Abstract
Objective
There has been tremendous growth in wearable technologies for health monitoring but limited efforts to optimize methods for sharing wearables-derived information with older adults and clinical cohorts. This study aimed to co-develop, design and evaluate a personalized approach for information-sharing regarding daily health-related behaviors captured with wearables.
Methods
A participatory research approach was adopted with: (a) iterative stakeholder, and evidence-led development of feedback reporting; and (b) evaluation in a sample of older adults (n = 15) and persons living with neurodegenerative disease (NDD) (n = 25). Stakeholders included persons with lived experience, healthcare providers, health charity representatives and individuals involved in aging/NDD research. Feedback report information was custom-derived from two limb-mounted inertial measurement units and a mobile electrocardiography device worn by participants for 7–10 days. Mixed methods were used to evaluate reporting 2 weeks following delivery. Data were summarized using descriptive statistics for the group and stratified by cohort and cognitive status.
Results
Participants (n = 40) were 60% female (median 72 (60–87) years). A total of 82.5% found the report easy to read or understand, 80% reported the right amount of information was shared, 90% found the information helpful, 92% shared the information with a family member or friend and 57.5% made a behavior change. Differences emerged in sub-group comparisons. A range of participant profiles existed in terms of interest, uptake and utility.
Conclusions
The reporting approach was generally well-received with perceived value that translated into enhanced self-awareness and self-management of daily health-related behaviors. Future work should examine potential for scale, and the capacity for wearables-derived feedback to influence longer-term behavior change.
Keywords
Introduction
In the last decade, there has been tremendous growth in the development of wearable technologies (“wearables”) for both consumer and research or healthcare applications. Despite some challenges to implementation, the use of wearables for clinical research and care is expanding as digital endpoints are validated.1–3 In older adults and persons living with complex health conditions such as neurodegenerative disease (NDD), objective measures of health captured outside of clinic visits may be particularly relevant for guiding therapeutic development and treatment decisions.4–6 Within the field, sights are set on integrating information derived from wearables into clinical trials and healthcare to capitalize on objective, patient-generated, health information captured in daily life.7,8 These approaches also hold promise for encouraging active patient engagement and shared clinical decision making. 9 Important strides have been made in wearable device and analytic development in support of physiological and behavioral monitoring during everyday activities,2,10 but despite these advances, there have been limited efforts to develop effective methods for sharing information derived from wearables (“feedback”) with clinicians and patients, particularly those with complex health conditions. The absence of evidence-informed recommendations addressing “what, when and how” wearables-derived information should be shared remains a critical gap in the field.
In some use cases, information generated from wearables is relatively interpretable, and the utility of the feedback in clinical decision-making is well-established. This is the case for feedback that targets a specific clinical outcome such as information obtained from Holter or continuous glucose monitors.11–14 Many wearables, however, have been designed to monitor more general, lifestyle-related, health behaviors such as physical activity, sleep or periods of stress for which evidence in support of the utility of feedback is mixed.15–21 Varied results may be linked, in part, to differences in the timing of feedback delivery (e.g. immediate or delayed)22–25 or whether feedback is delivered with or without the support of an expert (e.g. healthcare professional and trainer).23,26–28 Evaluation outcomes used to examine the utility of feedback (e.g. improved health or positive change in health-related behavior, changes to self-efficacy or motivation) 29 may also impact conclusions regarding uptake and effectiveness. For example, older adults in an exercise group who received smartwatch-derived feedback plus cognitive behavioral counselling reduced sedentary behavior more than those in an exercise-only group. 30 However, a group of older adults enrolled in a lifestyle intervention program who received immediate feedback from a wrist-worn activity tracker about number of steps and calories burned showed no change in physical activity self-efficacy. 31
To optimize uptake and effectiveness of wearables-derived feedback for lifestyle-related health behaviors and symptoms of disease, several practical considerations prioritizing the safe, appropriate and meaningful use of feedback must be considered, all of which extend current options afforded by many consumer and commercial devices. Of particular importance for accurate and reliable clinical reporting are including only those wearables-derived endpoints that have been validated for use in the population of interest 1 and which have clear guidelines for interpretation that allow comparisons with similar people22,28 and direct appropriate action. 32 Commonly used consumer and commercial devices provide limited opportunity to tailor feedback to the individual because of constraints associated with proprietary methods used to derive and share data. 33 These restrictions impact the potential to provide meaningful feedback and are particularly problematic for older adults and clinical cohorts, since a “one size fits all” approach does not consider the impact of unique age- or disease-specific features on reported outcomes. For example, clinical features of Parkinson's disease (PD) such as reduced arm swing, shuffling gait or akinesia can impact established metrics characterizing physical activity and mobility.34,35 In these instances, algorithms developed in healthy adults are not likely to provide accurate, reliable or meaningful information to a person living with PD (e.g. Weber et al. 36 ).
