Abstract
Objectives
This study aims to develop a deep learning-based classification model for early detection of dementia using a wearable device that can measure the electrical conductivity of the skin, temperature, and movement as factors related to dementia, interlocking them with an application, and analyzing the collected data.
Methods
This study was conducted on 18 elderly individuals (5 males, 13 females) aged 65 years or older who consented to the study. The Korean Mini-Mental State Examination survey for cognitive function tests was conducted by well-trained researchers. The subjects were first grouped into high- or low-risk group for dementia based on their Korean Mini-Mental State Examination score. Data obtained by wearable devices of each subject were then used for the classification of the high- and low-risk groups of dementia through a smart healthcare-based system implementing a deep neural network with scaled principal component analysis. The correlation coefficients between the Korean Mini-Mental State Examination score and the featured data were also investigated.
Results
Our study showed that the proposed system using a deep neural network with scaled principal component analysis was effective in detecting individuals at high risk for dementia with up to 99% accuracy and which performance was better compared with commonly used classification algorithms. In addition, it was found that the electrical conductivity of skin had the closest correlation with the results of the Korean Mini-Mental State Examination score among data collected through wearable devices in this study.
Conclusions
Our proposed system can contribute to effective early detection of dementia for the elderly, using a non-invasive and easy-to-wear wearable device and classification algorithms with a simple cognitive function test. In the future, we intend to have more subjects participate in the experiment, to include more relevant variables in the wearable device, and to analyze the effectiveness of the smart healthcare-based dementia classification system over the long term.
Introduction
The recent development of digital technology and wearable devices has made primary prevention and management easier with respect to the improvement of personal health, especially the provision of individually personalized health services through the analysis of information collected in daily life. 1 As the paradigm of medical service has shifted from disease treatment to health and wellness through prevention and management, the scope of healthcare services has expanded. Hood, L. and Flores, M. suggested 4 Ps: predictive, preventive, personalized, and participatory expansion as future medical keywords, 2 suggesting that the importance of self-directed health care will continue to increase along with the demand for technology that combines information technology (IT) and Bio-Technology. Smart healthcare (or digital healthcare) is an industrial field that deals with personal health and medical information, devices, systems, and platforms, representing comprehensive medical service that combines health-related services and medical IT.3–5 Mobile health (m-Health) continues to attract attention in the field of smart healthcare by improving treatment efficiency and reducing medical costs. 6 Mobile health refers to medical services and health information provided through information and communication technology and mobile devices such as smartphones, smart wearable devices, and personal digital assistants. 7 Such mobile-health and smart-health care systems are representative of a rapidly growing medical-industry field whose strength is being able to help patients manage their own health even after discharge and to accumulate and monitor data of normal people or people at the disease borderline.
Dementia, rapidly increasing as society ages, is difficult to cure once its symptoms progress, 8 so early detection of its symptoms, slowing its progression, and attention to safe and continuous care in the daily life of patients are very important. According to data from the National Institutes of Health in the United States, the number of dementia patients is rapidly increasing worldwide, and this disease has a terrible ripple effect than diseases such as cancer, emphasizing the urgent need to prepare countermeasures for dementia management.8,9 In Korea, a new dementia patient occurs on average once every 12 min, and caregivers spend about 20 million won a year caring for them. 10 Dementia patients are responsible for problems in daily life, work, and interpersonal relationships due mainly to symptoms of imperfect memory, thinking, judgmental ability and physical function deterioration, speech disorders, and personality changes. 11 If there is only mild cognitive impairment, a person may still be able to lead an independent life and exhibit relatively intact judgment. 12 Previous studies suggest that if dementia is detected and managed at an early stage, symptoms can be improved in about 35%–50% of patients and their daily-life functions can be maintained to some extent. 13
This results in a great deal of attention focused on smart healthcare technology that can detect and manage dementia patients at an early stage, and many related studies are being conducted, with wearable smart devices the fastest-growing industry, especially in the healthcare field. 14 An example of a wearable device developed in Korea is the Y-Band. This device, aimed at the treatment and management of dementia, and is an electroencephalography sensor that analyzes a patient's collected brain signals and automatically applies current stimulation to the forehead to control the activation of specific brain regions. 15 In addition to developing applications for caring for dementia patients and providing their locations, applications are being developed for identifying and monitoring meals and treatment status for the elderly. 16 There is a previous study on the user experience in establishing a health care application strategy to form an exercise habit, 17 and also a study on a smart medically customized health care service in which an EMR system is linked to a tertiary hospital. 18 A study was also conducted that collected and analyzed users’ physiological data from a chronic disease group and a dementia patient group using smart wearable devices, providing notification of needed care when the result value exceeded a threshold. 19 However, research related to application-based wearable smart devices focused on the prevention and early detection of dementia patients is sparse.
