Computer-Aided Diagnosis (CAD) framework using TL that can classify normal and pneumonic chest X-rays with an accuracy of 98.81%. This framework utilizes a weighted ensemble that accounts for decision scores obtained from the GoogLeNet, ResNet-18, and DenseNet-21 pre-trained DL networks. Similarly, Manickam et al. 25 compared the performance of three pre-trained architectures, namely ResNet50, InceptionV3, and InceptionResNetV2 to distinguish between normal, bacterial, and viral pneumonia classes in approximately 2300 chest X-rays obtained from a publicly available dataset. The authors applied data augmentation techniques such as rotation, horizontal and vertical flipping and shifting, and Gaussian blurring to overcome the class imbalance present in their data, yielding an accuracy of 93.06%, 92.67%, and 92.40% for the ResNet50, InceptionV3, and InceptionResNetV2 networks, respectively.
Moreover, Vrbančič et al. 26 employed a deep ensemble method based on Stochastic Gradient Descent with warm restarts to classify pneumonia in chest X-ray images using 10-fold cross-validation which produced an accuracy of 96.26%. Despite their acceptable accuracies, the DL models presented above utilize deep features that are automatically extracted from the input data which decreases the interpretability of the acquired results. In addition, such models have several limitations since they require high computational power systems and a large training dataset, both of which are often absent in existing medical systems.13,27
Even though TL attempts to reduce the volume of data required to produce acceptable accuracies, it is still more time consuming than ML models. Thus, various efforts have explored the effectiveness of utilizing a hybrid AI approach in which the feature extraction is performed by DL and the pneumonia classification is performed by ML in attempt to reduce the required classification time. This is illustrated in 22 where Masad et al. proposed a hybrid model in which a pre-trained Convolutional Neural Network (CNN) was used for deep feature extraction while the binary pneumonia classification was performed by various classifiers including Softmax, K-Nearest Neighbor (KNN), Support Vector Machine (SVM), and Random Forest (RF). The overall reported accuracies of these classifiers are 99%, 99.3%, 99%, and 98.6%, respectively where the RF classifier consumed the largest classification time. Similarly, Zein et al. 15 paired the EfficientNetB0 pre-trained network with an SVM classifier to classify normal and pneumonic chest X-rays with an accuracy of 97%. It is observed that the accuracies resulting from TL and the hybrid approach are relatively similar despite the reduced classification time in the hybrid approach. Additionally, it should be noted that the hybrid approach does not contribute to increasing the output's interpretability.
Furthermore, identifying a region of interest (ROI) for the purpose of feature extraction through lung segmentation or image cropping has the potential to increase the interpretability of AI model outputs while decreasing the required time due to reducing the number of utilized features. In addition, Chandra and Verma 28 suggest that utilizing a feature-extraction ROI can contribute to enhancing the performance of the model by disregarding irrelevant anatomies in chest X-rays such as the heart and diaphragm, as this may ultimately reduce the probability of yielding false positive results. In fact, the current literature contains various efforts that utilized lung segmentation to predict pneumonia and other diseases in chest X-rays. 29
To illustrate, pertaining to TL, Hasan et al. 30 applied vertical cropping and image processing techniques such as Contrast Limited Adaptive Histogram Equalization (CLAHE) to 5856 chest X-rays to encourage the pre-trained networks to extract features from the lung nodule area exclusively. Using an 80:20 training to testing ratio, the achieved accuracies are 96.2% and 95.9% for the VGG-16 and VGG-19 networks, respectively. Moreover, pertaining to ML, Chandra and Verma 28 utilized lung segmentation to extract first-order statistical features from a dataset comprising of 412 chest X-rays. Subsequently, the authors evaluated the performance of several ML classifiers such as Multi-Layer Perception (MLP), RF, Sequential Minimal Optimization (SMO), Logistic Regression (LR), and classification via regression, where LR yielded the highest accuracy of 95.39%. Since this accuracy is comparable to the accuracies achieved via DL in 30 despite the utilization of a significantly smaller dataset, combined with the fact that smaller datasets often result in a poorer model performance, it can be inferred that ML is quite promising when it comes to the fast, accurate, and interpretable detection of pneumonia in chest X-rays.
In addition to specifying feature-extraction ROIs, using different feature extraction and selection techniques can also minimize the computational effort of a ML model and enhance its performance. For instance, Akgundogdu 31 proposed the utilization of feature extraction based on two-dimensional Discrete Wavelet Transforms (DWT) to detect pneumonia in chest X-ray images. This method was evaluated on the Artificial Neural Network (ANN), KNN, SVM, and RF classifiers yielding an accuracy of 95.85%, 94.5%, 93.41%, and 97.11%, respectively. Moreover, Ebiele et al. 32 compared the performance of several ML classifiers before and after utilizing Principal Component Analysis (PCA) for feature extraction and selection, and the results support that utilizing PCA improved the accuracy, precision, recall, and F1 scores. Nevertheless, explicitly exploring the impact of feature selection on the ML model performance is not sufficiently addressed in the literature that addresses pneumonia detection in chest X-ray images. Therefore, exploring how ML models requirements, such as feature extraction, can be altered to provide an enhanced performance is a main focus of this paper.
