Abstract
Case summary
A 4-year-old male castrated domestic shorthair cat presented with a 3-month history of coughing and progressive dyspnea. The cat was diagnosed with idiopathic chylothorax after serum biochemistry, thoracocentesis, cytology of pleural effusion, echocardiography and thoracic imaging were performed. After failure to respond to medical management with repeated thoracocenteses, the oral administration of rutin and a low-fat diet, the cat underwent staged interventions consisting of CT lymphangiogram, subtotal pericardiectomy and pleural access port (PAP) placement, followed by thoracic duct ligation (TDL) and cisterna chyli ablation (CCA) 2 months later. The cat made a clinical recovery based on resolution of chylous pleural effusion 1 month after TDL and CCA, and the PAP was removed 1 month later. The cat remained free of clinical signs 3 years postoperatively.
Relevance and novel information
This is the first report documenting the use of a PAP for the management of feline chylothorax to disease resolution. Employing this device enabled frequent, low morbidity drainage of the pleural effusion as an alternative to repeated thoracocenteses and facilitated staging the interventions throughout management of idiopathic chylothorax in a cat. PAPs can be used successfully in the management of chylous pleural effusion in cats.
Keywords
Case description
A 4-year-old castrated male domestic shorthair cat was presented to its referral veterinarian with a 3-month history of coughing and worsening dyspnea. One month before the original presentation, thoracic radiographs demonstrated moderate pleural effusion. Amoxicillin–clavulanate (125 mg PO q12h for 14 days) was empirically prescribed. No clinical improvement was noted, the coughing persisted over the subsequent 2 weeks and the cat was referred for further evaluation.
At initial presentation, adequate body weight (5.8 kg), tachypnea (60 breaths/min) and inspiratory dyspnea were observed. Heart sounds and bronchovesicular sounds were muffled bilaterally. Thoracic ultrasonography revealed bilateral pleural effusion and 145 ml of opaque pink fluid was drained via thoracocentesis; dyspnea subsequently improved (Figure 1). Pleural effusion analysis and blood parameters were consistent with a chylous effusion (Table 1). Echocardiography and abdominal ultrasonography were unremarkable, ruling out other causes of chylous effusion. These findings were most consistent with a diagnosis of idiopathic chylothorax.

Photograph of a sample of the slightly blood-tinged chylous pleural effusion aspirated via thoracocentesis on initial presentation
Relevant laboratory results from pleural effusion and serology analysis

The cat was treated with rutin (250 mg PO q8h; Solgar) and a low-fat prescription diet (w/d Prescription Diet; Hill’s Pet Nutrition). At the recheck 7 days later, tachypnea (56 breaths/min) and dyspnea were again noted, and 160 ml of chylous effusion was evacuated via thoracocentesis. Two weeks later, a third thoracocentesis yielded 110 ml of chylous effusion. The cat was referred to a tertiary hospital for surgical intervention.
Upon presentation to the tertiary hospital, the cat’s physical examination findings were similar to the initial presentation and thoracocentesis yielded 108 ml of chyle. A staged surgical plan was developed for three reasons: (1) to ascertain if pericardectomy alone would resolve the chylothorax; (2) to allow full interpretation of the CT and surgical planning; and (3) to minimize anesthesia time to two shorter events. The first surgical intervention would consist of CT lymphangiogram, subtotal pericardectomy and placement of a pleural access port (PAP) (PP-202K, Le Port Kit PleuralPort; Norfolk Vet Products). Pleural effusion would be monitored by regular drainage through the PAP, and if clinical resolution of the chylothorax was not noted within 3 months, a second intervention involving thoracic duct ligation (TDL) and cisterna chyli ablation (CCA) would be performed.
The cat was sedated with alfaxalone (2 mg/kg IM) and midazolam (0.2 mg/kg IM), anesthesia was induced with alfaxalone (4 mg/kg IV) and the cat was maintained on inhalant isoflurane. A left popliteal lymph node cutdown was performed and 5 ml (1 ml/kg) of water-soluble contrast (iohexol, Omnipaque; GE Healthcare) was injected through a 25 G needle via a syringe pump into the lymph node over 5 mins, followed by whole-body CT scan to obtain a CT lymphangiogram. Apart from a scant amount of pleural effusion, no other abnormalities were noted. Contrast enhancement in the efferent lymphatic ducts leading from the left popliteal lymph node to the cisterna chyli and subsequently to the thoracic duct dorsal to the aorta was appreciated (Figure 2).

