Abstract
Case summary
A cat aged 12 years and 7 months was referred to a multidisciplinary hospital for investigation of feline injection site sarcoma (FISS) on the left thoracolumbar region. A CT examination of the mass revealed a multi-lobulated mass affecting the body wall, extending from the level of lumbar vertebrae L2 to L4. The mass was excised with 5 cm lateral margins, including resection of the 13th left rib, the caudal edge of the latissimus dorsi (LD) muscle, full-thickness abdominal wall and sections of the lumbar epaxial muscles. To reconstruct the defect, a combination of muscle flaps was used. This included diaphragmatic advancement and lateralisation, rotation of the LD, and creation of transposition flaps from the internal abdominal oblique and external abdominal oblique muscles, ensuring closure without tension. Skin closure required mobilising an inguinal flank fold flap. The cat was discharged from hospital 3 days postoperatively. Histopathology confirmed a diagnosis of FISS with clean wide margins. A gradual return to normal activity and complete healing of the surgical site was reported on follow-up, with one minor complication related to the skin flap (bruising at the base of the inguinal flank fold flap).
Relevance and novel information
This report describes the use of the aforementioned combination of muscle flaps to close a major abdominal wall defect in a cat with an excellent outcome. Practitioners can consider this technique when planning tissue reconstruction after FISS resection.
Introduction
Feline injection site sarcoma (FISS) is a neoplasia of mesenchymal origin that manifests as a subcutaneous mass at a site of injection1 –3 that is usually a vaccination but chemotherapy administration and microchip application have also been described.4,5 These tumours typically exhibit locally aggressive behaviour but have a relatively low metastatic rate. 1 They occur most commonly at frequent injection sites (interscapular, lateral thoracic wall or abdominal wall). The current literature shows that FISS can occur between 4 months and 2–3 years after injection. 2
The literature reports recurrence rates in the range of 26–59% after surgical excision, attributed to infiltration of local tissues.6 –9 The current recommended treatment comprises radical or wide surgical excision with 5 cm lateral margins and two fascial planes deep.6,10 Local recurrence is a negative prognostic indicator and is 10 times more likely if the neoplasm was incompletely excised. 6 In one study, 3% of FISSs were removed with incomplete margins, all of which were grade III fibrosarcomas. Of the cats, 14% had local recurrence after excision with 5 cm lateral margins and no adjunctive treatment, including 1/3 patients with incomplete margins. 10
Various muscle flaps have been described for the closure of large abdominal defects in dogs, but there is a lack of literature in cats. Large abdominal defects are common after the excision of infiltrative masses. Internal (IAO) and external abdominal oblique (EAO) muscle flaps, diaphragm advancement and latissimus dorsi (LD) muscle flaps have been described in dogs, resulting in good to excellent outcomes.11 –14 Complications typically reported in muscle flap reconstruction techniques include wound dehiscence and necrosis. 15 The use of polypropylene mesh has been described with a caudal superficial epigastric axial pattern flap in six cats with radical excision of fibrosarcoma, with good to excellent outcomes in all cats and no evidence of local recurrence. 16 Griffin et al 17 described closure after hemipelvectomy without the use of native muscular tissue and reported good outcomes in these cases. In one case, polypropylene mesh was used. 17
The objective of this case report was to describe the use and outcome of a combination of IAO and EAO muscle flaps, LD muscle flap, and diaphragmatic advancement and lateralisation to close a large body wall defect in a cat.
Case description
A 12-year-old domestic shorthair cat was referred for management of a soft tissue mass on the left lateral thoracolumbar region. The cat was previously diagnosed with diabetes mellitus, and at the time of writing, was currently in remission with no treatment for 2 years. The mass was first noticed 2 months before referral, and was described as a large, multi-lobulated, firm, indiscrete, ulcerated soft tissue mass. Fine-needle aspiration (FNA) cytology was suggestive of a sarcoma, with areas of necrosis. Two simultaneous incisional biopsies were performed by the referring veterinary surgeon 1 month after the initial presentation, revealing a fibrosarcoma compatible with a FISS. After diagnosis, the patient was referred for wide surgical excision.
