Abstract
Objectives
Corneal transplantation is the most common transplant worldwide and its success critically depends on the management of corneal graft rejection through topical steroid therapy during the first 12 months after surgery. There is currently no published data on adherence after keratoplasty. This pilot study aims to explore the adherence to topical steroid after penetrating keratoplasty using a smart electronic device.
Methods
Thirty patients undergoing penetrating keratoplasty were included to evaluate the adherence to topical dexamethasone medication for 12 months after surgery. Patients received the usual post-transplantation treatment (topical dexamethasone) and follow-up after surgery (day 15, months 1, 2, 3, 4, 5, 6, 9, and 12). Adherence to treatment was monitored using the KaliJAR device (Kali Care, Santa Clara, CA, USA), which recorded the number of single-dose units (SDU) discarded. At control visits, data recorded by the device were compared to the manually count of SDU. Adherence ratio and individual adherence curve were explored for all patients.
Results
Data from 27 patients showed a high agreement between adherence ratio calculated based on the device data and obtained from manual counting of the discarded SDU (intraclass coefficient correlation of 0.87 [95% CI: 0.738–0.938]). Mean adherence to the treatment over the 12-month study period was 95.2 ± 4%.
Conclusions
Adherence to topical dexamethasone for 12 months after corneal transplantation was high. The connected device was able to record accurately the discarded SDU. This approach would be a particular interest in the early identification and personalized follow-up of poorly adherent patients.
Introduction
Corneal graft allows visual function restoration in case of corneal blindness and represents the most common transplantation worldwide. Despite leading developments in lamellar grafts, penetrating keratoplasty (PK) remains a widespread surgical technique worldwide. 1 With many decades of hindsight, this well mastered approach still represents the only therapeutic option for many corneal diseases.
Corneal graft immune rejection is the most frequent complication after PK 2 that seriously compromises the short-term, medium-term, and long-term outcome of the graft. The occurrence of rejection episodes in patients treated by PK varies among reports from 20% to 60%.3,4 These episodes most often occur during the first 12 months after transplantation. 5 In order to reduce the risk of immune-mediated allograft rejection, a preventive treatment is used from the first post-operative days to control or alleviate the host immune response against the donor cornea. In patients without risk factor of rejection, this treatment consists solely of repeated instillation of steroid eye drops (dexamethasone, prednisolone, or betamethasone depending on the country, 3–6 times daily) with a progressive decrease over 12 months,6,7 and/or possible maintenance of the treatment for the long term, without international consensus on dosage and duration.
Despite this, adherence to this treatment is currently poorly known and there is no published data on adherence to steroid eye drops after keratoplasty. More generally, adherence to eye drops is poorly assessed in ophthalmology. 8 A complex relationship exists between the patient, their disease, treatment and their relationship with the ophthalmologist 9 and can impact adherence to the treatment. Previous studies have explored therapeutic adherence in glaucoma patients, who must instill eye drops daily and for life. Data collection is usually retrospective using a questionnaire, and is therefore associated to memory and subjectivity biases, and can thus be improved.10–12 A study using an electronic drop counter found a poor compliance in 45% of glaucoma patients. 13
In order to explore objectively adherence to topical steroid after PK, we proposed to follow patients’ treatment using a smart connected electronic device that counts the disposable single-dose units (SDU) eye drops.
Materials
Study design
This single-center prospective observational study was conducted at the University Hospital of Saint-Etienne, France. The study complied with the Declaration of Helsinki guidelines for research involving human subjects and was approved by an Institutional Review Board (IRB00010220, Comité de Protection des Personnes Sud-Est 1). The trial was registered with identifier NCT04107246 at https://clinicaltrials.gov. Each patient provided written informed consent before enrollment.
Patients undergoing PK were followed up to evaluate the adherence to topical dexamethasone medication for 12 months after surgery. All included patients received the usual post-corneal transplantation treatment and follow-up. Control visits occurred at day (D) 15 post-surgery, then monthly from months (M) 1 to 6 and finally at M9 and M12. Steroid therapy consisted in topical dexamethasone (Dexafree, Laboratoires Thea, France) with a gradually tapered dose: one drop thrice a day during the first 3 months, one drop twice a day from M3 to M6, one drop once a day from M6 to M9 and finally, one drop every other day out of two from M9 to M12. This therapeutic scheme represents our routine treatment protocol after PK.