Internet of things (IoT)-based artificial intelligence (AI)-related research is being used in various fields. Prediction and classification models using deep learning and machine learning approaches are also being actively used in various domains.20–24 Li et al. 20 conducted a study to accurately identify real alerts for network attacks by combining machine learning and deep learning. Gaurav et al. 23 suggested a hybrid combination of existing health care methods and IoT technology for effective health management in the digital era. Based on such development of IoT-based AI technologies, many studies25–28 using machine learning and deep learning methods have been conducted for predicting diseases. However, research on data collection and detection of diseases through wearable devices and applications are in their early stage and are as of now insufficient to apply to the early detection and classification of patients’ diseases, especially for dementia.
Based on the evaluated cognitive function of the elderly and their health data collected via wearable devices and applications, individuals at a high- or low-risk for dementia are required to be classified early with the help of an AI-based model. Therefore, this study aims to develop a classification model for early detection of dementia using a wearable device that can measure the electrical conductivity of the skin, temperature, heart rate, and movement as factors related to dementia, interlocking them with an application, and developing different prediction models by analyzing the collected data. The specific purposes of this study are as follows:
Analyze the status of data collected from the Korean Mini-Mental State Examination (K-MMSE) related to cognitive function and wearable devices in situ. Determine and analyze correlation between the K-MMSE scores and related variables collected from wearable devices. Suggest and evaluate a deep learning model for the classification of the elderly at risk for dementia.
Methods
Participants
This study was conducted on 18 elderly people (5 males, 13 females) aged 65 years or older who consented to the study. The experiment using a wearable device was conducted from November 1, 2020 to February 28, 2021, targeting 18 elderly people of ages greater than 65 and living in Busan and Ulsan, South Korea. The K-MMSE survey for cognitive function tests was conducted by well-trained researchers. The criteria for participant selection were:
Those 65 years of age or older and capable of communication. Those with K-MMSE scores of 20 or higher. Those understanding the procedure and purpose of the study and who have signed the consent form. Those having the capability to perform activities of daily living.
Ethics
After being informed about the purpose and methods of the study, all study subjects and their guardians voluntarily agreed to participate. This study was approved by the University Institutional Review Board (YSUIRB-202004-HR-064-2).
Data preparation
For the development of a deep-learning model for the classification of a high-risk for dementia, data should be obtained and analyzed. The system used in this study measures data through a wearable device with three integrated sensors providing data into a data logger. Accumulated data is then analyzed in the system. Figure 1 shows the used system comprising measurement, data log, and data analysis for the early detection of dementia.

Measurement, data log, and data analysis for the early detection of dementia.
Measurement of cognitive function of the elderly and their bio-signals via sensors of the wearable device in situ has been conducted and logged into a data storage system.
Korean Mini-Mental State Examination
The most commonly-used K-MMSE was used to determine a correlation between cognitive function and data collected from the wearable devices. The Mini-Mental State Examination (MMSE) is the most commonly-used cognitive function screening test in the community and clinical setting,29,30 and its reliability and validity have been proven through many previous studies. The K-MMSE, developed and used in Korea,31,32 has questions that are time-oriented (5 points), place-oriented (5 points), memory registration (3 points), attention and calculation (5 points), memory recall (3 points), and language and spatiotemporal composition ability (9 points). 31 Although K-MMSE has the advantage of being able to screen relatively easily, additional tests such as neuropsychological tests are required for an accurate diagnosis of dementia. However, in this study, as it was used to understand the relationship between cognitive function and risk factors in normal subjects, it is considered that it can be used as an indicator of overall cognitive function. The total maximum score is 30, and a score less than 24 is judged as suspected dementia in Korea. 17 In this study, the subjects were grouped based on 25 points based on the participants’ K-MMSE score distribution. We classified dementia risk into high-risk and low-risk groups based on the K-MMSE scores, with a score less than 25 classified as representing a high-risk-for-dementia group and other scores classified as representing a low-risk-for-dementia group.