Methodology
The proposed ML approach for pneumonia detection in pediatric chest X-rays is summarized in Figure 1 and guided by the following steps:
In Figure 2, the leftmost image represents a “Normal” preprocessed chest X-ray image before data augmentation, while the rightmost image represents the same image after data augmentation. For every scheme, the fault detection explained in step 4 will be conducted to determine the scheme, which yields the highest pneumonia classification accuracy.
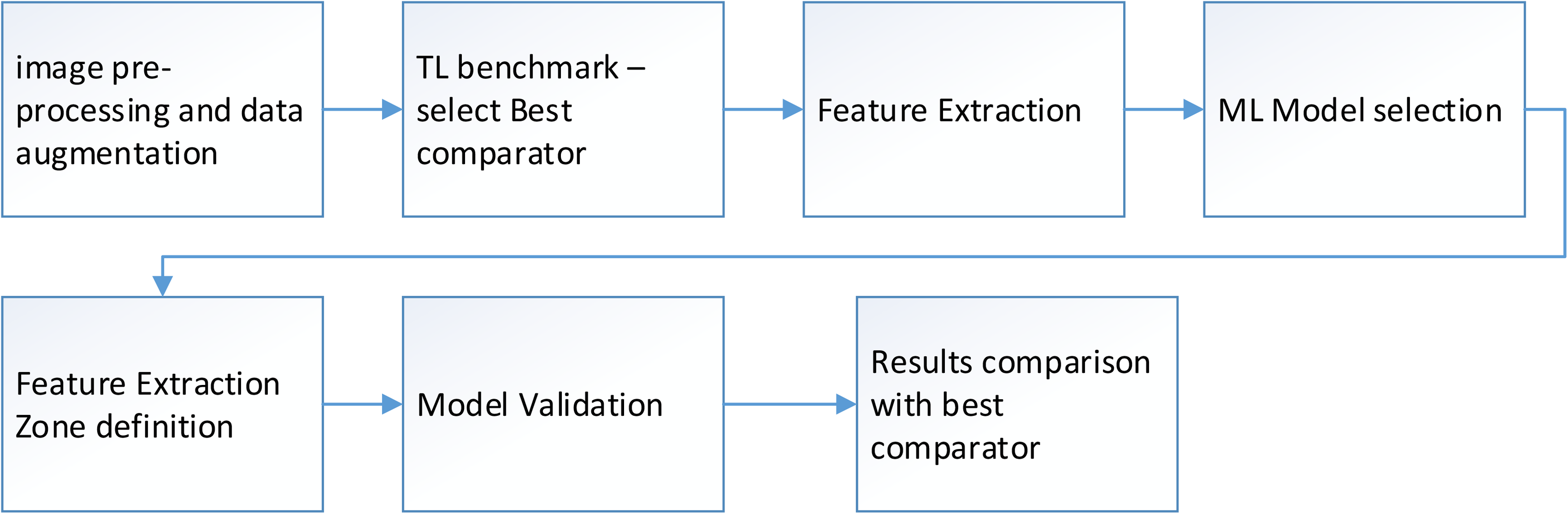
Methodology block diagram.
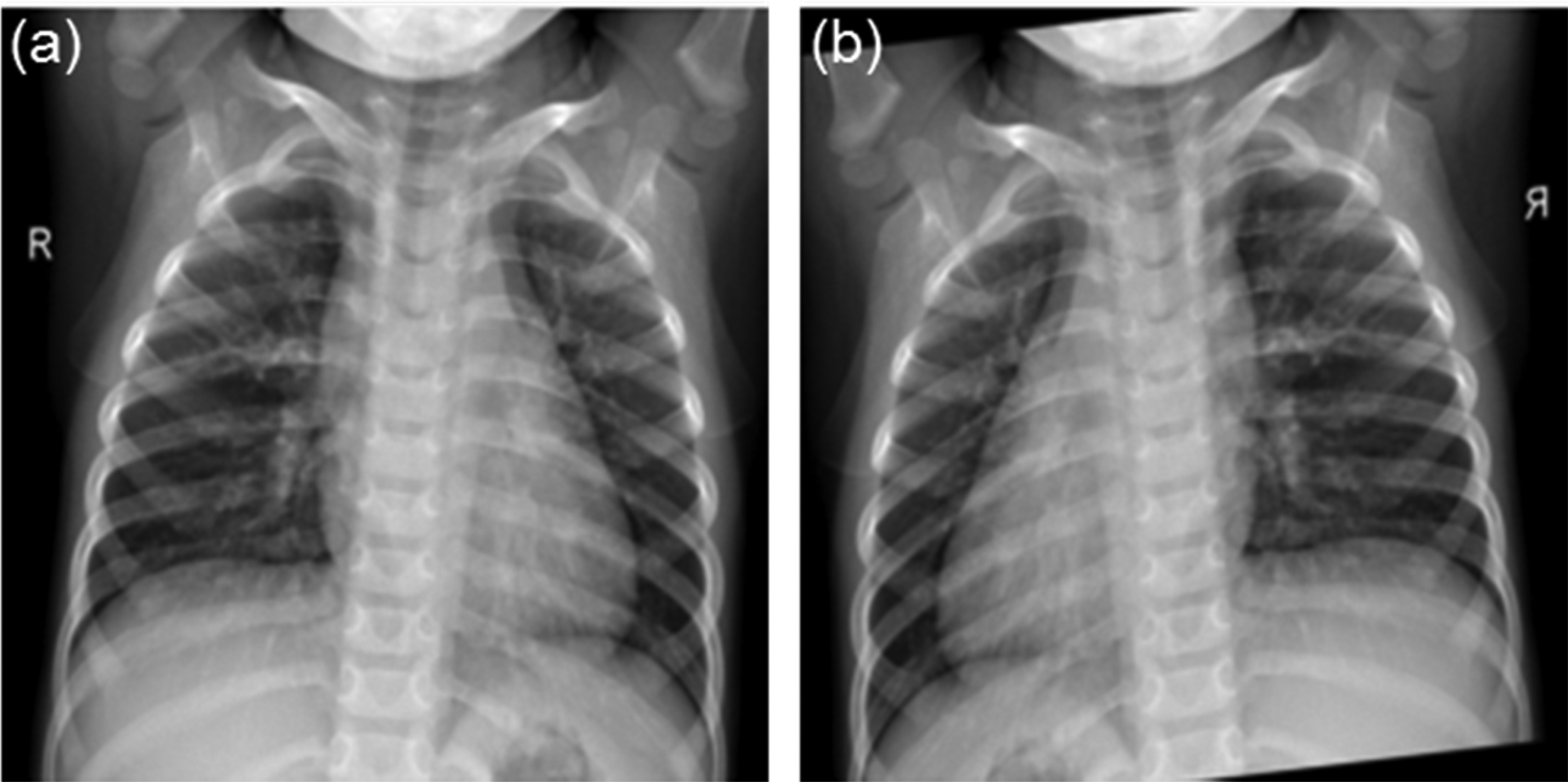
An example of a normal chest X-ray image before (a) and after (b) data augmentation.

A schematic of the investigated ROIs; (a) 4 ROIs, (b) 16 ROIs, (c) 64 ROIs, and (d) 256 ROIs.
Proposed data augmentation techniques and their corresponding parameter values.

Moreover, the proposed methodology will be evaluated using a desktop computer with an Intel® Xeon® Processor E5–1650 at 3.2 GHz and 16 GB of RAM.
Dataset