The protocol also allowed for adjustment of the dosage and type of steroid, in the event of elevated intraocular pressure (IOP) and after an episode of rejection. In this case, the shift to a steroid not available in SDU will result in patient's exclusion due to the impossibility of following the adherence of the new treatment.
As the occurrence of a rejection episode necessarily modifies the patient's attitude and adherence to treatment, patients presenting early rejection episodes were excluded to avoid potential associated bias.
At each visit, patients underwent a complete ophthalmic examination including slit lamp examination with IOP measurement by Goldmann applanation tonometry. Any adverse events and/or patient difficulties in using the collection box were recorded.
Adherence to treatment was monitored using the KaliJAR device (Kali Care, Inc, Santa Clara, CA, USA). The device is a container allowing the precise counting of SDU by recording the movements of the opening valve of the box, with a maximal storage capacity of 100 SDU (Figure 1). The container's lid is equipped with three infrared sensors able to detect and count the number of passages through its lid of SDU with or without caps (for more accurate data) discarded by the patient. The sensors, placed along a spiral-like pattern, are oriented at three different angles to reduce the risks of missing the reading of a vial (multiple angles to catch a flat object) and to measure the speed and the length of the object being inserted. The signal duration varied depending on the angle of the sensor and its position. A dedicated online software (Kali Care, Inc) 14 analyzed the three signals and detected the difference between the body and the caps of SDU according to the duration of the signal. The detection of the cap alone did not count as an instillation. The device transmitted SDU number and timing to a user-friendly interface in real-time when located in an area with 2G/3G/4G coverage. The device could be transported to follow the patient everywhere. An internal memory of about 24 h allows the storage of data when the device is moved from one localization to another. Patients were asked to discard the SDU in the device instead of the trash can after each eye drop application. The device recorded the number of SDU as well as the day and time of discarding.

The smart connected KaliJAR device designed for counting the single-dose units of eye drops. Source: reproduced with permission from the manufacturer.
The primary objective of the study was to accurately monitor the adherence to topical steroid after PK. At each visit, KaliJAR electronic data were analyzed by the physician by using the dedicated online software. The inter-visit data were further confirmed by manual counting of the SDU. The associated primary end point was the ratio between the number of instilled drops and the number of prescribed drops for the 12-month follow-up period. Adherence ratio was calculated at each visit for each patient.
The secondary objectives consisted first in the classification of patients’ adherence patterns by analyzing daily individual treatment hours. Accordingly, an individual adherence scatter plots was established for each patient by plotting the recorded instillation data (hour and date) of each patient over time for the whole study period. Each compliance curve was analyzed based on the fitting between recorded instillations and therapeutic prescription on each period, and the regularity of treatment (hours of instillation).
In addition, the evaluation of patients’ self-perception of their adherence to treatment was assessed using a visual analog scale (VAS) of 0 to 10, and the 8-item Morisky Medication Adherence Scale questionnaire (MMAS-8) at month 1, 6, and 12 after surgery. Briefly, the questionnaire consisted in eight questions with dichotomous responses (yes/no) for the first seven items, and a five-point Likert response for the last question. 15 This Morisky scale was previously translated and validated in French 16 and was adapted here for the use of eye drops. Scores on the MMAS-8 range from 0 to 8, with scores of <6, 6 to <8, and 8 reflecting low, medium, and high adherence, respectively.
Patient selection
Male and female patients aged 18 years or older undergoing PK were eligible for enrollment. Only patients able to properly apply topical steroid therapy or helped by a third party for instillations (family, healthcare support) were included. Patients under guardianship or unable to use the collection box properly (i.e. with a physical or mental disability that prevented normal use of SDU and/or the device and who do not have well identified reliable third party aid) were excluded from the study. No other treatment was allowed during the study and patients receiving multiple eye drop therapy were excluded from inclusion. Only the use of on-demand lubricant artificial tears was allowed. Figure 2 showed the Consolidated Standards of Reporting Trials (CONSORT) diagram of participant recruitment, screening, and enrollment.