Personalizing feedback to ensure relevance to the individual or a sub-group of individuals and developing a communication strategy that supports delivery of information to the target audience are also critically important for successful implementation.22–24,28,37 To ensure relevance, assumptions should not be made about what an individual will value, and end users (patients and clinicians) should be engaged in decisions surrounding feedback, so that the information shared is driven by what has been deemed important to personal or clinical decision-making.23,27,38 To ensure the safe and appropriate use of feedback by individuals, it is critical to have an effective communication strategy that supports interpretation of the data, 25 including contextualizing the results and clearly noting the data's limitations to safeguard against overinterpretation.22,26,28,39 Consideration must also be given to feedback methods that will optimize consumption by end users with diverse needs, capabilities and resources (i.e. format, structure, language level, etc.).22,25,27,39
To address the need for accurate, meaningful and digestible information to guide personal and clinical decision making, we designed an iterative co-development and implementation project focused on providing feedback derived from wearables data for older adults and those with cerebrovascular disease (CVD) and NDDs including PD, Alzheimer's disease (AD), mild cognitive impairment (MCI), frontotemporal dementia (FTD) and amyotrophic lateral sclerosis (ALS). We view provision of feedback to these groups to be critical for two reasons: 1) the application of wearable technology is particularly relevant to these cohorts where monitoring health-related behaviors for purposeful action may positively impact health trajectory, health outcomes and overall quality of life;35,40–42 and 2) heterogeneity within these groups emphasizes the need for approaches that address individual differences in patient-generated data and individuals’ capacity to access, understand and use feedback. The overarching principle of this work was to develop an approach for user feedback that was suitable for older adults and those with complex health conditions, and which did not rely on a particular device or technology. The latter was achieved by using raw data from the wearables to derive validated measures for our populations of interest, rather than relying on standard outputs from proprietary algorithms that can limit confidence in the data and constrain how the data are presented.
The primary objective of this study was to co-design and develop a user-centered approach for reporting outcomes from continuous monitoring with wearables to enhance individuals’ self-awareness and self-management activities related to daily health-related behaviors. While this work targeted specific cohorts, the products of this work have broad applicability related to effective wearables data-sharing for personal and clinical decision making.
Methods
The study was conducted in two phases (Figure 1). The development phase focused on an iterative co-design and development process with the goal of generating a person-centered feedback report targeting health behaviors, using data derived from wearable sensors. In the second phase of the project, the team evaluated the acceptability, useability and impact of the feedback report in a sample of older adults and persons living with CVD or NDD. The study was conducted within the Ontario Neurodegenerative Disease Research Initiative (ONDRI; ondri.ca), namely the Remote Monitoring in Neurodegenerative Disease (ReMiNDD) study 43 and the Health in Aging, Neurodegenerative Diseases and Dementias in Ontario (HANDDS-ONT) study (Figure 1).

Timeline for feedback reporting development and evaluation activities.
