Abstract
Case summary
This report describes the diagnostic findings, natural history and genetic analysis of the candidate gene Forkhead Box F1 (FOXF1) in a young cat with developmental lung disease and high probability of pulmonary hypertension. A 1-year-old male entire Chartreux cat was referred for cardiac murmur investigation and exercise intolerance. Echocardiography identified a high-velocity tricuspid regurgitant jet with right-sided cardiac changes, supporting a high probability of pulmonary hypertension. No congenital cardiac shunts or left-sided cardiac changes were found to support a primary cardiac cause of pulmonary hypertension. Extensive laboratory work, thoracic radiographs and CT were performed. Histopathological characterisation (lung biopsy and later post mortem) was necessary to reach the final diagnosis. Eight months after diagnosis, the cat developed right-sided congestive heart failure, eventually leading to euthanasia. Survival from diagnosis to death was 12 months.
Relevance and novel information
Developmental lung disease belongs to a group of diffuse lung diseases in humans associated with pulmonary hypertension. The veterinary literature describing lung growth disorders in cats is sparse, and the present report provides information on clinical presentation and progression alongside a thorough diagnostic workup, which may aid clinicians in identifying this condition. Lung biopsy was pivotal in reaching the final diagnosis. No causal variants in FOXF1 were identified.
Introduction
In children, developmental lung disease (DLD) falls under the broad category of diffuse lung disease, which encompasses a group of heterogeneous disorders characterised by impaired gas exchange and diffuse infiltrates on imaging.1–3 This condition in cats has yet to be thoroughly described and classified.4 –7 A subset of DLD in children results in the development of pulmonary hypertension (PH), which is associated with a guarded prognosis and reduced survival.8 –10
Genetic variants may be associated with certain diffuse DLDs in children, including genomic alterations in the gene Forkhead Box F1 (FOXF1), which is a transcription factor critical in embryonic lung development.11 –15
This case report describes the clinical findings, diagnostic evaluation and natural history of a young cat with histologically confirmed DLD leading to PH. Genetic testing was also performed to determine if a FOXF1 variant contributed to DLD in this cat.
Case description
A 1-year-old male Chartreux cat was referred to the cardiology department of Ospedale Veterinario San Francesco after presenting with exercise intolerance, open-mouth breathing and a heart murmur. The cat had been an indoor-only pet in a single-cat household since being acquired from the breeder at 8 weeks of age. Physical examination revealed a grade IV/VI left basilar holosystolic murmur with a normal heart rate and pulmonary auscultation.
Echocardiographic evaluation showed severe right ventricular concentric hypertrophy, moderate right atrial enlargement, no evidence of right interventricular or infundibular obstruction, normal pulmonic valve anatomy and pulmonary flow (0.61 m/s), minimal structural tricuspid valvular changes (mild thickening and elongation of the valve leaflets without obvious chordal or papillary muscle abnormalities) and a high-velocity tricuspid regurgitation jet (4.5 m/s). The left ventricle was underfilled with equivocal left ventricular wall thickness (maximal end-diastolic wall thickness: 5.6 mm interventricular septum and 5.8 mm free wall) and paradoxical septal motion. The left atrium was normal. Systolic anterior motion of the mitral valve was noted with a high velocity, turbulent scimitar-shaped aortic blood flow and associated high-velocity mitral regurgitation. No intracardiac shunts were noted (Figure 1). A bubble study excluded intracardiac or extracardiac right-to-left shunts.

Composite echocardiographic images in a Chartreux cat referred for heart murmur, exercise intolerance and open-mouth breathing during exercise. (a) Right parasternal, four-chamber view optimised for the right chambers showing right atrial enlargement with atrial septal bowing toward the left atrium and right ventricular hypertrophy. (b) Right parasternal five-chamber view, colour Doppler image showing findings compatible with systolic anterior motion of the mitral valve (mitral regurgitation and left ventricular outflow tract turbulence). (c) M-mode image taken from the right parasternal short-axis view at the papillary muscles level. Significant right ventricular dilation and hypertrophy were noted, with paradoxical septal motion and reduced left ventricular internal diameters. (d) Pulsed-wave Doppler flow taken at the pulmonary valve: laminar flow with fast acceleration time and longer ejection time (aorta-like flow profile). (e) Left parasternal apical view optimised for the right chambers. Colour Doppler flow showing the presence of tricuspid regurgitation. (f) Continuous-wave Doppler flow from tricuspid regurgitation jet identifying severe tricuspid regurgitation (4.5 m/s). These findings were compatible with high probability of severe pulmonary hypertension
Laboratory analysis included haematology (ProCyte Dx; IDEXX) with manual smear evaluation, serum biochemistry (BT3500 Vet; Futurlab), coagulation profile (Stago Compact Max), urinalysis and testing for feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) (SNAP Combo Plus; IDEXX). The only clinically relevant abnormalities on the serum biochemistry panel were a mild increase of alanine transaminase (ALT 241 U/l; reference interval [RI] 16–111) and aspartate aminotransferase (AST 62 U/l; RI 9–28). Faecal flotation, Baermann test on three fresh faecal samples and serum antibody against Dirofilaria immitis (IDEXX Laboratories) were performed and tested negative.
