Abstract
A 14-month-old male Armenian hamster (Cricetulus migratorius) presented with a spontaneous, subcutaneous, firm mass (4.0 × 2.0 × 1.5 cm) on the ventral neck extending towards the cheek pouch causing multifocal small oral ulcerations. This animal was immunized subcutaneously on the dorsal neck for the development of monoclonal antibodies seven months before presentation. The animal was euthanized and necropsy was performed. Histopathology of the mass showed a well demarcated, multilobulated, unencapsulated, highly cellular, neoplastic mass composed of spindle cells arranged in interlacing streams and bundles, with a moderate amount of fibrovascular stroma. The neoplastic cells exhibited indistinct cell borders and a moderate to large amount of eosinophilic, fibrillar cytoplasm, marked anisocytosis and anisokaryosis, binucleated and multinucleated cells, and high mitotic rate. Based on the histomorphologic features of the mass, and the presence of renal tubular hyaline globules and myeloid hyperplasia in the bone marrow, a diagnosis of histiocytic sarcoma was made. The presumptive diagnosis was confirmed by immunohistochemistry, upon which the neoplastic cells showed strong immunoreactivity for the histiocytic cell markers Iba1 and CD11b. Histiocytic sarcomas have been reported in Syrian (Mesocricetus auratus) and Siberian dwarf (Phodopus sungorus) hamsters but, to our knowledge, the current report represents the first case of histiocytic sarcoma described in an Armenian hamster. It is plausible to consider the animal’s experimental immunization history and the development of the histiocytic sarcoma to be related. An association between adjuvanted vaccines and soft-tissue sarcomas has been described in cats and referred to as feline injection-site sarcomas.
A 14-month-old male Armenian hamster (Cricetulus migratorius) presented with a firm, multilobulated subcutaneous mass (4.0 × 2.0 × 1.5 cm) on the ventral neck extending cranially towards the cheek pouch causing multifocal small oral ulcerations. This animal was part of an Institutional Animal Care and Use Committee (IACUC)-approved animal use protocol for the development of monoclonal antibodies. This previously experimentally naïve animal was immunized subcutaneously on the dorsal neck with the target peptide antigens (50 µg) nine months before presentation, followed by three additional immunization boosts three to four weeks apart (last boost seven months before presentation). The immunogen, keyhole limpet hemocyanin (KLH) conjugated to a peptide, was administered with an adjuvant (TiterMax®, Sigma-Aldrich, St Louis, MO, USA) as an emulsion in a total volume of 100 µL. While small subcutaneous granulomas may develop at the immunization site, lesions were not observed in this animal on macroscopic evaluation. None of the other eight animals in this cohort who were injected with the same immunogen developed masses. Prior to clinical presentation of the subcutaneous mass this animal was apparently healthy with no previous history of health problems.
The hamster was single-housed in a solid-bottom, polysulfone, individually ventilated cage (IVC) (Thoren Caging Systems, Hazelton, PA, USA) with autoclaved aspen-chip bedding (PWI Industries Canada, Quebec, Canada) and provided γ-irradiated feed (LabDiet 5053, PMI, St Louis, MO, USA) and acidified reverse osmosis water (pH, 2.5 to 2.8) ad libitum. The cage also contained bagged crinkle-cut paper strips (EnviroPaks®, W.F. Fisher and Son Inc., Branchburg, NJ, USA) and a running wheel as environmental enrichment. The IVC system is ventilated at 30 air changes hourly. HEPA-filtered room air is supplied to each cage and the rack effluent is exhausted directly into the building’s exhaust system. Cages are changed weekly in a Class 2 Type A biological safety cabinet. The animal holding room is maintained at 72 ± 2°F (21.5 ± 1°C), relative humidity between 30% and 70%, and a 12:12 hour light:dark photoperiod. The animal was maintained in accordance with the US Department of Agriculture Animal Welfare Act and Regulations and the Guide for the Care and Use of Laboratory Animals.1,2 The animal care and use program is fully accredited by AAALAC International.