Sensor-based data
The three sensors of the smart wearable device were an electrodermal activity (EDA) sensor to measure the electrical conductivity of the skin, an accelerometer (Acc X, Y, Z) sensor to measure wrist movement, and a temperature sensor. A wearable device that implements a galvanic skin response (GSR) sensor and a photoplethysmogram sensor for measuring bio-signals was used. For dementia risk factors collected through smart wearable devices, refer to the previous studies.33–37 Participants were asked to wear the developed smart wearable device on their wrists for a week, and the data collected through the application were analyzed.
Data analysis
The collected data is time-series data in various forms, and in order to analyze it, a data preprocessing algorithm was applied to obtain primary feature values for converting time-series data into feature variables. In order to use the variables obtained through the preprocessing algorithm more efficiently, principal component analysis (PCA) was applied to transform them into principal component variables, and reduced dimensions to obtain optimized secondary feature values and converted them into input variables for classification.
Classification method
Many studies have used deep-learning methods to solve a variety of problems. We used a Deep Neural Network (DNN) model with scaled PCA to extract features from the time-series data and identify factors associated with dementia.
PCA is a commonly used method to extract meaningful information from the raw data by converting it into new coordinate systems. 38 It generally reduces the dimensions of the initial raw data and reveals optimal hidden features of the data. It can generate inputs for its following classification algorithms.39,40 In this paper, diverse scalers, such as a min-max scaler, a quantile scaler, and a standard scaler, were selected and applied to the combinations of principal components for improving the performance of the suggested method. As the back-end classification algorithm, a DNN model38,40 was selected and the classification results were compared with those of Random Forest, 41 AdaBoost Classifier, 42 Gaussian Naïve Bayes, 43 Multi-Layer Perceptron Classifier, 44 and Support Vector Classifier. 45 The proposed classifier was implemented on Python Keras 46 and Tensor Flow. 47
Figure 2 illustrates the proposed overall process of the DNN with scaled PCA. The details of the process are as follows: (a) 32 input feature variables were extracted from 18 subjects, and the resulting dataset was divided into training (66%) and testing sets (34%); in the training process, 30% of the training sample was used for validation; (b) as a preprocessing step, PCA and a scaler were applied to convert diverse variables into feature variables, and models of the PCA and scaler were generated for testing; (c) we trained the DNN model using the scaled PCA variables; and (d) compared the predicted results with ground truth data provided by clinicians. The testing data were separated from the training data for testing.
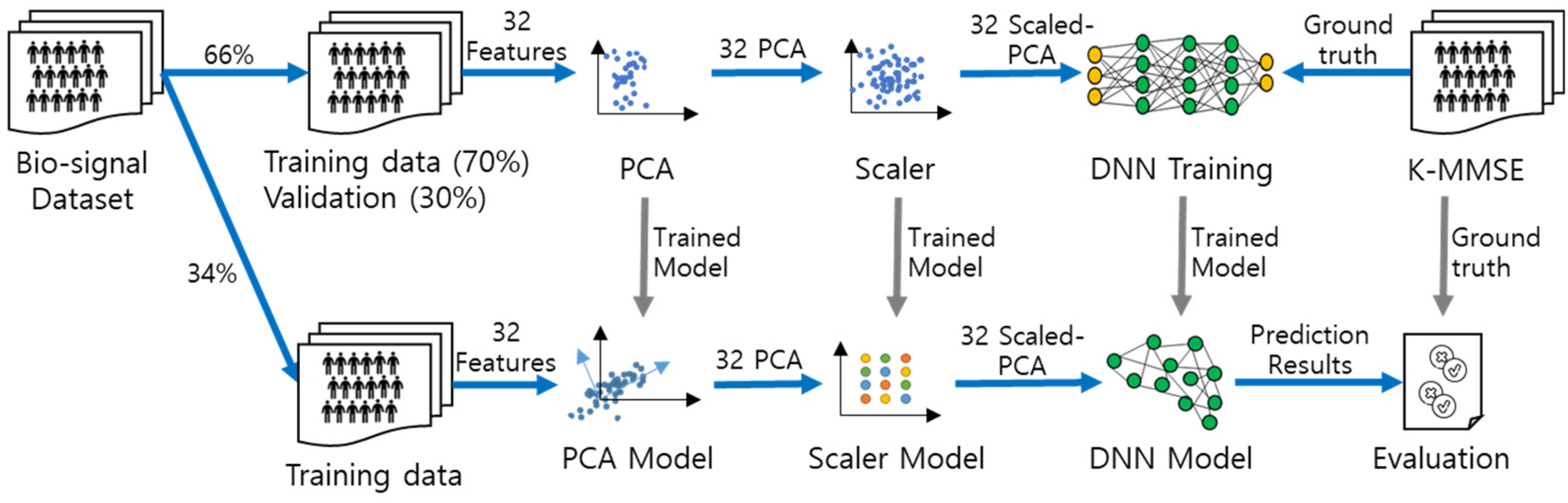
The overall process of the deep neural network with scaled principal component analysis.
