Abstract
Background
Childhood myopia, intensified by rapid technological advancements, is an escalating public health issue. Shared decision-making (SDM) facilitates collaborative treatment engagement and adherence, contributing to the mitigation of myopia progression.
Objective
This study developed and evaluated a web-based decision aid aimed at assisting parents in making informed, value-aligned decisions.
Methods
Following the International Patient Decision Aid Standards framework, we conducted a two-phase process: development (scoping, stakeholder engagement, and content planning) and testing (prototype creation and alpha and beta testing with external users) to develop the Childhood Myopia Shared Decision-Making Aid (CMSDM) to support informed and value-congruent treatment decisions. The study involved 55 parents, 12 ophthalmologists, 3 school nurses, 2 health education teachers, and 20 optometrists.
Results
CMSDM comprises two decision aids (myopia treatment and optical correction) with six components: (1) Introduction—stating the purpose of the tool; (2) Comparison—outlining treatment options with pros and cons; (3) Consideration—prompting value reflection; (4) Understanding—clarifying key points; (5) Decision—guiding informed choice; and (6) Submission—sharing the decision with the ophthalmologist. A review from five experts provided the aid with a content validity index of 0.85, demonstrating consensus on content relevance. Users reported a mean System Usability Scale score of 85, indicating excellent usability. The use of CMSDM was associated with improved parental knowledge (t = 2.31, p < 0.01; Cohen's d = 0.60) and reduced decisional regret (t = 2.01, p < 0.05; Cohen's d = 0.52) in preliminary testing.
Conclusions
CMSDM, the first SDM aid for childhood myopia, supports informed parental choice. Its effectiveness should be further examined in randomized controlled trials.
Background
The World Health Organization (WHO) has identified childhood myopia as a major global health concern. According to WHO reports, 1 by 2050, myopia will affect approximately 50% of the global population (4.9 billion people), with 925 million individuals (10%) developing high myopia, defined as a spherical equivalent (SE) of ≤–5.0 diopters (D). Moreover, a recent systematic review of myopia epidemiology reported that the global myopia prevalence has increased from 24% in 1990 to 36% in 2023 and is estimated to approach 40% by 2050, 2 highlighting the urgent need for effective strategies in myopia prevention and management.
Globally, countries aim to prevent early-onset myopia and manage its progression to high myopia, which can result in irreversible complications in adulthood. 3 Potential complications associated with myopia include macular degeneration, increased risk of retinal detachment, development of posterior subcapsular cataracts, higher incidence of open-angle glaucoma, 4 and reduced productivity due to severe vision impairment and blindness. 5 Parents and children must adhere to treatment regimens to prevent the progression of myopia to high myopia. 6
The primary objective of myopia control is to reduce abnormal elongation of the eye axis.3,7 Numerous experimental studies have confirmed effective methods for controlling myopia progression, 6 including optical, 8 pharmacological, 9 and environmental interventions. 10 Various myopia treatment modalities have been proposed for the effective control of myopia progression and reduction of adverse reactions. These include 0.01% 11 and 0.05% 12 low-concentration atropine, optical correction combined with low-concentration atropine, 13 orthokeratology combined with atropine, 14 MiSight, 15 and peripheral defocus design glasses. Clinical guidelines emphasize that eye care professionals (ECPs) must consider baseline refractive error and myopia progression when formulating appropriate management strategies. 16
Ophthalmologists, optometrists, parents, and children have different preferences and attitudes toward myopia treatment. 17 Parents make treatment decisions for their children's myopia. 18 Choices are mainly influenced by recommendations from ophthalmologists or optometrists 19 and from friends and family. 20 Studies indicate discrepancies between ophthalmologists and parents regarding myopia treatment selection, 19 potentially leading to noncompliance and impacting treatment outcomes. Previous studies have addressed that adherence to myopia control treatments remains challenging in children due to the inconvenience of long-term spectacle 21 or orthokeratology wear 22 and the burden of nightly atropine use. 23 Poor adherence diminishes treatment efficacy and elevates the risk of high myopia. To achieve effective myopia control, the decision-making process must emphasize both patient satisfaction with treatment choices and adherence to prescribed regimens. Decision aids (DAs) may mitigate decision regrets by enhancing parental knowledge and supporting shared decision making (SDM), thereby fostering better adherence. 24
