Abstract
Case summary
A 4-year-old castrated male domestic shorthair cat was presented with lethargy, anorexia and vomiting that did not improve with symptomatic treatment. Blood tests revealed markedly elevated liver enzymes. Abdominal ultrasound showed gallbladder abnormalities, including a small gallbladder, indistinct fundus, a continuous spiral-shaped morphology forming C- and reverse C-shaped curves, and hyperechoic fat surrounding the gallbladder. An exploratory laparotomy revealed gallbladder torsion without hepatic lobe torsion. Cholecystectomy was performed, and the prognosis was favourable.
Relevance and novel information
Gallbladder torsion without hepatic lobe torsion has not been previously reported in cats. The clinical signs, diagnostic imaging features and intraoperative findings differed from those reported in humans and dogs. The unique ultrasonographic appearance seen in this case may assist with future diagnoses of this rare condition in cats.
Introduction
Gallbladder torsion is a rare and potentially life-threatening condition that results from rotation of the gallbladder on its mesentery, leading to vascular compromise and subsequent necrosis. In human medicine, it is considered an uncommon cause of acute abdomen and is often associated with a congenitally mobile gallbladder, with precipitating factors such as increased intra-abdominal pressure, intestinal peristalsis or organ ptosis.1 –5
In veterinary medicine, gallbladder torsion has been reported in dogs and is typically associated with concurrent hepatic lobe torsion.6,7 To date, only one case involving gallbladder torsion in a cat has been described, which was also associated with hepatic lobe torsion. 8 Cases of gallbladder torsion without hepatic lobe involvement have been documented in dogs,9 –11 where potential aetiologies include congenital anomalies, previous abdominal surgery, trauma or sudden movement. However, the underlying pathophysiology in feline patients remains poorly understood.
Delayed diagnosis can result in complications such as gallbladder necrosis, rupture or biliary peritonitis, which may be life-threatening.9,10
This case report describes the first known feline case of gallbladder torsion without concurrent hepatic lobe torsion, confirmed via exploratory laparotomy. The report emphasises the ultrasonographic findings, surgical observations, histopathological features and clinical outcome, contributing valuable information to the current limited literature on this condition in cats.
Case description
A 4-year-old castrated male domestic shorthair cat weighing 4.3 kg was referred to our hospital because of persistent anorexia, lethargy and vomiting (once daily for 2 days). The referring veterinarian had performed blood tests that showed markedly elevated liver enzymes; bilirubin had been detected in the cat’s urine. The cat had a history of idiopathic cystitis and no prior abdominal surgeries.
On presentation, the cat was lethargic, with a heart rate of 184 beats per min, respiratory rate of 20 breaths per min and rectal temperature of 39.3°C. The mucous membranes were pink without jaundice and abdominal palpation revealed no apparent pain. Complete blood count (CBC) (Procyte DX; IDEXX) was within the reference intervals (RIs). Serum biochemistry (FUJIDRI-CHEM 7000V; FUJIFILM) revealed the following abnormalities: total protein 8.4 g/dl (RI 5.7–7.8); albumin 3.9 g/dl (RI 2.3–3.5); alanine transaminase (ALT) 3272 U/l (RI 22–84); aspartate transaminase (AST) 440 U/l (RI 18–51); alkaline phosphatase (ALP) 588 U/l (RI 38–165); total bilirubin (TBIL) 1.0 mg/dl (RI 0.1–0.4); total cholesterol 286 mg/dl (RI 89–176); glucose 182 mg/dl (RI 71–148); and calcium 12.2 mg/dl (RI 8.8–11.9). Feline leukaemia virus and feline immunodeficiency virus tests were negative. Urine sediment revealed bilirubin crystals. The laboratory data are summarised in Table 1.