Statistical analysis
Statistical assessment of the performance of a binary classification test, especially for an imbalanced dataset, requires two general performance metrics, sensitivity (Sn) and specificity (Sp). If the dataset is balanced, accuracy (ACC) alone can reflect the performance of the model, but since our dataset was evidently imbalanced, we used three additional metrics: Sn, Sp, and positive predictive value (PPV). Sn reflects the probability of detecting a high-risk-for-dementia group, Sp indicates the probability of detecting a low-risk-for-dementia group, and PPV is the probability that scores status was correctly classified. We used four status parameters: true positive (TP), true negative (TN), false positive (FP), and false negative (FN), to generate a confusion matrix. TP is the number of data points correctly identified as a high-risk-for-dementia group and the TN represents those correctly detected as a low-risk-for-dementia group. FP and FN are the data incorrectly detected as a high-risk-for-dementia group and a low-risk-for-dementia group, respectively. Sn, Sp, PPV, and ACC can be calculated as follows:
Results
Korean Mini-Mental State Examination
We first demonstrate the result of the evaluated cognitive function of the elderly. After each participant took the K-MMSE test, well-trained researchers analyzed the results. Table 1 shows the number of participants in a low- or high-risk group for dementia. There were 14 participants in a low-risk group (scores equal to 25 or more) and 4 individuals in a high-risk group (scores less than 25).
The number of participants in low- or high-risk groups for dementia.

Sensor-based data
We then show the results of the bio-signals measured and the processed data. Figure 3 shows an example of the measured physiological signals of a participant. The measured bio-signals were the electric conductivity of the skin, the movement, and the temperature of participants. By analyzing the basic time-series data, the initial feature data was produced as shown in Figure 4. The EDR data was separated by the vertical amplitude (OD) and the horizontal duration (OM). Each feature value was converted into basic statistical values (sum, mean, standard deviation, max, min, median, skew, and kurtosis) in 30-min increments and used as deep-learning data input. In particular, as local maximal and minimal values for EDA data are important, peak and valley values were stored together with time values.

Physiological signals from the wearable device (time series data and initial feature data received from the sensor). EDA: electrodermal activity; Acc: accelerometer.

Preprocessed data from physiological signals. (OD: The values of the horizontal duration.).
Classification
We demonstrate the classification results of the proposed algorithm, which is a DNN with scaled PCA including a min-max scaler, a quantile scaler, and a standard scaler. The scaled PCA was used to extract features from the time-series data and identify factors associated with dementia. The proposed DNN model comprises 180 nodes in each layer with a depth of four layers. A 30% dropout was applied to avoid overfitting.
As performance metrics of the classification algorithm, Sn, Sp, PPV, and ACC were considered. As the ACC cannot explain the measure for sparse datasets of two groups of high- and low-risk for dementia, the area under the receiver operating characteristic curve (AUC) was also applied. The AUC can be used for evaluating overall performance using a performance metric without any thresholds of the calculated probabilities from the classification algorithms. 38 The equal error rate value was also checked to confirm the overall error value.
Table 2 shows the performance characteristics of three scalers using a DNN algorithm for classification. The overall result indicates a high AUC value of 0.99 or more. When the AUC is closer to 1, the model is better. When Quantile scaled PCA was used, the overall sensitivity and specificity values were high. The Standard scaled PCA exhibited a relatively low value. In conclusion, the combination of DNN and Quantile scaled PCA achieved the best result with the AUC value of 0.997.