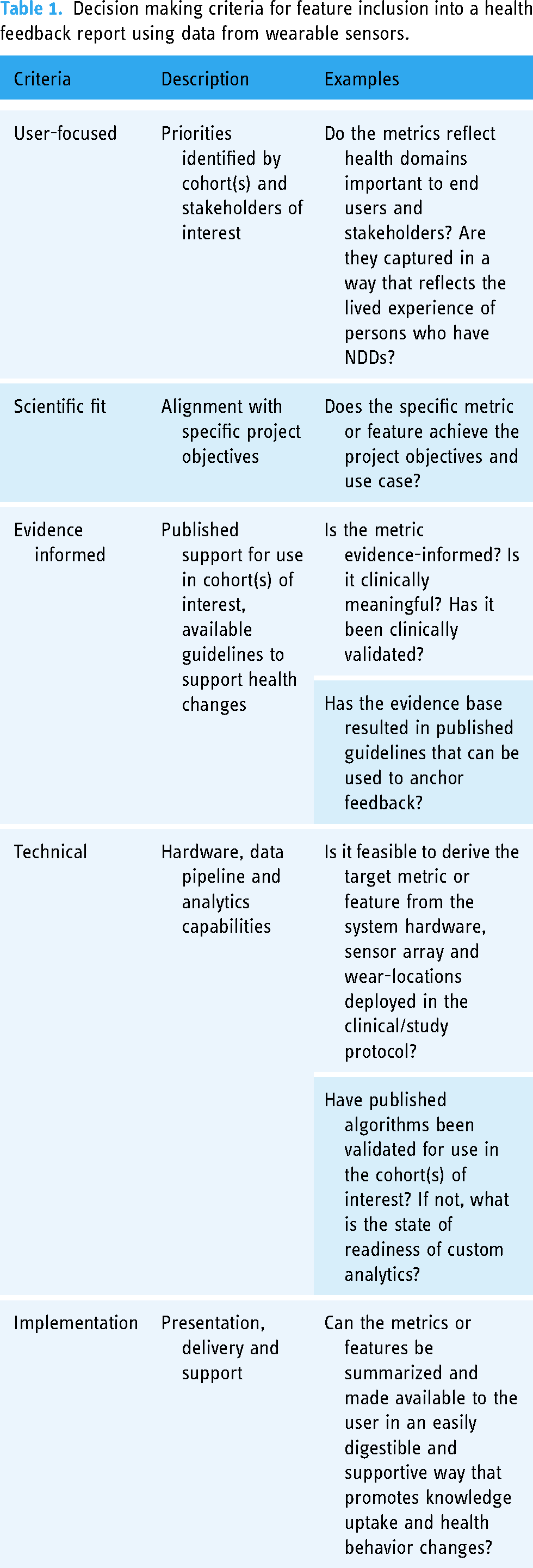
Our approach consisted of iterative cycles of stakeholder input. Each cycle consisted of planning, consolidation, implementation and evaluation stages (Figure 2) where stakeholder perspectives were integrated into report versions following a consolidation process to confirm that proposed design features aligned with project objectives using established criteria (Table 1).

Feedback reporting co-design and development approach grounded in a model of embedded stakeholder engagement.
Decision making criteria for feature inclusion into a health feedback report using data from wearable sensors.

Sample summary data frame generated for review as part of personalized feedback reporting. Data are included from multiple wearable sensors to integrate information about sleep (purple), gait (green), activity (yellow/orange), and sensor removal (grey), within and across days, for each participant. Each row represents a 24-hour period (days 1–7 stacked), from midnight to midnight. Duration of a specific behavior is represented by the width of the block.
When identified, any additional flags were logged, and processing steps reviewed before determining the necessary action. Unresolved instances (i.e. true behavioral data) were addressed with customized feedback that was entered into the report on a per participant basis. Once the data review was complete, a personalized feedback report was generated.
Phase 1: feedback development
Cycle 1—prototype report
Planning and consolidation
In Spring 2019, a voluntary survey was made available at an ONDRI community outreach event for an audience comprised persons with lived experience, family care partners and persons from the community seeking information on dementia, CVD and NDDs. The survey asked respondents to identify potential targets for inclusion in a feedback report (see Appendix 1—Initial Survey). Five attendees completed the survey (three NDD, two care partners). Responses were used as a starting point and considered alongside our additional criteria for reporting (see Figure 2 and Table 1) to determine what outcomes to include in a prototype feedback report. To check the face validity of the prototype report, we triangulated its content with our stakeholder group (Figure 4). Stakeholder reflections on the prototype report were integrated as part of the consolidation process (Figure 2).

Patient and Community Advisory Committee stakeholder representation. Clinicians include specialty, community-based, and allied health. Twelve members overlapped from prototype to version 1. Prototype development also included input from trial survey respondents with lived experience (n = 5; 3 NDD, 2 care partner) while draft version 1 was informed by ReMiNDD study respondents (n = 13 NDD). NDD: neurodegenerative disease.
Wearables data collection and prototype report generation
The prototype version of the feedback report was implemented and evaluated as part of the ONDRI ReMiNDD study. ReMiNDD was conducted between May 2019 and March 2020 and included 39 participants living with CVD or any of the following NDDs as diagnosed by a neurologist: PD, AD, MCI, FTD, or ALS (see Godkin et al. 43 for participant and protocol details including devices used). Importantly, all devices captured and logged raw data, which were subscribed to a custom analytics pipeline to derive outcomes related to physical activity, sleep and mobility (i.e. outcomes generated via proprietary software provided by device manufacturers were not included in the report). At the time of enrollment, participants were told that their participation included receipt of a personalized feedback report based on data collected during a week-long period of sensor wear. The ReMiNDD study received ethics approval through the Sunnybrook Research Institute in Toronto, Ontario (REB approval: 007–2019), and all participants provided written, informed consent.