The publicly available dataset used to validate the proposed methodology is published by Kermany et al. 33 It is comprised of 5856 pediatric chest X-ray images belonging to pediatric patients from Guangzhou Women and Children's Medical Center, where the patients’ age ranges between one and five years old. Notably, the dataset is highly imbalanced where 4273 images are labeled as pneumonic while 1583 images are labeled as normal. Moreover, Figure 4 provides a comparison between a normal and a pneumonic chest X-ray from the utilized dataset.
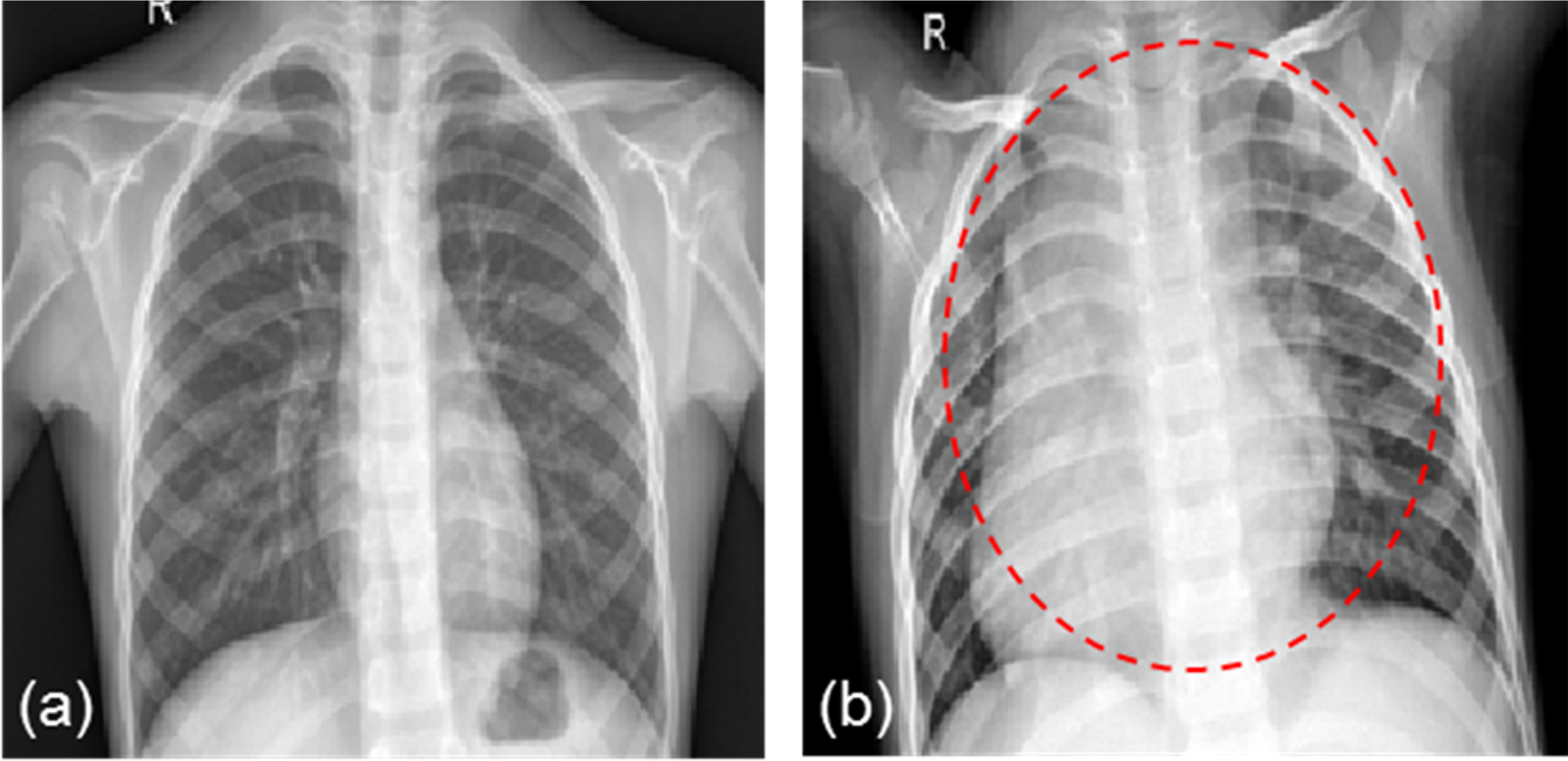
An example of a normal (a) and a pneumonic (b) chest X-ray from the utilized dataset.
It can be observed that the lung nodule area in the pneumonic image appears brighter than that of the normal image due to the accumulation of fluid.
Results and discussion
This section presents and discusses the results of the TL benchmark and the proposed ML technique.
Transfer learning benchmark