Given the size, location, and unexpected nature of the subcutaneous mass, the animal was euthanized via carbon dioxide overdose and submitted for a complete necropsy the day following lesion detection. The animal underwent a complete post-mortem examination, including macroscopic organ inspection and dissection. Histologic examination was performed on an extensive panel of tissues including all major organ systems and draining lymph nodes associated to the mass (submandibular). Additionally, multiple cross sections of the subcutaneous mass were collected and processed. All tissue samples were fixed in 10% neutral buffered formalin, routinely processed in alcohol and xylene, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin (H&E). Immunohistochemistry (IHC) for Iba1 (ab5076, Abcam, Cambridge MA, USA; applied at concentration of 1:500 following heat-induced epitope retrieval (HIER) in a pH 9.0 EDTA buffer), CD11b (ab133357, Abcam; HIER, 1:20000, pH 6.0 citrate buffer), Mac-2 (CL8942B, Cedarlane, Burlington, ON, Canada; HIER, 1:100, pH 6.0 citrate buffer), CD3 (ab135372, Abcam; HIER, 1:250, pH 6.0 citrate buffer), B220 (550286, BD Biosciences, San Jose, CA, USA; HIER, 1:200, pH 6.0 citrate buffer), and CD19 (90176, Cell Signaling, Danvers, MA, USA; HIER, 1:400, pH 9.0 EDTA buffer) was performed on the mass. Mac-2 staining was performed manually, using an avidin-biotin detection system (Vectastain ABC Elite Kit, PK-6100, Vector Laboratories, Burlingame, CA, USA). All other stains were performed on a Leica Bond RX automated stainer (Leica Biosystems, Buffalo Grove, IL, USA) using the Bond Polymer Refine detection system (DS9800, Leica Biosystems). The chromogen used was 3,3 diaminobenzidine tetrachloride and sections were counterstained with hematoxylin. Normal Armenian hamster tissues were stained and examined as controls, and confirmed the expected pattern of immunoreactivity for all antibodies. Slides were evaluated and lesions recorded by a board-certified veterinary pathologist (SM).
