Abstract
Case summary
A 12-year-old spayed female Ragdoll cat was treated with chemotherapy for gastric large B-cell lymphoma. Adverse effects included neutropenia and thrombocytopenia (day 132) and transient prednisolone-induced diabetes mellitus (day 209). On day 209, urinalysis revealed yeast, and Candida tropicalis was cultured. Concurrent findings included mild azotaemia, hyperglobulinaemia, right renomegaly and renal pelvic dilation. Chemotherapy was discontinued, and fungal pyelonephritis was treated with oral fluconazole (50 mg q12h). By day 244, progressive right renal pelvic and ureteral dilation with echogenic ureteral material was noted. Management included placement of a subcutaneous ureteral bypass device (SUB), extended oral fluconazole (137 days), fluconazole administration into the renal pelvis via the SUB (10 days) and intravenous caspofungin (10 days). At final follow-up (day 1306), the cat remained clinically well, non-azotaemic, with a patent SUB, reduced kidney size and no recurrence of fungal urinary tract infection, lymphoma or diabetes.
Relevance and novel information
This is the second report of feline Candida species pyelonephritis treated with a SUB, and the first of topical antifungal treatment using a nephrostomy tube, resolution of predisposing factors and a successful long-term outcome.
Case description
A 12-year-old female spayed Ragdoll cat was referred for lymphoma (day 0) to a multidisciplinary specialist hospital (hospital 3). The cat presented 2 weeks earlier to a first opinion practice (hospital 1) for chronic vomiting, haematemesis, melena, weight loss of 900 g (weight 5.6 kg, body condition score [BCS] 5/9), inappetence and pyrexia (40°C). Haematology and biochemistry revealed neutrophilia with a left shift and hypoalbuminaemia (Table 1). Pyrexia resolved with amoxicillin-clavulanate (18 mg/kg PO q12h). The cat was initially referred to a different specialist hospital (hospital 2) 1 week before presentation to hospital 3, where ultrasound identified a focal, mucosal gastric mass with a diameter of 3 cm at the pyloric antrum/lesser curvature. Gastroscopic biopsy identified gastric large B-cell lymphoma on histopathology and immunohistochemistry. Urinalysis was unremarkable.
Selected haematology and biochemical parameters

Abnormal results are shown in bold
C/A = clumped and adequate; PCV = packed cell volume; RI = reference interval
Prednisolone was commenced on day 0 (5 mg PO q12h) (Figure 1). The patient’s appetite improved by day 7, a multi-agent cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP)-based chemotherapy protocol was commenced and prednisolone was discontinued by day 37 (Figure 1). 1 Treatment included maropitant (1.3 mg/kg PO q24h), ondansetron (0.7 mg/kg PO q12h), omeprazole (0.8 mg/kg PO q12h) and mirtazapine (1.875 mg PO q48h), as required. During CHOP, the cat gained 400 g (body weight 6 kg) and ultrasound suggested partial remission with mild focal gastric wall thickening (4.2 mm). On day 114, vomiting, diarrhoea and a new 4.2 mm focal gastric nodule on abdominal ultrasound suggested progressive disease. CHOP was replaced by lomustine (Figure 1). 2 Grade 1 neutropenia and grade 3 thrombocytopenia occurred on day 132 (Table 1). 3 Subsequently, grade 4 neutropenia, inappetence, lethargy and pyrexia (39.9°C) occurred on day 139 (Table 1). Treatment included intravenous fluid therapy (IVFT) for 24 h (Hartmann’s solution, 20 mmol/l potassium chloride, 2 ml/kg/h), amoxicillin-clavulanate (17 mg/kg PO q12h) and marbofloxacin (2 mg/kg PO q24h) for 10 days. Because of persistent neutropenia, lomustine was discontinued. Subsequently, L-asparaginase was administered on days 143 and 150, considering its relatively low risk of myelosuppression (Table 1).