(a) Slow injection of the water-soluble contrast into the popliteal lymph node using a syringe pump to control the rate of infusion, and an intravenous line extension to facilitate maintaining the position of the 25 G needle. The entire procedure, including the cut-down to the lymph node, is performed in the CT room after the cat has been positioned for CT. (b) Reconstructed CT image showing the lymphatic drainage from the pelvic limb to the CC and TD. Reconstructed CT image showing the lymphatic drainage from the pelvic limb to the cisterna chyli (CC) and thoracic duct (TD)
After routine preparation for aseptic surgery, cefazolin (22 mg/kg IV) and a fentanyl bolus (3 µg/kg IV) were administered, followed by a fentanyl (3 µg/kg/h IV constant rate infusion [CRI]). Subtotal pericardiectomy was performed via a right-sided intercostal thoracotomy at the fifth intercostal space. The mediastinum was punctured digitally and right-angle forceps used to pass the 7 Fr radiopaque, fenestrated silicone tubing of the PAP through the sixth rib space. The PAP was secured using the provided boot to the titanium access port itself with 3-0 polypropylene per the manufacturer’s instructions (Figure 3). The thorax was closed routinely. A 19 G right-angle Huber needle (Norfolk Vet Products) was inserted transcutaneously into the PAP, 10 ml of air was aspirated until negative pressure was achieved and 1.5 ml of taurolidine-citrate catheter solution (TCS; Access Technologies) was instilled into the port. The cat recovered uneventfully, was maintained on maintenance fluids and the fentanyl CRI overnight and transitioned to buprenorphine (0.03 mg/kg TM) the following morning. The cat was discharged the morning after surgery on gabapentin (10 mg/kg PO q8h for 5 days), buprenorphine (0.03 mg/kg TM q6h for 5 days) and amoxicillin–clavulanate (12.5 mg/kg PO q12h for 7 days). Rutin was discontinued at that time.

(a) The pleural port device, consisting of the port that is placed subcutaneously, the white fenestrated silicone drain that is situated in the pleural cavity and the blue ‘boot’ that aids in securing the drain to the port. (b) The pleural port device in situ. The silicone drain is in the pleural cavity and the blue ‘boot’ is over the connection between the port and the drain. The port is being sutured to the latissimus dorsi with five interrupted sutures of 2-0 polypropylene (Prolene; Ethicon)
At a scheduled follow-up 4 days later, the cat had been doing well at home clinically, but its respiratory rate was 40 breaths/min. The skin over the PAP was aseptically prepared and 50 ml of chylous effusion was drained via the Huber needle, followed by flushing with TCS as previously described. The cat continued to do well clinically and subsequently presented every 1–2 weeks for PAP drainage. The effusion volume increased and 65–124 ml of effusion were aspirated from the PAP on each presentation.
On postoperative day 91 after the first intervention, the cat was anesthetized as before and underwent TDL and CCA. A left-sided paracostal approach caudal to the 13th rib was performed to enter the abdominal cavity, following a previously published technique for dogs. 1 The left dorsal diaphragm was incised and methylene blue (0.3 mg/kg; BioPharm) was diluted 1:1 with sterile saline and administered into the right colic lymph node (Figure 4). The dye was visualized entering the cisterna chyli and traversing into the thoracic duct dorsal to the aorta in the retropleural space. The thoracic duct was dissected away from the aorta and ligated with two small titanium clips (Hemoclip Plus; Teleflex). Cyanoacrylic tissue glue (0.5 ml, GLUture; Zoetis) was injected into the cranial aspect of the cisterna chyli in an attempt to obliterate any undetected collateral vessels. The entire visible cisterna chyli, apart from the portion adhered to the aorta, was then carefully dissected from the aorta with tenotomy scissors and removed. The dorsal aortic surface was then scarified with a dry cotton-tipped applicator. The diaphragm was closed with a continuous suture pattern of monofilament polydioxanone (PDS; Ethicon), the pleural cavity evacuated with a red rubber catheter and three-way stopcock, and the abdomen was closed routinely. Postoperative management was the same as for the previous intervention and the cat was discharged the following day.

Slow injection over approximately 1 min of the diluted methylene blue into the right colic lymph node through the left paracostal incision, using a 25 G needle
At the scheduled recheck 2 weeks later, the cat had healed appropriately and was doing well clinically, with no evidence of tachypnea or dyspnea. Drainage of the PAP yielded 6 ml of serosanguinous fluid. On a planned recheck at postoperative day 42 after the second intervention, the cat continued to do well and remained eupneic. The PAP yielded 2 ml of grossly serous effusion, with no persistent effusion appreciated on thoracic ultrasonography (Figure 5).

