Abstract
Case summary
A 14-year-old spayed female domestic shorthair cat was referred for evaluation of a laryngeal mass. The cat had a 2-month history of stridor and hoarseness, without any reported decrease in appetite. Laryngeal ultrasonography, fluoroscopy, endoscopy and contrast-enhanced CT revealed a 1.7 cm laryngeal mass without metastasis. A biopsy obtained under laryngoscopic guidance confirmed squamous cell carcinoma (SCC). The cat underwent stereotactic radiation therapy (SRT) in three 10 Gy fractions on consecutive days (total 30 Gy). Mild drooling occurred during treatment and was controlled with prednisolone. A partial response was achieved 45 days after SRT, with improvement in presenting signs. The cat maintained remission without late adverse effects until day 520; however, progressive disease was evident on day 603, and the cat died on day 644.
Relevance and novel information
Feline laryngeal SCC is rare, carries a poor prognosis and lacks established treatment protocols. Traditional surgery, such as laryngotomy with or without tracheostomy, often leads to severe or fatal complications. Evidence on radiation therapy in this tumour type is limited. To the authors’ knowledge, this is the first report of long-term remission in a cat with laryngeal SCC treated with SRT.
Case description
Signalment, history and clinical findings
A 14-year-old spayed female domestic shorthair cat weighing 3.55 kg (body condition score 4/9) was referred to the respiratory department of the Veterinary Medical Teaching Hospital at Nippon Veterinary and Life Science University for evaluation of a laryngeal mass detected on radiographs at the referring hospital, with a 2-month history of stridor and hoarseness. The owner reported that the cat’s appetite did not decrease during this period. On the initial examination (day 1), hoarseness was present, but no palpable lymphadenopathy or abnormalities were noted on routine haematology, plasma biochemistry or venous blood gas analysis. Radiography and fluoroscopy revealed a laryngeal mass (Figure 1), with no thoracic abnormalities suggestive of secondary effects from upper airway obstruction. Echolaryngography showed a mass (longest diameter [LD] 1.3 cm) on the ventral side of the arytenoid cartilage and reduced mobility of the left arytenoid cartilage, whereas the glottic structure remained preserved, and tracheal intubation was considered feasible (Figure 2). To reduce anaesthetic risk, prednisolone (0.7 mg/kg PO q24h) was administered until endoscopy.
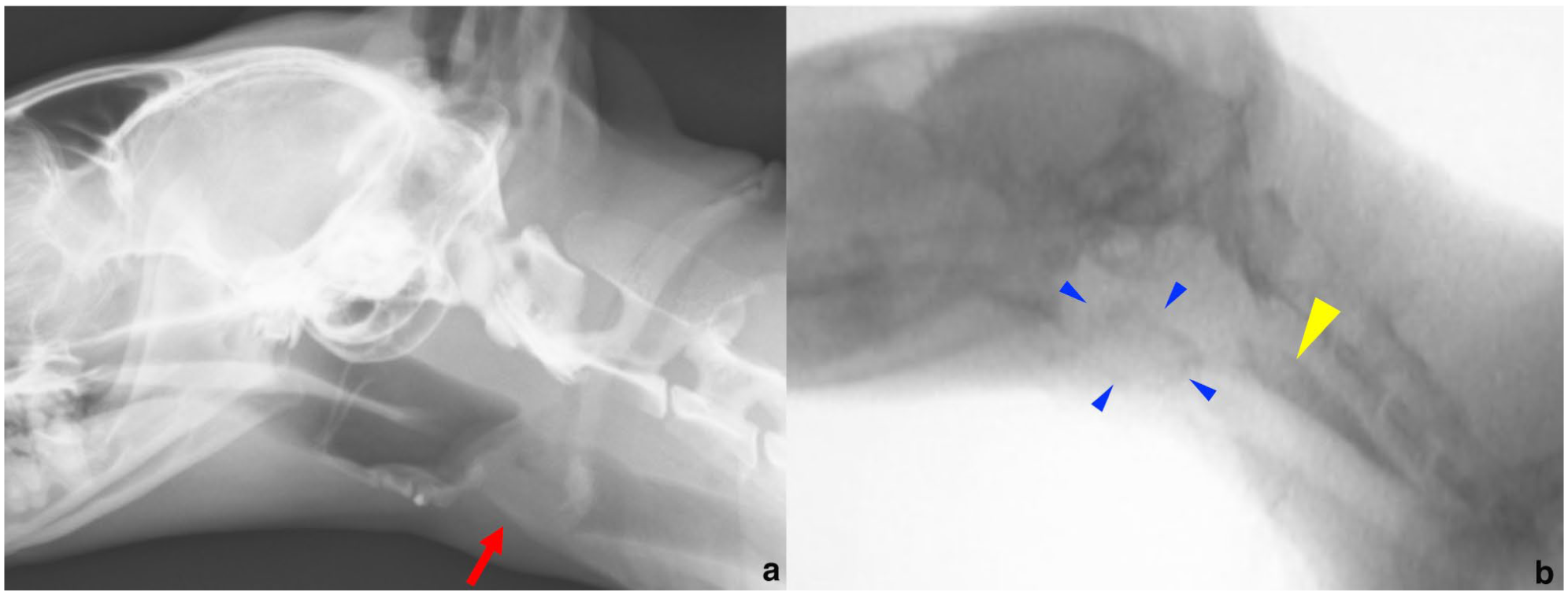
(a) Lateral laryngeal radiograph at presentation (day 1) showing a laryngeal mass (red arrow). (b) Fluoroscopic image after oral iopamidol showing a laryngeal mass (blue arrowheads). Contrast medium is visible in the oesophagus (yellow arrowhead) with partial leakage into the airway

Echolaryngographic image showing a hypoechoic mass (white arrows) in the laryngeal region. The arytenoid cartilages (right, blue arrowheads; left, red arrowheads) appear hyperechoic. The glottic structure (white asterisk, glottic cleft) was preserved