Confusion matrix and comparative performance of DNN with diversely scaled PCA (min-max scaler; quantile scaler; standard scaler).

DNN: deep neural network; PCA: principal component analysis; PPV: positive predictive value; Sn: sensitivity; Sp: specificity; TP: true positive; TN: true negative; FP: false positive; FN: false negative; AUC: area under the receiver operating characteristic curve; EER: equal error rate.
To compare and analyze the performance of the proposed model, it was applied to commonly-used deep-learning algorithms, which are Random Forest, AdaBoost Classifier, Gaussian Naïve Bayes, Multi-Layer Perceptron Classifier, and Support Vector Classifier, and the results were compared. Figure 5 shows the comparison results of the ROC curve of each algorithm with different scaled PCA. If a ROC curve of a model is closer to the left upper side of the plot, the model is better. 25 It means that a model is better as the AUC higher. Compared to the results of other algorithms, the one of DNN is significantly better for all scalers used.
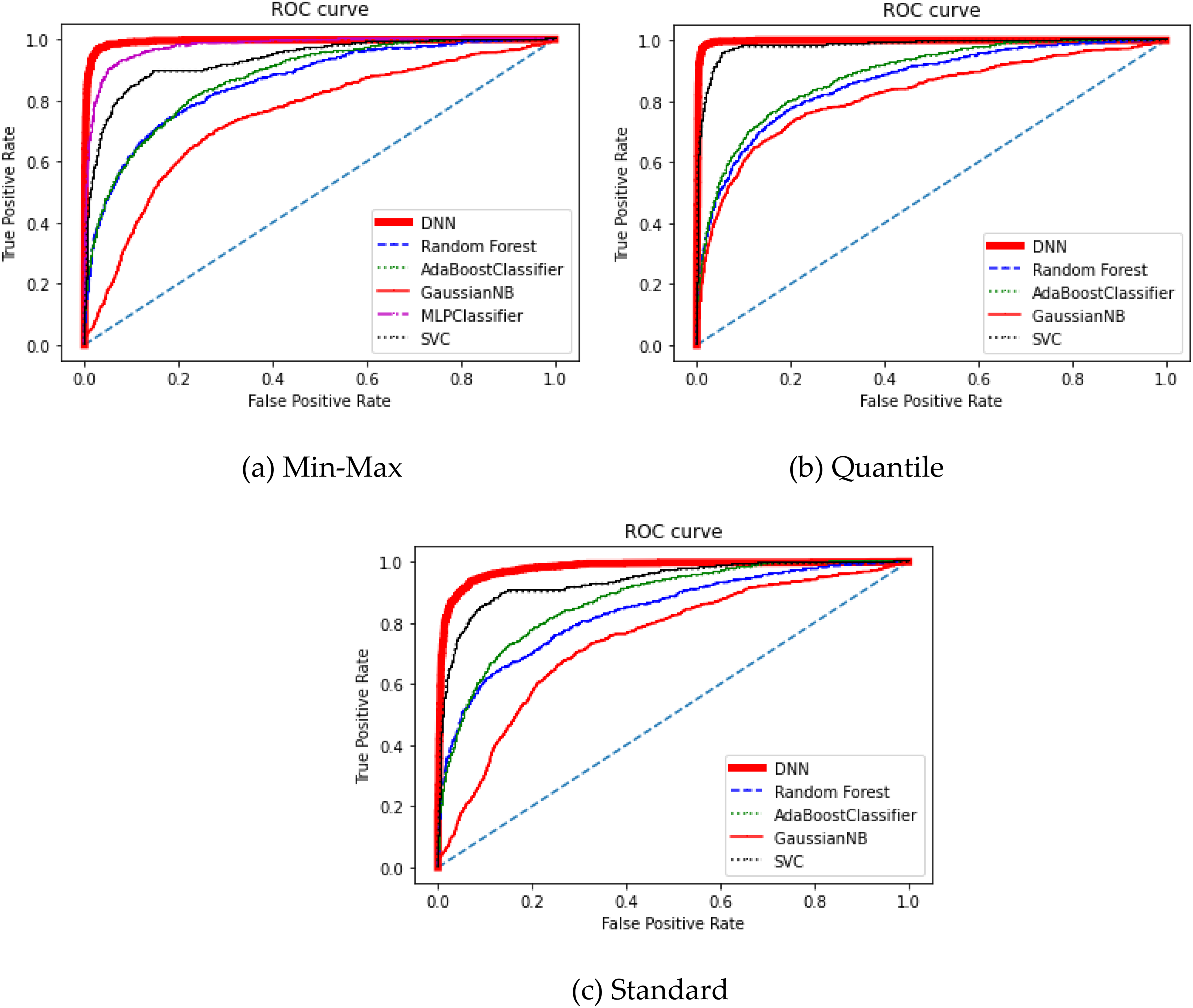
ROC curves of classifiers. (a) Min-max, (b) quantile, (c) standard.