SDM is increasingly recognized as a collaborative model in which healthcare providers and patients (or families) jointly consider available evidence alongside the patient's individual values and preferences, aiming to arrive at informed and personalized healthcare decisions. Patient DAs are instrumental in facilitating SDM by simplifying complex treatment options and enhancing clarity, ultimately supporting more confident and value-based choices. 21 To guide their application, the International Patient Decision Aid Standards (IPDAS) Collaboration proposed a structured framework for their development, later refined to integrate user-centered design approaches. 25 Given the high demand for myopia control interventions and the limited time ECPs have for patient education, web-based DAs present an accessible and scalable solution. 26
Online DAs offer real-time updates, interactive tools, and personalized recommendations based on current clinical evidence, surpassing traditional pamphlets or in-person consultations. Web-based DAs are designed for online use and feature interactivity (enabling user navigation of content and responses), dynamic content (adapting to user input or clinical evidence), and personalization (tailoring recommendations based on patient demographics and clinical conditions).27,28 These features provide patients and families with current, tailored treatment information and enhance engagement in SDM beyond conventional approaches. Studies have demonstrated that web-based DAs effectively improve decision-making processes for various health conditions, highlighting their potential to enhance patient education and promote informed choices. 29
This study describes the development and preliminary evaluation of a digital DA designed to assist parents in navigating treatment options for myopia control. The platform functions as a dynamic, evidence-informed support tool aimed at facilitating informed parental choices, increasing confidence in selected interventions, and promoting consistent adherence to recommended myopia treatments.
Methods
Study design and protocol
A web-based DA to support parental decision making regarding myopia control was systematically developed in Taiwan between September 2023 and February 2024, following the IPDAS. 25 The process comprised two main phases: development (scoping, stakeholder engagement, and content planning) and testing (prototype creation, alpha testing, and beta testing with external users) (see Figure 1). The Institutional Review Board of Chang Gung Memorial Hospital approved the study protocol (No. 202102324B0C502). All participants provided written informed consent before inclusion in the study.

Study design procedure.
Design and development
Development phase
Scoping and stakeholder engagement
Guided by the IPDAS framework and informed by the International Myopia Institute (IMI) guidelines, 30 we first conducted a scoping process to define the decision context, which involved supporting parents of children newly diagnosed with myopia in selecting appropriate treatment options during the follow-up period.
To ensure that the content and design of the DA aligned with user needs and clinical practice, input was collected from key stakeholders, including parents of children newly diagnosed with myopia, ophthalmologists, and optometrists. To validate the treatment options and clinical accuracy, semistructured interviews were conducted with 10 ophthalmologists and 18 optometrists specializing in pediatric myopia. Additionally, parents provided feedback through interviews and a survey to identify their values, concerns, and informational preferences. These insights were incorporated into the development process to enhance the relevance, clarity, and acceptability of DA. Parents were asked what factors and support would help them choose among different treatment options, while ophthalmologists provided perspectives on what information should be communicated to parents and what factors parents should consider before participating in treatment decisions. Purposive sampling was employed, and the interviews continued until data saturation was reached. A three-round review process and thematic analysis were performed using MAXQDA version 24. The qualitative component of this study was reported in accordance with the Consolidated Criteria for Reporting Qualitative Research (COREQ) checklist. 31
A literature review was also conducted using PubMed, MEDLINE, the Cochrane Library, Web of Science, and Scopus to identify systematic reviews and meta-analyses related to myopia control published between 2015 and 2023.
Content development and structuring
This stage comprised three steps. First, based on the literature review, evidence-based treatment options, clinical indications, and potential side effects were synthesized and organized into a comparison table. To ensure the table's clinical accuracy and clarity, 8 ophthalmologists and 14 optometrists who had already participated in the interviews were subsequently invited to review and refine its content. Second, qualitative interview data were analyzed to identify key considerations and informational needs among parents regarding treatment decisions. These insights were integrated with findings from a previous survey on SDM among parents of children with myopia, 32 resulting in a consolidated list of factors that influence parental decision making.
Third, the research team collaboratively discussed the website layout and finalized the content structure of the Childhood Myopia Shared Decision-Making Aid (CMSDM), following clinical priorities and user-centered design principles.
