Abstract
Case summary
A 4.5-year-old male castrated, predominantly indoor-housed domestic shorthair cat presented with lethargy, anorexia and fever, progressing rapidly to severe respiratory distress and neurologic dysfunction. Clinical evaluation and advanced diagnostics revealed widespread systemic disease involving the lower respiratory tract, nervous and hepatic systems. Despite aggressive supportive care, including intravenous fluids, antiemetics and non-steroidal anti-inflammatory drugs, the cat’s condition deteriorated, necessitating humane euthanasia. Post-mortem gross findings included consolidated lungs with peribronchiolar pallor, multifocal to coalescing hepatic hemorrhage and multifocal gastrointestinal ulcerations. Histopathologic examination revealed necrotizing and neutrophilic meningoencephalitis and fibrinonecrotizing bronchointerstitial pneumonia alongside necrotizing lesions in the vascular, hepatic, lymphoid, pancreatic and gastrointestinal tissues. Influenza A/H5 subtype (highly pathogenic avian influenza) was confirmed in the brain and pulmonary tissues using real-time RT-PCR. Rabies was excluded via negative indirect fluorescent antibody test performed on brain tissue. Feline calicivirus and feline herpesvirus-1 were not detected in the lung tissues via RT-PCR. No other organisms were observed in tissues by microscopy after histochemical staining.
Relevance and novel information
This case documents highly pathogenic avian influenza infection in a domestic shorthair cat with minimal known outdoor access, confirmed by real-time RT-PCR in both lung and brain tissues. Low cycle threshold values indicated exceptionally high viral loads and pronounced neurotropism. The rapid onset of necrotizing meningoencephalitis and severe bronchointerstitial pneumonia, concurrent with low cycle threshold values, highlights the relationship between high tissue viral burden and fulminant multisystemic pathology. This case expands the current understanding of feline exposure risk by supporting the possibility of indirect environmental contamination or involvement of unrecognized transport hosts.
Keywords
Introduction
Highly pathogenic avian influenza (HPAI) H5 viruses, enzootic in wild waterfowl, have repeatedly spilled into domestic and wild mammals since first appearing in poultry in the USA in February 2022. H5N1 infection has been associated with high mortality in cats, foxes, seals and mink, with only rare cases in dogs.1 –5 Cats appear particularly susceptible because of their predatory tendencies and dietary exposure to high-risk materials, including infected birds, rodents, raw meat diets (eg, chicken byproducts) and unpasteurized milk.1,2,5,6 Moreover, feline susceptibility to neuroinvasive HPAI may be influenced by the presence of alpha-2,3-linked sialic acid receptors in central nervous system tissues, which facilitate viral entry and replication, as demonstrated in both natural and experimental infections.7 –9 Infections often follow direct ingestion of infected tissue or contact with contaminated fomites, and clinical signs can range from subclinical to fulminant multisystemic disease.1,2,5,6 In this case, we describe a domestic shorthair cat with minimal outdoor access that developed rapidly progressive HPAI-induced multisystemic illness.
Case description
A 4.5-year-old male castrated domestic shorthair cat, weighing 5.9 kg, presented to a tertiary veterinary medical teaching hospital (tVMTH) with a 4-day history of lethargy, anorexia and persistent fever. The cat was housed exclusively indoors but experienced a single brief unsupervised escape approximately 1 week before presentation. According to the owner, the escape was limited to the backyard, and the cat was recovered and brought back inside within approximately 10 mins. During this escape, the owner reported observing the cat chewing on an Amaryllis species houseplant but did not note significant ingestion. There were no other known escapes, exposures to toxins or wildlife throughout the cat’s life, and nor was the cat reported to be consuming a raw food diet, raw food treats or exposed to raw milk. The cat was also reportedly up to date on all vaccines, including rabies virus.
