Abstract
This study describes a lateral thoracotomy approach for thoracic duct cannulation and lymphatic fluid collection in a feline model. The thoracic duct was cannulated via a left lateral intercostal thoracotomy in 12 cats. Lymphatic fluid was collected for up to 16 days and analyzed on days 3, 9 and 16. The volume collected and duration of cannula patency were recorded. Contrast imaging of the thoracic duct was performed if fluid ceased to flow or at the end of the 16-day study period. In two cats, the cannula became dislodged within 24 h. For the remaining 10 cats, mean daily volume collected was 43.7 mL (median 41.0, range 2.3 to 152.4 mL), and mean duration of cannula patency was 8.2 days (median 6.5, range 3 to 16 days). Contrast imaging revealed that the cannula was patent in three cats, obstructed in two cats, and the thoracic duct had ruptured or had extravasation of contrast outside the duct in five cats. Cytological examination of lymphatic fluid from the three time points revealed normal appearing small lymphocytes (97%) and few (3%) non-degenerate neutrophils, macrophages, eosinophils, and plasma cells. Based on the results of this study, lateral thoracotomy approach for thoracic duct cannulation is a feasible technique for collecting lymphatic fluid in cats. This technique may have application as a model for short-term evaluation of thoracic fluid in cats; however, cannula patency was unpredictable and should be considered when utilizing this technique.
Chylothorax is among the more commonly diagnosed effusive pleural diseases in cats, the majority being of idiopathic rather than traumatic origin. 1 Cats with chylothorax suffer from tachypnea, dyspnea, lymphopenia, weight loss and anorexia. If left untreated pleural fibrosis may develop, which can lead to substantial respiratory insufficiency and potentially death.1,2 Conservative management including dietary modification or pharmaceuticals such as rutin have been largely unrewarding. Surgical treatment involving thoracic duct ligation is generally successful, but carries substantial morbidity highlighting the need for more effective medical treatments.3–5
Humans are similarly affected by chylothorax, and in many cases fluid accumulation can be persistent despite repeated thoracocentesis and medical therapy.5–9 As such, significant effort has been dedicated to the evaluation of various pharmaceuticals that reduce the production of lymphatic fluid including octreotide, midodrine and gefitinib.10–17 These same drugs may hold promise for the treatment of feline chylothorax, but due to the unique nature of hepatic metabolism in cats, therapies that have proven successful for humans should ideally be validated in a feline model prior to clinical application. 18
Thoracic duct cannulation is the most commonly used technique for lymph collection and flow analysis in research settings, and serves as the most predictable method for evaluating medical therapies used to alter lymph production or flow.19–24 Thoracic duct cannulation for collection of lymphatic fluid in cats has been described in only one previous report. 19 The technique involved a ventral cervicothoracic approach and reported long-term collection, but additional details such as the volume of production, mean duration of patency, fluid composition, and perioperative complications were not provided. 19
The purpose of this study is to describe a lateral thoracotomy approach for thoracic duct cannulation and lymphatic fluid collection in a feline model, including volume of fluid collected, duration of cannula patency, lymphatic fluid analysis, contrast imaging, and postoperative complications.
Materials and methods
Ethical statement
This protocol was approved by the University of Wisconsin, Institutional Animal Care and Use Committee. Animal use complied with the National Research Council Guide for the Care and Use of Laboratory Animals standard.
Animals
Twelve young, healthy intact female domestic shorthair cats (Liberty Research Inc, Waverly, NY, USA) were commercially sourced for use in this study.
Presurgical evaluation
Prior to surgery, all cats underwent a physical examination, complete blood count, and serum chemistry panel. Cats were deemed healthy on the basis of these results, and any cat that was found not to be systemically healthy was excluded from the study. Cats were fasted for approximately 12 h prior to anesthesia.