Testing phase
Based on the findings from the development phase, a draft prototype of CMSDM was created, along with a corresponding website flow diagram comprising six sequential modules:
Introduction: Provides an overview of childhood myopia, the importance of early intervention, and the purpose of the DA in supporting parental involvement. Comparison of choices: Presents evidence-based treatment options and their potential side effects. The content was informed by an expert consensus meeting and tailored to address key parental concerns. Consideration: Uses Likert-type self-assessment questions to help parents reflect on their needs and preferences after reviewing the pros and cons of each option. Understanding: Includes comprehension-check questions for each treatment option, designed to reduce bias by presenting one benefit and one drawback per method. Decision: Assesses parents’ readiness to make a decision and whether further information is needed, allowing them to indicate their preferred option. Submission: Enables users to document their decisions and generate a printable or electronic summary to support clinical discussions.
All content was structured according to DA standards and progressively uploaded to an online platform. The CMSDM, developed in Mandarin, comprises two modules: one for treatment options and another for optical correction methods. As the target population of this study was children newly diagnosed with myopia, the treatment options presented in the DA were designed to include pharmacological therapy and optical correction, in accordance with current clinical guidelines. 32 Although combined therapy is beneficial in myopia treatment, it was not incorporated into the website design as the present study focused on the initial management of newly diagnosed myopia. As shown in Figure 2, users are required to complete each module in sequence but may return to previous sections if needed. Upon completion, the system generates a personalized summary that includes treatment preferences and responses to the self-assessment sections, facilitating more informed and confident discussions with healthcare professionals.

Website workflow of shared decision-making website for parents of children with myopia.
Alpha testing
To evaluate the usability and functionality of CMSDM, alpha testing was conducted on 15 parents of children with myopia, who were distinct from those involved in previous stakeholder interviews. The participants were asked to navigate through the six sequential components of the web-based system and were instructed to complete the components in order, simulating typical usage. The tasks included reviewing background information, comparing treatment options, reflecting on personal values using Likert-type scales, answering comprehension questions, selecting a preferred treatment option, and finalizing their choice through the summary submission page.
In addition to usability testing, the textual content was evaluated by three school nurses, one health education teacher, and one ophthalmologist. Each rater assessed the relevance of the content using a five-point Likert scale (1 = not relevant, 5 = highly relevant). They assessed the clarity, simplicity, and relevance of the textual content. The item-level content validity index (I-CVI) was calculated as the proportion of experts rating each item as 4 or 5 on a 5-point Likert scale. The scale-level CVI using the average method (S-CVI/Ave) was then computed as the mean of all I-CVI values across items. The feedback was reviewed by the research team and further validated in consultation with an ophthalmologist to identify content-related errors and suggest improvements across different sections of the system.
Beta testing
Beta testing was conducted with 15 parents to assess whether users could follow the step-by-step logic of the DA without confusion or disruption. Beta testing was conducted with 15 parents to assess whether users could follow the step-by-step logic of the DA without confusion or disruption. Of these, 10 were newly recruited participants, while 5 had previously taken part in the stakeholder interviews. During the sessions, user interactions, including click behavior, time spent on each section, and navigation patterns, were recorded to evaluate system flow, efficiency, and clarity. Observations focused on users’ comprehension of each section's purpose, navigational challenges, and moments of hesitation when interacting with specific features.
Upon completion, the participants completed the System Usability Scale (SUS) and an acceptability questionnaire to assess the tool's clarity, relevance, and perceived usefulness. Qualitative feedback regarding the design, language, and ease of use was also collected. All feedback was used to refine the structure, wording, and interface before the final implementation.
Finalization of the CMSDM
Following the integration of the user feedback, two ophthalmologists and two health education teachers reviewed the final version of the CMSDM. The project team implemented the necessary revisions with a software engineer. This rigorous development process ensured that the CMSDM provided a positive user experience through easy navigation, accurate content, and effective functionality for informed decision making.
Instruments
System usability scale
Usability was evaluated using the SUS, 33 a 10-item questionnaire employing a 5-point Likert scale ranging from 1 (Strongly Disagree) to 5 (Strongly Agree). Odd-numbered items are phrased positively, whereas even-numbered items are phrased negatively. Scoring is calculated by subtracting 1 from each positive item's score and subtracting each negative item's score from 5 (i.e. 5—score). The total adjusted score is then multiplied by 2.5, resulting in a final SUS score ranging from 0 to 100, with scores above 68 indicating acceptable usability. Content validity was assessed by five experts (three nursing faculty members, one ophthalmologist, and one health education specialist), who rated each section of the website on a 5-point CVI scale (1 = not relevant to 5 = highly relevant). Ratings of 4 or 5 were considered valid; the item-level CVI (I-CVI) was calculated as the proportion of experts assigning these ratings, and the scale-level CVI (S-CVI) was derived as the average of the I-CVIs, yielding 0.95. Internal consistency reliability was also high (Cronbach's α = 0.90).