On day 14, laryngeal endoscopy under sedation was performed with a bolus of dexmedetomidine (0.5 μg/kg IV) and propofol (2 mg/kg IV to effect over 1 min). No endotracheal intubation was performed, oxygen saturation was closely monitored and sedative doses were carefully adjusted to prevent loss of spontaneous breathing. Doxapram was prepared in case spontaneous breathing stopped, and equipment for endotracheal intubation and emergency temporary tracheostomy was available if the biopsy resulted in airway obstruction. Endoscopic examination revealed a firm mass extending from the ventral to the left side of the arytenoid cartilage (Figure 3a). A biopsy confirmed squamous cell carcinoma (SCC) on histopathology. Overall, none of the aforementioned interventions (ie, doxapram administration, endotracheal intubation or temporary tracheostomy) were required. Spontaneous breathing was maintained throughout the procedure, and the cat recovered uneventfully after administration of atipamezole (2.5 μg/kg IM).

(a) Endoscopic image of laryngeal squamous cell carcinoma before stereotactic radiation therapy (SRT) on day 14. The tumour extended from the ventral to the left side of the arytenoid cartilage. (b) On day 33 (final SRT day), the mass showed mild erythema without size change. (c) On day 78 (45 days after SRT), the mass had decreased in size. (d) On day 249, the mass was slightly larger than on day 78 but remained smaller than at diagnosis. No radiation-induced damage was observed in the larynx or oral mucosa
Treatment and outcome
Treatment options offered to the owner included surgery (partial or total laryngectomy with permanent tracheostomy), stereotactic radiation therapy (SRT), chemotherapy and palliative care, of which the owner chose SRT. Contrast-enhanced whole-body CT was performed for SRT planning on day 15. To minimise the risk of laryngeal swelling and subsequent airway obstruction, the cat was intubated using an endotracheal tube with an internal diameter of 3.0 mm after propofol induction (2 mg/kg IV to effect over 1 min). In addition, spontaneous breathing was maintained with oxygen supplementation alone, without the use of any anaesthetic agents apart from propofol. The CT scan revealed a 1.7 cm tumour (LD) localised to the laryngeal region without evidence of metastasis. The cat was positioned in sternal recumbency using a vacuum-mattress immobilisation device (Vacuum Form 2.0; Muranaka Medical Instruments) and a customised bite block. The radiotherapy plan was generated by a CT-based treatment planning system (Monaco; Elekta). The gross tumour volume (GTV), defined as the tumour visible on contrast-enhanced CT, was 1.2 cm3. Organs at risk (OARs), including the skin, brain, oesophagus, submandibular gland and spine, were contoured. No margin was added around the GTV for clinical target volume, considering the risk of late radiation effects on OARs. The planning target volume (PTV), defined by a 2 mm margin around the GTV, was 2.9 cm3. The patient was prescribed three fractions over 3 consecutive days with a total dose of 30 Gy. The coverage of the GTV receiving at least 100% of the prescription dose (i.e., V100%) was 95%, whereas the PTV receiving V95% was 87%. The mean and maximum doses to the GTV were 32.7 Gy and 34.8 Gy, respectively, while the PTV mean and maximum doses were 31.2 Gy and 34.8 Gy, respectively (Figure 4).

