Abstract
Objective
Drug poisoning is the most prevalent type of poisoning throughout the world that can occur intentional or unintentional. Standard way for data gathering with uniform definitions is a requirement for preventing, controlling and managing of drug poisoning management. The purpose of this study was to develop a minimum data set, as an initial step, for a drug poisoning registry system in Iran.
Methods
This was descriptive and cross-sectional study that was performed in 2019. As the first step a comprehensive literature review was performed to retrieve related resources in Persian and English languages. For the second step the medical records of drug poisoning patients at Afzalipour hospital affiliated to Kerman University of Medical Sciences were assessed. Related data from these two steps were gathered by a checklist. Finally, a questionnaire that was created based on the checklist data elements and had three columns of ‘essential,' ‘useful, but not essential', and ‘not essential' was used to reach a consensus on the data elements. Then the content validity ratio and the mean of experts’ judgments were calculated for each data element. The Cronbach’s alpha value for the entire questionnaire was obtained 0.9.
Results
The minimum data set of a drug poisoning registry system was categorised into the administrative part with three sections including 32 data elements, and clinical parts with six sections including 81 data elements.
Conclusion
This study provides a minimum data set for development of a drug poisoning registry system. Collecting this minimum data set is critical for helping policy makers and healthcare providers to prevent, control and manage drug poisoning.
Introduction
Drugs are one of the most dangerous and commonly used poisonous substances. 1 Drug poisoning is the most prevalent type of poisoning worldwide which can occur intentional or unintentional. Unintentional drug poisoning is occurred due to drug interactions, physiological problems in the body such as liver and kidney failure, and misuse by children. 2 Intentional drug poisoning is happened due to much consumption of drugs by adults for suicide attempts. 3 Typically, drugs that are kept at home, such as sedative pills and analgesics, are the most common cause of poisoning. 4 Drug poisoning has occurred when drugs are taken in error, used for suicide and homicide intent, prescribed drugs for someone else are taken, and self-prescribed drugs are taken with over the counter drugs and intoxication. Actually, adverse drug events are happened when prescribed drugs are taken correctly, but an adverse reaction such as an allergic reaction, idiosyncratic reaction, and drug interaction is occurred. Unintentional drug poisoning or toxic effects of drugs could be types of adverse drug events. 5
One of the most important causes of emergency department visits in the USA, is drug poisoning.6,7 In Iran, about 70% of poisonings referring to drug information centres are drug poisonings. According to the Iranian Forensic Medicine Organization, nearly 5500 Iranian people die every year due to poisoning, of which 700 deaths are due to drug poisoning. 8
Due to the large number of referrals drug poisoning patients to hospitals, recording the patients' data in order to prevent, control and manage treatment can be very helpful and facilitate evidence-based decision making in health system policies and plans. 9 Electronic record of patient health information while having a dynamic and flexible structure provides for data collection and reporting evidence-based, up-to-date and accurate health information. 10 Drug poisoning registry systems, as types of health information systems, may facilitate reporting the incidence and prevalence of diseases, temporal and spatial changes, the quality of care assessment, and improve research by collecting, analysing and reporting regularly health information of a specific population. 11
Some developed countries such as the USA (Toxicology Investigators Consortium (ToxIC)) 12 and Australia (Hunter Area Toxicology Service (HATS)) 13 have constructed their poisoning registry for gathering data about poisoning cases; in addition, The World Health Organization has provided International Toxicology (INTOX) software for data management of poison control centres. 14 However, there is no specific drug poisoning registry in Iran and some other countries.
The initial step in managing disease information for quality of care improving and controlling disease, is designing a minimum data set (MDS). 15 Development of a MDS, as the core health data element, for gathering data with uniform definitions in a standard way to ensure comparability is very important. 16 Using an MDS in a disease registry system is necessary to access reliable and comparable information about the number of patients, therapeutic methods, and outcomes of the provided health services relating to a specific disease. 17
Since there is no registry system for drug poisoning in Iran, the design and creation of a MDS, as the first step, is essential for the collection, processing, analysis and reporting of appropriate drug poisoning information in order to maintain and improve the health of the community. The aim of this study was to develop a MDS for drug poisoning registry system in Iran.
Methods
This study was conducted in three phases including (a) identification of available drug poisoning MDS, (b) questionnaire development, and (c) final version of MDS development, and these phases are presented in the following sections.