Preliminary implementation and evaluation
Prototype feedback reports were mailed to each ReMiNDD study participant approximately 3 months following completion of the in-home sensor wear portion of the study. Included in the report was a link to an accompanying video that guided participants through reading and interpreting their report. A one-page survey was appended to the back of each feedback report. Thirteen ReMiNDD study participants completed the voluntary survey, which was offered to all participants (n = 39) (see Appendix 1 for Prototype Report Survey and Results).
Cycle 2—version 1 report
Planning and consolidation
Data from Cycle 1 resulted in several changes being made to the prototype feedback report to create a revised version of the report for implementation into a clinical study context. Additional details were layered into the report including supplementary graphs and additional evidence-based measures to address the desire for more information based on Cycle 1 feedback (e.g. inclusion of sedentary behavior and nighttime walking). Changes embedded within the draft report version also addressed the need to account for known processing challenges in our groups of interest by including redundancies in information and ensuring readability by using multi-modal communication supports (e.g. written and visual) and reducing language complexity.49,50 All changes were included in draft form for subsequent review by research partners before Cycle 2 implementation.
In Fall 2021, the revised feedback report was submitted to the ONDRI PCAC (Figure 4). A guided focus group process was used to elucidate collective feedback on key areas for report implementation including the volume and type of information, readability for persons living with NDD and those within their circle of care, and overall format, layout and organization of the report. Specific themes that emerged from their feedback included the value of having participants understand
Wearables data collection and version 1 report generation
The Version 1 feedback report was implemented and evaluated as part of the HANDDS-ONT study (Figure 1); an Ontario-based, adaptive clinical trial 51 in community-dwelling healthy adults (termed here as “controls”) and persons living with probable or diagnosed CVD, PD, AD, MCI, FTD or ALS (termed here as neurodegenerative disease or NDD). HANDDS-ONT was designed as a fully remote study connecting clinical data with cellular, protein marker and genetic data, free-living behavioral data measured by wearables and longer-term health outcome data. Individuals were eligible to participate in HANDDS-ONT if: (a) they were 18 years of age or older, with self-reported English proficiency at 7/10 on a modified version of the Language Experience and Proficiency Questionnaire (LEAP-Q); 52 (b) had internet or telephone access; (c) could attend a community-based laboratory to provide a blood sample; and (d) were under the care of a primary medical care provider and were willing to put forth their provider's name. Participants wore three devices continuously for up to 10 days: an inertial measurement unit on the dominant (or least affected) wrist and ankle (AX6, Axivity Ltd, UK) and a chest device including combined electrocardiography and a tri-axial accelerometer (Bittium 360, Bittium Faros, FI). Similar to the ReMiNDD study, all devices captured and logged raw data, which were subscribed to a custom analytics pipeline to derive outcomes related to physical activity, sedentary behavior, sleep and mobility. The pipeline also generated the summary data frames for review (Figure 3) and the participant feedback reports. These outcomes were chosen for their relationship to chronic disease and brain health, their importance for self-management of health 9 and their potential for early detection or monitoring of declines in health.9,44,53–56 Specifically, the report included the following parameters: daily volume and patterns of light and moderate-vigorous physical activity and sedentary behavior, daily volume and pattern of stepping, step count accumulated in walking periods of different durations, total sleep time, duration of additional time in bed and patterns of nighttime behavior. See Figure 5 for details regarding the Version 1 report. The HANDDS-ONT study received ethics approval through Clinical Trials Ontario (Project ID: 3684) with Sunnybrook Research Institute in Toronto, Ontario as the Board of Record (REB approval: 2021–1517), and all participants provided written, informed consent.

Sample pages from participant feedback reports. On the left, supporting materials including recipient orientation to tables and graphs, glossary with definitions of outcomes and a summary of published guidelines. On the right, samples showing layers of information from high level summaries through to detailed patterns of behavior. Along the bottom, a description of features and information provided within the report.