Photograph of the serous pleural effusion from the final aspiration of the pleural access port 42 days after thoracic duct ligation and cisterna chyli ablation. Note the minimal volume and lack of chylous appearance of the fluid
The cat continued to remain free of clinical signs attributable to pleural effusion over the subsequent several months. Approximately 6 months after initial presentation and 2 months after TDL and CCA, the owners requested removal of the PAP because of its prominence under the skin. A final outpatient procedure to remove the PAP under general anesthesia was performed and the cat was discharged later that same day (Figure 6).
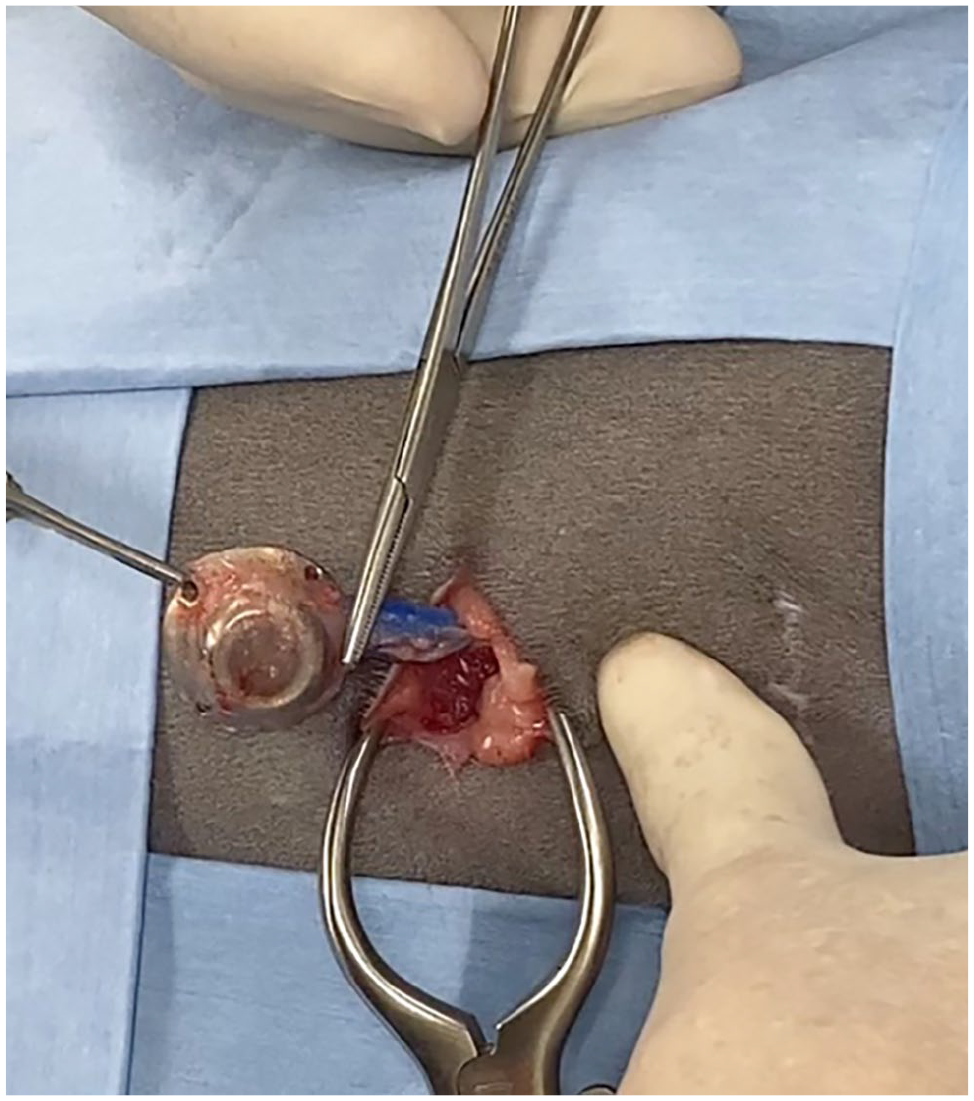
Removal of the pleural access port via a small incision directly over the port. The retaining sutures are cut, and the port and the associated drain removed. This is optional and is performed as an outpatient procedure under short general anesthesia
Two weeks later, the cat was clinically doing well, the physical examination was unremarkable and focused thoracic ultrasonography did not reveal evidence of pleural effusion.
At the last follow-up (via telephone) 3 years after removal of the PAP, the cat was reported to be doing well, with no unthrift or recurrence of tachypnea or dyspnea.