Serum biochemistry results at presentation
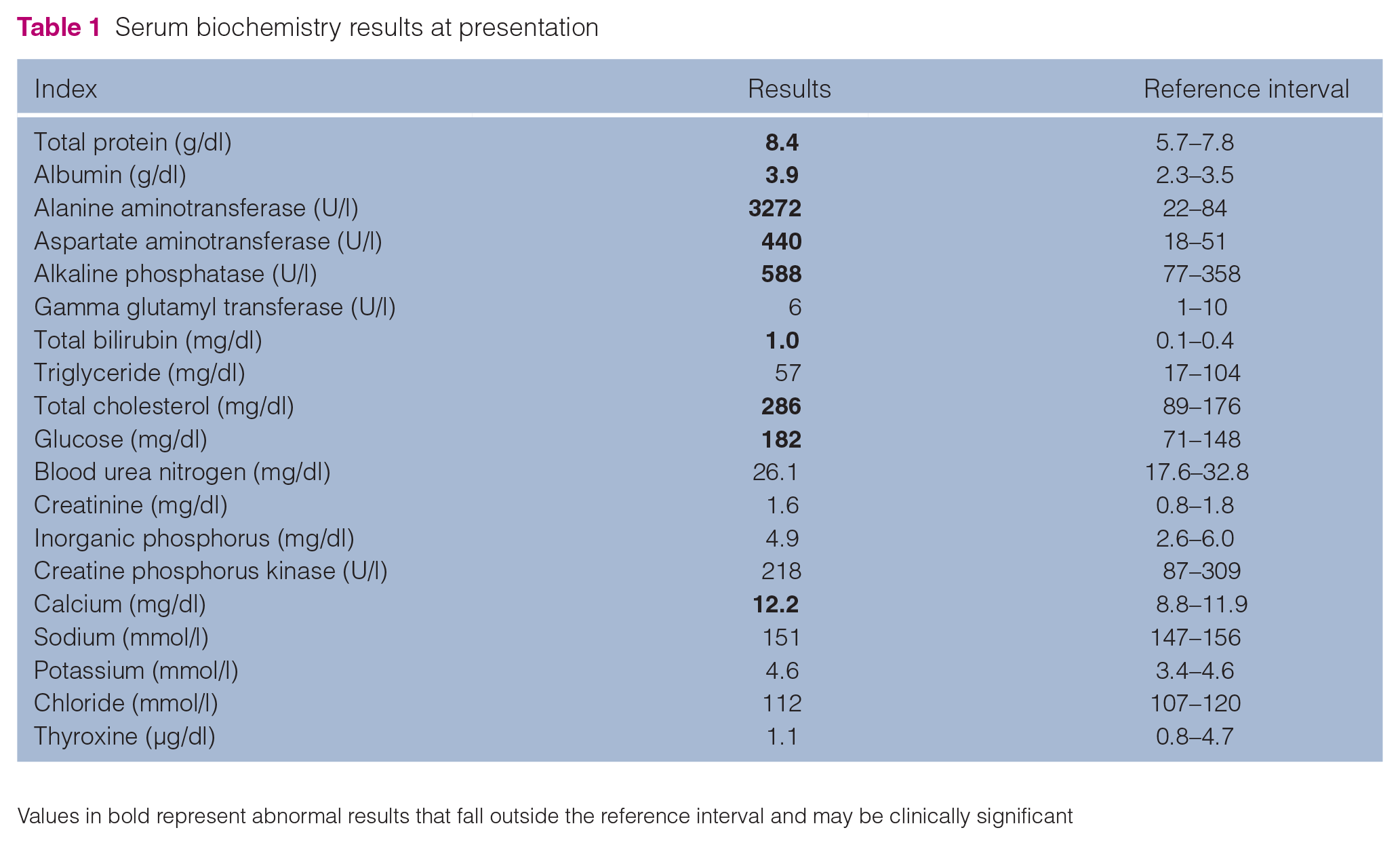
Values in bold represent abnormal results that fall outside the reference interval and may be clinically significant
Thoracic and abdominal radiographs were unremarkable. Abdominal ultrasound (ARIETTA 70; FUJIFILM) showed no gallbladder enlargement or wall thickening. The surrounding fat was locally hyperechoic. The gallbladder was small, the cystic duct appeared thin and straight, and the gallbladder body displayed a continuous spiral shape with alternating C- and reverse C-shaped configurations (Figure 1). No sludge, turbidity or gallstones were noted. Colour Doppler evaluation was not performed. The liver parenchyma was diffusely hypoechoic compared with the falciform fat. No hepatic lymphadenopathy was observed.