Dose distribution on (a) axial and (b) sagittal views from the stereotactic radiation therapy plan. (c) Dose–volume histogram showing percentage volume, total dose and relative dose. cGy = centigray; GTV = gross tumour volume; PTV = planning target volume
From the planning CT to the initiation of SRT (days 15–31), the cat received only prednisolone (1 mg/kg PO q24h). Although mirtazapine was available as an appetite stimulant if needed, it was not required, as no decrease in appetite was observed during this period. SRT was delivered on days 31–33 using a linear accelerator (Elekta Synergy; Elekta), with laryngeal ultrasound performed before each session to assess tumour size and confirm intubation feasibility. All sessions were performed with the patient intubated with the same anaesthetic procedure as that used for the planning CT. On the third day of SRT, drooling was observed, and an endoscopy performed on the same day showed no change in tumour size, with mild erythema (Figure 3b). Prednisolone was continued during SRT and tapered and continued until day 155. Although feeding-tube placement or administration of an appetite stimulant such as mirtazapine had been planned in case loss of appetite occurred after SRT, these procedures were not performed. At follow-up on day 50, presenting signs and drooling had improved, and echolaryngography showed slight tumour shrinkage (LD 1.0 cm). On day 78, ultrasound and endoscopy confirmed further shrinkage (LD 0.7 cm) (Figure 3c), achieving a partial response per the canine response evaluation criteria in solid tumours (cRECIST). 1 On day 155, the cat exhibited choking while drinking, and ultrasound showed a slight increase in tumour size (LD 0.9 cm) without changes in adjacent structures. Suspecting regrowth, toceranib phosphate (2.7 mg/kg PO q48h) was started. Two weeks later, plasma creatinine increased (2.17 mg/dl) and toceranib was discontinued. On day 249, ultrasound and endoscopy showed a tumour LD of 0.8 cm, while the cat was asymptomatic (Figure 3d). Toceranib was reintroduced at a reduced dose (2.2 mg/kg q72h) but stopped on day 301 because of vomiting and azotaemia (blood urea nitrogen 37.1 mg/dl, creatinine 2.75 mg/dl). On day 520, echolaryngography showed stable disease (LD 0.8 cm), and fluoroscopy with oral iopamidol confirmed no aspiration, indicating preserved swallowing. On day 603, ultrasound showed tumour enlargement (LD 1.4 cm) with worsening stridor. From day 630, the cat developed progressive dyspnoea, requiring oxygen therapy and prednisolone (1 mg/kg PO q24h), but died on day 644.
Discussion
SCC is one of the most common feline laryngeal tumours, along with lymphoma.2 –5 In limited reports of feline laryngeal SCC, treatments such as surgical excision,6 –8 tracheostomy,3,7,9 radiation therapy,8 –11 chemotherapy8,10 and palliative prednisolone4,5 have been attempted. Although metastasis is uncommon at diagnosis, laryngeal SCC usually carries a poor prognosis owing to local progression, causing dyspnoea, and euthanasia is often chosen soon after diagnosis.