Phase 2: version 1 report implementation and evaluation
Within the HANDDS-ONT study, feedback reports were returned to study participants in their preferred format (digital, hard copy or both). Given improvements in the automation of our personalized report processing pipeline, the target for turnaround from data offload to participant receipt of the report was 2–4 weeks, depending on format. Participants also received a 45-minute, one-on-one review of their report with trained study personnel via secure video conference. This study visit was structured to include the following:
Playback of a video, which highlighted key features of the report and oriented participants to the ways in which the data are presented (e.g. introduction to tables and figures). Study personnel-led page-by-page review of participant data highlighting key takeaways from the report, including anchoring detected behaviors to personal activity or sleep log recordings (where applicable), and to published guidelines (tailored to age group). To ensure a standardized approach across participants, no specific recommendations related to participants’ medical history or disease status were provided. Participants were encouraged to speak with their doctor before making health behavior-related changes and to share the report with others in their circle of care. Time for the participant to seek clarity or to ask questions about the information contained within the report.
Two weeks after the feedback report review visit, a final study visit was conducted to gather HANDDS-ONT participant perspectives on the report and probe whether behavior change and/or information sharing had occurred. Data were collected using a mixed methods approach, including both open-ended questions, and a coordinator-administered survey with questions displayed on-screen. Any additional comments provided by the participant were recorded verbatim within free-text fields (see Appendix 1 for Version 1 Report Survey and details of development).
Data analysis
A convenience sample including the first forty participants aged 60 or over enrolled in the HANDDS-ONT study was used to evaluate version 1 of the feedback report (participation from 09 December 2021 to 20 May 2022). Descriptive statistics were used to summarize the data. Data were summarized as whole group responses and stratified by cohort (i.e. control versus NDD) and cognitive status (COGhigh = MoCA ≥26, 57 COGlow = MoCA ≤22 58 ) using clinical data captured within the HANDDS-ONT study. Findings were also supplemented with illustrative examples akin to single subject analysis.
Results
Participants
HANDDS-ONT survey respondents included 25 control participants and 15 participants living with CVD or an NDD, who were sampled from the study's five cohorts as follows: seven CVD (one with MCI), four AD/MCI, and four PD. Table 2 summarizes participant demographic and clinical characteristics.
HANDDS-ONT participant demographics (survey data as of 20 May 22).

Data are presented as median range unless otherwise noted.
MoCA: Montreal Cognitive Assessment.
Higher score indicates greater functioning.
Feedback report evaluation
Results of the evaluation are shown in Figure 6. Most survey respondents (82.5%) found the report “somewhat” or “very easy” to read or understand, and 80% reported “just the right amount” of information/detail. Some differences emerged when data were examined by cohort (NDD versus control) and by cognitive status (COGlow (n = 7) versus COGhigh (n = 19)), specifically with respect to ease of reading or understanding, which was deemed “somewhat” or “very easy” for a greater proportion of NDD (93%) versus control (76%) participants, and for a slightly greater proportion of COGlow (86%) versus COGhigh (74%) participants. In the context of understanding daily health-related behaviors (i.e. activity, mobility and sleep), 90% found the feedback report “very helpful” or “helpful,” and 57.5% of participants made a behavior or lifestyle change within 2 weeks. When data were examined by cohort or based on low and high cognitive status, all participants living with an NDD or in the COGlow group found the feedback “very helpful” or “helpful” compared to a slightly smaller proportion of control participants (84%) and participants in the COGhigh group (89.5%). Finally, differences emerged between COGhigh and COGlow groups with respect to the proportion who reported a lifestyle or behavior change (68% (n = 13) versus 29% (n = 2), respectively). Examples of lifestyle or behavior changes reported by participants included joining a Pilates class and striving to move more throughout the day (i.e. taking movement breaks), as well as increased awareness of good sleep habits and monitoring of sedentary behavior. Of those participants who did

(A) Summary of feedback report evaluation with respect to readability, volume of information, and helpfulness of information, with rows representing results as follows: Row 1—overall cohort (n = 40); Row 2—neurodegenerative disease (n = 15) versus control (n = 25); and Row 3—COGlow (n = 7) versus COGhigh (n = 19) for cognitive status based on MoCA scores (COGlow ≤ 22, COGhigh ≥ 26). (B) Summary of results probing impact of feedback (n = 40), specifically behavior change and information sharing (friends or family, and medical practitioner).