Two days before referral, the cat was seen by the primary veterinarian for acute anorexia and lethargy. Physical examination revealed pale mucous membranes, 5–8% dehydration, tachypnea (60 breaths/min), tachycardia (150 beats/min) and hyperthermia (103.9°F [39.9ºC]). Serial blood work revealed marked increases in hepatocellular and pancreatic enzyme activities, hyperglycemia, hypocalcemia, hypophosphatemia, leukopenia and thrombocytopenia; however, it should be noted that there was no slide review (Tables 1 and 2). Urinalysis (via cystocentesis) showed specific gravity above 1.040, mild proteinuria and glucosuria, leukocyte esterase positivity and mild pyuria (6 white blood cells per high-power field [WBCs/hpf]). Abdominal ultrasound revealed gall bladder wall thickening and was otherwise unremarkable, with no evidence of pancreatitis or gastroenteritis. Feline leukemia virus, feline immunodeficiency virus and heartworm (SNAP Triple; IDEXX) testing was negative. Treatment included lactated Ringer’s solution (4.5 ml/kg/h), maropitant citrate (1 mg/kg SC once, Cerenia 10 mg/ml; Zoetis), amoxicillin-clavulanate (11 mg/kg PO q12h, Clavacillin; Dechra) and robenacoxib (1.05 mg/kg PO q24h, Onsior; Elanco). The cat worsened after 2 days and was referred to the tVMTH.
Complete blood count results in a 4.5-year-old domestic shorthair cat infected with highly pathogenic avian influenza before evaluation at the tertiary veterinary medical teaching hospital*
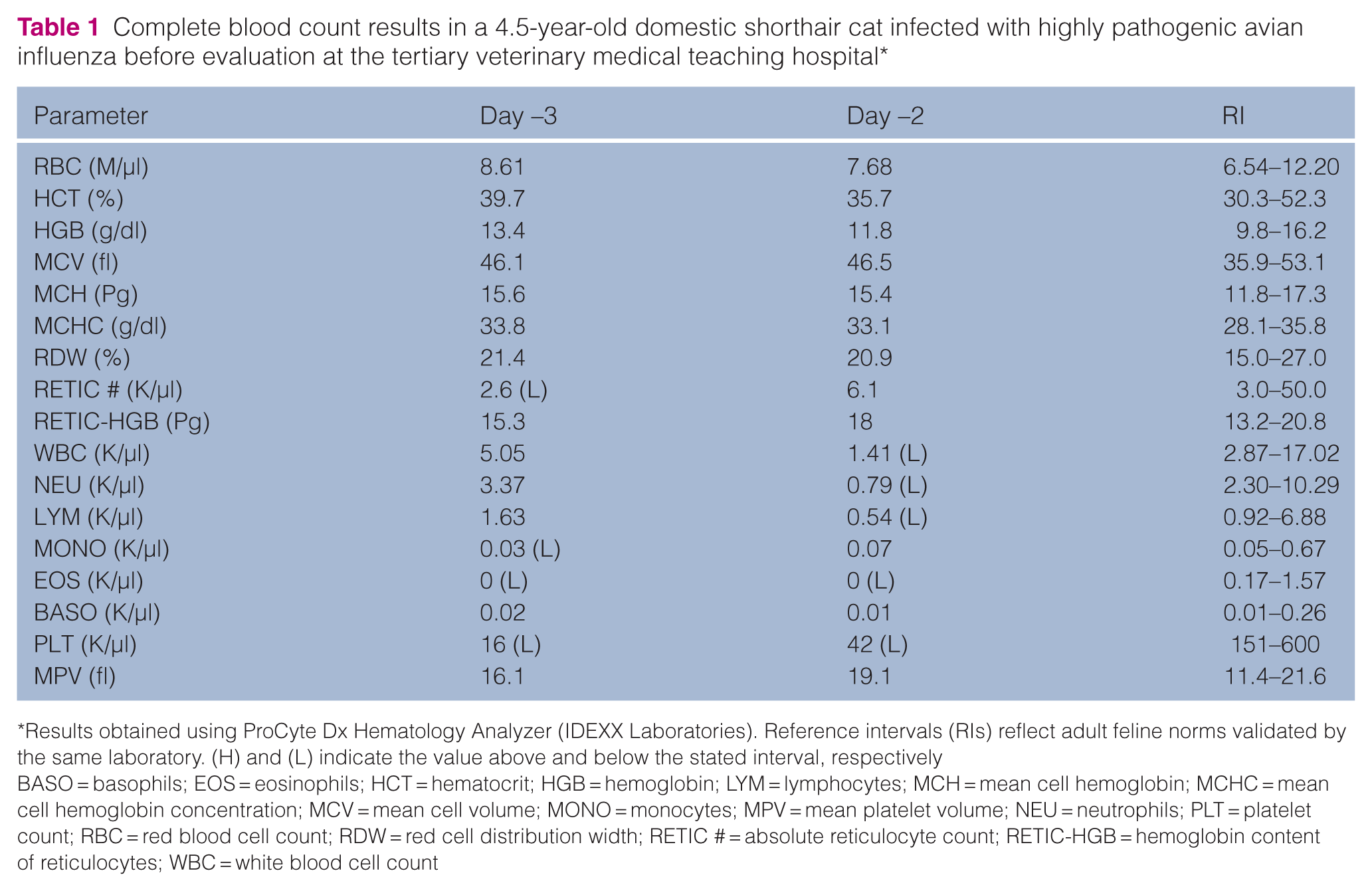
Results obtained using ProCyte Dx Hematology Analyzer (IDEXX Laboratories). Reference intervals (RIs) reflect adult feline norms validated by the same laboratory. (H) and (L) indicate the value above and below the stated interval, respectively
BASO = basophils; EOS = eosinophils; HCT = hematocrit; HGB = hemoglobin; LYM = lymphocytes; MCH = mean cell hemoglobin; MCHC = mean cell hemoglobin concentration; MCV = mean cell volume; MONO = monocytes; MPV = mean platelet volume; NEU = neutrophils; PLT = platelet count; RBC = red blood cell count; RDW = red cell distribution width; RETIC # = absolute reticulocyte count; RETIC-HGB = hemoglobin content of reticulocytes; WBC = white blood cell count