Macroscopically, the animal was over conditioned (body condition score – 4 out of 5) and a firm, multilobulated subcutaneous mass, measuring 4.0 x 2.0 x 1.5 cm (Figure 1(a)) was present on the right, ventrolateral neck. The mass extended towards the ipsilateral cheek pouch and there were multifocal, small-sized ulcerations (Figure 1(b) and (c)) on the adjacent oral mucosa. Additional findings included hepatomegaly with mild, diffuse parenchymal pallor and diffuse splenomegaly.
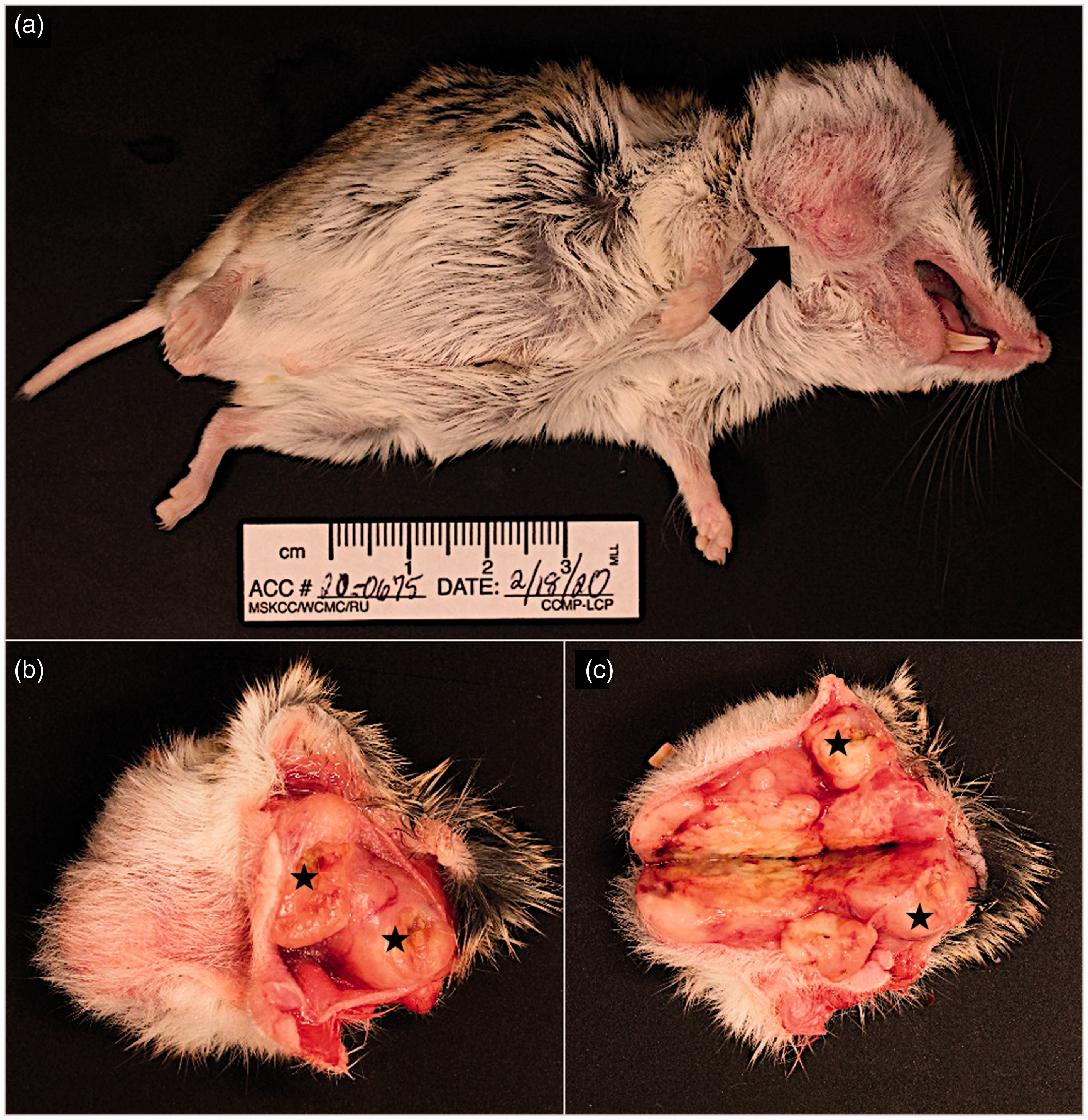
Armenian hamster, male, post-mortem macroscopic photographs. (a) Animal in dorsolateral recumbency, showing a subcutaneous mass on the right ventrolateral neck (black arrow). (b) Intact subcutaneous mass infiltrating the right cheek pouch with multifocal oral ulcerations (black asterisk). (c) Cut surface of multilobulated, tan-yellow, firm subcutaneous mass; multifocal oral ulcerations are also present (black asterisk).
Histopathological examination of the mass showed an infiltrative, multilobulated, unencapsulated, highly cellular mass composed of spindle to polygonal neoplastic cells arranged in interlacing streams and bundles, supported by a moderate amount of fibrovascular stroma, with indistinct cell borders and a moderate to large amount of eosinophilic, fibrillar cytoplasm. This morphology of the neoplastic cells was interpreted as histiocytic and no other neoplastic cell morphology was recognized. These cells had round to oval nuclei with finely stippled chromatin and one or two variably distinct nucleoli, and displayed marked anisocytosis and anisokaryosis, binucleation and multinucleation, and a mitotic rate of 27 per 10 400x fields (Figures 2(a) and 3(a), (b)). There were multiple, variably sized areas of hemorrhage, necrosis and mineralization, multifocal angioinvasion of neoplastic cells with thrombosis (Figure 3(f)), as well as local invasion of the oral squamous epithelium, causing focal oral epithelium ulceration with suppurative inflammation and intralesional coccoid bacterial colonies within the neoplastic mass.
