Abstract
Case summary
A 14-year-old spayed female Scottish Fold cat was presented to a private veterinary clinic for a health check. Radiography revealed a nodule measuring 1.0 cm diameter in the cranial segment of the cranial lobe of the left lung. On day 12, non-contrast-enhanced CT revealed a well-defined pulmonary nodule measuring 1.7 cm in diameter in the same area. On day 48, the nodule in the cranial segment of the left cranial lobe was resected by lobectomy. The nodule was firm and pale pink in surface colour. Microscopically, the mass was adjacent to the bronchi and bronchial glands and was composed of luminal cells forming the duct structure and an outer layer of non-luminal cells with squamoid differentiation. Tumour cells partially invaded into the surrounding parenchyma. Immunohistochemically, the luminal cells were positive for cytokeratin (CK) AE1/AE3, CK7, CK19, thyroid transcription factor-1 and human epidermal growth factor 2. Non-luminal cells were positive for CK AE1/AE3, p63, calponin, smooth muscle actin and p40. The Ki-67 indices of the luminal and non-luminal cells were 10.4% and 12.8%, respectively. Based on these findings, the tumour was diagnosed as a pulmonary epithelial-myoepithelial carcinoma with high-grade transformation as defined in the human World Health Organization classification.
Relevance and novel information
This is the first report of pulmonary epithelial-myoepithelial carcinoma with high-grade transformation characterised by morphology and immunohistochemistry.
Plain language summary
This report describes a rare type of lung cancer in a cat. A 14-year-old spayed female Scottish Fold cat was taken to a veterinary clinic for a routine health check. Chest imaging unexpectedly revealed a small mass in the left lung. Follow-up CT confirmed a well-defined lung nodule, and surgical removal of the affected lung lobe was performed. Examination of the removed tissue under the microscope showed that the tumour was made up of two different types of cells. One type formed small duct-like structures, while the other type surrounded these ducts and showed features similar to cells normally found in gland-supporting tissue. Some tumour cells had begun to grow into the surrounding lung tissue. Special laboratory staining tests were used to better understand the nature of the tumour cells. These tests confirmed that the two cell populations had different biological characteristics, supporting the diagnosis of a rare biphasic lung tumour. The growth activity of the tumour cells was higher than typically seen in low-grade tumours. Based on these findings, the tumour was diagnosed as a pulmonary epithelial-myoepithelial carcinoma with features similar to a more aggressive form described in human medicine. Although this type of lung tumour has been reported in people, it has not previously been described in cats. This case provides new information about rare lung tumours in cats and may help veterinarians and pathologists better recognise and diagnose similar cases in the future.
Introduction
Primary pulmonary salivary gland-type tumours are rare neoplasms in humans and include three biphasic carcinomas: adenoid cystic carcinoma, epithelial-myoepithelial carcinoma and mucoepidermoid carcinoma.1 –5 Epithelial-myoepithelial carcinoma is composed of an inner layer of epithelial cells forming duct-like structures and an outer layer of myoepithelial cells, and has previously been referred to as adenomyoepithelioma, pneumocytic adenomyoepithelioma, epimyoepithelial carcinoma, epithelial-myoepithelial tumour and malignant tumour comprising epithelial and myoepithelial cells.1,6 Among pulmonary malignant epithelial tumours are neuroendocrine tumours, including combined small cell carcinoma and combined large neuroendocrine carcinoma, which consist of a mixture of endocrine carcinomas and epithelial malignant tumours such as adenocarcinoma and squamous cell carcinoma.7,8 These tumours can be differentiated by morphology and immunohistochemical staining.1 –8
In cats, primary pulmonary tumours are extremely rare, accounting for approximately less than 1% of all tumours. Among these, pulmonary carcinomas are the most frequently reported, with adenocarcinoma accounting for approximately 70–85% of cases and often presenting clinically as lung-digit syndrome.9,10 Previous clinicopathological studies have demonstrated that feline pulmonary carcinomas are aggressive tumours with a high metastatic potential. 10 In domestic animals, pulmonary carcinomas have been classified based on histological features and cellular composition. Hahn and McEntee 11 also reported primary pulmonary tumours in cats and noted that feline pulmonary carcinomas may exhibit variable histological features, without proposing a specific classification system. Subsequently, D’Costa et al 12 demonstrated that feline pulmonary carcinomas frequently show marked morphological heterogeneity, with multiple histological patterns identified within individual tumours, making strict classification based on a single histological subtype difficult. Biphasic pulmonary carcinomas, however, are uncommon in cats, and their histological and immunophenotypic characteristics remain incompletely characterised. In veterinary pathology, tumours composed of epithelial and myoepithelial cells are well recognised in other organs, particularly in the mammary gland, salivary gland and apocrine gland, where they are classified as carcinoma and malignant myoepithelioma or complex carcinoma. In contrast, salivary gland-type tumours and epithelial-myoepithelial carcinoma of the lung have not been established as distinct diagnostic entities in feline oncology, and no specific veterinary classification or grading scheme exists for such pulmonary tumours.13,14 Therefore, in the present case, the human World Health Organization (WHO) classification of thoracic tumours was used solely as a comparative diagnostic framework to aid in the characterisation of a rare feline pulmonary neoplasm with biphasic epithelial and myoepithelial differentiation, within the context of established veterinary tumour classifications.