Chemistry panel results in a 4.5-year-old domestic shorthair cat infected with highly pathogenic avian influenza before evaluation at the tertiary veterinary medical teaching hospital*

All results were obtained using Catalyst One Chemistry Analyzer (IDEXX Laboratories). Reference intervals (RIs) reflect adult feline norms validated by the same laboratory. (H) and (L) indicate the value above and below the stated interval, respectively
ALB = albumin; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AMYL = amylase; BUN = blood urea nitrogen; CA = calcium; CHOL = cholesterol; Cl = chloride; CREA = creatinine; GGT = gamma-glutamyl transferase; GLOB = globulin; GLU = glucose; K = potassium; LIPA = lipase; Na = sodium; P = phosphorus; TBIL = total bilirubin; TP = total protein; – = not applicable
At the tVMTH, the cat was dull with tachypnea (100 breaths/min) and hyperthermia (106.2°F [41.2ºC]). Thoracic radiographs showed mixed pulmonary patterns, with interstitial, bronchial and alveolar involvement, most severe in caudal lung lobes (Figure 1). Mild pleural effusion and cardiomegaly raised concern for cardiopulmonary disease. A feline SNAP proBNP test (IDEXX Laboratories) was negative on serum (undiluted) but positive in diluted pleural fluid (1:1 with saline). 10 Pleural effusion cytology showed a high-protein modified transudate with mostly non-degenerate neutrophils, vacuolated macrophages and lymphocytes. Repeat blood work revealed panhypoproteinemia, hepatic injury, rhabdomyolysis, hyperbilirubinemia, electrolyte derangements, mild hyperglycemia, and worsening leukopenia and thrombocytopenia with clumping noted on slide review (Tables 3–5). Urine sediment showed transitional/renal epithelial cell loss (6–10/hpf), granular casts (0–1/lpf), glucosuria (3+), bilirubinuria (3+) and pyuria (4–8 WBCs/hpf). Serologic infectious disease testing, including Bartonella species, hemotropic Mycoplasma species, Ehrlichia/Anaplasma/Neorickettsia/Wolbachia species PCR and Toxoplasma species IgM/IgG ELISA, was negative.