Identification of available drug poisoning MDS
This descriptive and cross-sectional study was conducted in 2019. We aimed to develop a MDS for a drug poisoning registry system. In the first step to retrieve related resources (articles, reports, forms and theses), scientific databases such as the Web of Knowledge, PubMed, Scopus, Google Scholar, SID, MagIran and Google search engine were reviewed by keywords including ‘poisoning data', ‘drug poisoning data', ‘drug poisoning registry', ‘poisoning registry form', ‘drug poisoning registry system MDS', ‘minimum data set', ‘data set', and ‘data element from'. The inclusion criteria were articles, reports, forms and theses in Persian and English languages, published from 2000–2018. Letters to the editor, short communication, articles with no abstract and full text, not specifically related to the poisoning registry and not available MDS were excluded. Data were extracted from the related retrieved resources and entered into the checklist with two administrative and clinical sections. In the second step, the medical records of drug poisoning patients at Afzalipour hospital affiliated to Kerman University of Medical Sciences were assessed and related data were also entered to the checklist.
Questionnaire development
A questionnaire was developed using the data elements of the checklist and had three columns ‘essential', ‘useful, but not essential', and ‘not essential'. For adding necessary data by experts, a blank row was considered at the end of the questionnaire. The content validity of the questionnaire was assessed by an expert panel including two health information management and two clinical toxicologist experts. Cronbach's alpha was used to evaluate the reliability of the questionnaire. The collected data were analysed using SPSS 19, and the Cronbach’s alpha value for the entire questionnaire was obtained 0.9; then, the reliability of the questionnaire was confirmed.
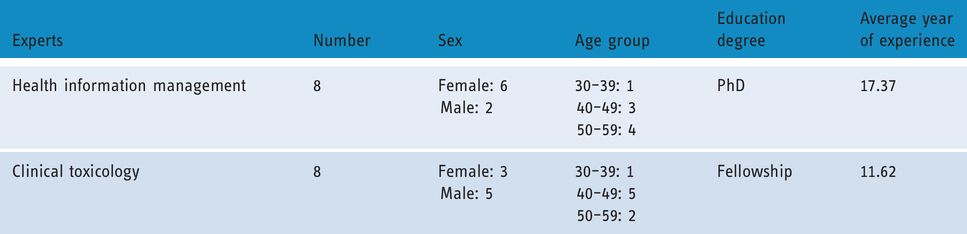
Research samples for determination of a MDS for drug poisoning registry system were eight experts of health information management departments of Tehran and Kerman Universities of Medical Sciences, and eight clinical toxicology experts working in Loghman Hakim, Baharloo and Afzalipour Hospitals affiliated to Shahid Beheshti, Tehran, and Kerman Universities of Medical Sciences, respectively. Table 1 shows the demographic characteristics of these experts.
Demographic characteristics of experts who participated to validate the preliminary data elements.
The questionnaire was given to the experts and we asked health information management experts to complete the administrative section and clinical toxicologists the clinical section.
Final version of MDS development
The questionnaire was analysed using the following formula for Content Validity Ratio (CVR):

‘Ne’ is the number of experts who have chosen the ‘essential’ option for an element, and ‘N’ is the total number of experts. In the first step, CVR was calculated for each data element and was compared with the Lawshe table value. 18 Additionally, the mean of expert judgments was also calculated for each data element by replacing the number 2 for ‘essential', the number 1 for ‘useful, but not essential', and the number 0 for ‘not essential' in the columns. Each data element that gained a value equal or greater than 0.75 for CVR (considering the number of eight experts) was accepted and used as starred item in the registry system that must be filled. If the value of CVR was between 0 and 0.75 and the mean of judgments was equal and greater than 1.5 for a data element, it was also accepted and used as usual item in the registry system that could be filled. The data elements for which the value of CVR was less than 0 and the mean of judgments was less than 1.5 were refused.
Results
Identification of available drug poisoning MDS
Final retrieved resources for the MDS collection phase included ToxIC of USA, 12 HATS of Australia, 13 Northern Ireland registry of self-harm, 19 National Poison Data System (NPDS) of USA, 20 National Poisons Information Centers (NPIC) of Australia, 21 and INTOX software of the World Health Organization. 14 The MDS for developing a drug poisoning registry system was categorised into the administrative and clinical parts. The administrative part has three sections including: demographic information (23 items), presentation information (9 items) and discharge information (6 items) (Table 2). The clinical part has six sections including: diagnostic information (3 items), exposure information (20 items), medical history (17 items), clinical findings (20 items), diagnostic intervention (22 items) and treatment information (6 items) (Table 3). The clinical data were related to control vocabularies of International Classification of Diseases 10th Revision (ICD-10) (T36–T50) and International Classification of Health Interventions (ICHI).
Administrative minimum data set (MDS) for drug poisoning registry system design.