Case description
A 14-year-old spayed female Scottish Fold cat presented to a private veterinary clinic for a health check. Radiography revealed a nodule, measuring 1.0 cm diameter, in the cranial segment of the cranial lobe of the left lung. On day 12, the cat presented to another private veterinary clinic for further detailed examination. No abnormalities were found in peripheral blood or blood biochemistry tests. Non-contrast-enhanced CT revealed a well-defined pulmonary nodule measuring 1.7 cm in diameter, located in the cranial segment of the cranial lobe of the left lung (Figure 1a). The tracheobronchial lymph nodes were not enlarged, and the other organs were normal. After 48 days, the nodule in the cranial segment of the left cranial lobe was resected by lobectomy, although it had not increased in size.
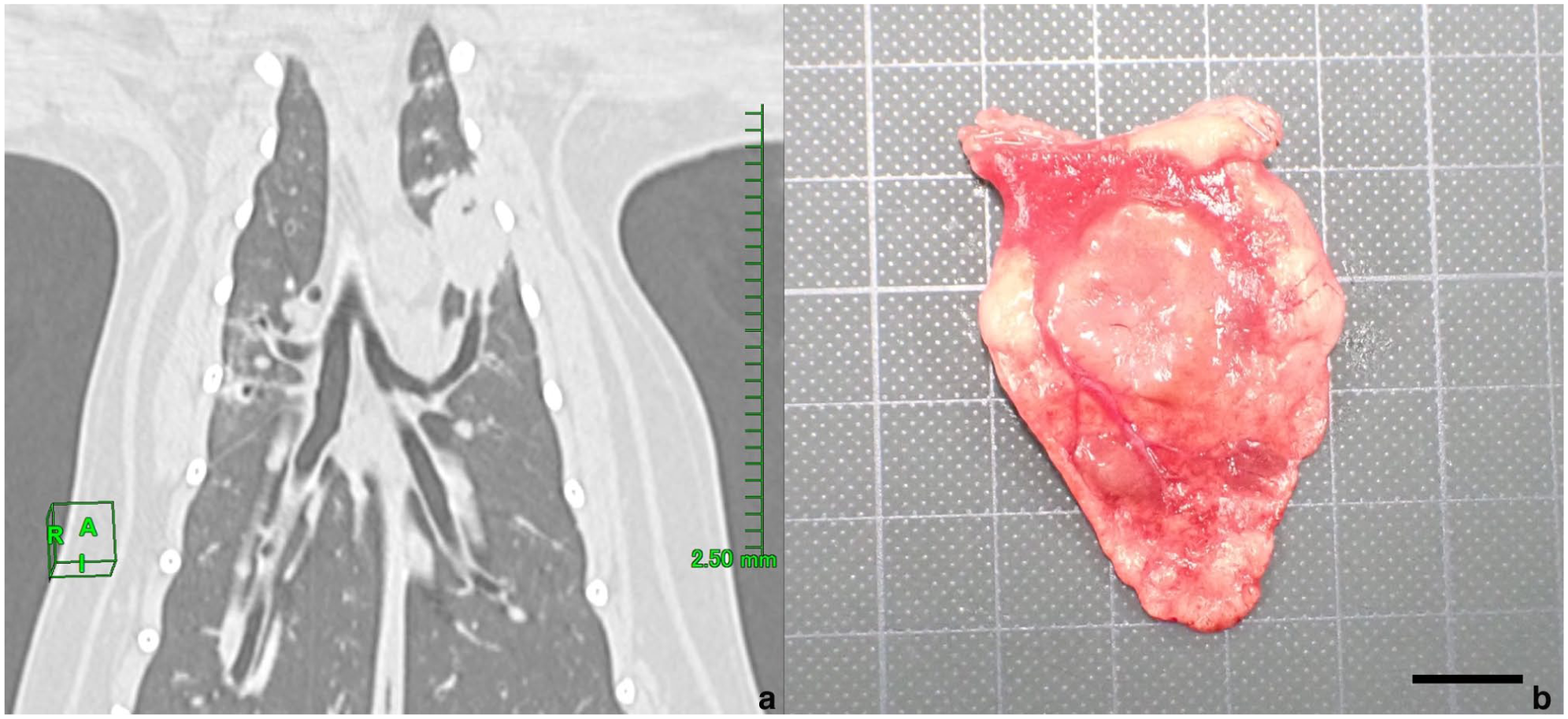
CT image and macroscopical appearance of the mass. (a) Non-contrast-enhanced CT demonstrated the presence of a well-defined pulmonary nodule measuring approximately 1.7 cm in diameter in the cranial segment of the cranial lobe of the left lung. (b) Resected mass. Bar = 1 cm
Grossly, the nodule was firm and pale pink in surface colour (Figure 1b). The resected tissue was fixed in 10% neutral-buffered formalin, processed routinely and embedded in paraffin wax, and sections stained with haematoxylin and eosin and periodic acid-Schiff (PAS). Microscopically, the mass was adjacent to the bronchi and bronchial glands, and its borders with the surrounding area were partially unclear (Figure 