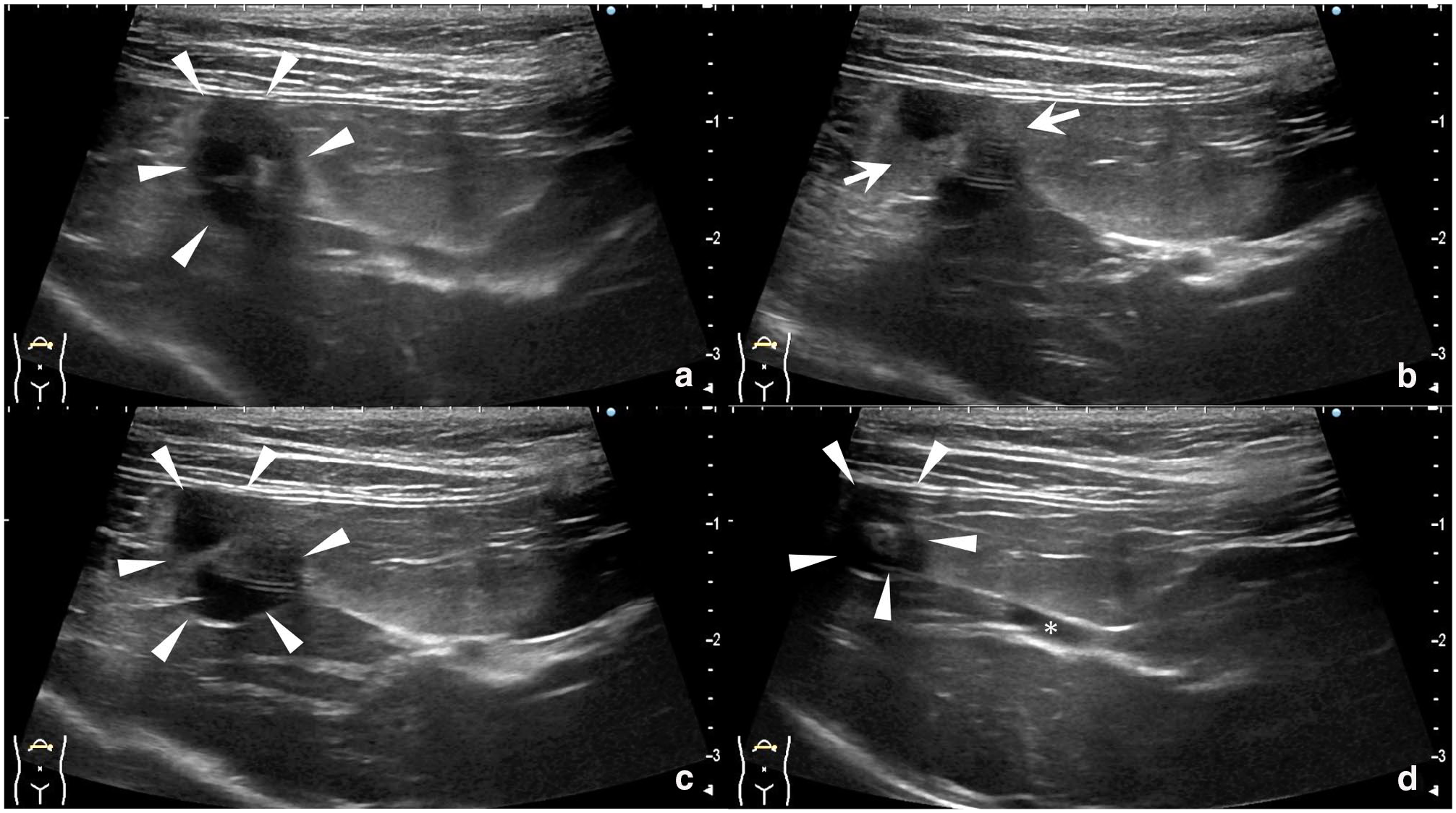
Transverse ultrasonographic images showing the gallbladder from fundus to cystic duct. (a) Gallbladder fundus with continuous C- and reverse C-shaped curves (arrowheads). (b) Hyperechoic fat surrounding the gallbladder body (arrows). (c,d) Spiral-shaped gallbladder body (arrowheads); the cystic duct appears narrow and straight (*)
Ultrasound-guided fine-needle aspiration of the liver revealed mild hepatocellular vacuolar change. Although hepatobiliary disease was suspected, a definitive diagnosis was not achieved. With the owner’s consent, exploratory laparotomy and tissue biopsy were performed on the same day.