Table 3 summarizes the comparison of specific performance indicators of AdaBoost Classifier, Gaussian Naive Bayes (NB), and support vector classifier (SVC) with differently scaled PCA. For algorithms except for the DNN, the SVC with Quantile scaler showed the best results regarding the AUC. However, the DNN with Quantile scaler still achieved the most significant results among others.
Confusion matrix and comparative performance of selected classifiers with diversely scaled PCA.

PPV: positive predictive value; PCA: principal component analysis; Sn: sensitivity; Sp: specificity; ACC: accuracy; AUC, area under the receiver operating characteristic curve.
Correlation
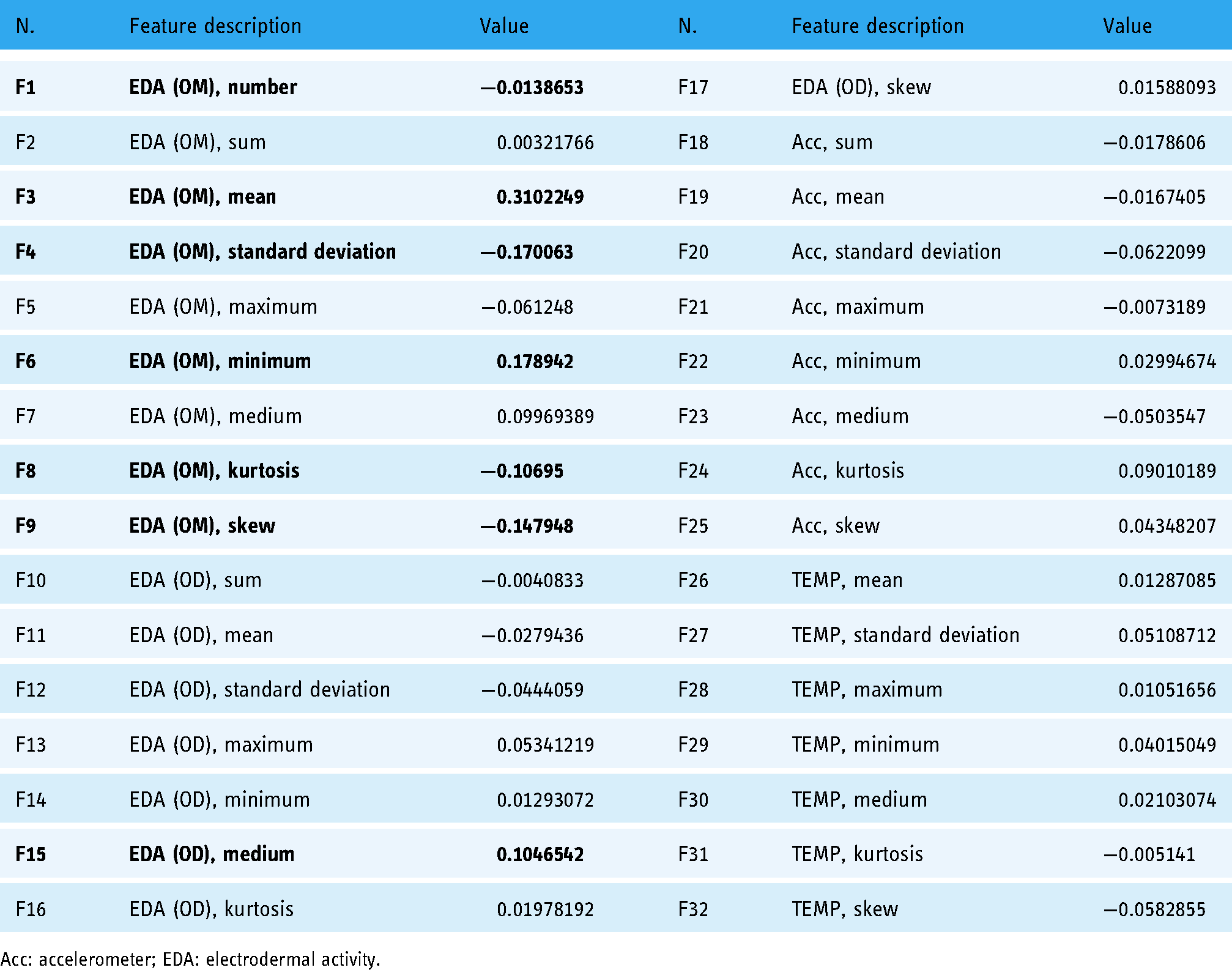
The features and the trained DNN have well classified the high- and low-risk for dementia with the AUC of 0.99, but have not described the detailed relationships between the measured variables and the K-MMSE diagnosis for dementia. It means that the results did not clearly exhibit relationships between principal components and dementia occurrence. Therefore, we then investigated the correlation coefficients (r-values) between each measured 32 features and the K-MMSE result. Table 4 lists the correlation coefficients of the 32 variables investigated. Even though the results show that there are weak correlations between the 32 features and the K-MMSE survey, the strongest correlation was obtained from the mean of EDA Sensor vertical amplitude (OM), as the value of +0.31. It is highlighted in bold and other strong results (over ± 0.10) are also shown in bold in the OM and OD values of the EDA sensor. This correlation result still seems less meaningful; we will discuss it in the “Discussion” section.
Correlation coefficients of the 32 features and the risk group for dementia (OM: vertical amplitude, OD: horizontal duration).
Acc: accelerometer; EDA: electrodermal activity.
Discussion
Future health care will be transformed into a paradigm of prediction and prevention, and early diagnosis and early treatment will become the core of health management. Within this trend, smart health and health-related big data are expected to play a key role in driving future health management paradigm, with utility values in the field of chronic diseases such as dementia, hypertension, and diabetes being particularly noteworthy. This study presented a smart healthcare-based system with a novel feature extraction system for monitoring and determining dementia risk in the elderly using classification algorithms and preprocessing methods such as PCA and various scalers.