The decisional regret scale
This is a validated five-item, single-dimension self-report tool rated on a five-point Likert scale. Items 2 and 4 are reverse-scored, and the total scores are converted to a 0 to 100 scale, with higher scores indicating greater regret. The Chinese version, translated by Xu et al., 34 has demonstrated acceptable psychometric properties.
Knowledge
The knowledge assessment consisted of six true/false/unsure questions covering key aspects of myopia management, including the risks of myopia progression, side effects of pharmacological and optical treatments, purpose of follow-up visits, treatment adherence requirements, and risks associated with discontinuing treatment. Each correct response was awarded one point, with higher total scores indicating better knowledge of myopia control.
Statistical analysis
The extracted data were transferred to an Excel spreadsheet for the organization and construction of tables and graphs. All data were analyzed using SPSS (version 28). Descriptive statistics (e.g. mean, standard deviation, and percentage) were used to summarize participants’ demographic characteristics and the distribution of key variables. The reliability of the scales was assessed using Cronbach's alpha, and content validity was evaluated using the CVI. Correlation analyses (such as Pearson or Spearman, depending on normality) were conducted to examine relationships between the main variables. To explore the effects of demographic or background variables, independent t-tests, one-way analysis of variance (ANOVA), or multiple regression analyses were performed as appropriate. The level of statistical significance was set at p < 0.05.
Results
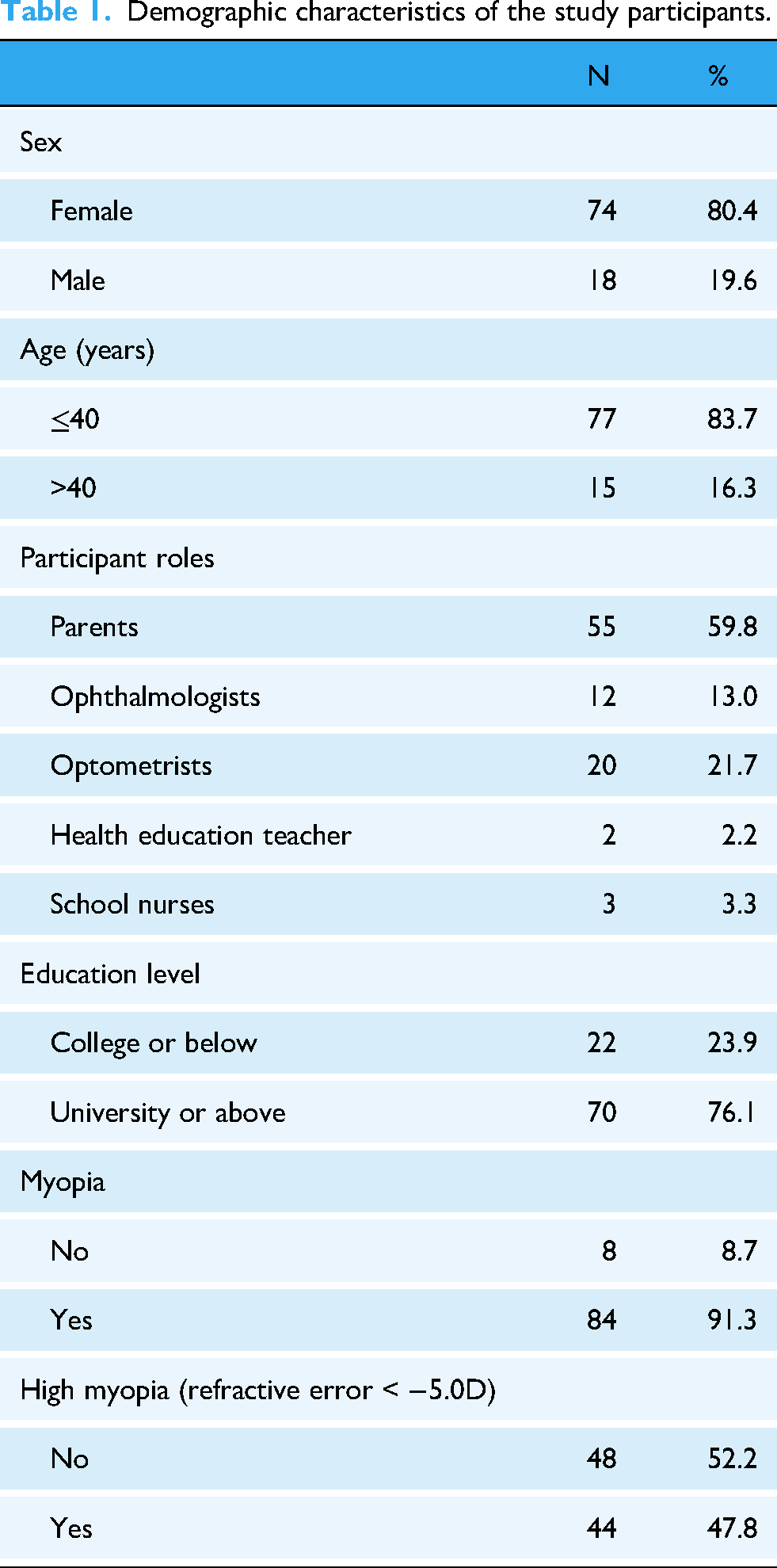
Ninety-two participants were involved in the development and testing of the CMSDM, including 55 parents, 12 ophthalmologists, three school nurses, two health education teachers, and 20 optometrists. The participants’ demographic information is presented in Table 1.
Demographic characteristics of the study participants.
Scoping and stakeholder engagement
We identified 11 systematic reviews and meta-analyses related to the diagnosis and treatment of myopia.6,35 A summary of these studies is presented in Table 2.
Literature review for evidence-based treatments for myopia control.

A structured literature review was performed to identify key components relevant to evidence-based interventions for childhood myopia. The content addressed both myopia definition and treatment domains, with a focus on clinically validated strategies, such as atropine eye drops, orthokeratology (Ortho-K), and peripheral defocus optics. 