Consolidated Standards of Reporting Trials (CONSORT) diagram of participant recruitment, screening, and enrollment.
Statistical analysis
As there was no data available on the adherence of transplanted patients, sample size was fixed to 30 patients, as we estimated that this first record will provide a sufficiently accurate estimate of therapeutic adherence after PK. Data normality was verified using the Shapiro-Wilk normality test. Agreement between manually and automated counted SDU for each patient was analyzed using intraclass correlation coefficient and Pearson's correlation coefficient. In addition, a Bland and Altman plot 17 was obtained by plotting the difference between both data according to the mean of manual and software count. For each patient, the data obtained at each visit concerned the period between the previous follow-up visit and the concerned visit. Analysis was performed using GraphPad Prism V5 (GraphPad Software, San Diego, CA); p < 0.05 was considered significant.
Results
A total of 30 patients were included in the study between November 2019 and October 2021 (Figure 2). One patient never brought back home the device and another patient finally declined to participate at the start of the study. One patient presented signs of graft rejection after surgery (7 days) and was withdrawn from the study (patient undergoing a third PK after two previous failures, in a context of corneal burn). Remaining 27 patients who followed the study consisted 17 men and 10 women, with a mean age of 58 years (range: 24–88) at inclusion. Of these 27 patients, 22 had previously undergone keratoplasty (alone or combined with cataract surgery) and 2 had simple cataract surgery.
During follow-up, one patient was withdrawn from the study because of a bacterial keratitis at M5 (Streptococcus oralis) and another one due to endophthalmitis following phacoemulsification at M9 (PK on a perforated cornea with no etiological pathogen agent found). Three other patients were withdrawn at M6 (n = 2) and M9 (n = 1) after medication shift due to IOP elevation (steroid treatment using fluorometholone, not available in SDU). The data of these five patients were analyzed until study discontinuation (Table 1). In total, 22 patients presented a complete data set over the 12-month study period.
Number of patients at each visit and of devices brought back at each visit.

Patients unable to come to the control visit due to COVID-19-positive PCR and/or symptoms or COVID-19-related restrictions in place at the time.
Same device presenting a dysfunction (no connection to 4G network).
The dosage of dexamethasone was adapted in few patients as follows: one patient was prescribed a posology of four times a day until M1 instead of thrice a day; posology was maintained to thrice a day for 13, 2 and 1 patients during M4, M5, and M6, respectively, to twice a day for 5 patients between M6 and M9, and to twice a day and once a day for 1 and 6 patients between M9 and M12, respectively. In each case, treatment adherence was calculated taking into account prescription changes.
Global use and functioning of the device
The functioning of the device was uneventful for 85% (23/27) of patients over the 12-month study period. Two devices failed to recover data at a given time of the study (4G network issue) and two presented a dysfunction and were replaced by a new one. Some data could therefore not be retrieved for several weeks. Despite replacement, one of these two devices still failed to record data from M6 to M12 (Table 1).
Agreement between manual and automated counting of SDU over the study period was represented in Figure 3(a). An intraclass coefficient correlation of 0.87 (95% CI: 0.738–0.938) showed a good agreement between the manual and automated methods. In total, 71% of SDU counting (in total 234 countings for the 27 patients) had a difference between automated and manual counts under 5%. In 10% of cases, the difference was comprised between 5% and 10%, in 9% of cases between 10% and 20%, and in 10% of cases over 20% (max = 66%). In 90% of cases, the observed difference between both counting techniques was an under-estimation of SDU number by the device.

Agreement between manual and automated counting of single-dose units over 12 months. (a) Scatter plot of the total number of single-dose units over 12 months. Lateral box plots represented the median, interquartile range, extreme and outliers and of each measure. Pearson's r = 0.87 (95% CI: 0.74–0.94), p < 0.0001. (b) Bland and Altman agreement plot. Solid central line represented the mean bias of −10, calculated as the mean of the differences between manual and automated counting. The 95% confidence interval (−153–132) was figured by the black doted lines.