Anesthesia
The anesthesia protocol consisted of premedication with hydromorphone (0.1 mg/kg IM), and acepromazine (0.025 mg/kg IM), induction with thiopental (10 mg/kg IV), and maintenance with isoflurane and 100% oxygen delivered via endotracheal intubation. Monitoring included indirect Doppler blood pressure, electrocardiogram, and hemoglobin saturation (pulse oximetry). Lactated Ringer’s solution was administered (5 mL/kg/h IV) during the procedure. If needed, a single dose of atracurium (Novartis Pharmaceuticals USA, East Hanover, NJ, USA) was administered (0.1–0.4 mg/kg IV, mean duration of effect 20–35 min) to paralyze the respiratory muscles during the critical phase of cannula insertion. Positive-pressure ventilation (10 breaths/min with a maximum peak airway pressure of 15 cmH20) was utilized during the thoracotomy part of the procedure. Return to spontaneous breathing was confirmed prior to completion of surgery and reversal of atracurium was not required in any cat.
Surgical technique for thoracic duct cannulation
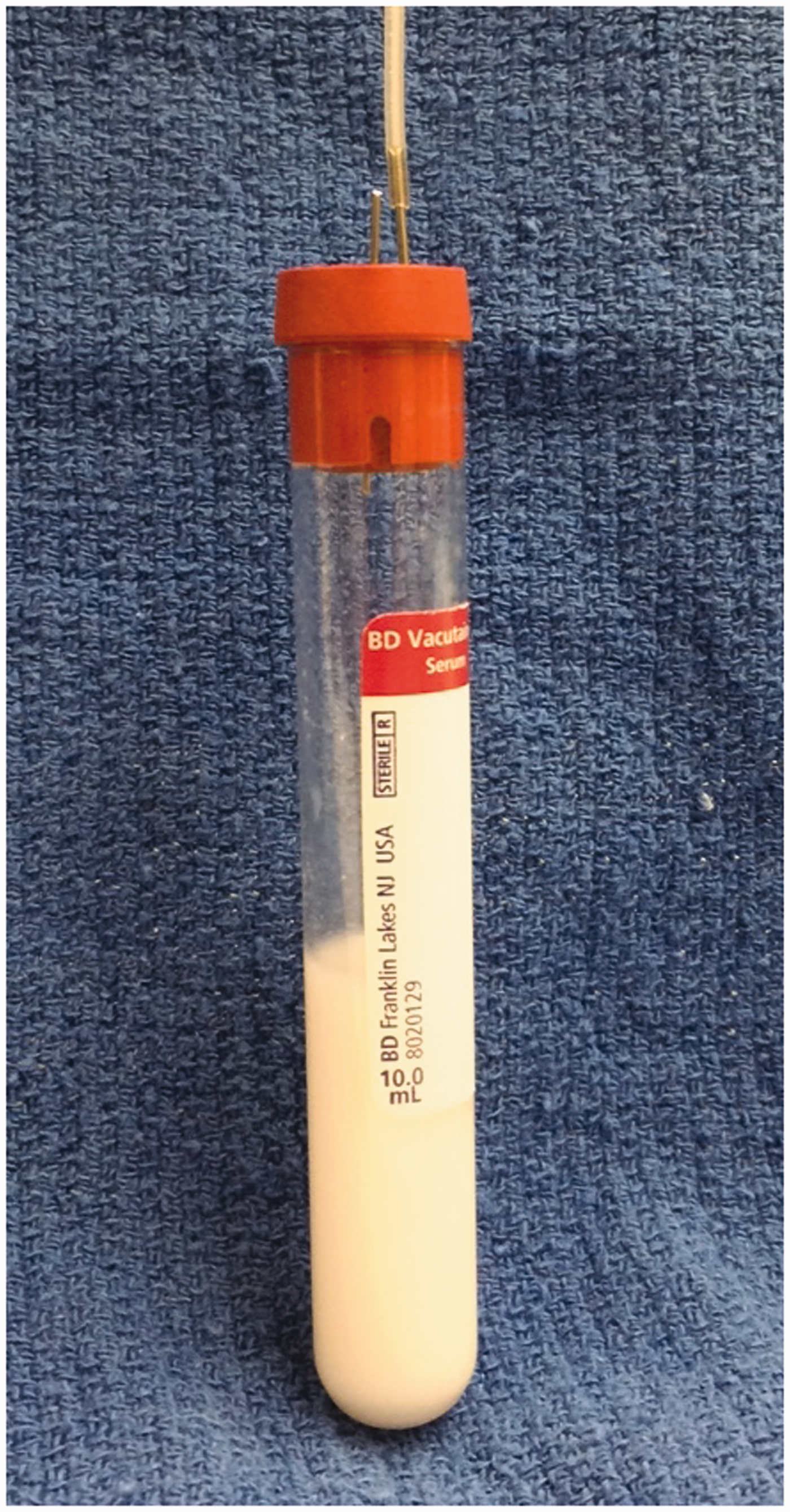
The cat was placed in right lateral recumbency and the left lateral thorax and abdomen were clipped and aseptically prepared. Surgery was carried out using the standard aseptic technique. A ninth intercostal thoracotomy was made. Upon entering the thorax, the lungs were retracted exposing the thoracic duct along the dorsolateral aspect of the aorta. Using an operating microscope (5–22 × magnification) the thoracic duct and any collateral branches were identified and carefully dissected from the surrounding pleura and adipose tissue. The thoracic duct and any collateral branches were ligated with 7-0 silk at the level of the eighth intercostal space to distend the thoracic duct caudally. A stay suture (11-0 nylon) was placed through the lateral wall of the thoracic