Abstract
Case summary
The cat described in this case report presented with progressive non-ambulatory tetraparesis. It was diagnosed with an occipitoatlantoaxial malformation (OAAM), which resulted in severe compressive myelopathy. This condition was caused by a flattening of the occipital condyles and shortening of the vertebral body of the axis. In addition, there was dysplasia in the atlantoaxial joint, which was observed on CT. The cat was treated surgically using a plate, which was custom-made with the help of a three-dimensional model of the malformation. Three weeks after surgery, the cat was able to walk without assistance; 5 years after surgery, the cat showed a normal neurological examination.
Relevance and novel information
Surgical planning using three-dimensional technology was useful in a complex case of OAAM. This is the first documented feline case that uses this type of technology for this condition.
Keywords
Introduction
Occipitoatlantoaxial malformations (OAAMs) are rare in dogs and even less frequently reported in cats. 1 These include atlanto-occipital fusion, atlas and odontoid process hypoplasia, axis malformation and modification of the atlantoaxial joint, resulting in vertebral canal narrowing observed in up to 50% of cases.2–11 Exposure to teratogenic substances during pregnancy has been suggested as a cause, although not proven. 12 Clinical signs develop during the first year of life and include neck pain, ataxia and ambulatory tetraparesis/tetraplegia from spinal cord compression.2 –20 Diagnosis is made through radiographs but myelography, CT and MRI offer greater insights into the extent of spinal cord compression. 1 Flexion radiographs of the neck can help diagnose conditions such as atlantoaxial instability; however, although this test diagnostic is well documented, it should be used cautiously owing to the risk of worsening spinal cord compression. 21
OAAMs has been documented in various species, including a calf, 13 two cats,14,15 a lamb, 16 a goat, 17 a rabbit, 18 dogs2–12,22 –24 and two domestic ferrets. 20 They are more frequent in Arabian horses, where they are well studied. 25 Similar to dogs, three presentations have been described in foals: death during birth; tetraparesis/tetraplegia at birth; or progressive ataxia. 19
Surgical stabilisation of the occipitoatlantoaxial (OOA) joint using Kirschner wires, screws, locking plates and custom-made plates has been described in dogs, where fixation of the OOA joint was achieved.22 –24 One cat with OAAM and atlantoaxial subluxation was stabilised with ventral transarticular pin placement, odontoidectomy and atlantoaxial joint arthrodesis, focusing solely on stabilising the atlantoaxial joint to address the instability at that level. 14
The aim of the present report was to describe the ventral stabilisation and dorsal decompression of an OAAM in a cat using a custom-made plate based on a three-dimensional model of the malformation and to report its long-term outcome.
Case description
A 4-month-old female Maine Coon cat presented with a 2-week history of progressive tetraparesis. A neurological examination revealed non-ambulatory tetraparesis with normal spinal myotatic reflexes and withdrawal reflexes in all four limbs, along with a normal cranial nerve examination. The lesion was localised at spinal cord segments C1–C5. Differential diagnoses included atlantoaxial subluxation or OAAM, immune-mediated or infectious myelitis or hereditary neurodegenerative disease. Complete blood count, serum biochemistry and electrolyte analysis were normal, as were thoracic radiographs; however, the hip radiographs demonstrated left coxofemoral incongruence.
Cervical radiographs revealed a flattening of the occipital condyles and bony overgrowth at the level of the atlantoaxial joint (Figure 1). CT was performed using 16-slice helical equipment (Brivo CT385; General Electric Healthcare) before and after an injection of iodinated iohexol contrast (Omnipaque 300 mg iodine/ml; 0.3 ml/kg injected into the lumbar subarachnoid space at the L5–L6 intervertebral space) to complete the myelo CT study. CT and myelography (Figure 2) showed bilateral occipital condyle hypoplasia, hypertrophy of the atlantoaxial joint, non-union of the centrum of the atlas and enlarged odontoid process, causing severe spinal cord compression at the point where the odontoid process and atlas centrum failed to fuse (Figure 3). A diagnosis of congenital OAAM with secondary compressive myelopathy was made. Decompressive surgery was recommended because of the progressive nature of the cat’s neurological deficits. Meanwhile, a neck splint was placed. 26 A three-dimensional-printed reconstruction of the caudal skull, atlas and axis (Figure 4) was used to manufacture a custom-made stainless-steel plate for ventral fixation (Figure 5).