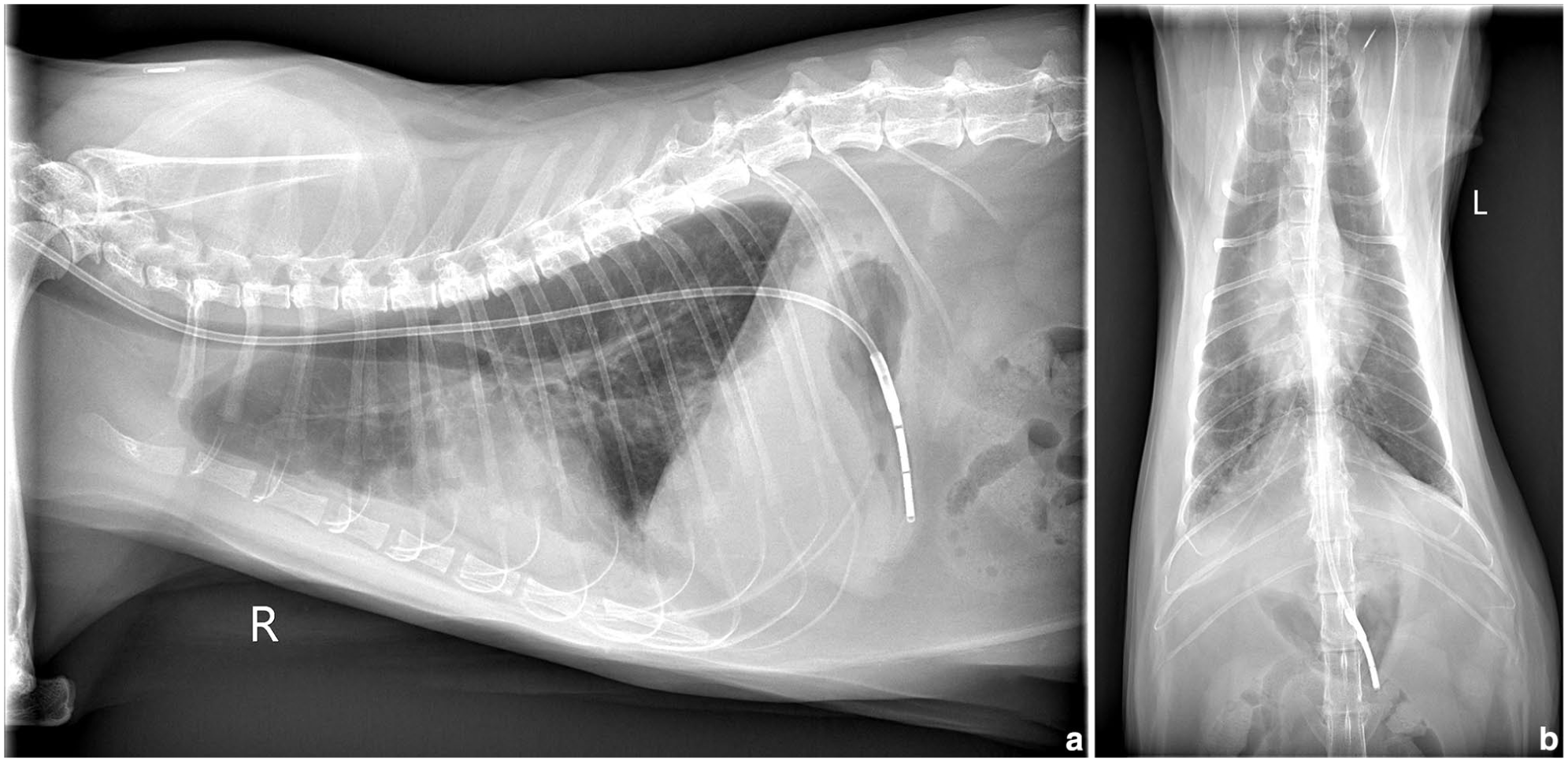
Thoracic radiographs showing the lower respiratory tract changes in a cat with confirmed highly pathogenic avian influenza (H5N1) disease. (a) Lateral view demonstrating diffuse mixed pulmonary patterns, including interstitial, bronchial and alveolar changes most pronounced in the caudal lung lobes. Mild pleural effusion and a moderately enlarged cardiac silhouette are also noted. (b) Dorsoventral view, highlighting the presence of pleural effusion and additional details of the lung and heart changes. L = left; R = right
Serial day 0 venous blood gas results in a 4.5-year-old domestic shorthair cat infected with highly pathogenic avian influenza at a tertiary veterinary teaching hospital*

Results were obtained using the ABL800 FLEX (Radiometer). Reference intervals (RIs) reflect adult feline norms validated by the same laboratory. (H) and (L) indicate the value above and below the stated interval, respectively
ABE = actual base excess; AG = anion gap; Cl = chloride; CREA = creatinine; GLU = glucose; HCO3 = bicarbonate; HGB = hemoglobin; iCa = ionized calcium; K = potassium; LAC = lactate; Na = sodium; pCO2 = partial pressure of carbon dioxide; pH = hydrogen ion concentration; PB = barometric pressure; T0 = initial presentation; T6 = 6 h after initial presentation; T10 = 10 h after initial presentation
Complete blood count results on day 0 in a 4.5-year-old domestic shorthair cat infected with highly pathogenic avian influenza at a tertiary veterinary teaching hospital*

Results were obtained using the Advia 120 (Siemens Healthcare Diagnostics). Reference internals (RIs) are those established by that institution for adult cats. (H) and (L) indicate the value above and below the stated interval, respectively