Surgery revealed a four-fold torsion centred on the body of the gallbladder, distal to the cystic duct. No hepatic lobe torsion was observed. The fundus adhered to the omentum. No gallbladder rupture, ascites or changes in colour were found (Figure 2). The gallbladder was retracted using forceps, ligated and transected at the cystic duct proximal to the torsion, and removed. A liver biopsy was also performed. Approximately 34 mins after induction, a decrease in mean arterial blood pressure (48 mmHg) was observed during gallbladder manipulation. A bolus of synthetic colloidal solution (10 ml/kg) was administered, leading to normalisation of blood pressure within 2 mins. A vagal reflex secondary to gallbladder traction was suspected.
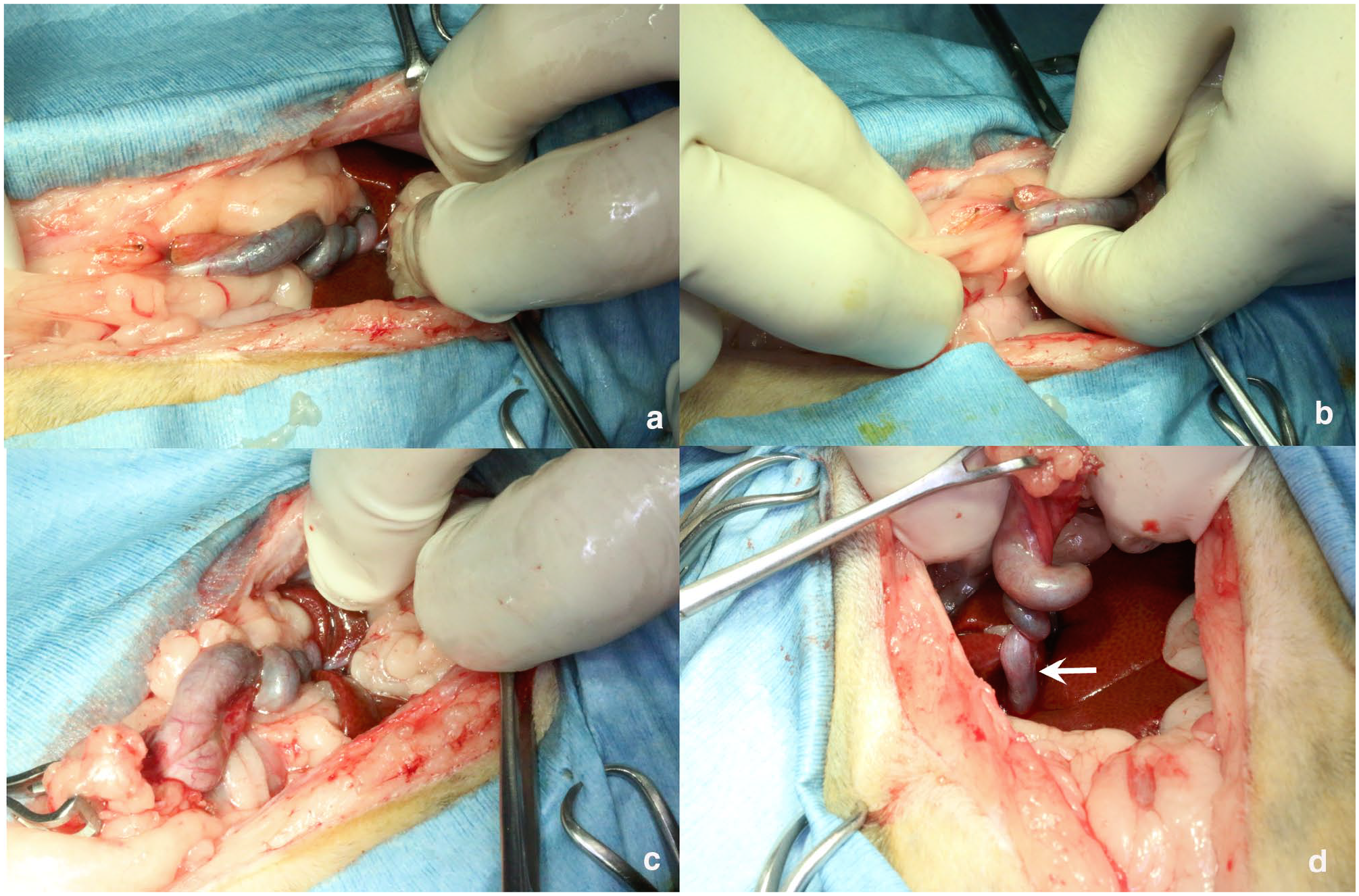
Intraoperative photographs. (a) Torsion of the gallbladder with four rotations observed from neck to body. (b) Adhesion of omental fat to the gallbladder fundus. (c) Hepatic lobes without evidence of torsion. (d) Traction of the gallbladder fundus revealing the torsion site. No torsion was seen near the proximal cystic duct (arrow)