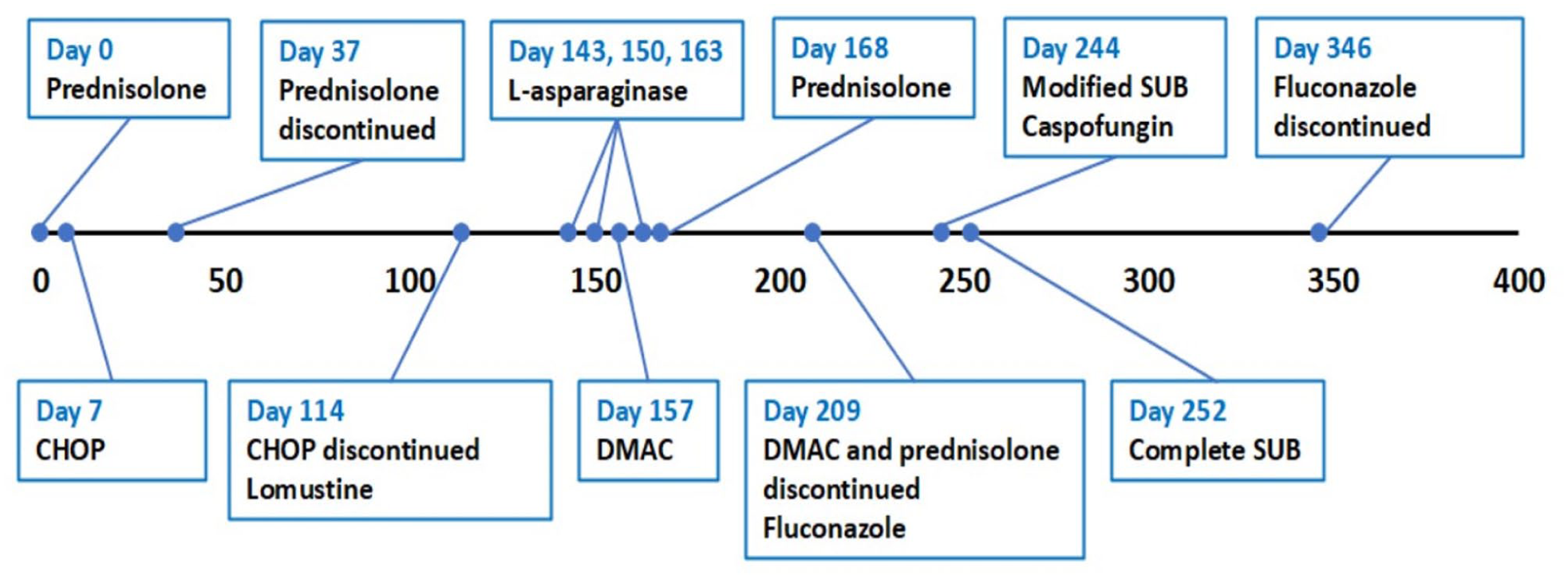
Treatment timeline (in days) showing when chemotherapy and antifungal treatments were commenced and discontinued. Surgical placement of the modified and complete subcutaneous ureteral bypass devices (SUBs) is included. CHOP (multi-agent CHOP-based chemotherapy protocol: vincristine [0.5–0.7 mg/m2 IV], cyclophosphamide [50 mg PO], doxorubicin [1 mg/kg IV]); lomustine (60 mg/m2 PO q4 weeks); L-asparaginase (non-pegylated form 400 IU/kg SC); DMAC (lymphoma chemotherapy rescue protocol: dexamethasone [1 mg/kg SC], cytarabine [300 mg/m2 SC], actinomycin D [0.6 mg/m2 IV]).
By day 157, adverse events from lomustine (neutro-penia and pyrexia) resolved. Haematology, biochemistry and urinalysis were unremarkable (urine specific gravity [USG] >1.050), and urine bacterial culture was negative (Table 1). As a result of the owner and clinician’s preference, a dexametasone, melphalan, actinomycin D and cytarabine (DMAC) rescue protocol was commenced (Figure 1). 4 On day 163, inappetence, diarrhoea and pale mucous membranes occurred. Moderate anaemia was suspected to be secondary to possible gastrointestinal haemorrhage (Table 1). DMAC was delayed and L-asparaginase, omeprazole (5 mg PO q12h) and sucralfate (0.25 g PO q8h) were administered. On day 168, neutropenia, mild anaemia and increased urea were documented (Table 1). Ultrasound identified persistent mild gastric focal wall thickening (3.2 mm). Prednisolone was recommenced (5 mg PO q12h). On day 176, haematology was unremarkable and melphalan (18 mg/m2 PO) was given. DMAC was continued on day 183, melphalan (18 mg/m2 PO) on day 198, and mitoxantrone (5 mg/m2 IV [substituted because of diarrhoea and lethargy despite actinomycin D dose reduction]) and prednisolone (2.5 mg PO q12h) on day 206.