duct at the level of the ninth or tenth intercostal space to enable manipulation of the duct and to prevent it from collapsing once incised. Using microdissecting scissors, an incision was made in the lateral wall of the thoracic duct just cranial to the stay suture. The cannula was inserted into the duct and advanced caudally for approximately 2 cm or to the level of the cisterna chyli. In some cases, the intercostal vessels overlying the thoracic duct were cauterized and transected to allow advancement of the cannula. The cannula was fashioned from a 40 cm length of silicone-coated silastic tubing (Silastic Laboratory Tubing, Fisher Scientific, Midland, MI, USA; 1.19 mm OD and 0.63 mm ID (n = 11) or 0.63 mm OD and 0.3 mm ID (n = 1)) with the tip cut to a 30° bevel. Immediately prior to insertion, the cannula was flushed with 0.1 mL of 1:100 sterile heparinized saline. Once positioned in the thoracic duct, the cannula was secured with two to three encircling ligatures using 7-0 silk (Figure 1). The cannula exited the thoracic cavity through a stab incision made in the dorsal aspect of the seventh intercostal space and tunnelled subcutaneously for 3 to 5 cm before exiting the skin on the craniolateral thoracic wall. The cannula was secured to the skin with a tape butterfly and 3–0 nylon suture. Prior to closing the thorax, an 8 F thoracostomy tube was placed through the twelfth intercostal space and secured to the skin with a 3–0 nylon suture. The thoracotomy incision was closed routinely in four layers. The cannula was attached to the collecting system fashioned from a 10 mL test tube, 20 gauge intravenous catheter, and a 22 gauge needle used to vent the tube (Figure 2). A jacket was placed on each cat to hold and protect the collecting system. The collecting system was emptied as necessary and the volume of fluid recorded.