Postoperative complications such as airway obstruction and aspiration pneumonia are frequently reported after laryngeal surgery.6,12 –14 Several studies have also described the concurrent use of tracheostomy during surgery.7,9 However, permanent tracheostomy in cats carries a high risk of fatal complications, including dyspnoea caused by mucous plugs at the stoma or elsewhere in the airway, and requires frequent care both during hospitalisation and after discharge.3,7,9,14 Most cats with laryngeal SCC that underwent permanent tracheostomy as the sole treatment died within 1 week.3,9
Little information exists on the use of radiation therapy for feline laryngeal SCC. A previous report described two cases treated with permanent tracheostomy and radiation therapy, of which one died from airway obstruction after 2 days and the other from anorexia after 281 days. 9 Reports also describe accelerated fractionated radiation therapy delivered twice daily for 5 or 9 days (10 or 14 fractions) with surgical excision and/or chemotherapy for feline laryngeal SCC.8,10 Two cats received neoadjuvant chemotherapy, resulting in tumour reduction, followed by surgical debulking and accelerated fractionated radiotherapy. 8 One cat died from the progression of another tumour 51 days after treatment, whereas the other died from metastasis after 144 days. Another study described a cat treated with accelerated fractionated radiation therapy and chemotherapy that achieved complete remission for 303 days but later died from pulmonary metastasis, with an overall survival of 348 days. 10 Two cats treated with single-fraction SRT at 20 Gy had poor prognoses, with progression-free intervals of 59 and 87 days and survival times of 84 and 106 days, respectively. 11 By contrast, the cat in this report achieved a progression-free interval of 573 days and overall survival of 614 days after SRT, which were markedly longer than previously reported. Possible reasons for the differences in outcomes between Yoshikawa et al 11 and this case include the smaller primary tumour size at treatment (<2 cm in this case vs ⩾2 cm in their cases) and the use of three fractions, which enabled a higher total dose (30 Gy). Compared with accelerated or conventional fractionated radiotherapy, SRT requires fewer anaesthetic sessions, a considerable advantage in cats with laryngeal tumours carrying high anaesthetic risks. As in this case, SRT as palliative treatment may be a useful option for feline early-stage laryngeal SCC. To detect such cases, radiography and ultrasonography of the larynx should be considered in cats showing signs suggestive of laryngeal disease, such as stridor, hoarseness or increased inspiratory effort. 15 Mucositis and pain during early radiotherapy phases for feline laryngeal carcinoma have been managed with gabapentin, prednisolone, buprenorphine, mirtazapine and oesophageal feeding tubes.10,16 In this case, although drooling suspected to reflect pain was observed on the final SRT session, it was manageable with prednisolone, and a feeding tube was not placed because the cat maintained adequate appetite before and after SRT. However, in cases where reduced appetite or malnutrition is observed at the start of treatment, placement of a feeding tube should be considered to ensure adequate nutritional support, given the possibility of anorexia after SRT. Prolonged prednisolone therapy was employed in this case to reduce the risk of airway obstruction or impaired food intake that could arise either from acute radiation-induced laryngeal swelling or from potential tumour regrowth after SRT. Although long-term corticosteroid administration carries the risk of adverse effects, its use was considered clinically justified given the potential for life-threatening airway obstruction. No late effects in the larynx or oesophagus have been reported in the few long-term survivors of feline laryngeal carcinoma treated with accelerated or conventional fractionated radiotherapy.10,16 Compared with conventional protocols, SRT delivers higher doses per fraction, theoretically increasing late-effect risk to surrounding organs. SRT for feline nasal tumours has generally been well tolerated, with few severe late effects.17,18 In humans with early-stage laryngeal SCC, although SRT provides high local control, severe late effects such as mucositis, chondronecrosis and mucosal necrosis have been described.19,20 In this case, while the oesophagus was excluded from the PTV, the entire larynx was included; nevertheless, no late effects were observed, as swallowing was preserved on fluoroscopy with oral iopamidol on day 520. Anecdotally, based on clinical experience, late effects in cats appear less clinically significant than in dogs. However, without histopathology, subclinical late effects cannot be excluded. Further studies with larger populations of feline laryngeal SCC are needed to assess outcomes and tolerability of standardised SRT protocols.
Toceranib has been reported as potentially useful in feline oral SCC;21,22 however, a recent study indicated it may provide no benefit in most cats with oral SCC when used as adjuvant therapy after radiation. 23 In this case, toceranib administration was short because of adverse effects, so its clinical benefit could not be assessed.
Conclusions
SRT as palliative treatment may be a useful option for feline laryngeal SCC, especially in early-stage cases without dyspnoea.
Footnotes
Acknowledgements
Author note
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Initiative for Realizing Diversity in the Research Environment grant from the Ministry of Education, Culture, Sports, Science and Technology, Japan, in 2025.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication; therefore, additional informed consent for publication was not required.