As a primary dementia screening tool, K-MMSE has widely been used in Korea. This tool could detect and quantitatively evaluate cognitive impairment. 48 Kim et al. 40 used the K-MMSE scores and the physical activity of the elderly to detect and predict dementia by using the combinations of eight classifiers and preprocessing methods. They also found correlations between dementia risk and physical activity via an IoT-based monitoring system. In our study, we collected the K-MMSE scores and bio-signals of the elderly to classify individuals at high or low risk for dementia and analyzed correlations between K-MMSE scores and related variables collected from wearable devices.
As a result of analyzing the correlation between the results of K-MMSE, that provides a simple look at cognitive function, and data collected through wearable devices, it was found that the electrical conductivity of the skin exhibited the closest correlation. This basically means that the skin conductivity is increased when the skin sweats, and this increase can be used as a useful indicator of irritation or stress. 49 These results are consistent with the results of previous studies suggesting that psychological stress is a mechanism leading to neurological and cognitive changes that can eventually result in dementia. 50 Over time, the brain loses its capability for regulating levels of hormones, including stress hormones, 51 so self-monitoring as a dementia risk factor is necessary.
Healey and Picard 52 identified relationships between stress and GSR (a measure of skin conductance level, the susceptibility of the skin to conducting electricity) features such as magnitude and duration. The values of the OM and OD, as shown in Figure 6, were considered to be the most important stress-related characteristics related.

Three vertical magnitudes (OM) and horizontal duration (OD) features of the galvanic skin response (GSR) signal (Healey and Picard 52 ).
Similarly, the OM of the electrical conductivity of the skin has the highest correlation with risk-for-dementia in our study. The results of this study suggested that the skin electrical conductivity related to stress was most relevant, but if it is expanded to more research subjects in the future, a clearer classification will be possible if there are two sensors (Acc, GSR) of the wearable device.