BASO = basophils; BANDS = band neutrophils; EOS = eosinophils; HCT = hematocrit; HGB = hemoglobin; LYMPHS = lymphocytes; MCHC = mean cell hemoglobin concentration; MCV = mean cell volume; MONO = monocytes; MPV = mean platelet volume; nRBCs = nucleated red blood cells; NEUTS = neutrophils; PLT = platelet count; PLT clumps = platelet clumping observed; Plasma TP = plasma total protein; RBC = red blood cell count; RDW = red cell distribution width; RETIC # = absolute reticulocyte count; WBC = white blood cell count, – = not applicable
Biochemistry panel results on day 0 in a 4.5-year-old domestic shorthair cat infected with highly pathogenic avian influenza at a tertiary veterinary teaching hospital*

Results were obtained using the Roche Cobas C501 (Diamond Diagnostics). Reference intervals (RIs) are those established by that institution for adult cats. H and L indicate value above and below the stated interval, respectively
AG = anion gap; ALB = albumin; ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; Ca = calcium; CHOL = cholesterol; CK = creatine kinase; Cl = chloride; CREA = creatinine; GGT = gamma-glutamyl transferase; GLOB = globulin; GLU = glucose; HCO3 = bicarbonate; K = potassium; Mg = magnesium; Na = sodium; OSM = osmolality (calculated); P = phosphorus; TBIL = total bilirubin; TP = total protein
Supportive care included oxygen therapy (60%), intravenous (IV) lactated Ringer’s solution (2 ml/kg/h), potassium chloride supplementation (KMAX 0.5 mEq/kg/h for 2 h), maropitant (1 mg/kg IV q24h), ondansetron (0.5 mg/kg IV q8h) and ampicillin-sulbactam (30 mg/kg IV q8h). Six hours later, blood work showed improved sodium, potassium and chloride, but persistent hypocalcemia, progressive hyperglycemia and hyperlactatemia (Table 3). The cat developed progressive neurologic signs, including ataxia, seizures and obtundation, alongside worsening respiratory distress. As a result of poor prognosis, humane euthanasia was elected and necropsy was performed.