By day 209, the cat had lost 300 g (body weight 5.7 kg), and lethargy and polyuria/polydipsia had occurred since restarting prednisolone. Mild anaemia, neutrophilia and hyperglobulinaemia, moderate hyperglycaemia and mild azotaemia were found (Table 1). Beta-hydroxybutyrate was within the reference interval [RI]. Urinalysis (cystocentesis, dipstick and cytology) showed 3+ glucose, 4+ blood, 1+ protein, pH 6.5, USG 1.038, two red blood cells per high-powered field (PHPF), five leucocytes PHPF and moderate numbers of yeasts. Ultrasound identified right renomegaly (length 4.7 cm, RI 3.0–4.3) with renal pelvic dilation (8 mm, RI 0–2) and echogenic contents, mildly hyperechoic perinephric fat and the right ureter was not visualised.5,6 The left kidney was unremarkable (length 4.2 cm). The assessment was complete remission of lymphoma (resolution of ultrasonographic and clinical changes), suspected prednisolone-induced diabetes mellitus and fungal pyelonephritis. Prednisolone was rapidly tapered then discontinued and fluconazole (50 mg PO q12h) was commenced (Figure 1). Urine dipsticks were monitored at home for glucosuria/ketonuria. Fungal urine culture identified a moderate growth of Candida tropicalis (Table 2).
In vitro antifungal sensitivity testing from the urine of an immunosuppressed cat with pyelonephritis

Interpretation using human derived breakpoints. Fungal urine culture identified a moderate growth of Candida tropicalis using MALDI-TOF MS (matrix-assisted laser desorption ionisation time of flight mass spectrometry) identification
I = intermediate; MIC = minimum inhibitory concentration; S = sensitive
During days 209–244, the cat improved with reduced creatinine, hyperglycaemia and resolution of glucosuria (Table 1). The cat was treated intermittently with IVFT and subcutaneous fluid therapy, maropitant, ondansetron and mirtazapine. By day 244, the anaemia resolved, mild neutrophilia persisted and creatinine had increased. Urinalysis revealed 1+ blood, pH 6, USG 1.022, one red blood cell PHPF and 15 leucocytes PHPF with no yeasts seen. Ultrasound showed progressive right renal pelvic dilation (1.1 cm) with distension of the proximal two-thirds of the right ureter (4 mm), which contained echogenic material and hyperechoic peri-ureteral fat. The gastrointestinal tract was unremarkable. The assessment was complete remission of lymphoma, resolution of clinical diabetes mellitus and right ureteral obstruction secondary to fungal pyelonephritis.
On day 244, under general anaesthesia, the nephrostomy catheter of a subcutaneous ureteral bypass device (SUB) (SUB 2.0; Norfolk Vet Products) was placed, with the catheter tip in the proximal right ureter, as previously described. 7 The nephrostomy catheter was passed through the abdominal wall and attached to a subcutaneous unidirectional access port (PleuralPort; Norfolk Vet Products) (Figure 2). We aimed to use this as a nephrostomy tube for renal pelvic decompression and for antifungal administration into the renal pelvis, to regain ureteral patency. Fluoroscopic pyelography during SUB placement confirmed complete right ureteral obstruction. Urine from the renal pelvis was grossly turbid with white plaque-like material. Bacterial and fungal culture was negative and cytology did not identify fungi. A jugular catheter was placed in anticipation of prolonged hospitalisation for intravenous fluid therapy, blood sampling and intravenous antifungal administration.

Photograph demonstrating the nephrostomy catheter (black arrow) of a subcutaneous ureteral bypass device (SUB 2.0; Norfolk Vet Products) attached to a subcutaneous unidirectional access port (black and white arrow) (PleuralPort; Norfolk Vet Products). This was used as a nephrostomy tube to allow renal pelvic decompression and antifungal administration into the renal pelvis to be performed
Postoperative management included IVFT, methadone (0.2 mg/kg IV q4h) for 4 days, ketamine (continuous rate infusion 0.12–0.17 mg/kg/h IV) for 3 days, followed by buprenorphine (0.01 mg/kg IV q8h) for 3 days and maropitant (1 mg/kg IV q24h) for 7 days. Systemic antifungals included caspofungin (IV q24h for 10 days) and fluconazole (50 mg PO q12h) (Figure 1). Caspofungin was given at 1 mg/kg once, then 0.75 mg/kg thereafter as a 0.2 mg/ml infusion over 2 h. 8 Topical treatment involved withdrawing urine from the renal pelvis via the SUB port (volume range 0.3–5 ml) and injecting the same volume of fluconazole (0.6 mg/ml solution q4h) for 7 days.
The cat tolerated use of the injection port; however, bruising developed. By day 252, urine from the port was grossly clear, haematology and biochemistry were unremarkable, postoperative inappetence persisted and pyel-ography (via the port) documented the lack of right ureteral patency. Under general anaesthesia, the cystostomy catheter of the SUB kit was placed and connected to the nephrostomy tube and a bidirectional access port according to manufacturer guidelines (Figure 3). 7 An oesophageal feeding tube was placed (removed on day 302). Analgesia included methadone (0.2 mg/kg IV q4h) for 3 days. Topical fluconazole was increased to 20 ml q12h until discharge on day 255. Fluconazole (50 mg PO q12h) was continued along with transmucosal bupre-norphine (0.02 mg/kg q8h) for 4 days and maropitant (8 mg PO q24h) for 5 days.