Discussion
Chylothorax is the accumulation of chylous effusion in the pleural space, which is typically caused by impaired lymphatic drainage. 2 Historically, the primary cause of chylothorax was thought to be secondary to traumatic injury and rupture of the thoracic duct, although subsequent studies document trauma as a rarer cause of feline chylothorax. 3 Lung lobe torsion, space-occupying lesions and cardiac disease have also been reported in association with chylothorax in cats.4 –6 The majority of the reported cases of feline chylothorax are diagnosed as idiopathic.2,3,7
Management of this cat initially involved feeding a low-fat diet and administration of rutin. Published results on the usefulness of rutin, a bioflavonoid, in the management of feline idiopathic chylothorax are disappointing. One study of four cats receiving rutin for the management of idiopathic chylothorax stated that 3/4 cats showed clinical improvement, although only one cat was associated with complete resolution of its chylous effusion and one cat failed to respond and was euthanized. 8 As there is the potential for spontaneous resolution of feline chylothorax, it is difficult to assess the actual efficacy of rutin in those cases where chylothorax resolved.9 –11
Surgical intervention was pursued as the cat was not improving clinically, there were concerns about restrictive fibrosing pleuritis developing as a result of duration of persistent effusion and placement of a PAP would be helpful to evaluate ongoing pleural effusion. Surgical management of feline chylothorax should be considered if medical management is unsuccessful, although some investigators recommend early surgical intervention in all cases of chylothorax in cats.2,3 Several surgical procedures and combinations thereof are used in cases of feline chylothorax: TDL, subtotal pericardiectomy and CCA have all been reported.1,2,12 Imaging of the thoracic duct before and after TDL with CT and lymphangiography, and direct visualization of the thoracic duct intraoperatively using methylene blue are recommended to improve surgical success.2,12 –24 A paracostal approach for combined TDL and CCA has been reported to aid in surgical visualization and avoidance of intercostal thoracotomy in dogs. 1 Success rate in cats for resolution of pleural effusion with TDL alone is varied (14–53%), but the success rate improved to 80% in cats with TDL and subtotal pericardiectomy combined.2,15,16 It is speculated that in the present cat, the combined surgical procedures performed in a staged fashion were key factors in successful management. Staging the interventions in this way allows the quicker thoracic procedures (PAP placement, subtotal pericardectomy) to be performed at the same time as the diagnostic CT lymphangiogram, which can take some time. The lengthier intervention (the paracostal transdiaphragmatic approach to identify and ligate the thoracic duct and ablate the cisterna chyli) could then be undertaken as a separate intervention. It also allows for the possibility that the subtotal pericardiectomy may prove curative as a sole intervention. 17
The use of PAPs for chronic pleural effusions has been documented in the cat.18 –21 Vascular access pleural ports are thoracostomy tubes attached to titanium access ports, placed during surgery to allow for repeated aspirations of the pleural space by inserting a right-angle Huber needle into the port septum. 2 Typically, the ports are flushed with taurolidine-citrate, a solution used to prevent microbial colonization of the port and act as an intraluminal blood anticoagulant. 22 A PAP was used for long-term medical management in a dog with persistent chylothorax after lung lobe torsion. 20 Two cats with chylous effusion and PAP placement have been reported in the literature; one cat developed PAP obstruction before disease resolution and the other cat was euthanized because of acute pneumothorax after PAP placement. 18 In contrast to that study, the cat in this report underwent PAP placement without issue, and the port remained patent and successfully aided in management of the disease to resolution without complication. Without PAP placement, persistent pleural effusion in this cat would have necessitated repeat thoracocentesis on multiple separate events. The development of pyothorax is a concern when repeated thoracocentesis is indicated.23,24 Early placement of the PAP allowed for pleural fluid drainage without the need for repeated thoracocenteses with little morbidity to the patient and allowed time to see if subtotal pericardiectomy alone would be effective in the management of the idiopathic chylothorax. PAP placement allows for the staging of longer surgical procedures, such as those used in the management of feline idiopathic chylothorax, providing time for the patient to heal and determine if further surgical intervention may be necessary.15 –17 Risks of PAP placement should be evaluated against the risks of serial thoracocenteses and considered in cases where feline pleural effusion may be chronic.
Conclusions
To our knowledge, this is the first case of feline chylothorax managed with a PAP to disease resolution. Surgical intervention was an important part of therapy with subtotal pericardiectomy, TDL and CCA performed. The PAP prevented the need for repeated thoracocenteses and associated risks of repeated pleural puncture, with minimal discomfort to the patient. PAPs can be considered in the staged management of chylous pleural effusion in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies).No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