The deep pre-trained AlexNet network is chosen to detect pediatric pneumonia in chest X-rays before and after image augmentation to serve as a time and accuracy comparison benchmark for the results obtained by utilizing the proposed ML methodology. The original and augmented datasets are divided into 70% training, 15% testing, and 15% validation sets as shown in Table 2.
Description of the original and augmented dataset division utilized for Transfer Learning.

Subsequently, AlexNet is trained then utilized to predict the classes of the testing set where Table 3 provides a summary of the obtained results.
A summary of the Transfer Learning benchmark results.

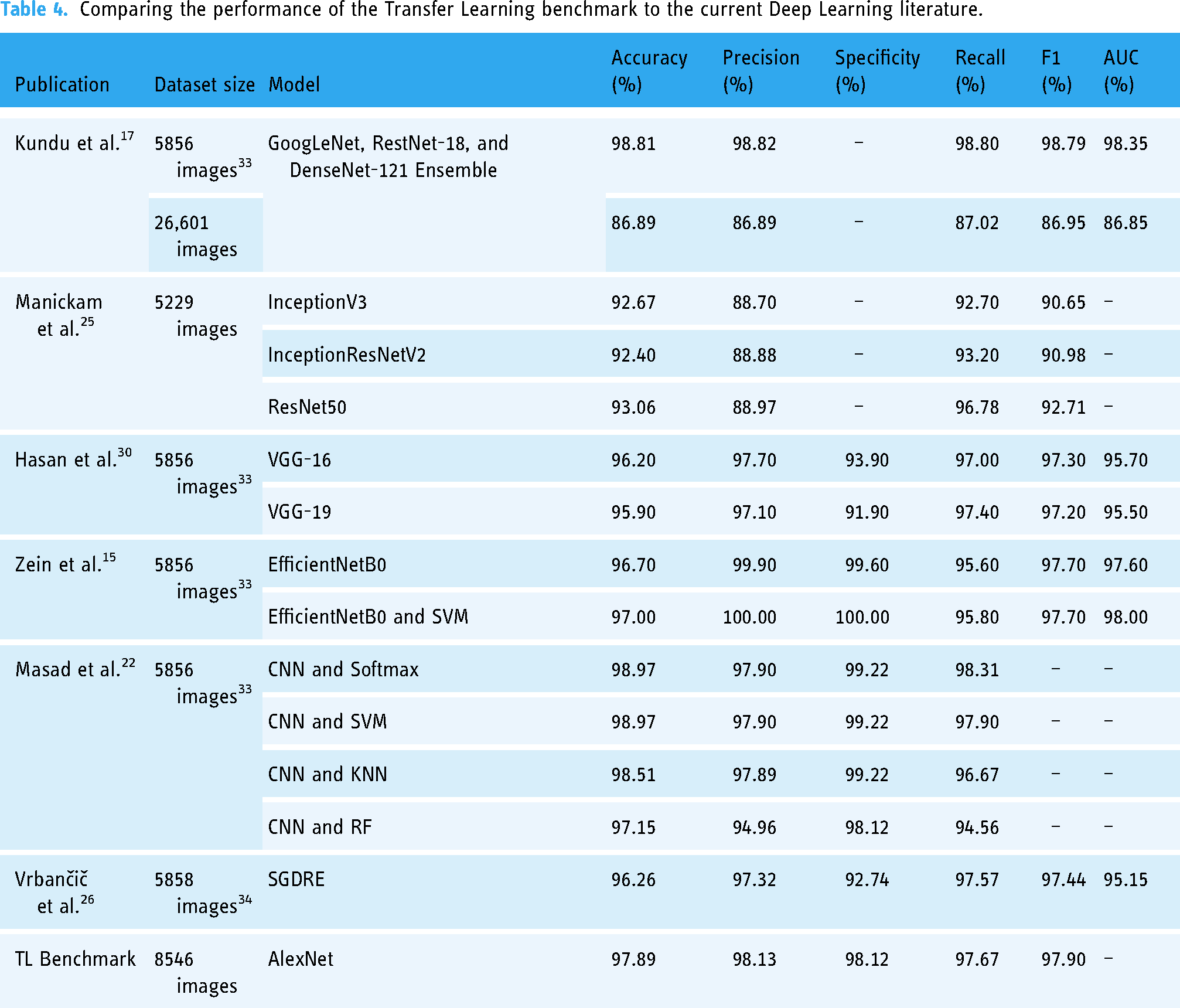
By analyzing the results presented above, it can be inferred that utilizing data augmentation contributed to a slight improvement in the testing accuracy at the expense of an added 50 min of training time. In addition, the performance of AlexNet is comparable to that of the models presented in the DL literature (see Table 4), which justifies utilizing it as an accuracy benchmark.
Comparing the performance of the Transfer Learning benchmark to the current Deep Learning literature.