On presentation, the cat was bright, alert and responsive, weighing 6.97 kg with a body condition score of 6/9. Thoracic auscultation and abdominal palpation were unremarkable. There was a firm, non-mobile mass on the left lateral thoracolumbar region measuring approximately 39 × 45 × 32 mm. Mild serous discharge from the previous biopsy sites was present. A CT analysis of the thorax and abdomen was performed, using a 64-slice helical scanner (SOMATOM go.All; Siemens), which revealed a mixed soft tissue and soft tissue-fluid attenuating, well-defined, multi-lobulated mass in the subcutaneous tissues and muscle layers of the left dorsolateral abdominal wall extending from vertebra L2 to L4 measuring 48 L × 31 W × 42 H mm (Figure 1). The mass was in contact with the deepest aspect of the left paraspinal muscles (longissimus lumborum and iliocostalis lumborum), causing deviation of these muscles and the adjacent abdominal wall but no obvious infiltration. There was a thick linear extension arising from the periphery of the deepest portion of the mass on the lateral side. The superficial cervical, deep cervical, axillary and accessory axillary lymph nodes were enlarged. No overt metastases were noted. Other incidental findings included a thymic remnant, subcutaneous nodules in the left lateral thoracic wall and lateral right thigh, a mild diffuse bronchial lung pattern, non-specific splenopathy, nephrolith/dystrophic mineralisation of the left kidney and diffuse dermal changes. FNA cytology of the enlarged lymph nodes evidenced reactive changes and no metastatic characteristics. Cytology of the subcutaneous nodules diagnosed benign keratinising lesions, and the owners declined concurrent excision of these nodules.

Multi-planar reconstruction CT images of the abdomen and thorax, showing the mass in the left dorsolateral abdominal wall, compatible with a feline injection site sarcoma (red arrow). Images are post-contrast and displayed in a soft tissue window: (a) sagittal, (b) transverse and (c) frontal
Wide surgical excision of the mass was recommended, which was performed 21 days later for client-related reasons. The patient was pre-medicated with medetomidine 0.035 mg IV and methadone 1.40 mg IV. Anaesthesia was induced with propofol to effect (30 mg IV) and maintained with isoflurane inhalant and oxygen. The patient was positioned in right lateral recumbency. The skin was incised around the mass with 5 cm lateral margins (Figure 2).

(a) Feline injection site sarcoma (FISS) present on left flank with sterile surgical pen markings around palpable edges of mass (inner continuous line) and 5 cm lateral margins (outer dotted line). (b) Body wall defect after excision of the FISS with 5 cm lateral margins
The underlying tissues were resected with 5 cm lateral margins, including resection of the 13th left rib, the caudal region of the LD muscle, full-thickness abdominal wall and part of the epaxial muscles of L2 and L3 (Figure 3a). A thoracostomy drain was placed under direct visualisation (MILA Guidewire Chest Tube 12 G × 20 cm; DMS Veterinary) and secured to the skin using a non-absorbable monofilament suture (size 2-0 nylon) in a Roman sandal friction pattern. Reconstruction of the thoracic and abdominal wall involved diaphragmatic advancement and lateralisation, by incising the serosa of the concave surface of the diaphragm and suturing this to the 12th rib using a combination of simple interrupted and simple continuous circumcostal sutures (absorbable monofilament polydioxanone, size 2-0). The dorsal fascia of the LD was incised, and the flap was rotated ventrally. A flap was also created caudally by incising the ventral fascia of the IAO muscle and rotating the flap dorsally and cranially. The IAO muscle flap was sutured to the free edge of the diaphragm and to the LD cranially (Figure 3b), to the remaining fascia of the transversus abdominis ventral to the epaxial muscles dorsally and to the remaining IAO fascia ventrally. The remaining EAO muscle was then brought dorsally and sutured to the ventral edge of the LD flap cranially, and to the IAO flap dorsally (completely covering the underlying IAO flap sutures). This combination of muscle flaps resulted in complete closure without tension (Figure 3c).

(a) Abdominal wall defect after wide excision of the mass. The lumbar epaxial muscles (1) and remnants of the transversus abdominis (5) are dorsal to the defect. Stay sutures are placed on the latissimus dorsi (LD) (2) caudal edge of the diaphragm (4) and internal abdominal oblique (IAO) (3). (b) The IAO muscle flap (3) was sutured cranially to the free edge of the LD (2) and diaphragm, and dorsally to the transversus abdominis (5). Stay sutures are placed on the external abdominal oblique (EAO) (6) and ventral remnants of the IAO muscle. (c) The EAO muscle (6) was sutured dorsally to the IAO muscle (3) and cranially to the LD (2) and to the remnants of the IAO muscle. This allowed complete closure of the defect
The iatrogenic pneumothorax was drained via thoracostomy drain. Closure of the skin without tension required transposition of an inguinal flank fold flap. Subcutaneous tissues and skin were tacked with multiple simple interrupted sutures of absorbable monofilament material (absorbable monofilament polydioxanone, size 2-0). The subcutaneous layer was closed with simple continuous absorbable monofilament suture (poliglecaprone 25, size 3-0) and the skin was closed with skin staples (Figure 4). The resected tissue was inked and submitted for histopathology.