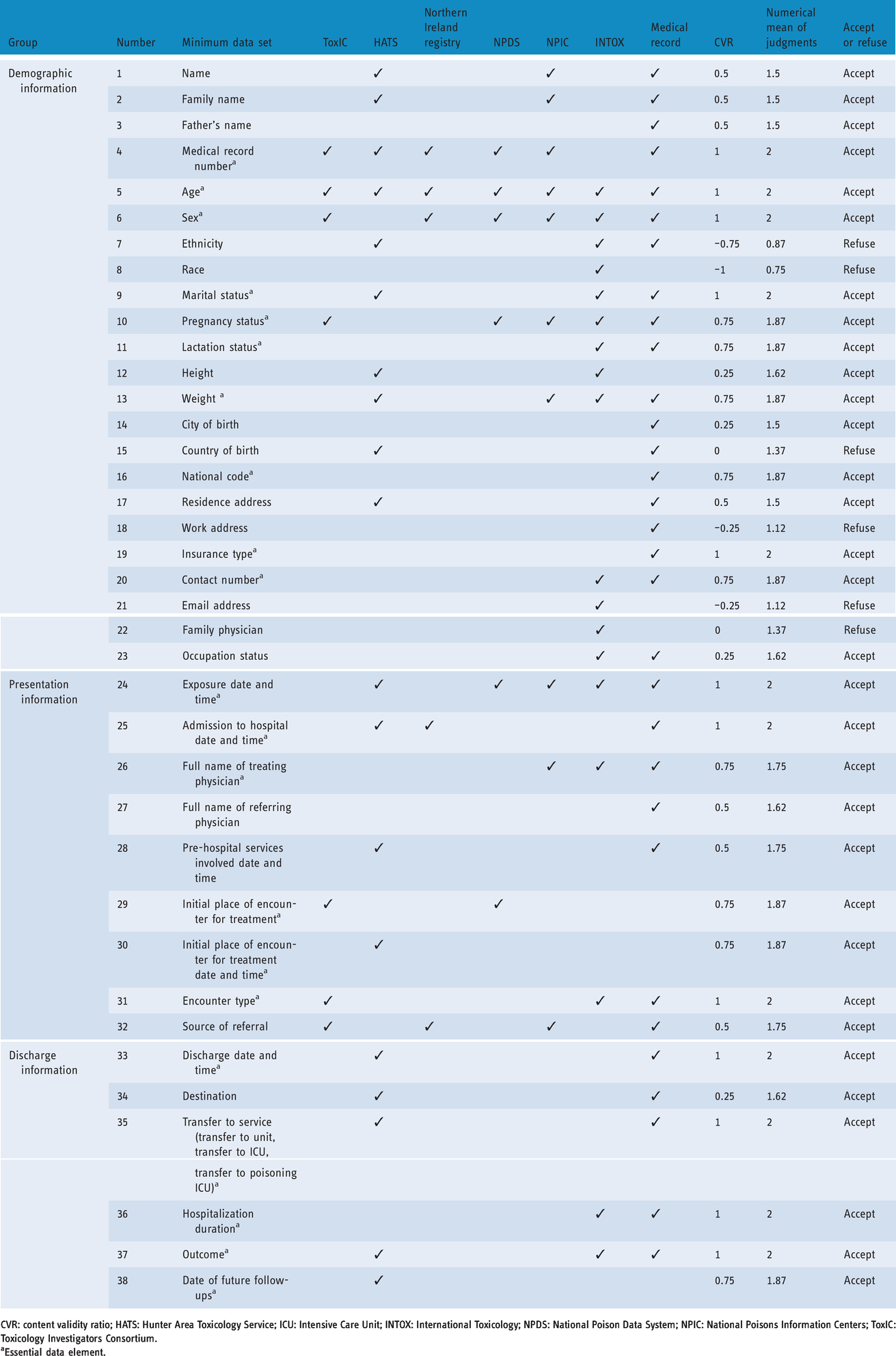
CVR: content validity ratio; HATS: Hunter Area Toxicology Service; ICU: Intensive Care Unit; INTOX: International Toxicology; NPDS: National Poison Data System; NPIC: National Poisons Information Centers; ToxIC: Toxicology Investigators Consortium.
aEssential data element.
Clinical minimum data set (MDS) for drug poisoning registry system.

AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; ALP: Alkaline Phosphatase; Bs: Blood Sugar; BUN: Blood Urea Nitrogen; CBC: Complete Blood Count; Cr: Creatinine; CVR: content validity ratio; EMS: Emergency Service; HATS: Hunter Area Toxicology Service; INR: International Normalized Ratio; INTOX: International Toxicology; NPDS: National Poison Data System; NPIC: National Poisons Information Centers; PT: Prothrombin Time; PTT: Partial Thromboplastin Time; ToxIC: Toxicology Investigators Consortium; U/A: Urine Analysis; VBG: Venous Blood Gases.
aEssential data element.
Final version of MDS development
Out of the total 126 data elements, 13 were refused (6 administrative and 7 clinical data elements) because the value of CVR was less than 0 and the mean of judgments was less than 1.5. Six refused data elements in the administrative part were ethnicity, race, country of birth, work address, email address and family physician. Seven refused data elements in the clinical part were chemical formula of drug, drug colour, drug constituent units, drug country of origin, drug legal category, drug hazards and contraindications, and drug provided place.