Right lateral radiograph of the occipitoatlantoaxial joint showing flattening of the occipital condyles, enlarged odontoid process and ventral bony overgrowth at the level of the atlantoaxial joint causing narrowing of the vertebral canal

Sagittal reconstruction of the myelo CT scan in a bone window showing a thickening of the ventral arch of the atlas, non-union of the centrum of the axis and narrowing of the vertebral canal

Three-dimensional reconstruction showing hypoplasia of the occipital condyles, thickening of the ventral arch of the atlas, and dysplasia and hypertrophy of the atlantoaxial joint

(a) Three-dimensional-printed reconstruction of the caudal cranium, atlas and axis of the cat (Beta Implants). The red arrow indicates the thickened odontoid process of the axis. (b) Three-dimensional-printed reconstruction of the caudal cranium, atlas and axis of the cat (Beta Implants)

Custom-made stainless-steel plate designed from a three-dimensional-printed model of the malformation (Beta Implants)
One month after diagnosis, the OAAM was surgically addressed. The cat was premedicated with methadone (0.2 mg/kg IV) and dexmedetomidine (0.5 mg/kg IV). Induction was with alfaxalone (1 mg/kg IV) and maintenance was with isoflurane (1.2%) in an oxygen flow of 0.6 l/min. Constant rate infusions of fentanyl (5 µg/kg/h) and ketamine (10 µg/kg/h) were administered during surgery.
The cat was positioned in dorsal recumbency with the neck extended. A modified ventral approach to the atlantoaxial junction as described by Shores and Tepper 27 was used, extending cranially to expose the atlanto-occipital joint. The joint capsule was removed; as a result of thickening, reduction was not feasible. The occipital condyles, the wings of the atlas and the body of the axis were exposed, ventrally stabilised with a custom-made plate (Figure 6) and anchored with six 2.5 mm stainless-steel cortical screws (Beta Implants): two in the axis, two in the atlas and two in the occipital condyles.

Intraoperative image of the first surgery showing the plate being anchored to the occipital bone, atlas and axis with screws through the previously created holes (cranial = left, caudal = right)