Skin closure with transposition of an inguinal flank fold flap
The cat recovered uneventfully from anaesthesia and was hospitalised receiving intravenous fluid therapy and a constant rate infusion of ketamine (range 14–35 µg/min). Medications consisted of meloxicam oral suspension (0.69 mg PO q24h for 14 days), gabapentin tablets (50 mg PO q8h for 14 days) and cefalexin (150 mg PO q12h for 10 days). The chest drain was removed 1 day postoperatively. The cat was discharged 3 days postoperatively.
Histopathology confirmed the diagnosis of a fully excised sarcoma compatible with FISS. The narrowest histological tumour-free distances from the surgeon-cut margins (in relation to the anatomical location of the neoplasm) were 6 mm deep, 32 mm cranial, 40 mm dorsal, 13 mm caudal and 48 mm ventral. The plane forming the deep margin was the second deeper fascial plane. The sections of bone (rib) included in the sample did not show pathological changes.
Good progress was reported on telephone follow-up 9 days postoperatively. The cat presented to the referring practice for staple removal 14 days postoperatively. Good wound healing was reported, with a patch of dry dermatitis over the left stifle (suspected clipper rash), with no signs of dehiscence or necrosis. At 16 days postoperatively, photographs were sent by the owner (Figure 5), and it was reported that the cat was very active and back to its normal self. A small area of superficial skin bruising next to the inguinal skin flap donor site (left stifle) was apparent, which was compatible with the area of skin rash reported by the referring veterinarian and was managed conservatively. The owner was pleased with the outcome of the surgery. A follow-up physical examination at the referring practice 36 weeks postoperatively reported the surgical site had completely healed, with no palpable body wall defects or signs of local recurrence. Verbal and photographic follow-up 47 weeks postoperatively (Figure 6) confirmed the cat remained its normal self with no loss of function and no obvious cosmetic changes as a result of the procedure.
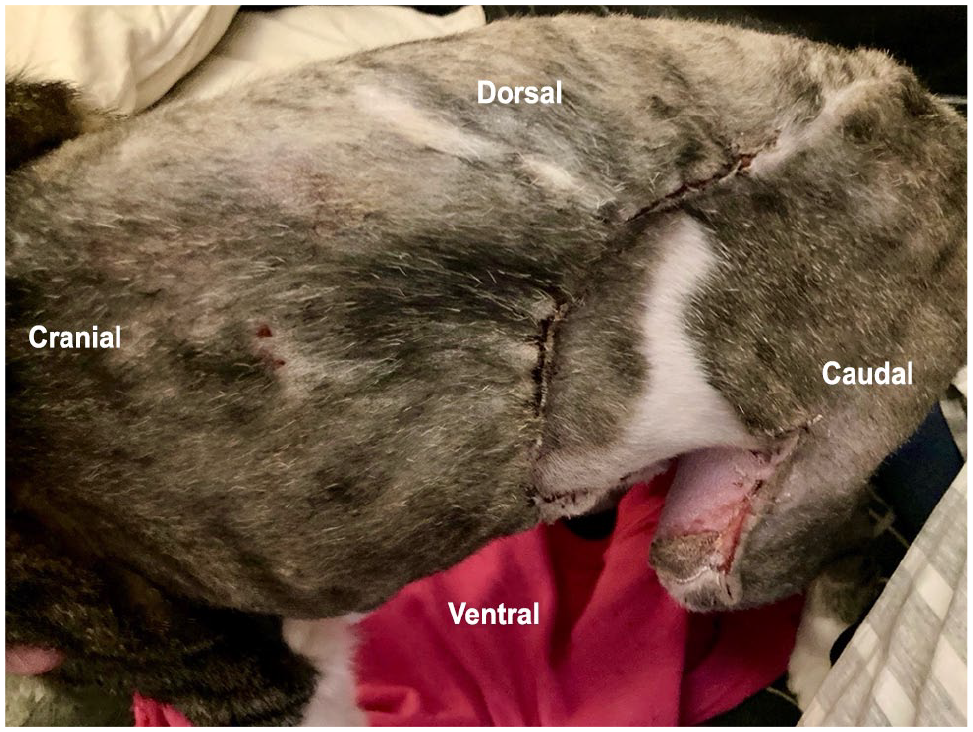
Photographs submitted by the owner 16 days postoperatively

Photographs submitted by the owner 47 weeks postoperatively
Discussion
A combination of diaphragmatic lateralisation and advancement, and LD, IAO and EAO muscle flaps were used for body wall reconstruction after wide resection of a FISS in the case described. Complete healing was achieved with no major complications. A minor local skin complication occurred at the inguinal skin flap donor site, which resolved without intervention.