2a). The tumour consisted of two cellular components: luminal cells forming the duct structure and an outer layer of non-luminal cells with biphasic or solid proliferation with infiltration into the surrounding parenchyma (Figure 2b). The luminal cells were cuboidal with round nuclei and pale cytoplasm (Figure 2b). Non-luminal cells had round or short spindle-shaped nuclei and scant eosinophilic cytoplasm, and exhibited cuboidal, short spindle-shaped or squamoid differentiation (flattened epithelial morphology without true squamous differentiation) (Figure 2b). In the solid proliferation area, squamoid cells were observed from the intermediate to the central region (Figure 2c), and necrosis was often observed in the centre. Mitotic figures were more frequent in non-luminal cells than in luminal cells. Both luminal and non-luminal cells were negative for the PAS reaction. No intravascular invasion or the spread through air spaces of the tumour cells was observed. In addition, mild lymphocytic infiltration around the bronchioles, mild macrophage infiltration within the alveoli and hyperplasia of type II alveolar epithelial cells was often observed in the parenchyma.
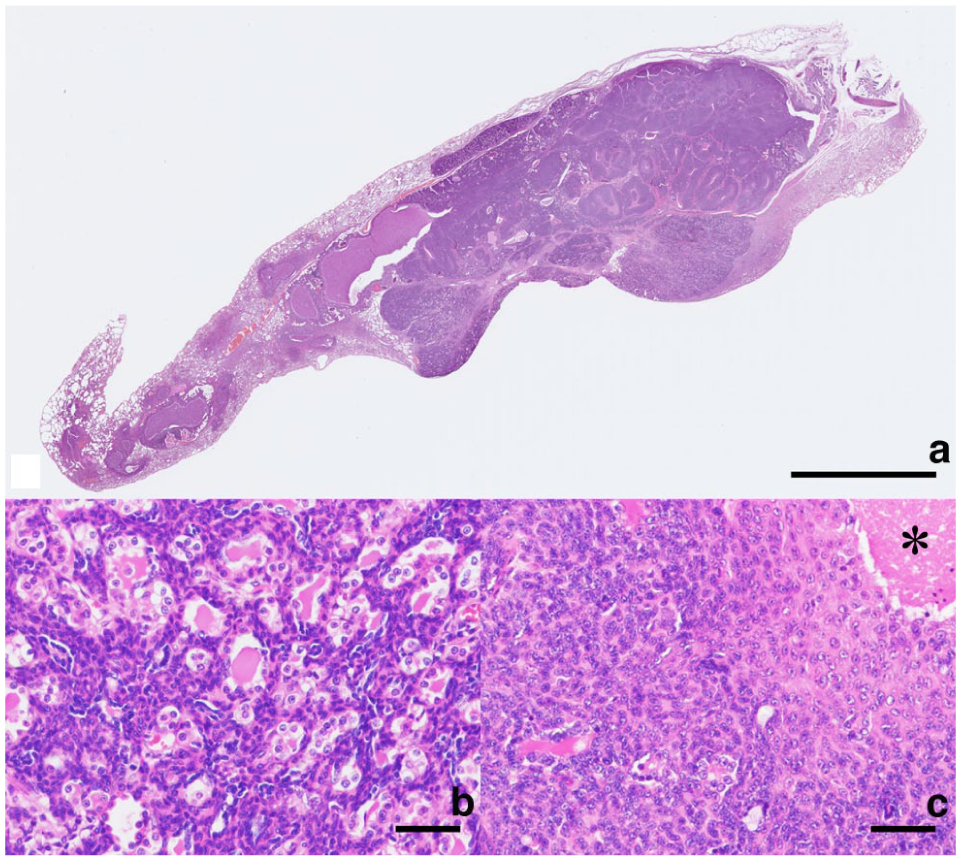
Histology of the pulmonary mass. (a) The mass is located adjacent to the bronchi and bronchial glands. Haematoxylin and eosin (H&E). Bar = 5 mm. (b) Basophilic tumour composed of an inner layer of epithelial cells forming duct-like structure and an outer layer of myoepithelial cells. H&E. Bar = 50 μm. (c) Areas of squamoid differentiation in the myoepithelial cells and necrosis (asterisk). H&E. Bar = 50 µm