A postoperative CT scan revealed that the right occipital screw had penetrated the cranial cavity, requiring revision surgery (Figure 7). Because of excess bone and fibrous tissue formation in the process, atlantoaxial reduction was not possible, and dorsal decompression was postponed to the following day because of hypothermia.
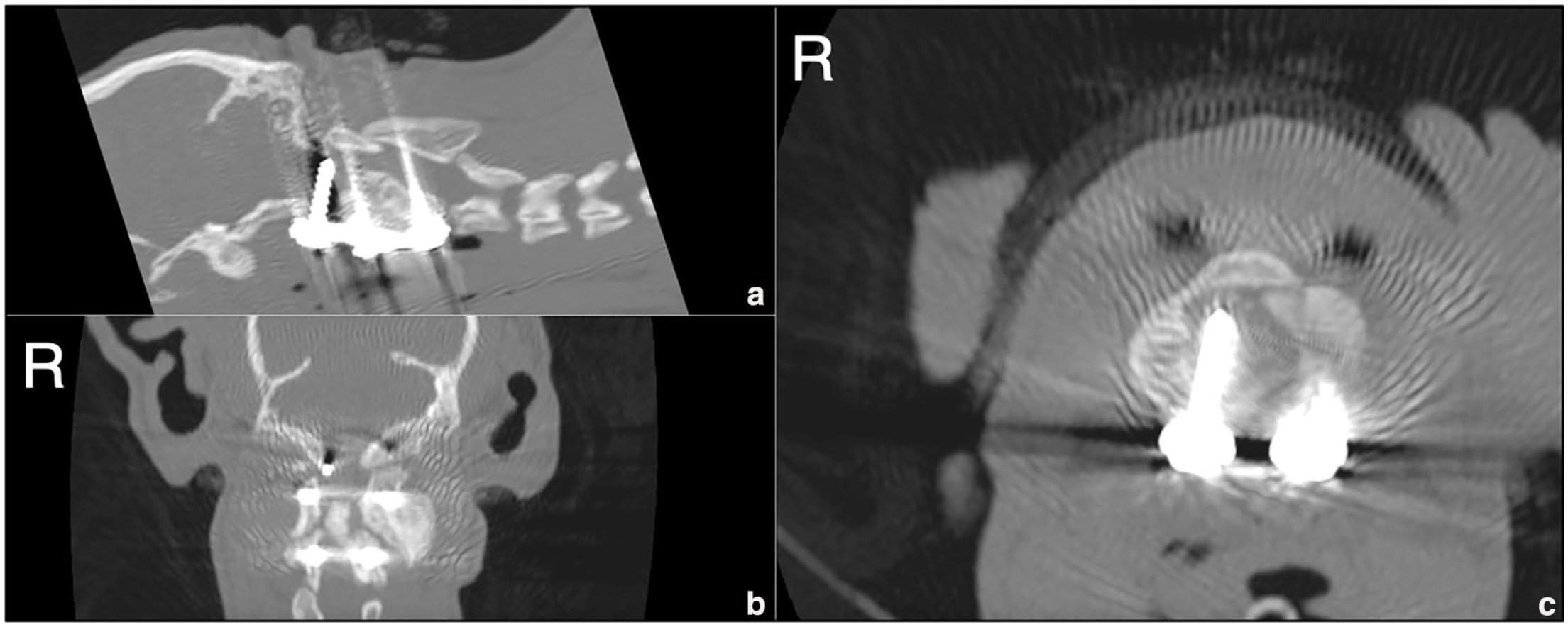
Postoperative CT scan in (a) sagittal, (b) dorsal and (c) transverse reconstructions showing the right occipital screw invading the cranial cavity
The cat was treated with dexamethasone (0.05 mg/kg IV q24h), gabapentin (15 mg/kg PO q8h) and pantoprazole (1 mg/kg IV q24h), alongside fentanyl (5 µg/kg/h) and ketamine (4 µg/kg/h) in a constant rate infusion. The following day, anaesthesia and aseptic preparation were repeated. The cat was positioned in sternal recumbency for a skin incision from the occipital protuberance to the caudal aspect of the axis.
The dorsal muscles were retracted to reveal the atlantoaxial space. Using a pneumatic motor, the caudal half of the atlas arch and the cranial third of the spinous process of the axis were removed to decompress the vertebral canal. Adequate spinal cord decompression was confirmed by a postoperative CT scan (Figure 8) and radiography (Figure 9).

Postoperative CT scan after dorsal laminectomy in (a) sagittal, (b) dorsal and (c) transverse reconstructions showing adequate spinal cord decompression
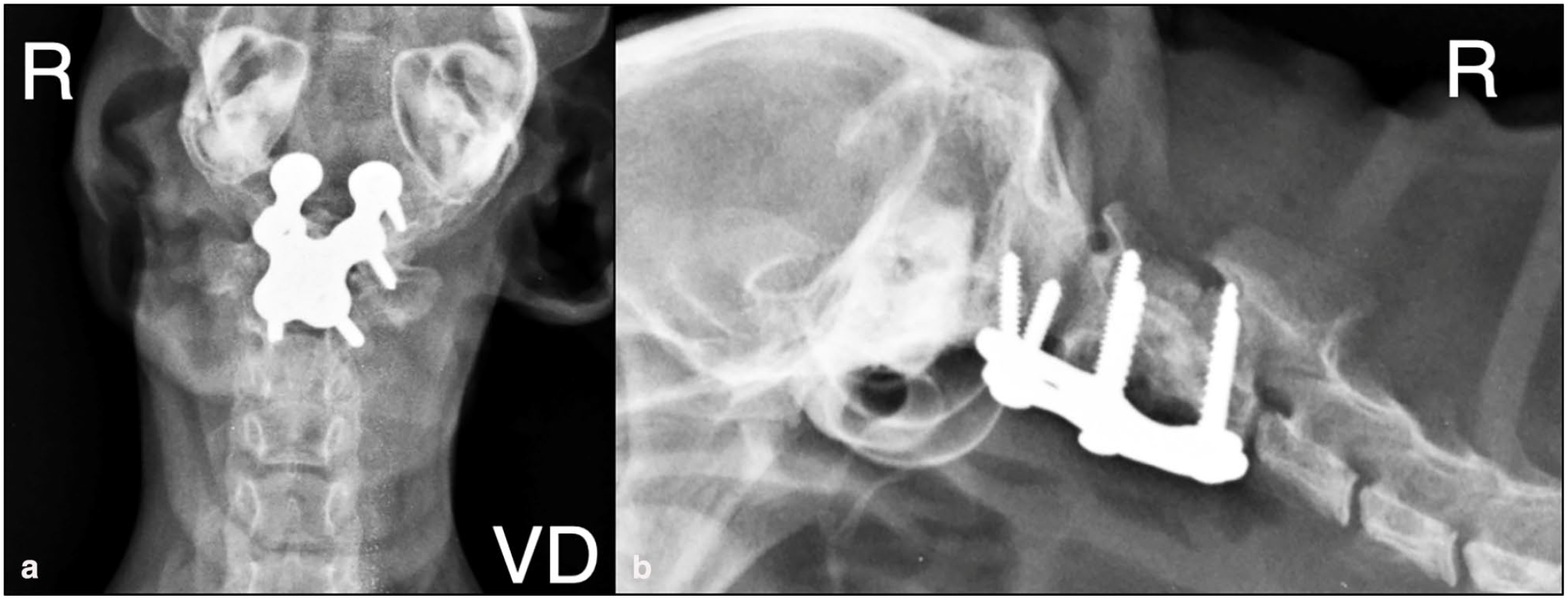
(a) Ventrodorsal and (b) right lateral radiographs of the occipitoatlantoaxial joint immediately after the decompressive surgery showing optimal placement of the plate and screws and the dorsal laminectomy
The cat experienced two episodes of respiratory difficulty, bilateral miosis and opisthotonus from suspected increased intracranial pressure (ICP) secondary to spinal cord swelling or spinal cord dysfunction. Dexamethasone (0.05 mg/kg) and mannitol (0.5 g/kg IV) were administered, resulting in significant improvement within 12 h. The cat remained stable, alert and non-ambulatory tetraparetic during the following week and was discharged with prednisone (0.5 mg/kg q12h) and omeprazole (1 mg/kg q12h) for 1 week.