Preoperative analgesia included buprenorphine (0.02 mg/kg IV). Postoperatively, the cat received intravenous fluids and cefazolin (20 mg/kg IV q8h). Although voluntary feeding resumed on postoperative day 1, assisted feeding with Hill’s a/d canned food was continued until day 4 to ensure adequate intake.
The excised gallbladder was shrunken and showed a spiral morphology from the body to the neck. Bile collected after cholecystectomy was submitted for culture and was negative.
Histopathology showed irregular gallbladder morphology with a thin smooth muscle layer, oedematous connective tissue and disorganised mucosal epithelium. Granulation tissue and haemosiderin-laden macrophages were observed where the omentum had adhered. Dilated lymphatics were noted around the bile ducts. Liver histology revealed preserved hepatocellular architecture without significant abnormalities (Figure 3).
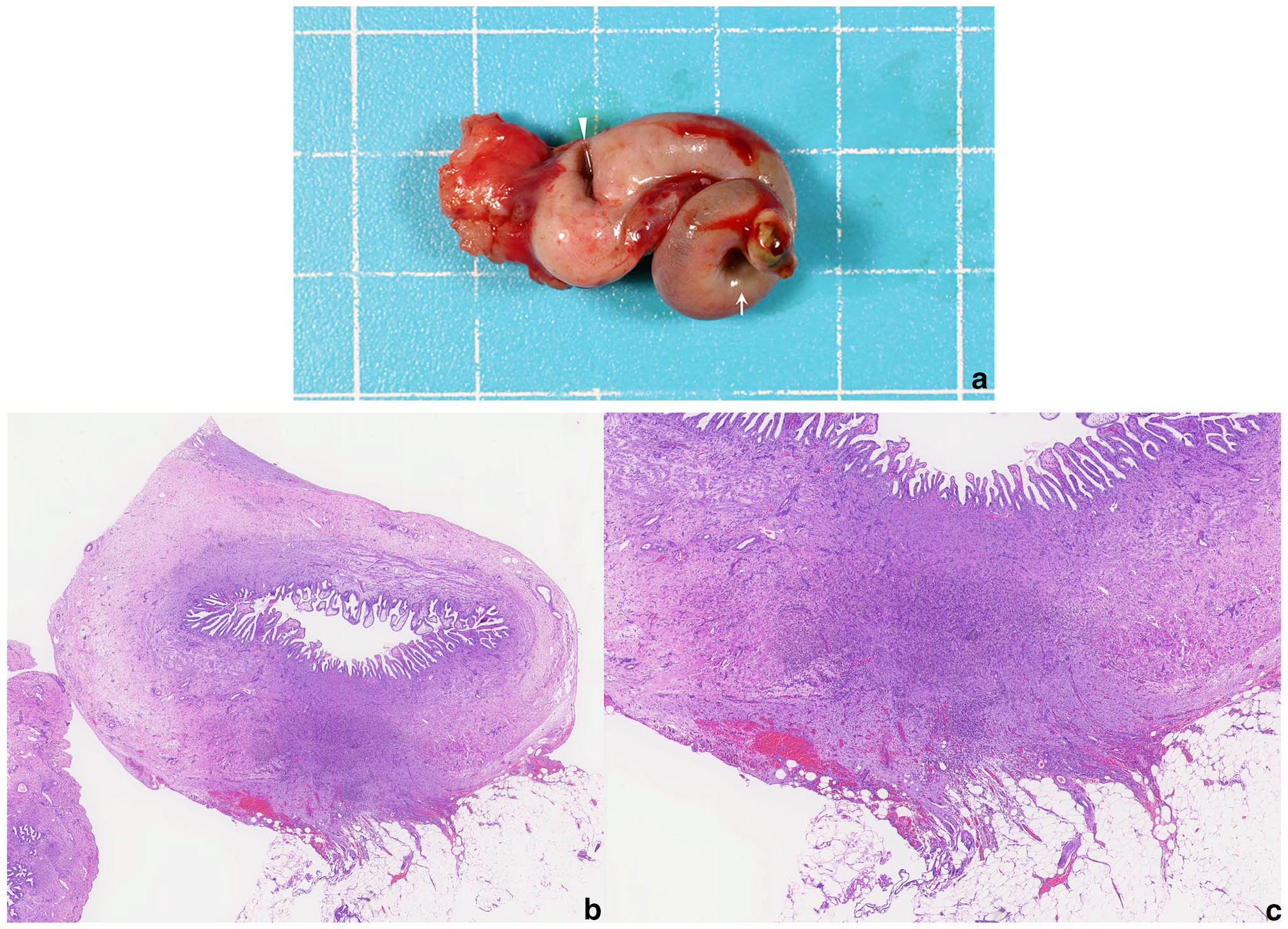
(a) Excised gallbladder showing spiral morphology and shrinkage. The neck shows mild greenish discoloration (arrow). (b, c) Histopathology of the gallbladder wall at the adhesion site with granulation tissue and haemosiderin-laden macrophages. Original magnification x 30 (b) and x 60 (c)
Postoperative recovery was uneventful, and the cat was discharged 4 days after surgery. At the 1-week follow-up, clinical signs had resolved and serum ALT, AST, ALP and TBIL had returned to within normal limits. Cholesterol, glucose and calcium concentrations had normalised by the day after surgery. Albumin levels were not re-evaluated postoperatively. The prognosis remained excellent.
Discussion
Previous reports have described isolated gallbladder torsion in dogs with a ‘wandering gallbladder’ lacking hepatic attachments.10,11 In contrast, other cases have involved concurrent torsion of the gallbladder and adjacent liver lobes – specifically the right medial and quadrate lobes – twisting in the same direction,6,7 suggesting that gallbladder torsion may have occurred secondary to hepatic lobe torsion. These observations imply that there may be two distinct pathophysiological forms: primary (isolated) gallbladder torsion and gallbladder torsion associated with hepatic lobe torsion.