6 For atropine, concentrations ranging from 0.01% to > 1% were compared in terms of efficacy, side effect profiles, appropriate timing of administration, and follow-up recommendations.11,36,37 The review of Ortho-K lenses included their mechanisms of action, application procedures,38,39 clinical effectiveness, and safety considerations. 40 Studies examining the combined use of atropine and Ortho-K were also included to assess potential enhancements in visual outcomes.14,38 Regarding peripheral defocus strategies, particular attention was paid to MiSight® 1 day, 15 a daily disposable dual-focus soft contact lens approved for pediatric myopia control. 8 In addition, specially designed spectacle lenses were evaluated. 41 Both approaches were reviewed for their effectiveness, suitability for specific age groups, follow-up protocols, and associated costs. A summary of the reviewed components, content categories, and reference sources is presented in Table 2.
To ensure that the content of the CMSDM addressed the needs of both users and clinicians, key information themes were identified through interviews with parents and ECPs. These themes informed the structure and features of the SDM website (see Supplemental Appendix 1).
Treatment efficacy: Parents expressed concern about whether the treatment would result in visible improvements, while ECPs emphasized clinical outcomes, particularly the effectiveness of atropine and orthokeratology. Accordingly, the website presents comparative data on the effectiveness of each treatment in controlling axial length and refractive error. Myopia progression: Parents were interested in whether the treatment could slow progression and axial length changes. ECPs emphasized the importance of using axial length and postcycloplegic refraction as monitoring indicators. To address this, the website includes visual explanations of myopia progression and highlights the clinical relevance of axial length. Children's lifestyle habits: Parents focused on ease of use and compatibility with their children's daily routines. ECPs suggested tailoring treatments based on individual preferences, activities, and atropine concentrations. The website includes guided questions to help parents assess treatment suitability based on lifestyle factors. Cost: Parents frequently cited it as a major factor, particularly for optical interventions, whereas ECPs acknowledged the issue but were less inclined to initiate discussions. Therefore, the website includes estimated cost ranges for each treatment option to facilitate transparent communication. Side effects and adherence: Parents were concerned about tolerability and whether their children could follow the regimen. ECPs stressed the need for parental involvement, especially in orthokeratology. In response, the website offers an FAQ section outlining the common side effects and adherence requirements for each treatment.