Several outlier points (patients, in red) could explain the differences observed between the two techniques of evaluation, as showed by the large confidence intervals in the Bland and Altman plot (Figure 3(b)), associated with four main outlier points.
Adherence to steroid therapy after PK
The participation of patients at control visits was impacted by COVID-19 pandemic between M2 and M5 follow-up visit, with 2 to 8 patients who did not participate to the planned control visit during this period (Table 1). As monitoring was carried out remotely by the connected device, the follow-up of patients was not significantly impacted. The transmitted data and SDU manual counting were cumulated and adapted in function of the duration of the unchecked period. The follow-up of included patients was globally satisfying despite the impact of the COVID-19 pandemic. From M2 to M5, nine, three and two patients did not show up for 1, 2 and 3 control visits respectively because they presented COVID-19 symptoms and/or positive PCR test to COVID-19 that required isolation, or because of confinement during these periods (Table 1).
Major part (95%) of patients seen at control visits brought back the device. Mean adherence to treatment over the 12-month study period was 95.2 ± 4% (range 80.4–100%). Adherence ratio was over 90% for 25 patients over 27 (Figure 4(a)). In addition, adherence was stable over the study period when analyzed at each time point (Figure 4(b)). Two other patients presented wide min–max interval in adherence ratio over the study period (patients at ranks 19 and 25 in Figure 4(c)). The first patient presented highly irregular profiles during the first study month and was not following the prescription during this period (repeated and alternating under and over-medication phases; individual medication profile shown in Figure 4(c)). The patient in rank 25 presented a low adherence ratio at M2 (48%) compared to the high adherence observed during the rest of the study period (100%).

(a) Distribution of the number of patients according to the adherence ratio calculated for the total study period. (b) Box plot showing the evolution of adherence during the study period. Data were shown as median, interquartile range with minimum and maximum values. Crosses represent the mean value. (c) Box plot showing the adherence of each patient for the whole study period, classified from the most to the less adherent (n = 27). The gray boxes represented the five patients withdrawn before the end of study period. Data were shown as median of all values of adherence calculated over the study period, interquartile range with minimum and maximum values.