At the post-mortem examination, the lungs were diffusely firm, poorly aerated and mottled red to dark red throughout all lobes, with faint airway-centered nodular masses that, on cut section, exuded mucohemorrhagic fluid and were accompanied by multifocal linear hemorrhages extending into surrounding parenchyma. The liver was uniformly ochre with rounded margins and a pronounced reticular surface. Scattered multifocal to coalescing, irregularly marginated cherry-red patches lacked normal lobular architecture. The heart weighed 25.2 g, with wall thicknesses of 3 mm (right ventricle), 13 mm (interventricular septum) and 14 mm (left ventricle), consistent with hypertrophic cardiomyopathy. Small volumes of serosanguineous fluid were present in both the thoracic (30 ml) and peritoneal (15 ml) cavities. Near the pyloric sphincter, a 1 cm × 0.3 cm linear chronic-active gastric ulcer was rimmed by firm fibrous tissue, and the small intestine contained moderate amounts of cherry-red blood transitioning to melanic fluid at the jejuno-ileal junction.
Histologically, severe necrotizing lesions were evident in the brain, lungs, liver, lymph nodes and pancreas. The brain exhibited gray-matter-centric necrotizing and neutrophilic meningoencephalitis with secondary neuronophagia, astrocytosis and microvascular hypertrophy (Figure 2). Pulmonary sections showed fibrinonecrotizing bronchointerstitial pneumonia (Figure 3), while hepatic foci corres-ponded to multifocal random areas of coagulative to lytic hepatocellular necrosis. Similar necrotic lesions were identified in exocrine pancreatic tissue and multiple lymph nodes. Myocardial disarray of the left ventricle confirmed the gross finding of hypertrophic cardiomyopathy. All other histologic changes were minimal and deemed clin-ically insignificant.

Photomicrographs demonstrating the described necrotizing lesions in the brain of a 4.5-year-old domestic shorthair cat with confirmed highly pathogenic avian influenza (H5N1) disease. (a) Affected neuropil exhibits regional rarefaction, increased numbers of glial cells, and occasional neurons with shrunken, angular, hypereosinophilic cytoplasm and fading or pyknotic nuclei. Microglial cells and rare neutrophils flank degenerate and necrotic neurons and neuronal debris. (b) Inflamed vasculature with segmental disruption of the internal elastic lamina and expansion of the vascular wall by brightly eosinophilic, amorphous material, which suspends cellular debris, and small numbers of a mixed inflammatory population (fibrinoid necrosis). Hematoxylin and eosin stain; bar = 50 μm
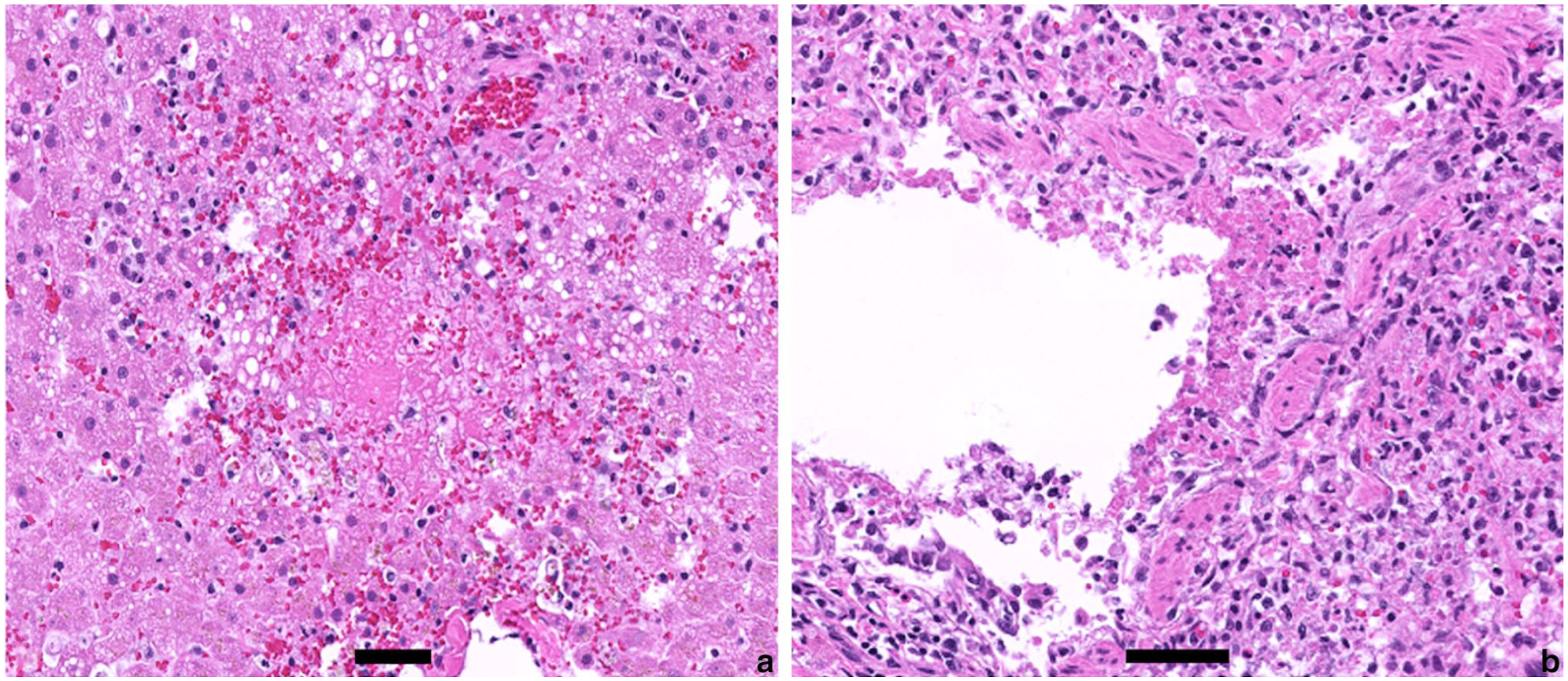