Immunohistochemistry (IHC) was performed on the resected mass using labelled streptavidin-biotin and primary antibodies specific for cytokeratin (CK) AE1/AE3, CK7, CK19, smooth muscle actin (SMA), Ki-67, p63, calponin, p40, thyroid transcription factor-1 (TTF-1), human epidermal growth factor 2 (HER2), chromogranin A and synaptophysin to identify the cellular origin and proliferative activity (Table 1). The primary antibodies were validated through positive reactions with appropriate feline tissue and negative reactions when normal mouse or rabbit immunoglobulin G was substituted for antibodies. IHC revealed that the luminal cells were positive for CK AE1/AE3, CK7, CK19 (Figure 3a), TTF-1 (Figure 3b) and HER2, and negative for p63, calponin, SMA and p40. Non-luminal cells were positive for CK AE1/AE3, p63 (Figure 3c), calponin (Figure 3d), SMA and p40, and negative for CK7, CK19, TTF-1 and HER2, suggesting a myoepithelial cell immunophenotype. Both the cell types were negative for synaptophysin and chromogranin A. The Ki-67 indices of the luminal and non-luminal cells were 10.4% and 12.8%, respectively. Ki-67 staining of the squamoid cells was mostly negative. Based on the morphological and immunohistochemical findings, the tumour was diagnosed as a biphasic pulmonary carcinoma composed of epithelial and myoepithelial cells, showing features consistent with epithelial-myoepithelial carcinoma with high-grade transformation as defined in the human WHO classification.