Different adherence profiles
The analysis of the eye drops instillation scatter plots allowed us to classify the patients into three main categories: (1) adherent patient with highly regular instillation scheme (n = 20, 74%), (2) adherent patients with irregular instillation scheme (n = 6, 22%), and (3) non-adherent patients (n = 1, 4%). Representative examples were presented in Figure 5. The eye drops instillation scatter plots of the non-adherent patient did not allow recognizing the different phases of prescription (dosage tapering over time) during the follow-up period (Figure 5(c)).
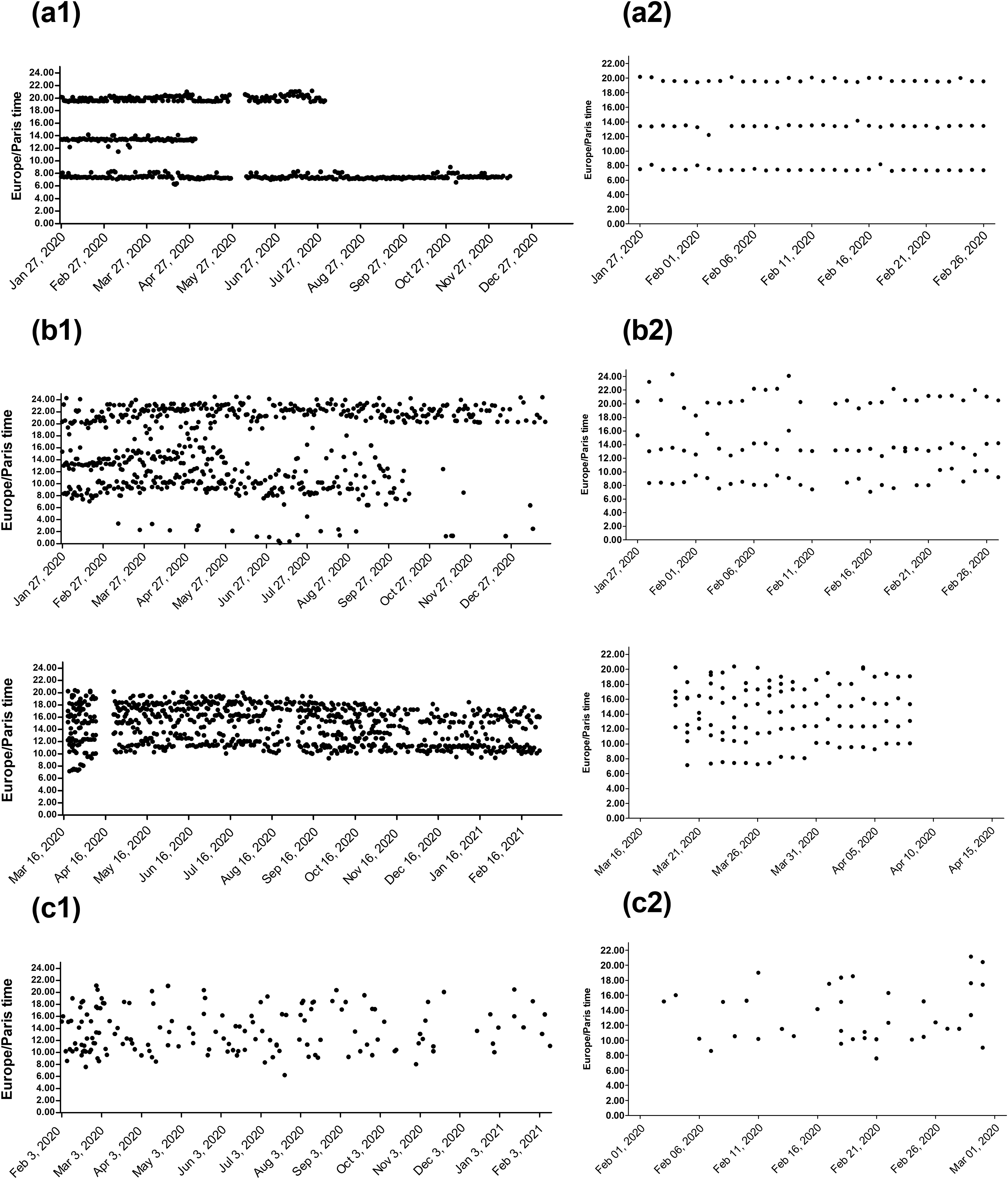
Representative examples of eye drops instillation scatter plots recorded by the electronic device during the 12-month follow-up period, (a) from an adherent patient with highly regular instillation scheme, (b) from two adherent patients with irregular instillation scheme, (c) from the non-adherent patient. Whole study period (1) and the first month of follow-up (2) are shown for each patient. Instillation scheme consisted in one drop thrice a day during the first 3 months, one drop twice a day from M3 to M6, one drop once a day from M6 to M9 and finally, one drop every other day out of two from M9 to M12. Periods with no data recording are observable in A1 profile (from 27 May to 04 June 2020) and in the second example of B1 profile (from 09 April to 20 April 2020) and were linked to 4G network connection issues.
Self-reported adherence to treatment
Self-reported adherence using the questionnaire was high. Only one patient, classified as non-adherent based on monitoring device data, rated a score of 7 for the questionnaire whereas the remaining 26 patients rated the maximum score of 8. Similarly, VAS score was high for all patients. The score provided by the VAS ranged from 6.5 to 10, with the score of 10 representing 91.8% of all the available scores. Taken together, these results support a homogenous and high self-reported adherence to steroid therapy.