When asked about sharing with others in the participant's circle of care, almost all participants (92%) shared their report with a family member or friend, with the proportion of participants who shared with a family member or friend slightly higher within the cohort of persons living with an NDD (100%) compared to the control cohort (88%). Motivation for sharing with a friend or family member included wanting to engage partners in making lifestyle changes, educating and advocating for change in others, and self-advocacy inclusive of helping others understand a symptom or requesting specific support. When probed about sharing with a home care provider, personal support worker, or other paid caregiver, 97.5% (n = 39) of survey respondents reported not having this kind of support person within their circle of care. Within 2 weeks of receiving their feedback report, three survey respondents (all living with NDD) had shared the report with a healthcare provider, with one respondent scheduling an appointment specifically to discuss the report. Of those who did
Although feedback regarding the report was generally positive, a range of profiles did emerge across participants from those who responded favorably across all dimensions (i.e. readability, volume, helpfulness, behavior change and information sharing) to those who reported mixed responses in terms of interest, uptake or utility. For example, one participant living with CVD indicated they found the report very easy to read or understand, and very helpful for understanding their daily health-related behaviors. This participant also made several changes to their behavior because of the feedback report, including increasing their daily activity levels by going up and down the stairs and starting to use a cane to increase balance confidence and facilitate more walking outdoors. This participant also shared the information with several people within their circle of care including a friend and family member, home care provider and multiple healthcare providers. When probed about motivation for sharing with a home care provider, this participant said, “I had things that I needed to discuss with them and couldn’t always remember…[the report] gave more clarity for what I wanted to talk about and what I was asking for” and suggested that “[the] home care provider shared this information with her boss to show where I needed more support in particular areas.” When probed about motivation for sharing with multiple healthcare providers, they said, “mainly because my memory and my ability to put words to what I want to talk about…the report helped keep my focus in the areas that I wanted to talk about” and “the report helped me get a referral to an occupational therapist…bringing the report to my occupational therapist will help her better understand where to start off.” A control group participant indicated they found the report somewhat easy to read or understand and very helpful for understanding their daily health-related behaviors. They made changes to their lifestyle including a commitment to walking four times per week and commented on an increased awareness of the need to break up long sedentary periods with movement. When probed about motivation for sharing with a friend or family member, they said, “[I shared the report] to invite [my] neighbour to help me increase my walking.” In contrast, another control participant indicated that they found the report somewhat easy to read or understand, somewhat helpful for understanding their daily health-related behaviors and reported that although they thought about it, they did not take steps towards making any behavior or lifestyle changes. This participant did not share the report with anyone in their circle of care, and when asked why they did not share, they said “[I] did not feel the need to share the report” with a friend or family member. In terms of reasons for not sharing with a medical practitioner, they said “[I] haven’t spoken to [my] doctor in 2 years due to COVID but would likely share at my next physical.”
Discussion
This study describes the co-design, development and evaluation of an approach to sharing information from wearables for the purpose of improving self-awareness and self-management of health-related behaviors. Feedback included a personalized report delivered in a supportive environment following a week-long period of multi- sensor wear and application of custom analytics to the raw sensor data. Evaluation of the approach suggested the feedback was well-received by participants and resulted in enhanced self-awareness and self-management of health, with most participants reporting increased understanding of health-related behaviors, engagement in purposeful actions to change behaviors and/or sharing (or intention to share) information with others in their circles of care.
Critical to the development of the report was ensuring its acceptability and useability. As a group, most respondents found their report easy to read or understand, with an appropriate volume of information, and helpful for understanding daily health-related behaviors and/or making lifestyle changes. It should be noted that embedded supports (1:1 consultation and video supplement) may have influenced participant responses; however, evaluating the impact of each of these features was beyond the scope of this study. Some differences did emerge when participants were stratified by cohort or when subgroups of participants were extracted to examine outcomes with respect to cognitive status. A slightly higher proportion of participants living with an NDD rated the feedback report as helpful compared to control participants, and a larger proportion of respondents in the group with higher cognition scores reported making a behavior or lifestyle change compared to those with lower scores. Lower cognitive ability, however, did not appear to impact participant ratings regarding ease of reading and helpfulness or volume of information, suggesting that reports were accessible to all participants. However, it should be noted that the COGlow group was small, with only seven respondents. Examining individual participants revealed variability in reported uptake (behavior changes and information sharing), even within the same cohort. For example, two participants from the control cohort both indicated that the report was helpful and yet, differed in their intent to make lifestyle changes and interest in sharing with others in their circles of care. This further emphasizes the importance of developing specific methods of feedback reporting to consider the unique needs and abilities of recipients. That said, participants’ largely positive responses to personalized feedback suggest that the process used to develop the content and methods for reporting should be considered a template for translating wearables data into meaningful and actionable outcomes. The current study examined short-term changes, or intention to change behavior, to investigate the potential for impact. There is need, however, to examine the capacity for wearables-derived feedback to influence longer-term changes in health-related behavior. Such work requires examining behavior change in the context of established models (e.g. transtheoretical model of behavior change). Previous work suggests that maintenance of a behavior change (e.g. engagement in a physical activity program) declines over time, 59 even following interventions that employ multiple behavioral change strategies and span several months (e.g. >6 months). 60 Wearables-derived feedback may provide a solution to promote long-term behavior change, and future work should aim to understand the impact of feedback on maintenance of behavior when it is delivered longitudinally, at time points when people are most susceptible to reverting their behavior.