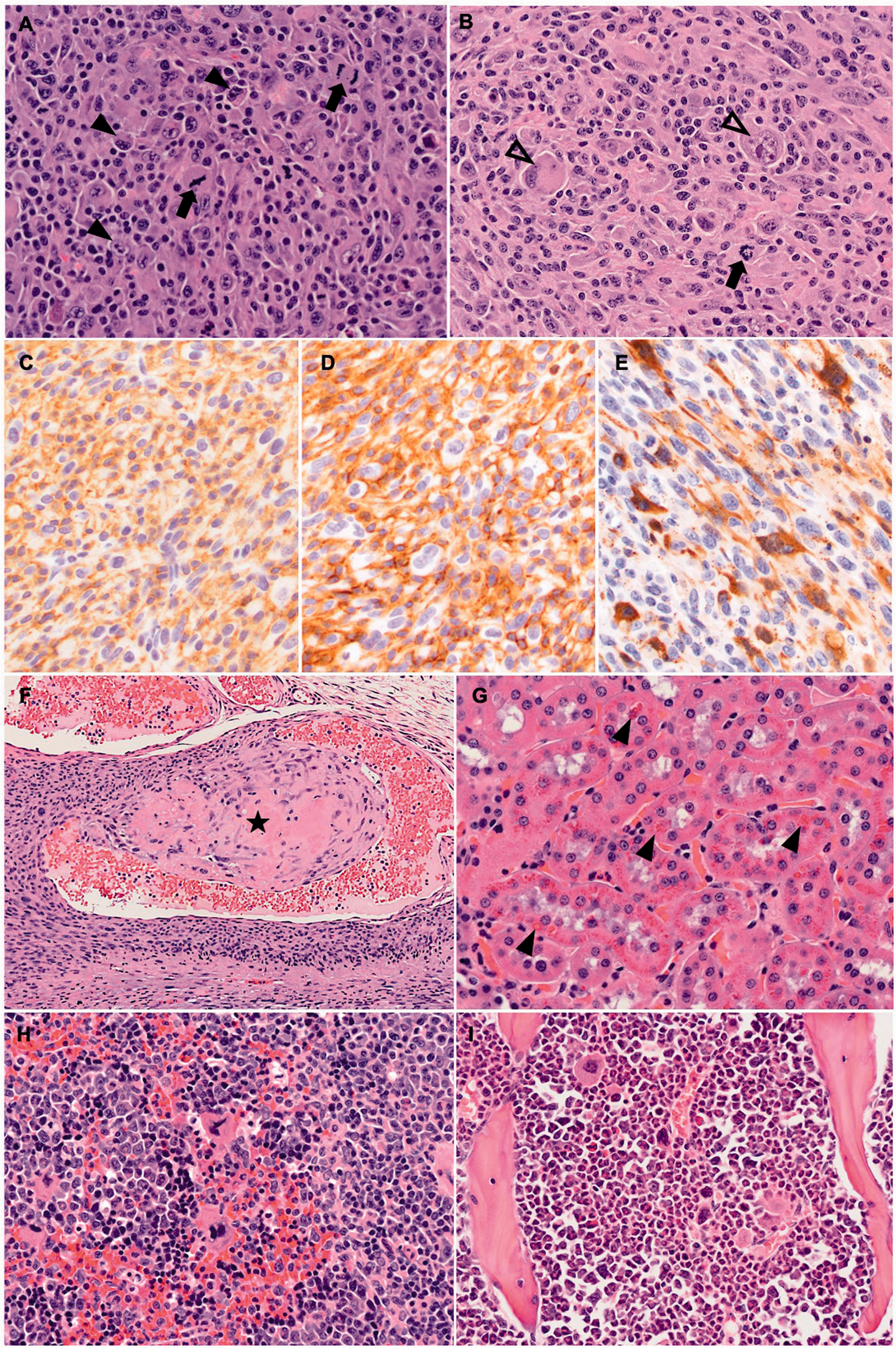
(a)–(f) Histiocytic sarcoma, right ventrolateral neck, Armenian hamster, male, photomicrographs. (a), (b) Mesenchymal neoplastic cells showing marked anisocytosis and anisokaryosis, mitotic figures (black arrows); binucleated (black arrowheads) and multinucleated giant cells (empty arrowheads) are also present. H&E; x400. (c) Most neoplastic cells show strong membranous immunopositivity for Iba1. IHC; x400. (d) Most neoplastic cells showing strong membranous immunopositivity for CD11b. IHC; x400. (e) A smaller proportion of neoplastic cells showing moderate to strong cytoplasmic immunopositivity for Mac-2. IHC; x400. (f) Angioinvasion and thrombus formation (black asterisk) of the neoplastic cells. H&E; x100. (g) Renal proximal tubules with intracytoplasmic, eosinophilic hyaline droplets (black arrows). H&E; x400. (h) Expansion of the splenic red pulp by marked extramedullary hematopoiesis characterized by the presence of erythroid and myeloid precursor cells, and megakaryocytes. H&E, x400. (i) Myeloid hyperplasia of the femoral bone marrow characterized by an erythroid to myeloid precursor cell ratio of 1:4. H&E; x400.