Discussion
To our knowledge, this study report for the first time, the adherence to topical steroid therapy during the first year after PK. With more of 93% of patients with a global adherence ratio over 80%, therapeutic adherence in our cohort was high compared to therapeutic adherence reported in other ophthalmologic disease like glaucoma. 18 Adherence to topical glaucoma treatment is by far the most explored field in ophthalmology. In a therapeutic point of view, glaucoma represents the most challenging chronic eye disease requiring a long-term preventive therapy on asymptomatic patients with no perception of any beneficial effect. A growing number of reports have explored the particular interest of electronic device for the monitoring of adherence to topical glaucoma treatment19,20 using the Travatan Dosing Aid, a monitoring device dedicated to a specific formulation of Travoprost that electronically stores data on the time, date, and number of drops administered. 20 Mean adherence to topical glaucoma therapy monitored by this device over a period of 2 to 3 months, ranging from 71% to 91%13,14,21,22 represents the upper level of the adherence rates reported by classical approaches (patient self-reports and pharmacy claims data). 23
The high therapeutic adherence observed after PK in our cohort can be explained by mutually non-exclusive hypotheses: (1) The path between PK decision and surgery requires a particular psychological investment of the patient. Indeed, patients are pre-selected before PK based on their self-motivation for PK. The decision to undergo PK is guided by the patient reflection, particularly as the patient will receive a cornea from a donation and can wait long periods before a donor cornea is available for the surgery. (2) The relatively short duration of topical steroid therapy after PK in comparison to glaucoma or life-long disease treatment duration, and the frequent control visits scheduled during the follow-up period. (3) A possible selection bias should have also contributed, as the study was based on voluntary patient participation and required patients with no signs of cognitive impairment. Dementia was indeed associated with lower glaucoma medication adherence in a recent report. 24
Finally, another contributing factor to high adherence, in this study and all studies reporting electronic monitoring of medication, is the direct incentive effect of using a connected monitoring device for the patient, as the patient is aware of being followed-up in real time. This aspect, most probably variable among patients and time, may represent an intrinsic lever of interest to improve adherence level compared to real day life. Patient undergoing PK represent a minority compared to glaucoma patients. However, the access to a donor cornea became scarce 1 and the consequences of a rejection are briskly deleterious for patient. In this context, the systematic electronic follow-up of patient during the first year(s) of post-graft treatment seems therefore realistic.
Poor adherence to topical steroid therapy after PK is very likely to be a crucial factor impacting surgery final success, as this therapy is the gold standard for preventing immune-mediated allograft rejection. Although we have recently demonstrated proof of concept that a subconjunctival dexamethasone implant can temporarily replace eye drops, 25 there are currently no validated approaches allowing a prolonged release of steroid after PK eye drops remaining the unique current available option. The discontinuous nature of instillations, associated with fluctuations in the local concentration of the steroid, is already considered as a contributing factor to graft rejection. Thus, any deviation from fixed application time and interruption in the therapeutic scheme may increase the risk of rejection after PK. Particularly, the initiation phase of medication, known to have important clinical consequences, may be crucial after graft surgeries like PK. 26
In this context, the ability of electronic monitoring device to supply accurate data on the time and date of treatment is highly valuable for the physician to understand each patient profile and have objective information to evaluate graft rejection risk. In this pilot study, we were able to observe several distinct patient profiles, as reported for glaucoma patients. 27
Nevertheless, adherence level and/or pattern seems to be an essential but not sufficient factor to foresee therapeutic efficacy and define adherence thresholds in the context of PK. 28 Drug properties, like pharmacological profile, and patients’ characteristics play a significant role in therapeutic efficacy. Thus, the upcoming studies on this field should focus on providing clues to understand the role of steroid adherence pattern and the impact of other factors in the context of prevention of corneal graft rejection after PK.
We confirmed what most of the studies using electronical-monitoring devices reported: there is a low agreement between the adherence provided from device data and the self-reported adherence.14,21 For example, glaucoma patients, especially new patients, were prone to over-reporting the number of doses taken and the timing adherence. 29 Additionally, a poor agreement between the physician assessment and the electronic monitoring was also observed for individual cases, 13 supporting a poor ability of the physician to identify which persons are poorly adherent from their self-report or from other subjective clues. Electronic Monitoring of Adherence is now considered as the gold standard to measure adherence. 28 By allowing real-time data transmission and follow-up of patient profiles, connected monitoring devices may revolutionize the way in which eye care providers follow their patients. These devices, combined to dedicated smartphone applications and/or smart health network could play crucial role in patient management to improve therapeutic efficacy and post-surgical outcomes. The high cost of connected electronic devices is still limiting their use not only in common practice, but also in clinical trials, particularly for extended period of patients’ follow-up. However, affordable technological solutions will promote the use of these tools in a near future.