Our general approach and the questions used to guide development of the report were grounded in a desire to address the “what and how” of information sharing for outcomes derived from wearable devices, with particular emphasis on: (a) the types of information to include for the specific purpose of improving self-awareness and self-management of health-related behaviors (the “what”); and (b) the method of delivery for older adults and persons living with NDD (the “how”). To this end, the goals of the report (self-awareness, self-management) and the specific needs and perceptions of the target groups were defined early, with intention, and were central to the development process, which included ongoing engagement of persons with lived experience. It should be acknowledged that the convenience sample (first 40 HANDDS-ONT participants) may have biased survey responses to include those from older adults and persons living with NDD who were keenly interested in receiving feedback, given that HANDDS-ONT study recruitment materials noted the inclusion of a personalized health and activity report. Further, although some differences emerged when respondents were stratified, these differences may have been greater, and the ability to interpret and use information may have been impacted if the sample included participants with a larger range of cognitive abilities and physical capacities and/or larger sub-group samples. Finally, since all participants received a 1:1 consultation and video supplement to aid in interpreting their data, it is possible that views on readability, helpfulness and volume of information would have been less favorable if the reports were delivered without these supports.
In addition to examining individual behavior change, the present study examined the potential for impact by probing the extent to which survey respondents shared or planned to share their data with others. Most respondents indicated sharing their report with a family member or friend and several shared or intended to share with a healthcare provider. It is important to note that the 2-week timeline for gathering participant responses specifically impacted the extent to which sharing occurred with healthcare providers, with participants reporting an intention or plan to share at their next scheduled visit (outside of the study period). Some participants did not deem the information contained within the report important for sharing with a healthcare provider. When probed further, these responses appeared to be driven by “good performance” that was viewed as unnecessary for follow-up or the feeling that their healthcare provider was already aware of the types of findings contained within their report. The former raises the matter of ensuring that outcomes perceived to be positive by end users are consistent with the data and its link to risk, while the latter emphasizes the need to: (a) consider the target audience when making decisions about what information or measures to report; and (b) ensure that potential relevance of the information for clinical decision making is understood by the recipient. Other participants identified real or perceived barriers to sharing with a medical practitioner, including limited access or appointment time constraints, which may further support the need to have clinical stakeholders embedded in the feedback development process when working towards creating information sharing pipelines that are effectively and sustainably integrated into health systems/clinical care. The reasons for sharing, which included a desire to engage partners in making lifestyle changes and educating/advocating for others and self, suggest that information sharing about modifiable health-related behaviors has the potential to empower recipients and to positively impact awareness or behavior of others.
As noted, two important and pragmatic aspects of the development and evaluation process detailed within this paper include the availability of appropriate guidelines to anchor feedback and use of validated measures suitable for the context (e.g. clinical versus free-living behavior) and populations of interest. Guidelines have been established surrounding activity and sleep that have been linked to reduced chronic disease risk and preserved mobility and health in older adults.32,61 However, continued work is needed to establish disease-specific guidelines between free-living behaviors and health outcomes in persons living with CVD or NDD that account for specific symptoms and comorbidities. In addition to evidence-informed, disease-specific guidelines, it is also imperative that appropriate analytic methods are used to ensure validity of reported outcomes. For example, despite the ubiquitous use of wrist worn accelerometers to derive physical activity intensity estimates in free-living settings, 62 this method may be unreliable in older adults or persons living with NDD due to age- and impairment-related changes in movement. 36 Instead, an approach that uses multiple body-worn sensors to resolve uncertainty, 43 as well as age- or disease-appropriate thresholds for activity intensity classification,63–65 can help to improve real-world activity estimates in these groups. Even where known features of a particular cohort (e.g. muted arm swing in PD) are accounted for, variability across individuals poses an additional challenge that requires person-level consideration of wearables-derived data. Within the current approach, this is addressed using case-based review of data prior to report generation as a quality assurance process.