Photograph demonstrating the placement of the cannula within the thoracic duct (white arrows), and secured with 7-0 silk ligatures. The thoracic duct lies dorsolateral to the thoracic aorta in this image. Note the intercostal vessels (white asterisks) overlying the thoracic duct. Photograph demonstrating the thoracic fluid collection system.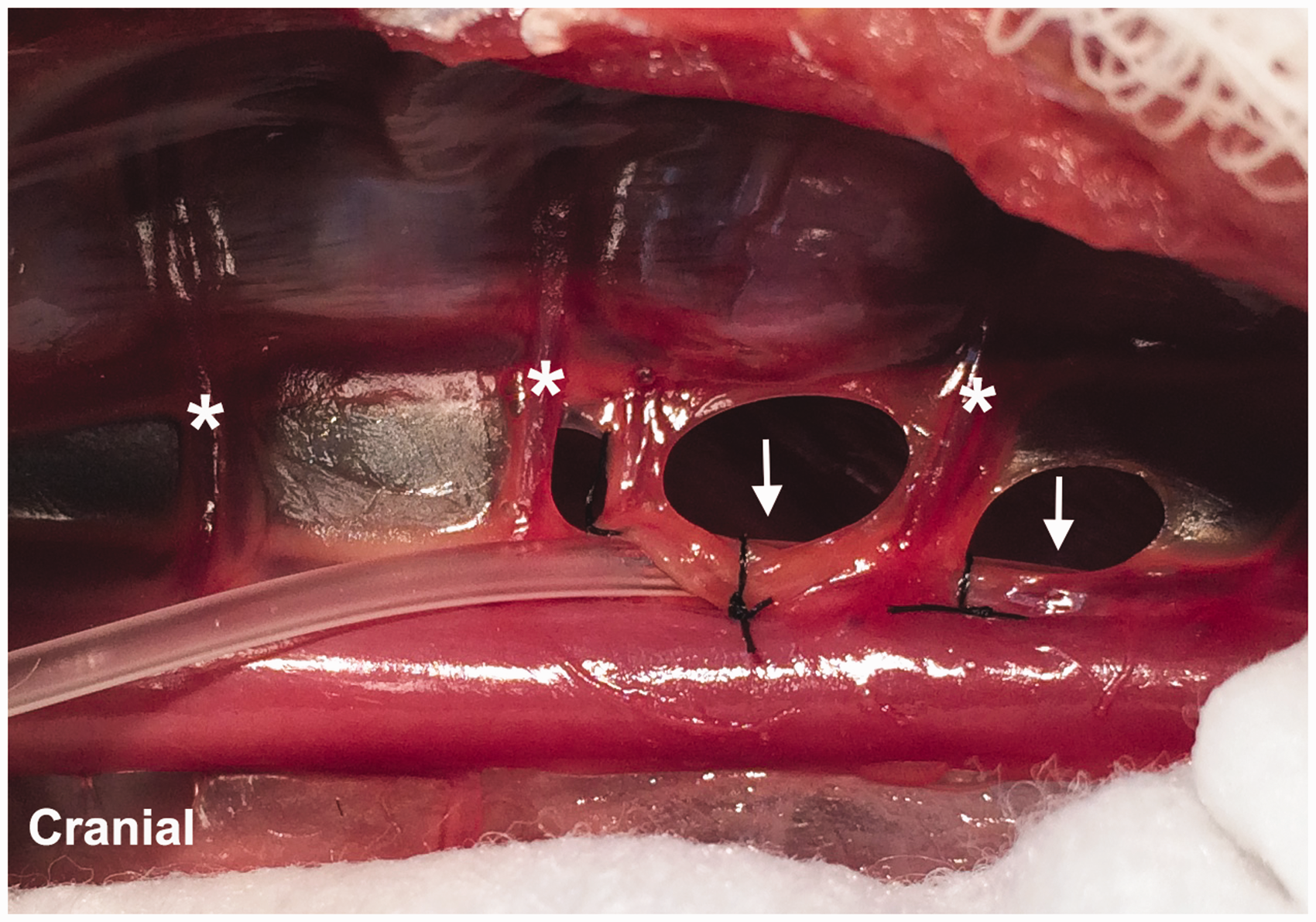
Postoperative care and evaluation
Vital signs, including rectal temperature, pulse rate, respiratory rate, mucous membrane color, and capillary refill time were monitored every hour after surgery until stable and then every 8 h for the first 24 h. The thoracostomy tube was aspirated every 1 to 2 h for the first 8 h and then removed at 24 h if no significant air or fluid was produced. Hydromorphone (0.1 mg/kg IV or SQ every 4–6 h) or buprenorphine (0.01 mg/kg IV or SQ every 8 h) was administered for the first 24 h and then on an as needed basis thereafter. To help prevent lymphatic fluid from clotting within the cannula, heparin (50–100 IU/kg SQ every 8 h) was administered for the duration of the study.