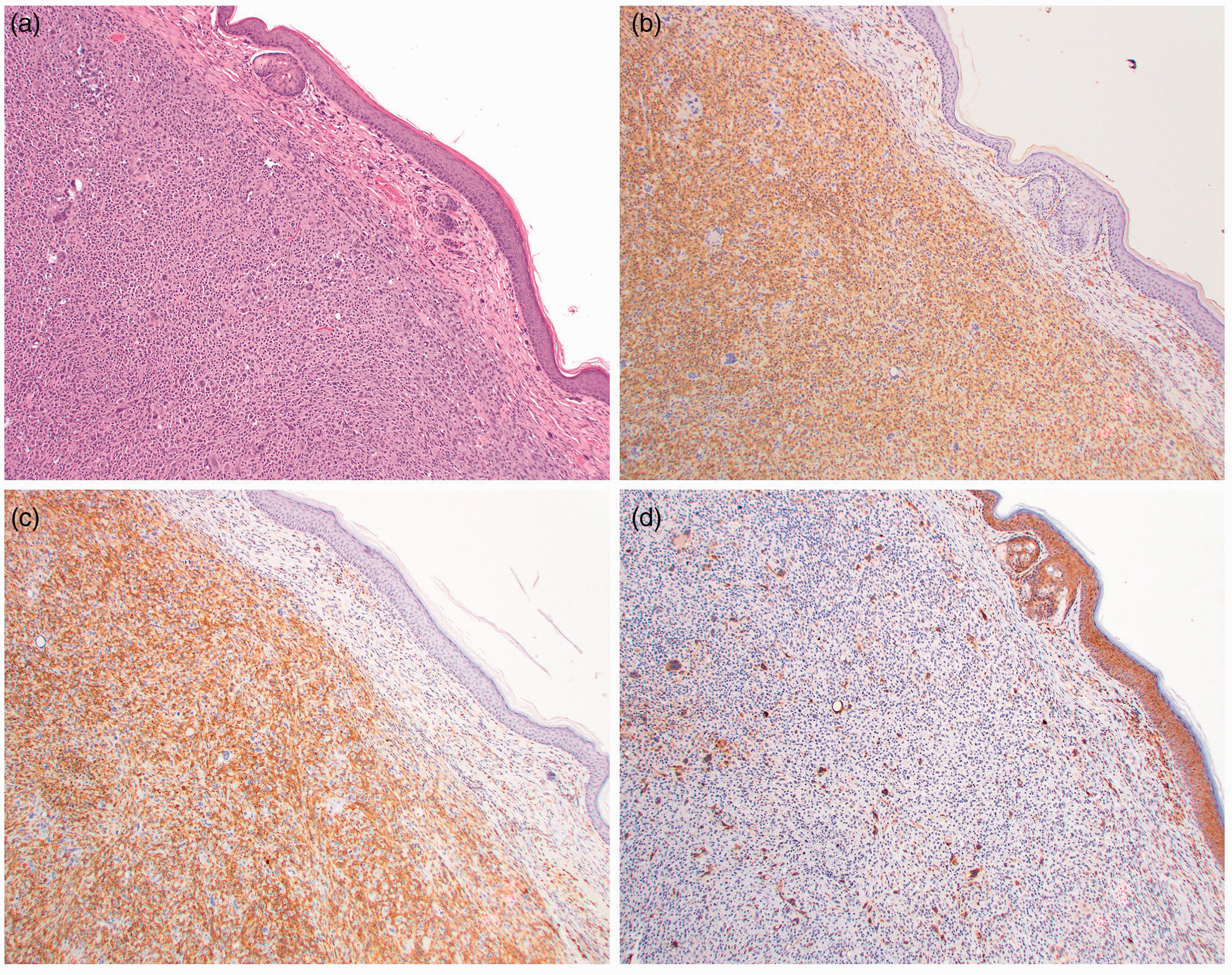
(a)–(d) Histiocytic sarcoma, right ventrolateral neck, Armenian hamster, male, low magnification photomicrographs. (a) Mesenchymal neoplastic cells showing marked anisocytosis and anisokaryosis, mitotic figures, and binucleated and multinucleated giant cells. H&E; x10. (b) Most neoplastic cells show strong membranous immunopositivity for Iba1. IHC; x10. (c) Most neoplastic cells showing strong membranous immunopositivity for CD11b. IHC; x10. (d) A smaller proportion of neoplastic cells showing moderate to strong cytoplasmic immunopositivity for Mac-2. IHC; x10. Epidermis is positive on Mac2 because, in addition to macrophages, Mac2 is expressed by some epithelial cells.