Follow-up revealed progressive improvement, with moderate proprioceptive ataxia and tetraparesis by the first check-up 7 days later and there were no further complications.
Five years postoperatively, the cat remained neurologically normal and follow-up radiography showed displacement in the right occipital screw; however, the OAA joint and C2 and C3 vertebrae appeared to be fused (Figure 10).

(a) Ventrodorsal and (b) lateral control radiographs of the occipitoatlantoaxial joint showing mobilisation of the screw implanted in the right occipital condile and fusion of basioccipital bone, C1, C2 and C3 vertebrae 5 years postoperatively
Discussion
OAAMs have been documented in dogs,1–12,22,23 horses,19,25 ferrets 20 and farm animals,13,16–18 in addition to humans; 1 however, in cats, this is limited to two cases14,15 and only one was surgically treated. 14 The present case represents the first cat with OAAM treated with combined ventral fixation using a custom-made plate based on a three-dimensional model and decompressive dorsal laminectomy.
OAAMs vary in osseous abnormalities, neurological signs and degree of spinal cord compression. Treatment should be tailored to the specific malformation and neurological grade on a case-by-case basis.
Treatment can be medical or surgical. The medical procedure relies on neck immobilisation with a splint and the use of anti-inflammatory and analgesic drugs. 15 A cat diagnosed with a congenital OAAM was treated medically with good results, presenting a normal neurological examination 12 months after diagnosis. 15 In this case, surgical treatment was not pursued as a result of the severity of the malformation (abnormal osseous tissue between the occipital bone and the atlas, absence of the dens and partial hypoplasia of the vertebral arches of the atlas and axis). 15
The evaluation of the surgical treatment of OAAMs in domestic animals is limited to reports of one cat 14 and four dogs,22 –24 with different implants and variable outcomes and complications.