Cats were housed individually in 20 × 28 inch stainless steel hospital kennels. Environmental control included temperature regulation (21.5℃) and light cycle regulation (14–10 h light–dark cycle). Free-choice water was available at all times, and a clean 8 × 11 inch litterbox with clay litter was provided. Each cat was fed one-half cup of dry feline maintenance food (Iams feline maintenance; Iams Co, Dayton, OH, USA) with two teaspoons of canned maintenance food (Iams feline maintenance; Iams Co) twice per day. Enrichment was provided in the form of daily human interaction. Housing of each cat complied with the National Research Council Guide for the Care and Use of Laboratory Animals standard. A physical examination was performed daily and incisions were monitored for evidence of swelling, redness, or drainage.
Blood and lymphatic fluid collection and analysis
Lymphatic fluid was collected for up to 16 days to determine the daily volume produced. Lymphatic fluid analysis (triglycerides, cholesterol levels) and cytological examination were performed on days 3, 9 and 16. Whole blood and serum were analyzed for triglycerides, cholesterol, albumin, total protein, packed cell volume (PCV), white blood cell count, and lymphocyte count on days 3 and 9.
Contrast imaging
Contrast thoracic radiographs were obtained by injecting 0.5 mL of iohexol (Amersham Health, Amersham, UK), diluted 50:50 with sterile saline through the cannula at the end of the study period or if lymphatic fluid ceased abruptly and failed to resume within 24 h.
Withdrawal criteria
Cats were withdrawn from the study if, based on the contrast study, the cannula had become dislodged from the thoracic duct or if the cannula was determined to be occluded based on the absence of the flow of lymphatic fluid and the inability to inject sterile heparinized saline through the cannula.
Cannula removal
Cannulae were removed at the end of the study using gentle traction on the tubing at the level of the skin with the cats manually restrained. Cannulae that could not be removed easily with traction were surgically removed during scheduled ovariohysterectomy. All cats were adopted into private homes after completion of the study.
Data analysis
Data for age, body weight, surgical time, volume of lymphatic fluid production, duration of cannula patency, whole blood, serum and lymphatic fluid analysis and lymphatic fluid cytological examination are reported in descriptive terms including mean, median and range, where appropriate.
Results
Animals
Mean age and body weight were 9.8 months and 3.23 kg, respectively. The thoracic duct was successfully cannulated in all 12 cats. Two cats were withdrawn from the study within the first 24 h due to dislodgement of the cannula, therefore results represent data from 10 cats only. During lymphatic fluid collection, body weights decreased a mean of 0.25 kg (range: gain of 1.01 to loss of 0.45 kg).
Anesthesia
Three cats were administered atracurium to control motion associated with spontaneous respiration during cannulation of the thoracic duct.
Surgical procedure
In four cats, collateral branches running parallel to the thoracic duct were identified and ligated separately from the main thoracic duct. In four cats, the eighth and ninth intercostal vessels had to be cauterized and transected to enable advancement of the cannula into the thoracic duct. Mean duration for the surgical procedure was 4:18 h (range 3:15 to 6:05 h).
Lymphatic fluid production and cannula patency
Mean daily production of lymphatic fluid was 43.7 mL (median 41.0 mL, range 2.3 to 152.4 mL). Cannulae were patent for a mean of 8.2 days (median 6.5, range 3 to 16 days).