Primary antibodies used in the immunohistochemistry analysis

HER2 = human epidermal growth factor receptor 2; TTF-1 = thyroid transcription factor-1
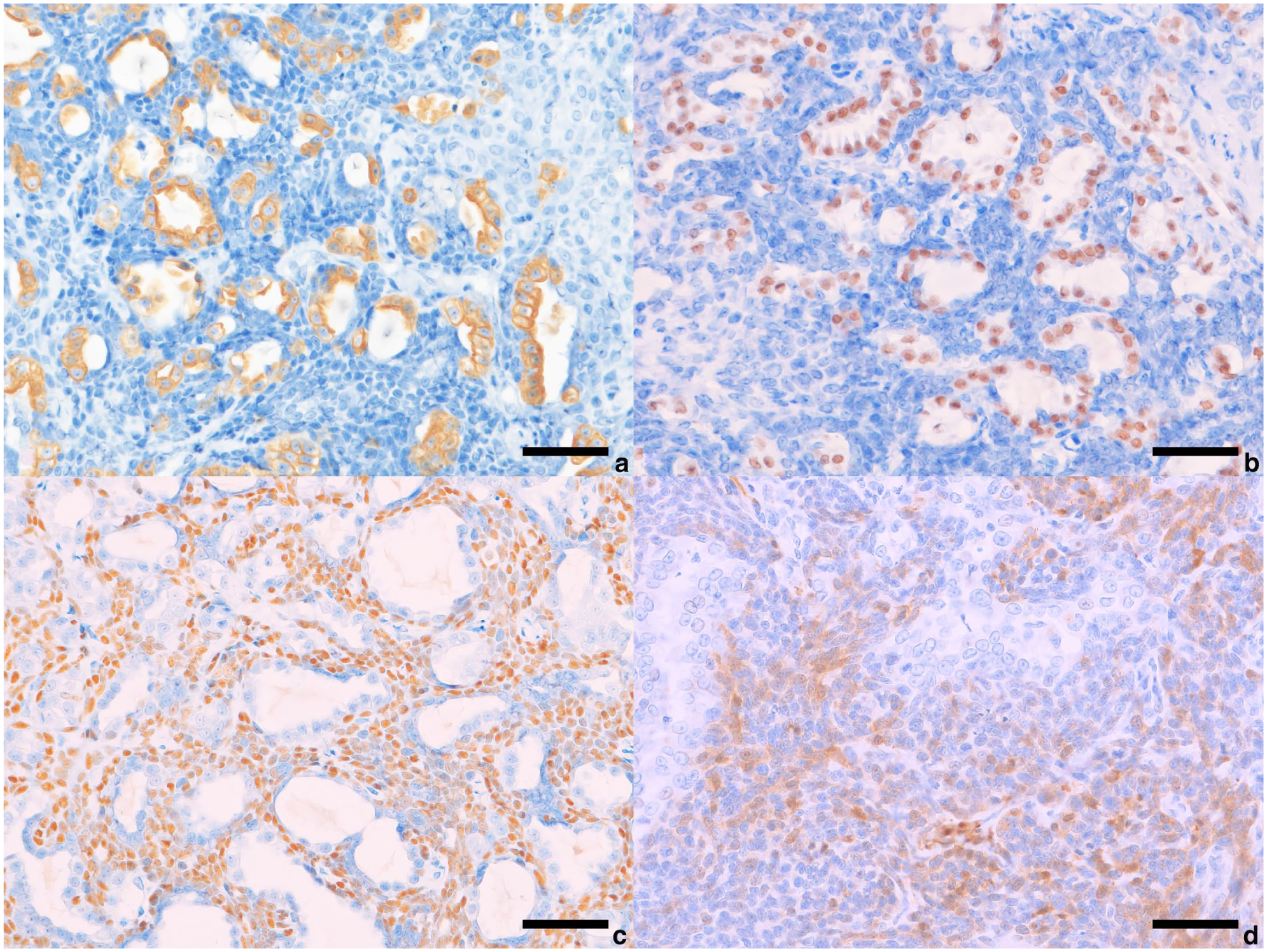
Immunohistochemistry of pulmonary mass. (a) Epithelial tumour cells immunopositive for cytokeratin 19. (b) Epithelial tumour cells immunopositive for thyroid transcription factor-1. (c) Non-epithelial tumour cells immunopositive for p63. (d) Non-epithelial tumour cells immunopositive for calponin. Bar = 50 µm
At 125 days after lobectomy, no recurrence or metastasis was observed on regular radiography.