Photomicrographs demonstrating the described necrotizing lesions in the liver and lung of a 4.5-year-old domestic shorthair cat with confirmed highly pathogenic avian influenza (H5N1) disease. (a) Affected hepatic parenchyma demonstrates necrosis and loss of hepatocytes with replacement by large numbers of extravasated erythrocytes, fibrin, cellular and karyorrhectic debris. (b) A small bronchiole exhibits segmental necrosis and sloughing of epithelial cells with adherent strands of fibrin. Hematoxylin and eosin stain; bar = 50 μm
Real-time RT-PCR of lung and brain tissues confirmed the presence of H5 subtype avian influenza and pan-influenza A viruses. Although absolute quantification was not performed because of the absence of a standard curve, low cycle threshold values were obtained, consistent with high viral loads (brain H5 cycle threshold [Ct] 12.84/Influenza A Ct 10.68; lung H5 Ct 17.55/Influenza A Ct 14.92). Subsequent sequen-cing performed at the National Veterinary Services Laboratories confirmed the virus as clade 2.3.4.4b-B3.13, consistent with strains associated with ongoing outbreaks in North American wildlife and poultry populations. Indirect fluorescent antibody testing for rabies antigen was negative, and PCR assays for feline calicivirus, herpesvirus-1 and Chlamydia species in pulmon-ary tissue were also negative. No bacterial organisms were cultured from brain tissue. No other infectious agents were noticed on microscopic evaluation of stained tissues. A collective summary of the clinical signs and diagnostic findings can be found in Table 1 in the supplementary material.
Discussion
HPAI H5Nx, particularly the 2.3.4.4b clade, has emerged as a significant pathogen across multiple species, including domestic cats.1,2,6,11 The cat in this case exhibited a rapidly progressive illness marked by lethargy, anorexia, and both neurologic and severe lower respiratory signs. This clinical trajectory parallels findings from global outbreaks, where high morbidity and mortality have been consistently reported in cats.2,5,6,11 The acute onset and rapid decline observed in this cat showcases the virulence of HPAI in feline hosts and its capacity to cause multi-organ involvement.
Neurologic manifestations such as tremors and ataxia have been commonly described in naturally infected cats and are supported in this case by histopathologic evidence of meningoencephalitis.1,2,5,6,11 Lower respiratory involvement, evidenced by severe bronchointerstitial pneumonia, pleural effusion and respiratory distress in the current case, is a hallmark of HPAI in mammals, driven by high viral loads in lung tissues.1,2,6,11,12 Detection of viral RNA in both lung and brain samples by RT-PCR further supports the extent of viral dissemination and replication.