Contrast imaging
Contrast imaging was performed in 10 cats. Imaging revealed that two cannulae were completely occluded, five had contrast leakage outside the thoracic duct, and three had normal filling of the thoracic duct and cisterna chyli. In one of these cats, a small collateral branch of the thoracic duct was identified to be bypassing the cannula site and entering the thoracic duct more cranially.
Systemic values
Complete blood count and serum chemistry values were within normal limits for all the cats on initial evaluation.
On day 3, two cats had a mildly elevated PCV (50% and 51%) (reference: 30–47%); four cats had a decreased lymphocyte count (0.806 × 103 cells/uL, 0.445 × 103 cells/uL, 0.449 × 103 cells/uL and 0.277 × 103 cells/uL) (reference: 1.5–7.0 × 103 cells/uL); and two cats had mildly elevated triglyceride levels (203 mg/dL and 263 mg/dL) (reference: 17–133 mg/dL).
On day 9, one cat had a decreased lymphocyte count (0.518 × 103 cells/uL); one cat had a mildly elevated PCV (53%); and two cats had mildly elevated triglyceride levels (176.6 mg/dL and 263 mg/dL).
Lymphatic fluid values and cytological examination
Cytological examination of lymphatic fluid samples on days 3, 9 and 16 were similar for all three time points. Each sample was milky white in color. The mean total nucleated cell count was 20.66 × 103 cells/uL (median 13.1 × 103, range 1.15 to 97.6 × 103 cells/uL). Cytology revealed predominantly (97%) small lymphocytes with normal morphology and varying proportions (3%) of non-degenerate neutrophils, macrophages, plasma cells, eosinophils and erythrocytes. Occasionally bacteria were noted in samples, but these were ruled to be contaminants due to the lack of evidence of neutrophilic or cytological response to the bacteria.
Mean fluid cholesterol levels were 88 mg/dL, 67.6 mg/dL and 100.5 mg/dL on days 3, 9 and 16, respectively. Mean fluid triglyceride levels were 2664 mg/dL, 3287.6 mg/dL and 2099.5 mg/dL on days 3, 9 and 16, respectively. Fluid cholesterol to triglyceride ratios were less than 1 on days 3, 9 and 16, consistent with normal lymphatic fluid.
Cannula removal
In 10 cats (including the two cats that dislodged the cannula immediately after surgery), the cannula was easily removed with gentle traction on the tubing at the level of the skin. In two cats, the cannulae were removed through the previous thoracotomy incision at the time of ovariohysterectomy.
Discussion
Thoracic duct cannulation via a left intercostal thoracotomy approach is a feasible option for lymphatic fluid collection in a feline model. The approach provided good access to the thoracic duct; however, the very delicate nature of the thoracic duct and variation in the number and precise anatomical location of the branches makes this a relatively challenging procedure even with the magnification of an operating microscope.
In most cats, the thoracic duct was readily identified lying just deep to the pleura on the surface of the mediastinal fat dorsolateral to the aorta. In others, the thoracic duct was located deeper within the fat and more dorsal to the aorta making positive identification more difficult. Four cats had multiple branches to the thoracic duct that had to be meticulously dissected in order to identify the largest branch for cannulation. In all the cats, the aorta had to be retracted ventrally to completely expose the thoracic duct and any collateral branches.
Other specific challenges with cannulation of the thoracic duct included compression of the duct by the overlying intercostal vessels, preventing caudal advancement of the cannula into the cisterna chyli. In those cats, the intercostal arteries had to be transected and the thoracic duct freed from the overlying adventitia to allow the cannula to pass the obstruction. Also, valves present within the thoracic duct hindered the caudal advancement of the cannulae in some cats and despite repeated attempts to manipulate the cannula past the valves, it was not possible, ultimately reducing the length of cannula that was secured within the thoracic duct.