Furthermore, since AlexNet is generally less computationally expensive than the other pre-trained models, 35 utilizing it as a benchmark for time comparison is justifiable.
Machine learning results
Pertaining to ML, the augmented dataset is divided using the scheme described in Table 5.
Description of the augmented dataset division utilized for Machine Learning.

After that, the sixteen proposed statistical features are extracted from five feature-extraction schemes, namely the whole image and 4, 16, 64, and 256 equally sized regions per image. This is conducted to obtain insights regarding the impact of the feature-extraction ROI size on the model output. Consequently, the performance of twenty-six ML classifiers is evaluated using the MATLAB Classification Learner and Neural Net Fitting apps on each feature-extraction scheme, and the resulting accuracy is displayed in Figure 5.
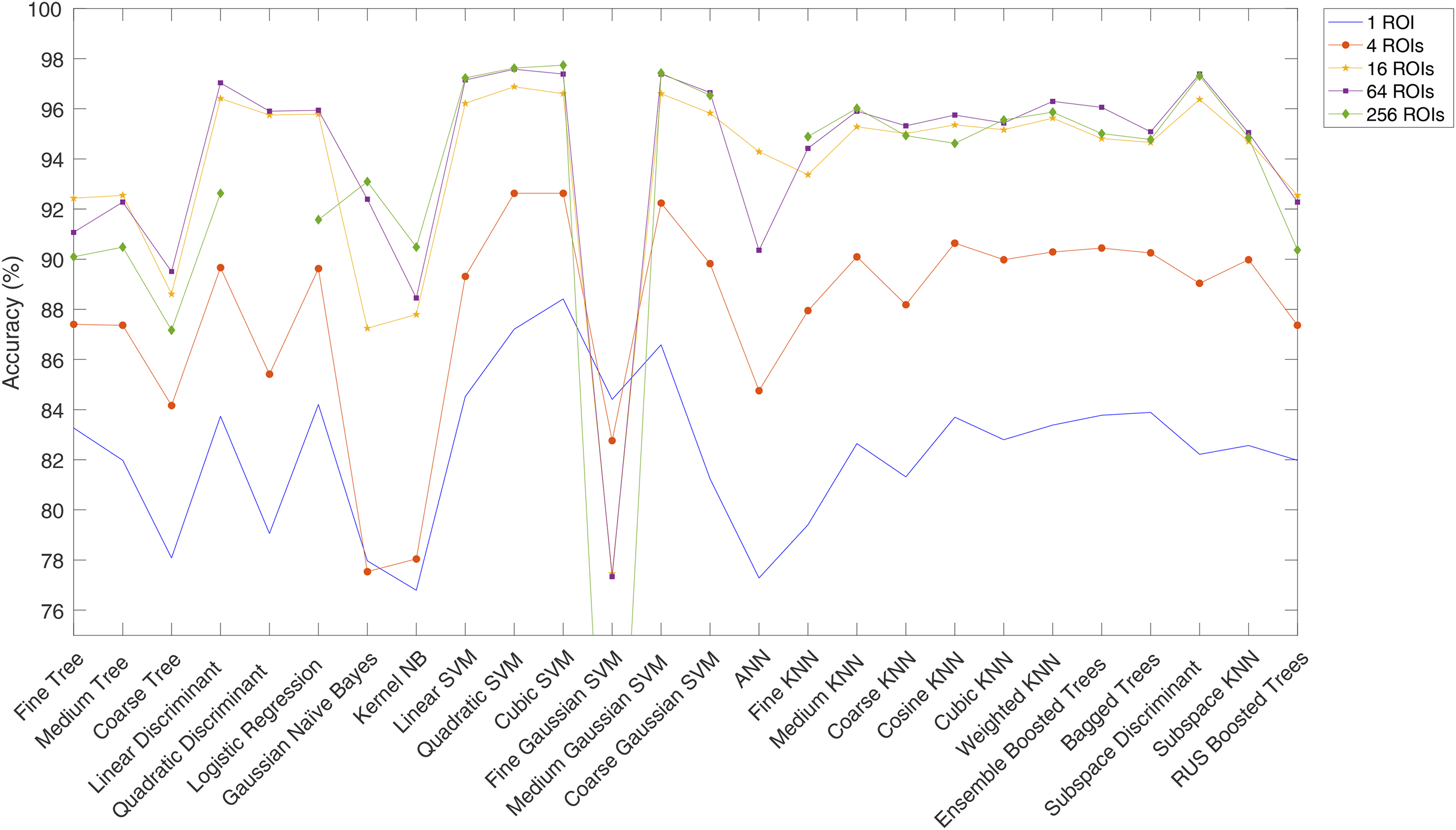
Plot of accuracy per ML classifier for each feature-extraction ROI scheme.