Three-view thoracic radiographs showed normally expanded pulmonary fields with no abnormalities of the pulmonary parenchyma. A severe generalised increase in the size of the cardiac silhouette was observed without obvious pulmonary vascular changes (Figure 2).

Thoracic radiographs, performed in lateral (right) and ventrodorsal (left) views at the time of presentation, showing severe generalised cardiomegaly without notable changes in lung parenchyma and/or pulmonary vessels
Advanced imaging with consideration for histopathological characterisation of the lung parenchyma was advised. A CT angiography examination was performed under general anaesthesia (Brivio 385CT; General Electric) using iodinated non-ionic contrast medium (2 ml/kg, Visipaque 320 mg I/ml; GE Healthcare). The main findings were severe generalised cardiomegaly (mainly affecting the right chambers), diffuse ground-glass opacity in the lung parenchyma, some parenchymal bands and subpleural interstitial thickening associated with mild bronchiolectasis (Figure 3).

Composite image showing relevant CT findings. (a) The lung parenchyma in a pulmonary window. (b) Severe cardiomegaly, in a pulmonary window, with the presence of ground-glass opacity (GGO). The degree of GGO was likely attributable to primary lung disease, but unlikely to justify the severe right-sided cardiomegaly and reported clinical signs. (c) Severe generalised cardiomegaly, in a soft tissue window, mainly involving the right atrium and ventricle. (d) The pulmonary trunk, in a soft tissue window. Although there are currently no standard measurements applicable to cats, the pulmonary trunk to aorta ratio measured normal (1.1).
A lung biopsy of the middle right lung lobe was performed using a mini-thoracotomy technique because of the diffuse nature of the tomographic changes. The tissue sample was submitted for histopathology to the Veterinary Diagnostic Laboratory at Michigan State University, which was consistent with a developmental abnormality of the pulmonary parenchyma.
Eight months after the original diagnosis, the patient presented with laboured breathing. Point-of-care ultrasound showed pleural effusion, which was drained, yielding approximately 150 ml of serosanguineous fluid that had a lymphocyte-rich effusion (total protein: 2.8 g/dl) with a predominantly small lymphocyte population (total nucleated cell count: 15,000 cells/µl). The most likely differential was pleural effusion due to congestive heart failure, which was presumed to be right-sided because of the high probability of severe PH and right-chamber remodelling.
Intravenous furosemide (Dimazon; MSD Animal Health) treatment at 2 mg/kg IV was started, with significant improvement in the patient’s general clinical status. Escalating doses of oral furosemide (Diuren; Teknofarma S.r.l.) were needed in the following 4 months to control congestive heart failure signs. Unfortunately, compliance with oral medication was poor and additional therapies could not be added. As a result of the patient’s decreased quality of life, the owners opted for euthanasia 12 months after the diagnosis. After euthanasia, the lungs, heart, kidneys, liver and spleen were placed in formalin and submitted to the Veterinary Diagnostic Laboratory at Michigan State University for histopathological examination.
Gross pathology confirmed right-sided cardiomegaly with marked right atrial enlargement and mild right ventricular dilatation with mild hypertrophy without any stenotic lesions affecting the right ventricle or pulmonary trunk. Widespread cardiomyocyte degeneration was noted. Capsular foci of neovascularisation and fibrin deposition on the liver surface were present, supporting right-sided cardiac insufficiency. The lungs were affected by diffuse lung pathology (Figure 4). All lobes were characterised by markedly enlarged alveolar spaces and airway abnormalities reflecting abnormal development of both conducting airways and alveoli (Figure 5).