Detection of N-terminal pro B-type natriuretic peptid (NT-proBNP) in pleural effusion despite an undetectable serum concentration represents an observation of uncertain diagnostic value, although it may offer insight into effusion composition and biomarker distribution. This disparity may reflect fluid compartment dynamics in heart failure, where elevated intracardiac pressures promote transudation of protein-rich fluid into the pleural space. In contrast, serum levels may be influenced by factors such as rapid renal clearance, hemodilution or fluctuations in peptide release.10,13,14 In such scenarios, pleural NT-proBNP measurement may offer added diagnostic utility, particularly when serum values are inconclusive and cardiac disease is suspected.
Although no direct contact with infected birds was confirmed, several potential exposure pathways were identified. The cat reportedly had a brief (<10 mins) outdoor excursion, during which contact with infected wildlife or ingestion of a transport host such as a rodent could have occurred. However, it is thought to be less likely given the owner’s reported observation of the event. Although the owner did not report feeding a raw diet, indoor cats are still known to consume small mammals that may carry HPAI. 5 Additional risk factors included the owner’s occupation as an avian researcher and her residence near a dairy farm with a large feral cat population. These environmental and household factors together represent plausible exposure routes for the cat and potential risk to close human contacts.
Since cats are known to shed avian influenza viruses and may act as intermediate hosts, their close contact with people remains a potential zoonotic concern. According to the owner, shortly before the cat became febrile and anorexic, their child had developed a transient illness characterized by fever, conjunctivitis, headache and gastrointestinal upset. The illness resolved within 3 days and was not evaluated with laboratory testing. Although there is no direct evidence of zoonotic transmission or coinfection in this case, the close timing raises the possibility of shared environmental exposure or undetected transmission between species. In households where humans and companion animals cohabitate during influenza outbreaks, concurrent infection with human and avian strains could create conditions that allow viral reassortment. Although this did not occur here, the scenario highlights the importance of continued One Health surveillance and risk awareness.2,6,11
Despite aggressive supportive care, including intravenous fluids, antiemetics and non-steroidal anti-inflammatory drugs, the cat’s condition deteriorated and euthanasia was elected. This outcome is consistent with reported mortality rates in feline HPAI cases, which often exceed 80% in those with severe neurologic or respiratory disease.2,5,6,11 However, some cats survive mild illness or remain subclinical, suggesting that disease severity and prognosis are influenced by viral load, host immune status and concurrent comorbidities.2,5,6,11 In this case, the presence of hypertrophic cardiomyopathy may have contributed to the severity of systemic disease.
Conclusions
This case demonstrates the severe clinical course and unexpected exposure pathways of HPAI in a predom-inantly indoor domestic shorthair cat. Real-time RT-PCR confirmed HPAI infection, revealing exceptionally high viral loads in pulmonary and cerebral tissues that account for the severe bronchointerstitial pneumonia and necrotizing meningoencephalitis observed. Detection of HPAI in an animal with limited outdoor access suggests indirect environmental contamination or overlooked transmission vectors. Given the owner’s occupation as an avian researcher and residence near a substantial feral cat colony, enhanced surveillance, timely diagnostic testing and rigorous biosecurity measures are imperative. The state health department facilitated diagnostic confirmation and is actively monitoring HPAI cases in domestic cats throughout the state.
Supplemental Material
Supplementary Table 1
Clinical and pathologic findings associated with HPAI infection in a domestic shorthair cat.
Footnotes
Acknowledgements
The authors would like to acknow-ledge the small animal diagnostic imaging service at the James L Voss Veterinary Teaching Hospital for their initial interpretation of radiographs. We would also like to thank the Colorado Department of Public Health and Environment and Dr Blaire MacNeill of the Microbiology Department of Colorado State University for their assistance in investigating this case.
Supplementary material
The following file is available online:
Supplementary Table 1: Clinical and pathologic findings associated with HPAI infection in a domestic shorthair cat.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Federal Agency USDA-APHIS-VS under the contract #12639521D0078 and purchase order #12639524F1025 for influenza A virus testing in cattle and mammals.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