In this case, the gallbladder torsion occurred independently and no evidence of a wandering gallbladder was found, indicating a pathophysiology distinct from previously reported cases. Grossly, the gallbladder fundus was adherent to the omentum, and histopathology revealed chronic inflammatory changes in the gallbladder wall. The absence of prior abdominal surgery argues against postoperative adhesions. These findings suggest that adhesion of the omentum to the fundus may have created an axis between the fundus and the neck of the gallbladder, leading to rotational instability and eventual torsion. Although the precise trigger remains unknown, possible contributing factors include gallbladder distension or contraction, body movement and fluctuations in intra-abdominal pressure caused by neighbouring organs such as the gastrointestinal tract or urinary bladder. No similar mechanisms have been reported in cats, dogs or humans.
Ultrasonographic findings differed from those reported in human and canine gallbladder torsion. In humans, common findings include gallbladder enlargement, wall thickening, malposition, pericholecystic fluid and the ‘cystic duct knot sign’. In some cases, absent blood flow in the gallbladder wall is seen on Doppler imaging.4,5,11 –15
In dogs, ultrasonographic features include ascites, gallbladder enlargement, wall thickening and displacement; however, some cases lack distinct features.6,9,10,16 The previously reported feline case involving hepatic lobe torsion showed marked ascites, echogenic fat, enlarged liver lobes, gallbladder displacement, wall thickening and intrahepatic duct dilation. 8
In contrast, the present case showed a small gallbladder with a spiral-shaped body, thin cystic duct, indistinct fundus and localised hyperechoic fat, without enlargement, wall thickening or displacement. No pericholecystic fluid or gallstones were detected. Doppler evaluation was not performed but should be considered in future cases to assess blood flow.
In human medicine, the absence of blood flow within the twisted gallbladder wall on colour Doppler imaging has been reported as a useful diagnostic indicator of gallbladder torsion.5,6,11,13 In contrast, such Doppler findings have not been described in canine reports. Although Doppler ultrasound has been used to evaluate vascular flow in torsed hepatic lobes in feline cases,6–10,16 there are no reports specifically assessing blood flow within the gallbladder wall during torsion in cats. Although Doppler was not performed in the present case, evaluation of gallbladder wall perfusion using colour Doppler may serve as a valuable diagnostic tool for gallbladder torsion in both dogs and cats in the future.
This case suggests that gallbladder torsion in cats may present with ultrasonographic features distinct from those in other species. In particular, a spiral-shaped gallbladder body and hyperechoic surrounding fat may be helpful diagnostic clues. Further accumulation of similar feline cases is necessary to clarify diagnostic criteria.
Gallbladder torsion in cats is extremely rare, with only one previously reported case secondary to hepatic lobe torsion. 8 This is the first report of gallbladder torsion without hepatic lobe involvement in a cat.
Further studies are needed to clarify the prevalence and predisposing anatomical features of gallbladder torsion in cats. The diagnostic utility of Doppler evaluation should also be explored.
In cats with elevated liver enzymes, ultrasonographic findings such as a spiral-shaped gallbladder body with continuous C- and reverse C-shaped curves, loss of the normal teardrop-shaped morphology of the gallbladder and pericholecystic hyperechoic fat may be indicative of gallbladder torsion.
In the present case, follow-up was available up to 2 years postoperatively, during which time the cat showed no signs of hepatic failure and remained clinically well. Previous reports suggest that early diagnosis and surgical intervention result in favourable outcomes;6,10 however, if diagnosis is delayed, complications such as gallbladder rupture or bile peritonitis may occur.9,10 These findings indicate that early diagnosis followed by prompt surgical management is crucial for preventing severe complications and improving prognosis.
Conclusions
Gallbladder torsion without hepatic lobe torsion can occur in cats and may present with unique ultrasonographic features. These include a spiral-shaped gallbladder body with continuous C- and reverse C-shaped curves and localised hyperechoic fat. Such features should prompt consideration of gallbladder torsion as a differential diagnosis in cats with elevated liver enzymes and non-specific gastrointestinal signs. Further studies are needed to establish the clinical and diagnostic characteristics of this rare condition in feline medicine.
Footnotes
Acknowledgements
The authors would like to thank all veterinary staff involved in the case management and care of the patient described in this report. Additionally, we would like to express our gratitude to Dr Nomura for their dedicated care and contribution to this case.
Accepted: 3 June 2025
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