The subjects of this study are normal people who have not been diagnosed with dementia and can communicate with a K-MMSE score of 20 or higher. It is useful as a study to determine whether it can be used as a monitoring tool for early diagnosis of dementia through the classification ACC of dementia risk group and normal group through the correlation between cognitive function and dementia related risk factors that can be collected from wearable devices.
Among 18 participants, four participants were diagnosed with high risk for dementia, while others were grouped with low risk for dementia by K-MMSE diagnosis. As bio-signals, we measured the electrical conductivity of the skin, the movement, and the temperature of the elderly. By using these data, we tested combinations of DNN with diversely scaled PCA. The used scalers were a min-max scaler, a quantile scaler, and a standard scaler. Among the combinations of the algorithms, the performance of DNN with scaled PCA with a quantile scaler was the best in terms of AUC. According to Chen et al., 25 the sensitivity rather than ACC is more highlighted in medical data. They explained that the higher the sensitivity rate, the lower the probability that a patient who will have the risk of disease is predicted to have no disease risk. In our study, both sensitivity and ACC of the proposed DNN with scaled PCA has higher than those of other algorithms. The sensitivity was also higher than ACC for all scalers. Previous studies on predictive algorithms for healthcare system construction also have mentioned high ACC in DNN.53–56
In addition, the proposed DNN with a quantile scaler and SVC with a quantile scaler outperformed other combinations. As the SVC is generally used for binary classification, it might show better performance than others except for DNN. The error rate can be managed by mapping the features into higher dimensional space in the SVC. 54 Ayon et al. 57 also indicated that the DNN and the SVC showed better performance in the classification of heart disease. Among the algorithms of classifiers, the algorithms using a quantile scaler showed better performance compared with those of other scalers. Abdulraheem and Ibraheem 58 explained that a quantile function is the best normalization function for imbalanced data. It means that this scaler can be used for imbalanced health data with high performance. Our study seems to fit this analysis.
While medical services in the smart health environment domestic and abroad have not yet been spread due to various regulations, 6 the frequency of use in more diverse disease groups is expected to increase in the future both due to changes in the social environment and consumer demand. Although there have been studies and developments using Apple Health, Google Fit, and Samsung Health as specialized applications for daily health management, this requires discussion of the efficiency and effectiveness of interworking services and services between smart devices and applications. 59 The biggest advantage of smart wearable devices is that all data is collected naturally in everyday life such that users are unaware aware of their presence. The key to active health management and preventive management is to provide analysis through user health information as meaningful data so that users can receive medical services early and help them develop healthy lifestyles. However, since most of the presently existing smart health care research for dementia patients is related to smart home systems or care support for dementia patients, 8 research on smart health systems for early detection and prevention of dementia should be expanded in the future.
This study has some limitations as follows: (a) As a result of experimenting with individuals within the elderly population over 65 years of age for one week, the sample size was small due to the nature of the experiment, and (b) more diverse clinical information related to dementia could not be reflected in the variables. Nevertheless, by suggesting a classification model for dementia patients using smart wearable devices, it can be used as a basic foundation for future research or policy decisions.
Conclusions
This study proposes a new smart healthcare system that provides dementia-classification services based on deep-learning analysis of collected data through smart wearable devices. Despite limited results due to the small sample size and the limitations of clinical factor-related variables, the proposed system (a) provides accurate detection and classification functions and (b) is a non-invasive and easy-to-wear device by the elderly, so it is considered useful as a dementia risk monitoring tool. In the future, we intend to have more subjects participate in the experiment, include more relevant variables in the wearable device, and analyze the effectiveness of the smart healthcare-based dementia classification system over the long term.
Footnotes
Acknowledgments
The author would like to thank Drs Jungyoon Kim and Songhee Cheon for their assistance and guidance in this research.
Author contribution(s)
JL conceptualized and designed the study. JL obtained funding, and JL gained ethical approval. JL led participant recruitment and data collection. JL wrote draft of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dong-A University research fund.
Ethical approval
This study was approved by the Youngsan University Institutional Review Board (YSUIRB-202004-HR-064-2).
Guarantor
JL.