Difficulties related to maintaining the cannulae included obstruction from clot formation, dislodgement, and leakage of chyle from the collecting system. The clot responsible for obstructing the cannula in two cats was not histologically examined, but appeared to be fibrin or congealed fluid that formed within the tubing. Although heparin was administered postoperatively to potentially reduce the incidence of clot formation within the thoracic duct and cannula system, its utility specifically within the lymphatic system of cats is not known. Two cats accidentally dislodged the cannula from the thoracic duct with their back legs during recovery from surgery, despite having the jacket in place to protect the tubing and collection system. Although relatively form fitting, the jacket could not completely protect the cannula from being scratched or pulled by the back legs if the cats were persistent or forceful. Leakage of fluid from the vent of the collecting system occurred occasionally in some cats if the tubes were inverted when the cats were lying down or rolling over. The 22 gauge needle used to vent the collecting tube, although small in diameter, did allow a small amount of fluid to leak as the level within the tube reached capacity.
The surgical times reported in this study included the entire procedure from initial skin incision to securing the collection system to the skin. There was a significant learning curve with this procedure and although the surgical times varied, they tended to decrease with experience. The dissection and cannulation of the thoracic duct were obviously the most challenging and unpredictable part of the procedure due to the very delicate nature of the thoracic duct and variation in the number of branches identified. The use of atracurium to paralyze respiratory muscles was useful in minimizing movement while attempting to cannulate the thoracic duct. Although there was still movement of the thorax due to the action of the ventilator, the consistency of the motion (10 breaths/min) was less disruptive than normal spontaneous respiratory muscle activity.
In this study, there was no change in the cytological characteristics of the lymphatic fluid collected over several time points, suggesting that this model does not result in significant inflammation or artifact to the samples. Additionally, the thoracic fluid collected in each cat at each time point had a fluid cholesterol to triglyceride ratio of less than 1, further supporting the classification of the fluid collected as normal lymphatic fluid.25,26
There was little deviation outside the normal reference range for the systemic values evaluated in this study (cholesterol, triglycerides, PCV, total protein and lymphocyte count) with the exception of relatively mild changes in PCV, triglycerides, and lymphocyte count. It is likely that the mild elevation in PCV seen in two cats on day 3 and one cat on day 9 (50%, 51% and 53%) was due to mild dehydration as those cats also had total protein values in the high end of the normal range. The lymphopenia identified in five cats was most likely due to lymphocyte loss from continuous thoracic duct fluid sampling.
To our knowledge there is only one other report describing a technique for cannulation of the thoracic duct in cats. 19 That report describes a ventral midline approach over the caudal cervical and cranial thoracic regions for access to the thoracic duct. Cannulae were reportedly maintained for up to six months; however, the total number of cats utilized in this study, as well as the mean and median duration of patency were not indicated, making direct comparisons between these techniques difficult.
A potential improvement to the technique described in this study may be to place the cannula directly within the cisterna chyli to reduce the challenge of having to cannulate the very small and fragile thoracic duct. The surgical approach to the cisterna chyli would be similar but positioned more caudally in the thorax, or alternatively, a paracostal abdominal approach could be used access the cisterna chyli and caudal portion of the thoracic duct. Placement of the cannula directly into the cisterna chyli would avoid problems associated with the valves of the thoracic duct and potentially decrease the concern for missing collateral branches and allowing more accurate determination of lymphatic flow.
Conclusion
The lateral thoracotomy approach for cannulation of the thoracic duct in cats is a challenging but feasible technique for evaluating lymphatic fluid in a non-terminal research model. Use of this model may have application for evaluating novel drugs designed to reduce lymphatic fluid production for treatment of chylothorax in cats; however, differentiating the effect of the treatment from complications with the cannula system remains a challenge.
Footnotes
Acknowledgement
We acknowledge Dr Marie-Claude Theoret for her involvement with this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt the following financial support for the research, authorship, and/or publication of this article: This research was funded by the American College of Veterinary Surgeons and the University of Wisconsin, School of Veterinary Medicine, Companion Animal Fund.