Content and formation
Based on the design objectives and functional content of the DA, the information was reorganized into six distinct decision-making steps. Each step was aligned with a specific goal in the SDM process for childhood myopia control. These steps guide users from a foundational understanding to preference-sensitive choices and final treatment decisions. Visual representations of each step, along with the corresponding content domains and intervention purposes, are presented in Supplemental Appendix 2.
Step 1: Introduction
The first step focuses on foundational knowledge, providing explanations of the mechanisms underlying childhood myopia, the objectives of myopia control, and diagnostic and monitoring procedures. These concepts are supported by illustrated animations and explanations grounded in axial length changes to enhance user comprehension.
Step 2: Evidence-based treatments
This section presents the current evidence-supported options for myopia control. It includes detailed descriptions of the principles, procedures, and potential side effects of each approach. The homepage features three primary treatment types, accompanied by visual explanations of their mechanisms and application processes.
Step 3: Treatment comparisons
To facilitate informed decision making, this step offers comparisons of treatment effectiveness based on published evidence, particularly axial length outcomes. It also outlines the specific requirements for each intervention and provides a clear summary of their respective advantages and disadvantages.
Step 4: Parents’ and children's considerations
This component invites users to reflect on their expectations regarding treatment outcomes, children's daily routines and learning habits, and financial constraints. The system incorporates models of student lifestyles tailored by grade level to help contextualize choices. Parents are also prompted to record and articulate their preferences.
Step 5: Treatment decision assessment
In this step, the system evaluates the user's understanding of the treatment options and introduces the estimated costs. It includes interactive questions related to treatment knowledge. A cloud-based algorithm then matches the expressed preferences with suitable treatment options.
Step 6: Recommended decision
The final step presents a list of treatment options that align with user preferences and clinical criteria. Parents are encouraged to select and prioritize these options for discussion with their eye care provider, facilitating a collaborative and informed treatment decision.
Prototype creation and alpha testing
After consultation between the research team and software engineers, the content of the CMSDM was sequentially structured according to each decision-making step, as illustrated in Figure 2.
The DA platform was designed to guide parents through a structured and sequential decision-making process. The process begins with the input of demographic and clinical data, including the child's refractive status and diagnostic information. Following this, users access an introductory section that presents foundational knowledge of myopia, including diagnosis, evidence-based treatment options, and potential adverse effects. Depending on the treatment category selected, additional information is provided regarding the principles, mechanisms, and effectiveness of pharmacological or optical correction methods. The next section offers comparative information and summarizes the effectiveness, benefits, drawbacks, target population, and financial considerations associated with each treatment. Parents are then invited to reflect on a range of personal and contextual factors, such as visual quality, possible side effects, convenience, treatment costs, and follow-up needs. A knowledge assessment is provided to ensure comprehension before making a final decision. The process concludes with the submission of selected preferences that can be used to support discussions with the ophthalmologist. Based on the ratings of five experts, the I-CVI values ranged from 0.60 to 1.00, and the overall s-CVI using the average method (S-CVI/Ave) was 0.80, indicating acceptable content validity.
Usability testing
Fifteen participants completed the alpha testing questionnaire. System usability was assessed using the 10-item SUS. The mean score was 85 (SD = 6.5), reflecting excellent usability based on standardized interpretation guidelines. We invited 15 parents of children who were newly diagnosed with myopia and were in the early stages of treatment adaptation to test the website. The mean decision regret score before using the website was 26.03 (SD = 10.13), which significantly decreased to 20.13 (SD = 11.03) 3 months later (t = 2.01, p < 0.05, df = 14; Cohen's d = 0.52). Similarly, knowledge of myopia control significantly improved after using the CMSDM, with the mean score increasing from 3.21 to 4.32 (t = 2.31, p < 0.01, df = 14; Cohen's d = 0.60).
Feedback on DA content
Qualitative feedback from both ophthalmologists and parents highlighted several areas that required improvement (Table 3). In the Entry section, they noted confusion around myopia degree selection and treatment options. The Introduction required clearer explanations of corneal structure and axial length. For Treatment Options, suggestions included simplified descriptions, age guidance for Ortho-K, larger fonts, and the use of color to emphasize key content. The benefit of combining treatments was also mentioned.