The device used in this study is specifically designed for SDU. Previous reports using electronic devices were adapted to eye drops bottles, with either simple approach, recording the handling date and time of the bottle from a container 21 or more evolved device allowing the additional monitoring of the number of dispensed drops.27,30 However, this last one is restricted to travoprost eye drops and thus cannot allow the follow-up of a great majority of patients with other or combined medication. Another limitation of these monitoring devices is the incapacity to detect the inability to correctly administer the eye drops, a frequent limitation to therapy efficacy as observed in glaucoma patients. 31
Several limitations in this study can be highlighted. The small number of patients included gives us a first insight in the global adherence to treatment after PK. This cohort did not allow us to study the impact of adherence to therapy on the outcomes of PK or the impact of specific parameters like patient's ocular surgery history (only three patients without ocular surgery history in our cohort), age or gender on the adherence to therapy. The exploration of potential risk factors for non-adherence will require the follow-up of a larger cohort of patients. In addition, we did not define quantitative metrics to define the limits between regular and irregular adherence patterns for this pilot study. In future works, such metrics will be required to characterize more accurately the instillation patterns. Considering the technology used for the therapeutic monitoring, the risk a false positive was expected to be null, as it would require a voluntary discard of purposely emptied single doses, which is unlikely in the context of the study. False negative results (SDU not recorded whereas present in the box) were the main risk due to a few boxes dysfunction, mainly related to unconnected boxes due to limited 4G network. These issues in network connection were observed in three boxes during the study period, leading to notable under estimation of SDU by the connected device compared to manual count. This risk of false negative was abolished by the systematic manual counting of the SDU at each visit control. Finally, only one particular case of under estimation of SDU by manual counting was observed during the study period. As the patient did not follow control visits between M2 and M6 due to COVID-19 restrictions, the patient emptied the device alone whereas SDU automated counting was well functioning during the same period and revealed high observance.
Patient inclusion and follow-up started in November 2019, prior to COVID-19 pandemic onset, and was thus conducted during the pandemic period. Recent reports have suggested that the pandemic contributed to alter health behaviors like medication adherence. In particular, patients with glaucoma followed during the pandemic presented lower adherence to ocular hypotensive medication. 32 In our study, up to eight patients were tested positive on nasopharyngeal swabs between M2 and M5 and could not attend the control visit. Taking into account the post-surgical context and relative short duration of the treatment compared to glaucoma therapy, as well as the high rate of adherence observed in the cohort and the clearly distinguishable patients’ profiles, the impact of COVID-19 pandemic on the adherence may be considered negligible whereas it clearly impacted the participation of patients to follow-up visits.
Conclusion
Electronic monitoring of topical steroid therapy over 12 months in patients after PK was efficient and reveals adherence over 95% in most of the patients. This approach, able to characterize specific pattern of adherence, would be of particular interest in early identification and follow-up of non-adherent patients, and/or as an educational incentive to help achieve good adherence. The study highlighted a risk of data loss in case of 4G network connection issues. This aspect should be used to implement the device with an internal memory that would improve its autonomy and accuracy in the future.
Footnotes
Acknowledgements
We thank all the participants who took part in the study, and Dr Sina Fateh and Philippe Cailloux from Kali Care.
Contributorship
MCT, ASG, GT, and PG researched literature, partnership, and conceived the study. ASG, SP, TG, MM, CL, PB, and MCT were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. MM, CL, SP, and TG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Conflict of interest
Kali Care, Inc, loaned the devices and provided the central computing service for data, but were not involved with data analysis or interpretation, or drafting of the manuscript. The authors have no financial interest in Kali Care, Inc, nor did the company have any control in the implementation, analysis, and writing of the report. The authors declare no conflict of interest.
Ethical approval
The ethics committee Comité de Protection des Personnes Sud-Est 1 approved this study (REC number: NCT04107246).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
GT.
Trial Registration
ClinicalTrials.gov, Identifier: NCT04107246.