Additional findings included vascular pulmonary and cerebral thrombosis, presence of renal tubular intracytoplasmic eosinophilic hyaline droplets (Figure 3(g)), splenic extramedullary hematopoiesis (EMH; Figure 3(h)), and myeloid hyperplasia of the bone marrow (Figure 3(i)). Dissemination of the histiocytic neoplastic process was recognized multifocally within the liver sinusoids and perivascular areas with small aggregates of neoplastic cells with a similar histiocytic phenotype as observed in the mass. No metastatic spread was recognized in the draining (submandibular) lymph nodes. Multifocal perivascular and interstitial, neutrophilic and lymphocytic infiltrates were observed in various organs, including the lungs, mediastinum, submandibular lymph nodes, salivary glands, liver, kidneys, and peritoneum. Lymphocytolysis was recognized in the Peyer’s patches of the ileum.
A diagnosis of histiocytic sarcoma was made based on the histomorphologic features of the mass, the presence of renal tubular hyaline globules, and myeloid hyperplasia in the bone marrow. As histiocytic sarcomas lack a distinct morphologic appearance, they must be distinguished from other types of sarcomas: soft tissue sarcomas, and lymphoid and myeloid neoplasms. The presumptive diagnosis was confirmed by IHC, upon which the majority of neoplastic cells showed strong membranous immunopositivity for the histiocytic cell markers Iba1 (Figures 2(b) and 3(c)) and CD11b (Figures 2(c) and 3(d)). A smaller proportion of the neoplastic cells showed a moderate to strong cytoplasmic immunoreactivity for Mac-2 (Figures 2(d) and 3(e)). The neoplastic process showed no immunoreactivity for the lymphoid cell markers CD3, T cell marker, and B220 and CD19, B cell markers. Therefore, the differential diagnosis of lymphoma can be excluded.
A pronounced secondary systemic inflammatory and hematopoietic cell response was observed in a variety of organs and tissues. Marked splenic EMH, marrow myeloid hyperplasia, and hematopoietic precursor cells present in the kidney are features seen in association with histiocytic sarcomas in mice and dogs.3,4 As the animal was used for monoclonal antibody development, it is unclear to what extent the presence and severity of the general inflammatory state is attributable to immunization. The presence of lymphocytolysis of the Peyer’s patches was likely secondary reactive to the systemic neoplastic and inflammatory processes.
Left atrium and auricle thrombosis is a common finding in female Syrian hamsters (Mesocricetus auratus), typically occurring in association with amyloidosis or with changes in fibrinolytic parameters. 5 In this animal there was no evidence of amyloidosis upon histopathological examination of the heart and any other tissue. Thrombus formation has also been described in association with neoplastic processes, including histiocytic sarcomas in hamsters. 6
Renal hyaline droplets in the proximal tubular epithelium are commonly observed in association with myeloid leukemia, lymphoma and histiocytic sarcomas in mice and rats, and are also present in this hamster, corroborating the diagnosis of a histiocytic neoplasm.7–9 In cases of histiocytic sarcoma in mice and rats, hyaline droplets were shown to be positive for lysozyme. The overproduction of lysozyme by neoplastic cells followed by renal tubular uptake has been proposed as the mechanism for their presence. 9
Historically, hamsters were commonly used for experimental induction of tumors using xenogeneic viruses – for example, adenoviruses, papillomaviruses, polyomaviruses; however, spontaneous tumors are generally rare in these species. 5 Most information regarding neoplasia in hamsters has been obtained from Syrian hamsters as they are the most commonly used species in research. 10 Marked variation in the prevalence and incidence of neoplasms has been reported in different colonies and is most likely due to the influence of genetic, age, sex, environmental conditions, and extent of the gross and histologic examinations in each of these reports.5,10–12
Lymphomas and epithelial tumors are the most common neoplasms observed in hamsters, and can be associated with hamster polyomavirus (HaPyV) infection. HaPyV is the known cause of transmissible lymphoma, which is epizootic in young Syrian hamsters, affecting mainly the mesenteric lymph nodes and gut-associated lymphoid tissue.5,13,14 However, HaPyV infections are uncommon in laboratory hamsters. Spontaneous, not viral-induced lymphoma occur in aged hamsters and are often multicentric, affecting the lymphoid tissues, including thymus, lymph nodes and spleen, as well as the liver and skin. Mycosis fungoides-like cutaneous lymphoma have been described in adult Syrian hamsters.5,13,15,16