Surgical treatment has been reported in four dogs and one cat with secondary atlantoaxial subluxation, which focused on stabilisation of the subluxation.14,22 –24 One dog, stabilised with multiple implants, cement and a bone graft had implant failure and was euthanased. 23 Another dog, stabilised with two cortical screws occiput–C1–C2 and bone graft, showed postural deficits without pain 26 months later. 22 In the third dog, stabilisation with odontectomy, Kirschner wires and a bone graft showed a good prognosis and 40% bone fusion after 15 weeks of follow-up. 23 The last dog, treated with a customised three-dimensional-printed titanium locking plate that was placed ventrally on the atlantoaxial joint surface, showed a good recovery after 9 months, with only subtle left-sided cervical torticollis. 24 The only reported cat with a surgically-treated OAAM suffered a sudden neurological deterioration due to atlantoaxial subluxation. 14 Ventral stabilisation using transarticular Kirschner wires, atlantoaxial arthrodesis and odontoidectomy were performed, with the cat responding well and it was neurologically normal 18 months postoperatively.
In the present case, subluxation was not observed; however, the enlarged odontoid process and the atlanto-occipital joint produced a severe narrowing of the vertebral canal due to overgrowth. The initial goal of surgery was to reduce the compression by atlantoaxial traction and ventral surgical stabilisation; however, fibrosis and bone remodelling of the atlantoaxial joint prevented reduction and subsequent decompression. For that reason, a second procedure was necessary to decompress the spinal cord dorsally.
Surgery in this case was elected owing to the cat’s worsening neurological status despite medical treatment.
The use of three-dimensional-printed models has recently been introduced in veterinary medicine to facilitate the visualisation of complex anatomy. Using these techniques, it is easier to understand the spatial relationship between adjacent structures. 28 In our cat, we opted for this type of technology since it allowed us to understand this complex malformation while calculating the angles and lengths of the necessary implants for correct positioning.
Postoperative complications in similar surgeries include respiratory difficulty from hypoventilation and increased ICP, as seen in our case. Treatment focused on improving ventilation, oxygenation and fluid balance, with diuretics or corticosteroids considered for severe cases. A screw loosening was noted but did not cause complications, and the cat made a full recovery. Other rare postoperative complications include seroma formation, Horner’s syndrome and implant failure. 14
The prognosis of patients with surgically-treated congenital OAAMs appears to be good if the animal survives the postoperative period.14,22 –24 A good outcome is based on the resolution of neurological signs or the interruption of the progression of neurological deficits. A recurrence of neurological signs is possible as a result of implant failure, which, to date, has not occurred in the cat in this case report, despite the displacement of one occipital screw.
Given the scarce number of described cases in small animals, it is not possible to establish a standard treatment protocol. These are malformations that occur during development and subsequent calcification of the ossification centres; therefore, many animals are not skeletally mature at the time of initial evaluation. In skeletally immature animals with mild neurological signs, it might be advisable to postpone any surgical intervention until growth is complete to reduce the possibility of implant failure as a result of insufficient calcification. The cat in the present report was not fully grown at the time of the surgery; however, the severity and progression of the signs warranted early intervention. At 4.5 years postoperatively, repeat radiography showed that the OAA joint appeared completely fused. At 5 years postoperatively, the cat was skeletally mature and had fully recovered.
This case represents the first reported cat with a complex OAAM treated with a combined surgical approach, consisting of ventral fixation using a custom-made plate based on a three-dimensional-printed reconstruction followed by a decompressive dorsal laminectomy. Despite the severity of the presenting clinical signs and the skeletal immaturity at the time of surgery, the cat has achieved an excellent long-term outcome, with a normal neurological examination and an excellent quality of life.
Conclusions
Surgical intervention for OAAMs should be considered even in young immature cats presenting with neurological signs or progressive deterioration. The use of three-dimensional technology might help to design custom-made implants and minimise the risks of such complex and delicate surgeries, with excellent long-term results, as in this case. However, the stainless-steel implant used in this case caused significant artefacts in the CT images. The use of alternative materials for three-dimensional-printed plates and screws may help reduce these artefacts and improve advanced imaging quality.
Footnotes
Acknowledgements
The authors would like to acknowledge all the staff involved in the treatment of this cat for their dedication and commitment to the animal’s care, as well as that of its owners.
Author note
The preliminary results of this paper were presented as a poster and abstract at the annual congress of the European Society of Veterinary Neurology/European College of Veterinary Neurology (ESVN-ECVN), Majorca, Spain, 23–24 September 2022.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required