Thus, it can be inferred that a higher number of feature-extraction ROIs results in a higher testing accuracy where the top performing ML classifiers include several classifiers belonging to the SVM family as well as the subspace discriminant classifier. This is in line with 36 that demonstrates SVM's candidacy in medical applications as it tends to yield the highest classification accuracy. It should be noted that the Quadratic Discriminant and Artificial Neural Network (ANN) models did not converge for the 256 ROI scheme as they were computationally expensive. In addition, the accuracies achieved by the top performing classifiers for the 64 and 256 ROI schemes surpass those provided in the ML literature as summarized in Table 6.
Comparing the performance of the top Machine Learning models to the proposed Machine Learning model.

When comparing these two schemes, it is observed they are quite similar in term of the testing accuracy, especially when it comes to the top performing classifiers. However, when factoring in the training time, the performance of the 64 ROI scheme is superior as illustrated in Figure 6. Hence, the 64 ROI scheme is optimal.
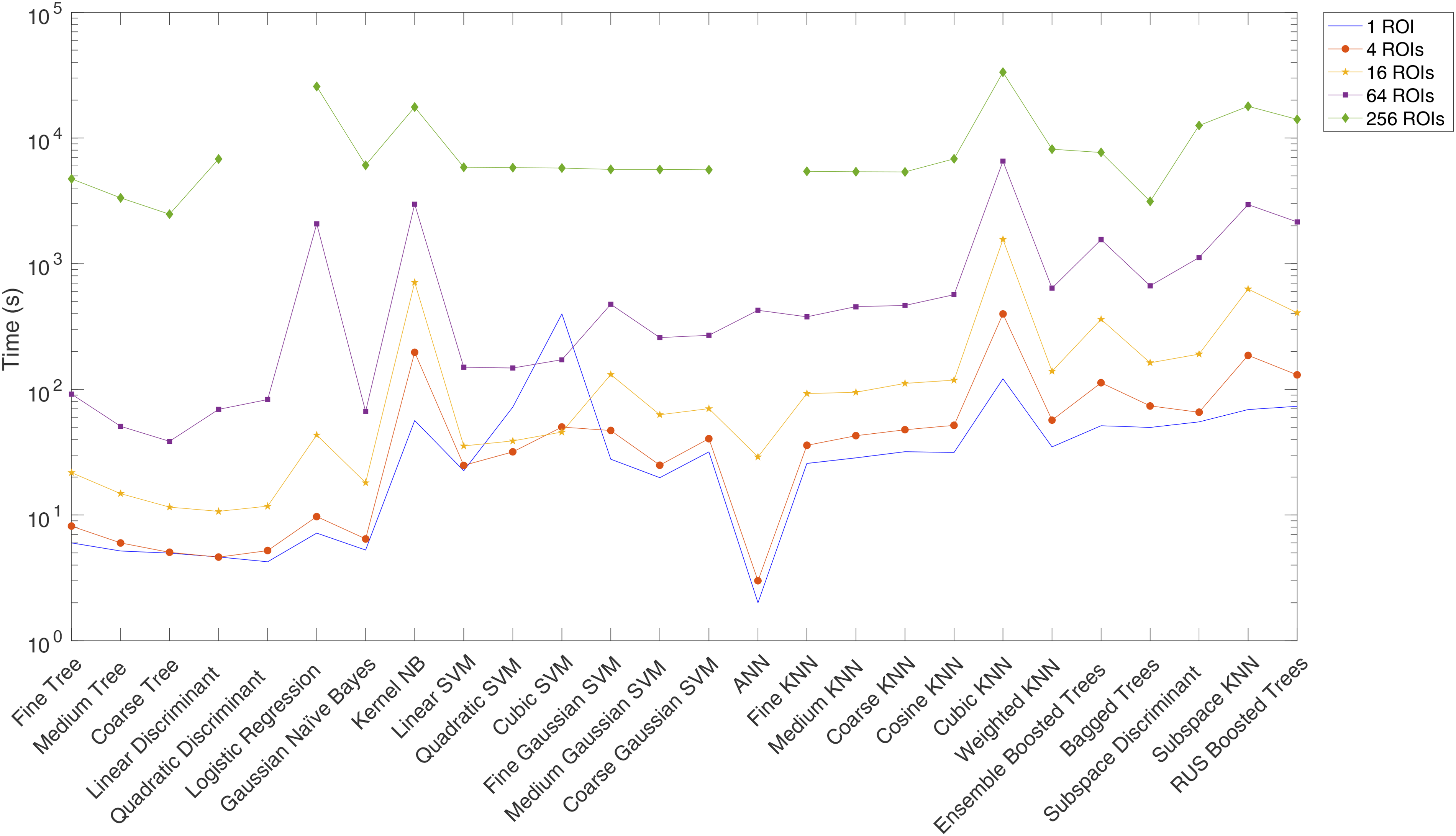
Plot of training time per ML classifier for each feature-extraction ROI scheme.