Summary of feedback on each section of the decision aid.

In the Preference Assessment, where parents selected decision factors, the original three-point scale was considered unclear. The respondents recommended removing the neutral option. In the Treatment Decision section, both groups suggested showing selected options as decision outputs and prompting follow up if no decision was made.
Following user testing, the CMSDM was revised based on participant feedback and finalized by the research team (see Supplemental Appendix 3).
Website security and privacy considerations
The CMSDM was intentionally developed following a Privacy by Design approach, 42 emphasizing data minimization and user protection. The platform requires no account registration or password authentication and does not collect, store, or process any personally identifiable information. Users access only general educational content, and no sensitive health or demographic data are retained. All communications between users and the platform are secured through HTTPS encryption, ensuring data integrity and confidentiality during transmission. This design choice reduces potential privacy risks commonly associated with digital health websites that embed third-party requests or complex consent controls, as demonstrated previously. 42 By eliminating unnecessary data collection and simplifying user access, the CMSDM minimizes privacy concerns while maintaining usability, accessibility, and compliance with contemporary digital health privacy principles.
Discussion
To our knowledge, this is the first study to develop a DA to support SDM in pediatric myopia control. It incorporates IPDAS standards and SDM while evaluating usability, decision regret, participation, and visual outcomes and offers interactive, personalized support to enhance informed parental decision-making and treatment adherence. Owing to time constraints in clinical consultations, ECPs must clearly explain myopia treatment options, making DAs essential. Liverpool and Edbrooke-Childs 43 noted that this DA system was designed to prioritize the assessment of patients’ readiness for decision making. For instance, in the DA system developed for refractive surgery, Aghdam, Zarei 26 adopted an exploratory needs assessment approach, which integrated ophthalmologists’ clinical expertise with patients’ perspectives and aimed to enhance decision readiness by identifying essential “must-know” information for informed choices. The system prioritized content that reflected both professional knowledge and patient priorities, thereby ensuring that the DA effectively supported both informed decision making and SDM.
Treatment options for childhood myopia have been extensively synthesized through systematic reviews and discussed in clinical and academic forums. The incorporation of preferences from both children and parents in the decision-making process is increasingly emphasized. 44 DAs augment treatment discussions by integrating evidence-based outcomes with the subjective experiences of families. 45 Recent research has explored the distinctions between pharmacological and optical interventions for myopia control, supported by a growing body of clinical evidence. 46 The current study structured the DA around two separate modules, pharmacological treatment and optical correction, to better align with varying visual needs and progression patterns. This structure allows parents to select an appropriate DA module based on their child's myopia progression, facilitating personalized and informed decision making. This study focused on the treatment of children within 1 year of an initial myopia diagnosis. According to Gifford et al. 30 and Leshno et al., 47 initial management is typically initiated with a single treatment, and therefore, combined therapy options were not included in the present DA. The necessity of incorporating such options warrants confirmation in future study.
In addition, the integration of the literature and clinical guidelines in developing DAs allowed for a more objective incorporation of both parental preferences and ophthalmologists’ recommendations. This approach enabled parents to better understand myopia treatment options and make informed decisions based on their needs and those of their children. This aligns with findings from the systematic review by Salaky et al., 44 which demonstrated that DAs across various healthcare contexts improve patients’ understanding of available options, enhance awareness of risks, increase perceived knowledge, and strengthen confidence in decision making. These findings align with the user feedback and outcome indicators from the pilot testing phase of this study. Specifically, the issue of treatment costs, often considered by ECPs but rarely discussed directly, has been highlighted. 48 Using DAs facilitates more transparent communication between parents and ECPs, shifting the treatment decision-making process from a focus on efficacy to one that considers family needs, children's daily routines, and the overall visual quality. Although axial length is often referenced in research as a key indicator of myopia control efficacy, many parents in clinical settings report a lack of information regarding changes in axial length. 6 Consequently, they often cannot evaluate treatment effectiveness and are less likely to attend regular follow-up appointments, especially if they are unaware that axial length is crucial for monitoring myopia progression. 49 In this study, ophthalmologists highlighted the importance of explicitly presenting information in the treatment comparison section of DAs. Given that “effectiveness” is a primary concern for parents in treatment selection, it is crucial to provide clear, evidence-based data on treatment efficacy to support informed decision making. Integrating evidence-based research into a DA website is the most compelling component of an SDM platform.