Right lateral abdominal radiograph from a different patient demonstrating the complete subcutaneous ureteral bypass device (SUB 2.0; Norfolk Vet Products) in place: nephrostomy catheter (black arrow), subcutaneous bidirectional access port (white arrow) and cystostomy catheter (black and white arrow)
Urine fungal cultures on days 290, 346 and 459 were negative. Fluconazole was discontinued on day 346. From day 346 onwards, the patient’s body weight was in the range of 5.8–6.17 kg and the cat was well. Rechecks every 3–6 months included creatinine, ultrasound, SUB flushing to assess patency, urinalysis and bacterial/fungal culture. Intermittent cystitis, managed with meloxicam (0.05 mg/kg PO q24h as needed), was likely mechanical given the negative bacterial and fungal cultures. Intermittent haematology and biochemistry were unremarkable. On day 1306, the cat was well, her body weight was 5.86 kg, BCS was 6/9, urinalysis was unremarkable (USG 1.028), urine bacterial/fungal culture was negative, urea was mildly increased, the SUB was patent, right kidney length was 2.64 cm with a renal pelvis of 6.5 mm and there was no evidence of recurrence of lymphoma (Table 1).
Discussion
Candida species urinary infections are uncommon in cats,9 –16 with pyelonephritis rarely reported.10,17 Optimal treatment is not defined and outcomes may be influenced by the predisposing factors.12,13,15,17 Antifungal drug choices, doses and routes in this case were based on sensitivity results, published pharmacokinetic/safety data and extrapolation from human medicine.8,18 Two antifungals and oral/intravenous/topical routes were used as our aim was to achieve cure and right ureteral patency.
Predisposing factors in this patient included immunosuppression, antimicrobial use and diabetes mellitus. 16 Retroviral infections may predispose to fungal infection and lymphoma and not testing for this was an oversight. Resolution of infection was likely achieved by resolving all predisposing factors and appropriate antifungal use. Diabetes mellitus was diagnosed, rather than stress-induced hyperglycaemia, based on persistent hyperglycaemia and glucosuria documented at home. Insulin therapy was not initiated, and prednisolone was discontinued, with close monitoring for either progression or remission of the condition.
Systemic antifungals were used first to try to regain ureteral patency. We aimed to discontinue prednisolone/chemotherapy and manage diabetes before a potential surgery. We had concerns for lymphoma recurrence and fungal biofilm formation within a SUB. However, retrospectively, immediate surgical intervention should have been considered to maximise renal function preservation. Our surgical approach aimed to retain as much total renal function as possible. Another approach to this case, however, which was also offered to the client, could have been right ureteronephrectomy combined with systemic antifungals, with a risk of potentially greater reduced total renal function.
Both topical and systemic antifungals are used to treat fungal pyelonephritis in humans.19 –23 Nephrostomy tubes allow for lavage and drug administration.19 –22 We modified a SUB into a nephrostomy tube because of concerns that an external tube would require higher patient care and risk ascending bacterial contamination. Removal of renal pelvis debris was possible; however, optimal decompression was not achieved using drainage every 4 h, potentially at the detriment of kidney function. Another approach would have been to place a complete SUB initially. We elected not to do so, owing to concerns that the SUB’s bidirectional flow might not facilitate debris removal or optimise delivery of topical treatment to the renal pelvis.
This is the second report of feline Candida species pyelonephritis treated with a SUB. 17 The previously reported case had pre-existing chronic kidney disease (CKD) and cardiomyopathy, antimicrobial use, ureteroliths, postoperative urethral obstruction/oliguria/fluid overload (day 4), postoperative SUB obstruction (day 6), bacterial urinary tract infection (week 12) and euthan-asia because of progressive CKD (month 11). The fungal infection resolved with systemic fluconazole and CKD was the limiting factor, with acute kidney injury (AKI) secondary to ureteral/SUB/urethral obstructions likely worsening the long-term outcome. In our case, pre-treatment with antifungals, removal of renal pelvic debris with a nephrostomy tube, lack of CKD/AKI and resolution of predisposing factors likely led to longer survival.
Conclusions
This is the second report of feline Candida species pyelonephritis treated with a SUB, and the first to use topical antifungals through a nephrostomy tube and the first to report resolution of predisposing factors and a successful long-term outcome.
Footnotes
Acknowledgements
The authors would like to thank the cat’s owners, the staff and students of the University Veterinary Teaching Hospital Sydney, and Professors Vanessa R Barrs and Richard Malik for case management advice.
Author’s Note
Lara A Boland is now affiliated to MediPaws, Leichhardt, NSW, Australia.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