Abdominal wall resection and reconstruction of large defects have been previously reported in the veterinary literature, including a variety of muscle flap techniques. Other previously reported muscle flaps were also considered in this case, such as a cranial sartorius or rectus abdominis muscle flaps, but these were deemed less appropriate owing to the location of the defect.15,16,18,19 The specific combination of reconstructive techniques used in this patient employed the most accessible options given the location and size of the defect without need for further separate incisions or patient repositioning. In this case, the use of a synthetic mesh was considered as a backup option, should primary reconstruction prove unfeasible. The use of mesh has been reported in human and veterinary literature for closure of large defects, with a wide range of materials available, most commonly polypropylene.12,19-22 Complications include repair failure and infection, and autologous tissue is preferred when possible. 23
In the current case, wide excision resulted in significant loss of abdominal and thoracic wall tissue. Closure exclusively with subcutaneous tissues and skin has been previously reported to provide successful closure of large defects after a hemipelvectomy procedure. 17 That option was ruled out in this case as a result of the size of the defect and the need to achieve a reliable hermetic closure of the thoracic cavity.
The recommended treatment for FISS involves radical or wide surgical excision including 5 cm from the palpable lateral edges of the mass and two fascial planes deep. 24 Marginal resection and incomplete margins carry a higher risk of recurrence. 10 Adjuvant or neoadjuvant radiation therapy has also been described for local management, particularly in cases where wide clean surgical margins were not achieved or when tumour size reduction was preferable before surgical excision.9,24,25 In the current case, radiation therapy was not administered as the mass was completely excised with wide clean histological margins. No significant difference in disease-free intervals has been found with adjuvant multimodal chemotherapy treatment vs surgical treatment alone.8,25 –29 However, the majority of these studies are underpowered; therefore, caution is advised when drawing conclusions. Wide or radical surgical excision is still considered the first treatment of choice in the current veterinary literature.
The use of the terms ‘wide’ and ‘radical’ excision has been inconsistent in the veterinary literature. In the present case, the term wide excision has been used, based on the Enneking surgical dose descriptions by the Musculoskeletal Tumor Society (MSTS). The MSTS defines radical margins as the removal of all normal soft tissue included in the anatomical compartment(s) involved with the mass, and wide margins as intracompartmental en-bloc excision of the mass with a cuff of normal soft tissue (irrespective of margin length or depth). Considering that in this case, strictly speaking, radical excision would imply resection of the entire extent of all body wall muscles infiltrated by the mass, from origin to insertion, we think ‘wide excision’ more accurately represents this procedure, since any remnants of muscle beyond 5 cm from the mass were preserved.
Complications of muscle flaps include surgical site infection, dehiscence and seroma formation.14,18,30 Closure of such large defects can lead to tension and increase morbidity. 12 In this case, multiple tissues were incorporated in the reconstruction, with the aim to achieve tension-free closure. However, if incomplete margins were reported or local recurrence occurred, all tissues involved would potentially be seeded with neoplastic cells. In such cases, revision surgery would lead to increasingly aggressive resections with minimal autologous tissue remaining for closure. This limitation should be considered when planning primary autologous reconstruction.
Another limitation of this report is that it describes a single case. Despite the outcome being considered excellent in this patient, larger studies would be needed before drawing any conclusions about the techniques used. Lastly, most of the follow-up available for this case relied on the physical examinations performed at the referring veterinary practice and on verbal and photographic updates provided by the owners via telephone and via email. Re-examination at the referral hospital would have been desirable for the purpose of this case report; however, this was not possible owing to the owners having relocated and declining travel.
Conclusions
This case report describes the use of a combination of IAO and EAO muscle flaps, LD muscle flap, and diaphragmatic advancement and lateralisation after major body wall resection in a cat, with an excellent outcome. This combination could be considered a viable option in the reconstruction of major body wall defects in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Linnaeus Veterinary Limited supported the costs of the open access publication charges.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