Limitations and future implementation
This study describes the development of a web-based DA platform, the CMSDM. However, the relatively small sample size used for usability testing limits our ability to conduct subgroup analyses (e.g. by age group, education, or digital literacy) and restricts the generalizability of the findings. Nonetheless, this preliminary pilot study offers critical insights. Future large-scale and demographically diverse studies are warranted to mitigate this limitation and strengthen the generalizability of the findings.
Moreover, the current version of the CMSDM was developed solely in Mandarin to address the needs of Taiwanese parents. While this ensured cultural and contextual relevance, it limits the aid's broader applicability. Translation and cultural adaptation into other languages will be essential for wider dissemination and use across diverse populations. Furthermore, as the DA was designed to be used within 1 year of an initial myopia diagnosis in children, and in accordance with prior studies that recommend initiation with a single treatment modality, combination therapies, such as low-dose atropine plus orthokeratology, were not included in the current version. The necessity and feasibility of incorporating such integrated options should be evaluated in future studies.
Future large-scale, randomized controlled trials should evaluate the effectiveness of the tool by comparing CMSDM with standard educational materials, focusing on differences in knowledge acquisition, decision-making quality, and adherence. A larger sample size and a more demographically diverse parent population would also provide deeper insights into subgroup variations. Collectively, the insights gained from this pilot study will inform the ongoing optimization of CMSDM, enhancing its relevance, inclusivity, and utility across diverse clinical and cultural contexts.
The CMSDM is mobile-compatible, and future applications may incorporate interactive features, such as progress tracking, feedback loops, and improved mobile optimization to enhance accessibility, parental engagement, and adherence. Since the CMSDM is a preparatory tool for parents to be used independently before clinical consultations with ophthalmologists or optometrists, thereby facilitating more informed discussions during consultations, treatment adherence was not evaluated in this study. Future research should incorporate real-world measures of adherence—such as clinic follow ups, continuation or drop-out rates, and compliance with orthokeratology or atropine regimens—to rigorously assess the CMSDM's impact beyond decision-making support.
Conclusions
This study demonstrated the potential of CMSDM as a DA for parents of children with myopia, especially regarding optical correction. By providing relevant knowledge, the tool enhances parents’ capacity for informed decision making. The CMSDM supports ophthalmologists in the preconsultation process by streamlining discussions, especially with parents who already possess a basic understanding. CMSDM streamlines the explanation of treatment options, enabling more focused and efficient clinician-parent communication and ultimately improving interactions between parents and ECPs. Future studies involving larger and more diverse populations are warranted to further establish the role of CMSDM in supporting parental decision making regarding childhood myopia.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076251395560 - Supplemental material for A web-based, user-centered decision aid to support parental choices in myopia control: A development and usability study
Supplemental material, sj-docx-1-dhj-10.1177_20552076251395560 for A web-based, user-centered decision aid to support parental choices in myopia control: A development and usability study by Li-Ling Hung, Chi-Chin Sun, Li-Ling Liao, Li-Chun Chang and Hsiao-Jung Chen in DIGITAL HEALTH
Footnotes
Acknowledgments
The authors express their appreciation to the participating parents, healthcare professionals, and research staff, whose contributions were essential to the design, implementation, and assessment of the CMSDM decision support tool.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Chang Gung Memorial Hospital (IRB No. 202102324B0C502). All participants provided written informed consent prior to their inclusion in the study.
Author contributions
Material preparation was performed by LLH, CCS, LLL, LCC, and HJC. LLH conceptualized the study and supervised the project. CCS, LLL and HJC conducted data analysis and system testing. The first draft of the manuscript was written by LCC. All the authors contributed to the study conception and design, commented on posterior versions of the manuscript and also read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Chang Gung Memorial Hospital, National Science and Technology Council, (grant number BMRP 978, NSTC 111-2314-B-255 -004 -MY3).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Access to any information such as the study protocol or anonymized data can be available upon reasonable request.
Guarantor
Dr L-C Chang and H-J Chen are the guarantors of this work and take full responsibility for the integrity of the data and the accuracy of the analysis and interpretation.
Peer review
This manuscript was peer reviewed by independent experts through a double-blind process.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