Finally, there is critical need to address scale and sustainability of methods for providing feedback that accommodate the heterogeneity that exists in older adults and persons living with CVD or NDD (including diversity of background, education, data literacy and experience) and which considers the critical elements needed for successful patient and clinical implementation (i.e. end user education and uptake, data flow and health system integration that ensures privacy, respects patient autonomy, etc.). In this study, methods included 1:1 consultation with trained personnel, which has previously demonstrated success with respect to uptake of information 22 and behavior change 66 but which is time and resource intensive. Consideration should be given to alternate approaches that meet the need for assistance with data interpretation while reducing demands, with sensitivity to the possibility that information contained within the report may cause an individual distress or that a delivery model that provides insufficient interpretive support could lead recipients to make decisions that pose a risk to their health or well-being. Such alternatives may include triaging individuals based on level of support required as determined by relevant health/clinical data, with additional methods including providing professional support on an as-needed basis, leveraging care partner support, and automating where possible, without compromising privacy, clarity of the information or access to resources. The report presented here has been designed to include layers of information and modules that can be inserted or removed as well as a video supplement as methods for managing scope, scale and sustainability.
As advances in this space continue, focus should remain on ensuring end-user needs are met by grounding work in co-design frameworks and processes. In the context of a knowledge-to-action (KTA) framework, the current study addressed several KTA phases including identifying the problem of being able to derive meaning from wearables data and considering barriers and facilitators to adapt traditional metrics and implementation processes for the purpose of positively impacting behavior change in our cohorts of interest.
67
In addition, where the goal of reporting is the use of wearables’ data for specific
Conclusion
This study provides a person-centered framework and template for reporting wearables-derived information that considers the needs and perspectives of older adults and persons living with NDD. The reporting system serves as a viable foundation to improve the utility of wearables-derived information in older adults and individuals with complex health conditions, including those with cognitive impairment, and has been built to accommodate continued development. A key feature of the approach is the use of raw device data, which permits tailored analytics to ensure relevant and accurate outputs for different cohorts and use cases. Continued work within our research group will result in a publicly available data processing pipeline for raw data processing and analysis by individuals with limited programming expertise. Continued analyses of data from HANDDS-ONT will enable us to better understand the needs of individuals and to describe the acceptability and utility of the report more richly by assessing responses from participants with a broader range of capabilities and grounding them in a published model of health behavior change. Future work will investigate methods for scaling the approach in a manner that preserves a commitment to personalization, as well as methods for implementing and evaluating feedback longitudinally to support behavior change.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231179031 - Supplemental material for User-centered design of feedback regarding health-related behaviors derived from wearables: An approach targeting older adults and persons living with neurodegenerative disease
Supplemental material, sj-docx-1-dhj-10.1177_20552076231179031 for User-centered design of feedback regarding health-related behaviors derived from wearables: An approach targeting older adults and persons living with neurodegenerative disease by Karen Van Ooteghem, F Elizabeth Godkin, Vanessa Thai, Kit B Beyer, Benjamin F Cornish, Kyle S Weber, Hannah Bernstein, Soha O Kheiri, Richard H Swartz, Brian Tan, William E McIlroy and Angela C Roberts in DIGITAL HEALTH
Footnotes
Acknowledgements
The authors wish to thank ReMiNDD and HANDDS-ONT participants and study partners, as well as the Ontario Neurodegenerative Disease Research Initiative (ONDRI) patient and community advisory committee (PCAC) members, ONDRI investigators, ONDRI research coordinators and the ONDRI project management team for their input and administrative support of this project.
Contributorship
KVO, ACR, RHS and WEM conceived the study and were involved in protocol development and gaining ethics approval. KVO, FEG, BT, KBB, ACR and WEM developed the data analytics plan and assisted with data interpretation. VT, FEG, KVO and ACR developed feedback delivery supports and conducted literature review, data collection and data analysis. KBB, BFC and KSW led algorithm and analytics pipeline development and data processing and review for outputs included in feedback reporting. HB, SOK and KBB were involved in development of pipeline for feedback report generation. KVO, FEG and ACR wrote the first draft of the manuscript. All authors critically reviewed and edited the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The human research ethics committee of Sunnybrook Research Institute, Toronto, ON, Canada, approved both the ReMiNDD (REB approval: 007-2019) and HANDDS-ONT (REB approval: 2021-1517) studies, the latter which included the study protocol described within this manuscript.
Guarantor
KVO.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ontario Brain Institute, an independent non-profit corporation, funded partially by the Ontario government. The opinions, results, and conclusions are those of the authors and no endorsement by the Ontario Brain Institute is intended or should be inferred.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