Statistical analysis
The SVM ML family provided the best results in terms of accuracy and time. However, the differences among the SVM methods were not that obvious. The analysis was repeated 10 times for all SVM tools results and the accuracy (training and testing), training time, TN, TP, FN, and FP were obtained. Each time, the data was split to 30% test and 70% train randomly, then the normal class was augmented to match the pneumonia class (augmentation is also random). Next, ANOVA analysis was conducted for each outcome among the SVM family ML methods. Based on the p-value of 0.625, there is no statistically significant difference among the methods in terms of time, training accuracy, and true negatives (TN). However, there is strong evidence of a difference in terms of training and testing accuracy, false positives (FP), and true positives (TP) with p-values of 0.00. Figures 7 and 8 depict box plots for the results.

Box plot of training time.
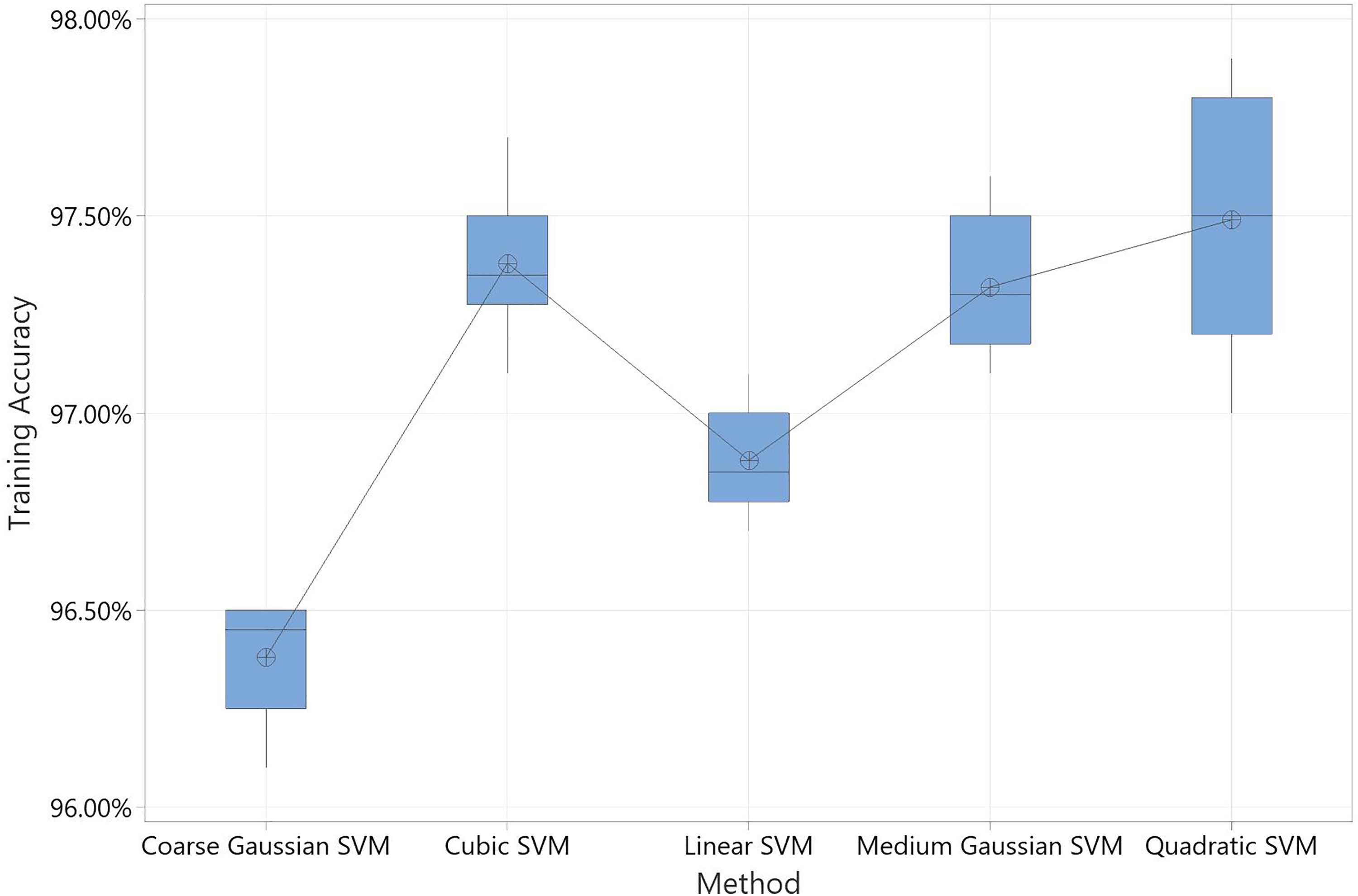
Box plot of training accuracy.