Discussion
Pulmonary combined carcinoma characterised by biphasic cell proliferation is uncommon in domestic animals but occurs more commonly in cats than in dogs.13,14 However, the immunohistochemical characteristics of feline combined carcinoma include pan-cytokeratin, CK5 and TTF-1, and the immunophenotypes of myoepithelial and endocrine markers remain unclear. 14 Therefore, the WHO classification of human lung tumours was useful to determine the diagnosis of this tumor.1 –3,7,8 The differential diagnoses of this tumour included three biphasic tumours, including adenoid cystic carcinoma, epithelial-myoepithelial carcinoma and mucoepidermoid carcinoma, and two combined neuroendocrine tumours, including combined small cell carcinoma and combined large cell neuroendocrine carcinoma. Adenoid cystic carcinomas are composed of epithelial and myoepithelial cells, and have three growth patterns: tubular, cribriform and solid.3,6 Mucoepidermoid carcinomas are composed of mucin-secreting cells, squamoid cells and intermediate-type cells.2,6 Combined small cell carcinoma and combined large cell neuroendocrine carcinoma are characterised by a mixture of tumour cells expressing for synaptophysin and chromogranin A and malignant epithelial tumours such as adenocarcinoma and squamous cell carcinoma. These tumours were excluded based on their morphological and immunophenotypic characteristics.
Primary pulmonary salivary gland-type tumours usually originate from the epithelium of the submucosal tracheobronchial glands and typically present as endoluminal lesions, with approximately 20–30% occurring in the peripheral lung parenchyma.1,6 This tumour formed a mass in the lung parenchyma adjacent to the bronchus, corresponding to a rare type of tumour in humans. The cellular origin of this tumour is thought to be derived from the submucosal bronchial gland epithelium.
Pulmonary epithelial-myoepithelial carcinoma is generally low-grade malignancy; however, cases exhibiting high-grade features or so-called high-grade transformation have been described in human pathology.1,6 In humans, high-grade transformation is characterised by a constellation of histological findings, including marked cellular pleomorphism, anisonucleosis, loss of the typical biphasic architecture due to solid proliferation, increased mitotic activity, infiltration into surrounding tissues and tumour necrosis.1,4,15 –19 As the degree of malignancy increases, the myoepithelial component may display diverse morphological features, such as spindle-shaped, plasmacytoid, squamoid and/or cleared cytoplasm phenotypes.15,19 In addition, the Ki-67 index is typically below 10% in low-grade epithelial-myoepithelial carcinoma, whereas values exceeding 10% have been reported in high-grade cases. 4 In the present case, the tumour showed infiltrative proliferation into the surrounding tissues, morphological heterogeneity of the myoepithelial cells including squamous differentiation, and a Ki-67 index exceeding 10% in both luminal and myoepithelial cell populations. Although definitive evidence of biological transformation cannot be established in a single feline case, and no formal grading system exists for epithelial-myoepithelial carcinoma or related biphasic tumours in animals, these findings are comparable to histological and immunohistochemical features used to define high-grade transformation in the human WHO classification.
Clinically, epithelial-myoepithelial carcinoma with high-grade transformation arising in the salivary glands in humans is associated with aggressive behaviour, including lymph node metastasis and recurrence, whereas primary pulmonary epithelial-myoepithelial carcinoma with high-grade transformation has not consistently been reported to show lymph node metastasis or recurrence.16,19 In the present case, no recurrence, lymph node enlargement or distant metastasis was observed at 125 days after surgical resection. Taken together, these observations suggest that the tumour may share certain pathological features and short-term clinical behaviour with human pulmonary epithelial-myoepithelial carcinoma exhibiting high-grade features. Further investigation of cases and long-term follow-up are required to better characterise the pathological spectrum and biological behaviour of pulmonary epithelial-myoepithelial carcinoma in cats.
The application of the human WHO tumour classification in this case does not aim to introduce or establish a human disease entity, such as pulmonary salivary gland-type tumour, within feline oncology. Rather, it is used strictly as a comparative diagnostic framework to describe the morphological and immunophenotypic features of a rare feline pulmonary neoplasm composed of epithelial and myoepithelial cells, for which no specific veterinary classification or grading scheme currently exists. Importantly, the tumour in the present case is discussed primarily within the context of established veterinary pathology concepts, including biphasic and combined pulmonary carcinomas as described in domestic animals. Reference to the human WHO classification is limited to facilitating clearer communication of histological patterns and immunohistochemical characteristics and does not imply direct transposition of human tumour entities into veterinary classification systems.
Conclusions
This is the first report of feline primary pulmonary biphasic carcinoma composed of epithelial and myoepithelial cells, showing histological features consistent with epithelial-myoepithelial carcinoma with high-grade transformation as defined in the human WHO classification. Recognition of this tumour expands the spectrum of feline pulmonary neoplasms and highlights the value of a comparative pathology approach when established veterinary classifications are lacking.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