Of the 113 remaining data elements 88 were determined as essential and starred in the registry system that must be filled (21 administrative and 67 clinical data elements), and 25 were determined as usual items in the registry system that could be filled.
Discussion
This study showed that, despite the prevalence of poisoning in Iran, there is no registry system in this specific field, particularly in the drug poisoning area. Based on the study findings, the MDS for a drug poisoning registry system is divided into an administrative part with demographic information, presentation information and discharge information sections, and a clinical part with diagnostic information, exposure information, medical history, clinical findings, diagnostic intervention and treatment information sections. These results are consistent with the other study that determined the MDS for poisoning with acidic and alkaline substances information management system. 22
Considering the importance of the poisoning issue and the high prevalence of poisoning, other countries in the world have started to set up service centres in the area of poisoning; thus, they provide specialised information to the public and medical staff. In these poison control centres, toxicological information, counselling services for the management of people with poisoning, providing analytical laboratory services, training and research for the prevention and treatment of poisoning is provided. 23 The World Health Organization has introduced software called the INTOX Data Management System, specifically designed for poison control centres. 14 This software provides the ability to create integrity in various databases and facilitates tracking of contacts, storing information and statistical analyses. The information contained in this software includes demographic information, the main reason for referring, how to refer, exposure information, exposure reason (intentionally and unintentionally), the type of toxic substance, clinical symptoms, treatment information and clinical outcomes. 14 Sex, age, ethnic origin, marital status, pregnant status, lactating status, height, weight and occupation are considered as demographic data elements in this software. These elements are almost consistent with the demographic information in the present study; however, ethnic origin element was refused because it attained a CVR value less than 0 and the mean of judgments less than 1.5. Product colour, product constituent units, product country of origin, product legal category, product hazards and contraindications clinical data elements that collected in INTOX Data Management System, were refused in the present study.
The ToxIC registry was established by the American College of Medical Toxicology for surveillance and research purposes. 12 This registry collected the following MDS: demographic information, exposure information, agent information, clinical syndromes, symptoms and signs information, and treatment information. 12 The demographic and exposure information of this registry is more limited than the present study. In the ToxIC registry only the name of the institution, age, sex and patient code of the demographic information is collected. Almost other collected information is consistent with the results of the present study.
The HATS in New South Wales, Australia was developed as a database for gathering data about poisoning cases. 13 This database collected the following MDS: demographic information, exposure information, presentation information, past history, clinical examination, psychiatric consultation, treatment information, outcome, discharge and follow-up information. 13 The demographic information in this data base includes medical record number, name of patient, date of birth, country of birth, ethnic origin, marital status, height, weight, employment status and address. The demographic information in the present study is wider, but the element of country of birth and ethnic origin were refused from the MDS because they achieved a CVR value less than 0 and mean of judgments less than 1.5. Other main collected information is consistent with the drug poisoning MDS in the present study.
Similar to the other countries, the most common cause of poisoning in Iran is the consumption of drugs in excess of the amount for treatment.8,24 This can be because of the accessibility of pharmaceutical drugs in Iran. For example, the availability of benzodiazapines without prescription in some cases is a major cause of using them for suicide commitment. 25 Using a drug registry system that collects standardised data to identify the causes and control this type of poisoning is necessary.
The development of a MDS is the most basic task in the data collection of a disease registry system for guiding and supporting healthcare providers and using the data for administrative and research goals. Data sharing and interoperability can improve by using a proper MDS.26,27 Therefore, the development of MDS by standardization of data element is essential.
In this study, the sample size of expert panel involved eight experts of health information management departments of Tehran and Kerman Universities of Medical Sciences, and eight clinical toxicology experts working in Loghman Hakim, Baharloo and Afzalipour Hospitals. However, these experts were selected from two provinces of Iran; the addition of more expert participation from other medical universities across the country is suggested to yield higher validity for national MDS development.
Conclusion
A registry system for drug poisoning data is needed which has two main components that are administrative and clinical with three and six subcomponents respectively. Collecting these data elements as accurate epidemiological data about drug poisoning is critical for helping policy makers and healthcare providers to prevent, control and manage this condition.
Footnotes
Acknowledgments
The authors thank all of the clinical toxicologists and health information management experts that cooperated with them to complete questionnaire.
Conflict of interest
The authors declare that there is no conflict of interest in this study.
Contributorship
S.HG and SH.R.NK participated in the collection and analysis of data. All authors contributed in the design of the study, drafting the article and approval of the final version of the article.
Ethical approval
This study does not need any ethical committee approval and patient consent.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Guarantor
Sharareh R Niakan Kalhori.
Peer review
This manuscript was reviewed by reviewers, the authors have elected for these individuals to remain anonymous.