Histiocytic sarcomas have previously been reported in Syrian hamsters and Siberian dwarf hamsters (Phodopus sungorus).6,12,17 The occurrence of neoplasia in Armenian hamsters is not well characterized and prior to this report was limited to the description of hepatocellular carcinoma in animals exposed to estrogens. 10 This report represents the first case of histiocytic sarcoma in an Armenian hamster. Histiocytic sarcoma is a malignant proliferation of cells with morphologic and immunophenotypic features similar to those of mature histiocytes. 6
Since the 1980s, Armenian hamsters have been used to generate interspecies mouse:hamster hybridomas that secrete hamster monoclonal immunoglobulin. As a result, Armenian hamster monoclonal antibodies are now commonly used reagents for many immunological assays. 11 It is plausible to consider that the animal’s experimental history of immunizations and the development of the histiocytic sarcoma may be associated. The immunogen used in this animal, KLH, is highly immunogenic and would likely have caused a potent localized inflammatory response after each immunization. There is a well-documented association between adjuvanted vaccine injection sites subsequent abnormal tissue reaction to chronic inflammation (i.e. uncontrolled proliferation of fibroblasts and myofibroblasts), and development of soft tissue sarcomas in cats including fibrosarcomas, malignant fibrous histiocytomas, osteosarcomas, and rhabdomyosarcomas.18–22 Histiocytic sarcoma, a hematopoietic neoplasm that is generally not classified as a soft tissue sarcoma in laboratory rodents, has not been associated with vaccination in cats or other species, although it does occur in the subcutis in laboratory rodents. A recent report showed the association between the use of vaccines including aluminum-based adjuvants and the appearance of injection site granulomas in lambs and macrophage-driven translocation of the adjuvant into regional lymph nodes and its potential association with systemic signs. 23
The adjuvant used in this case, TiterMax®, is an alternative to Freund’s Adjuvant as it has shown similar reliability and effectiveness without the associated toxic adverse effects. 24 It contains three main ingredients: a proprietary block copolymer CRL-8941 (immunomodulator), squalene (a metabolizable oil), and a unique microparticulate stabilizer that allows for the formation of a stable water-in-oil emulsion when mixed with the immunogen. 24 The copolymer binds antigen and activates macrophages and complement, which influences the localization and retention of antigen in lymphoid tissue and the activation of immunoreactive cells. Subcutaneous granulomas develop at the immunization site in response to the adjuvant/immunogen emulsion resulting in a local antigen-antibody-complement type III hypersensitivity (Arthus) reaction. Subsequent activation of complement activates the recruitment of polymorphonuclear cells and the degranulation of tissue mast cells.24,25 The recruitment and activation of macrophages and dendritic cells for antigen processing by adjuvant immunomodulators can lead to other inflammatory consequences. 25 Activated macrophages secrete proinflammatory cytokines that modulate the adaptive immune response and antibody production, recruit neutrophils, basophils, and lymphocytes, increase vascular permeability, and cause local tissue destruction. However, severe adverse reactions have not been reported in hamsters in association with TiterMax®. 24
Given the limited information on the pathogenesis of vaccine-associated granulomas or feline injection-site sarcomas (FISS), it is possible the experimental immunization protocol in this hamster induced or facilitated the development of histiocytic sarcoma. Repeated immunogen administrations might have triggered an excessive reparative response at the injection site, subsequently cumulating in malignant transformation of the mesenchymal cells. Although the location of the immunization (dorsal neck) and the tumor (ventral neck) were distinct, drainage from the area and their relative proximity support their association. However, no reports of a phenomenon similar to FISS has been reported in vaccinated or immunized hamsters, nor have we observed sarcomas in other immunized hamsters; therefore, it is unclear if such an association exists, and if it does it is rare. Furthermore, these immunization reactions tend to be soft tissue sarcomas thought to be derived from the mesenchymal stem cell, whereas histiocytic sarcomas are a hematopoietic tumor and have not been commonly associated with these vaccination/injection reactions. Whether spontaneous or experimentally induced, this is the first reported case of histiocytic sarcoma in an Armenian hamster.
Footnotes
Acknowledgements
We thank Drs Frances Weis-Garcia who provided key experimental details and Neil S Lipman for his editorial assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by a NIH/NCI Cancer Center Support Grant (grant number P30 CA008748).