Notably, the time required for this scheme is significantly lower than the time needed by the TL benchmark as shown in Table 7. In addition, the quadratic SVM yielded an accuracy of 97.58% for the 64 ROI scheme, which is comparable to the 97.89% obtained via TL.
Comparing the performance of the proposed Machine Learning approach with the Transfer Learning Benchmark.

Hence, the results highlight the candidacy of ML as an accurate and computationally inexpensive tool for pediatric pneumonia detection in chest X-ray images. As mentioned earlier, the motivation behind utilizing ML in the proposed technique lies in its low computational expense and high potential for interpretability. While the former has been successfully confirmed by the obtained results, the latter possesses potential for further improvement. Thus, after realizing the optimal feature-extraction scheme, namely the 64 ROI scheme, the most important features are identified using the Classification and Regressions Tree (CART) method using Minitab and traced back to their corresponding ROI locations in the chest X-ray images as shown in Figure 9.

A schematic illustrating the ROIs containing the most important features obtained from Minitab.
This adds an interpretable dimension to the proposed approach as it brings critical ROIs from the X-rays to the radiologists’ attention while making the diagnosis decision using our model. Moreover, to further enhance the interpretability, the future work will tackle reducing the X-ray images to a centered square enclosing the locations that contain the identified important features as illustrated in Figure 10.

An example of a reduced X-ray image.
Then, step 5 of the methodology will be repeated for the reduced X-ray images considering a higher number of feature-extraction regions than those in the optimal ROI scheme. This step will be reiterated until a satisfactory model performance is achieved, where this will answer whether focusing on a specific region in the X-rays is more efficient than considering the image as a whole.
Conclusion and future work
In this paper, a novel ML approach is proposed to reliably detect pediatric pneumonia in chest X-rays. This approach entails performing data augmentation to balance the classes of the utilized dataset, optimizing the feature extraction scheme, and evaluating the performance of several ML models. Moreover, the performance of this approach is compared to a TL benchmark to evaluate its candidacy. After investigating the effect of varying the number of the feature extraction ROIs on the classification time and accuracy, it is inferred that the 64 ROI feature extraction scheme is optimal. Using this scheme, the Quadratic SVM model yielded an accuracy of 97.58%, which surpasses the accuracies reported in the current ML literature. In addition, this scheme's classification time is significantly smaller than that of the TL benchmark, making it practical and less computationally expensive. Hence, the results highlight the candidacy and potential of the proposed approach in detecting pediatric pneumonia in chest X-rays.
Moreover, the future work will investigate refining the optimal feature-extraction scheme to enhance the proposed approach's performance and interpretability. This will be conducted by reducing the X-ray images into a centered square enclosing the locations that contain the lungs. Then, the feature extraction scheme will be optimized for the reduced images by varying the number of ROIs. This step will be reiterated until a satisfactory model performance is achieved, as this will provide insights on whether focusing on a specific region in the X-rays is more accurate and efficient than considering the image as a whole. Then, the most important features will be determined using several methods such as CART. Subsequently, these features will be traced back to their corresponding locations in the chest X-rays in order to aid medical practitioners in diagnosing pediatric pneumonia in an interpretable manner.
Lastly, it is important to note that this research is limited to the binary classification of pneumonia using pediatric X-rays with a certain resolution and a sufficient sample size. It is worth mentioning that the proposed method has a potential of being used for other applications such as detection of COVID-19 or shaft defects in a motor or other binary classification applications using X-ray images or any other different imaging modality. Similarly, the proposed method may be used on adult X-rays that vary anatomically from pediatric X-rays. Nevertheless, each one of these applications might have its own specific challenges and opportunities. As a result, generalizing this method to other applications needs to be evaluated.
Footnotes
Acknowledgements
The authors would like to thank Dr Hussam Alshraideh from the industrial engineering department at the American University of Sharjah for his support and technical advice. The work in this paper was supported, in part, by the Open Access Program from the American University of Sharjah. This paper represents the opinions of the author(s) and does not mean to represent the position or opinions of the American University of Sharjah.
Author contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Natalie Barakat, Mahmoud Awad and Bassam Abu-Nabah. The first draft of the manuscript was written by Natalie Barakat and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Consent to participate
Not applicable- A publicly available dataset is used in this study is. Data set is published by Kermany et al. 33
Consent to publish
Not-applicable- A publicly available dataset is used in this study is. Data set is published by Kermany et al. 33
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a scholarship fund provided by the Engineering Systems Management department at the American University of Sharjah.
Guarantor
Mahmoud Awad.