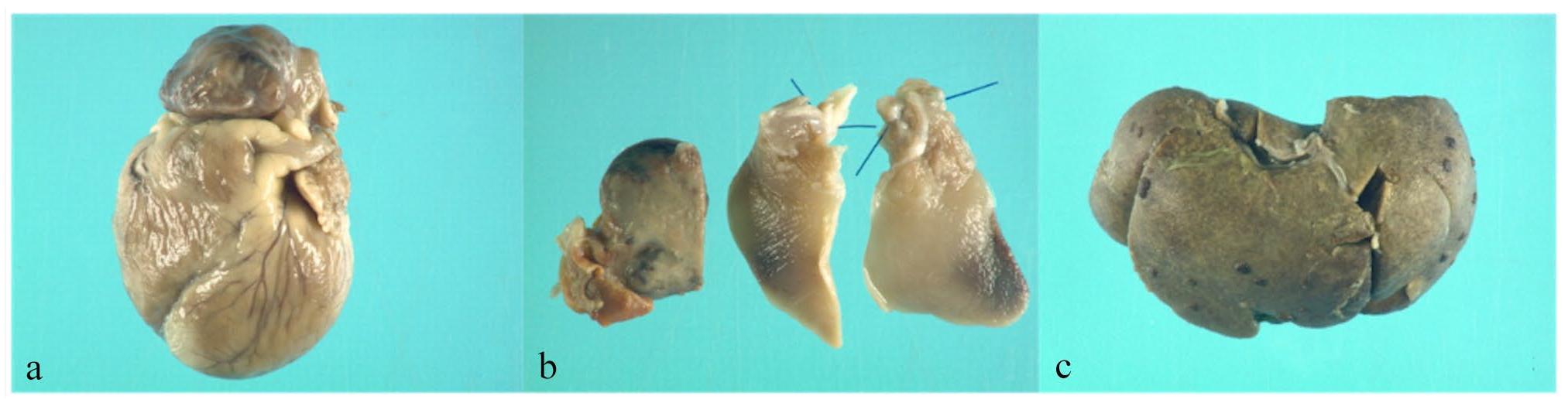
Relevant gross pathology findings: (a) prominent right-sided cardiomegaly with dilated right auricle, right ventricular dilation and normal pulmonary trunk; (b) lung lobes, fixed in formalin, with diffuse mottled areas in all lung lobes; and (c) macroscopic appearance of the liver, which appears enlarged with fibrin deposition and foci of neovascularisation
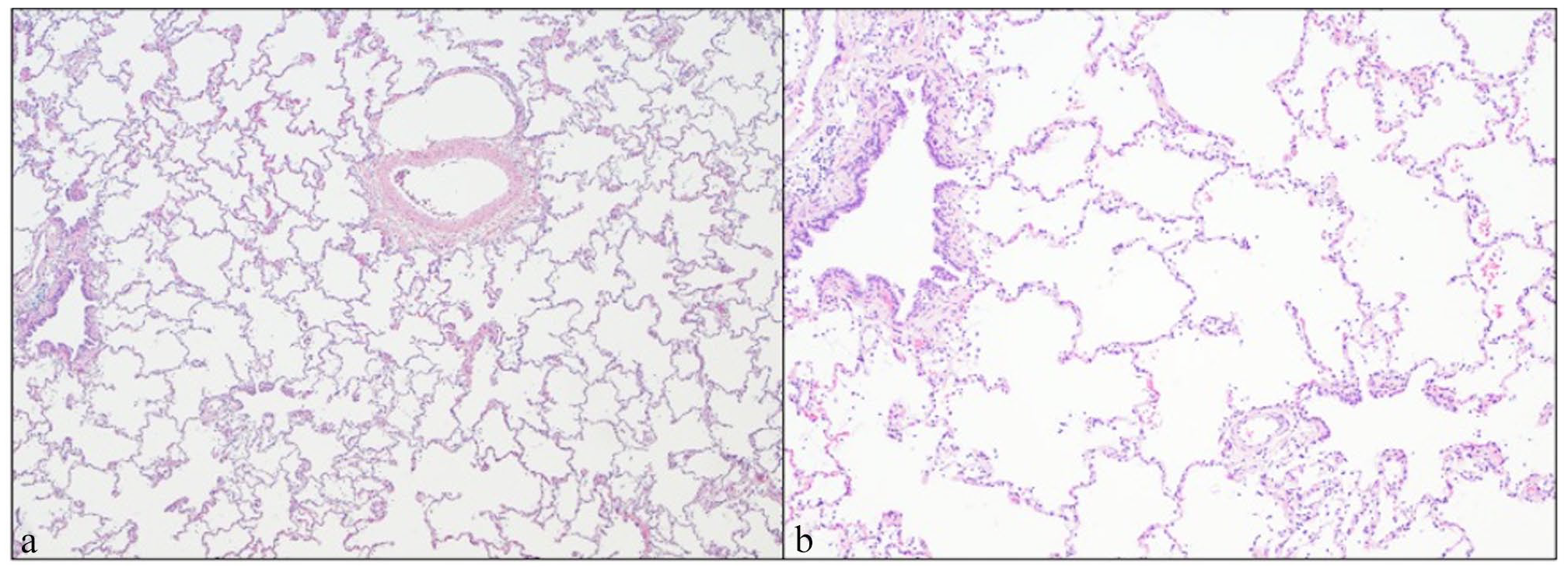
Lung histopathology. The alveoli are markedly dilated in the uncollapsed portions of the lung. Alveolar haemorrhage was identified in many alveoli but was considered artefactual. (a) Low magnification showing the lung alveolar parenchyma is diffusely affected. Individual alveolar diameter is equivalent to or larger than adjacent terminal bronchioles. (b) High magnification slide showing alveolar parenchyma enlargement. Bronchi and bronchioles had prominent smooth muscle bundles, occasionally surrounded by prominent lymphofollicular aggregates (bronchus-associated lymphoid tissue hyperplasia) and often contained large numbers of foamy alveolar macrophages and eosinophilic cellular debris
Whole blood EDTA and kidney tissues were submitted to the University of Milan for DNA extraction and were shipped to the Feline Genetics and Comparative Laboratory at the University of Missouri. Approximately 3 µg of DNA was submitted for variant detection in FOXF1 using Sanger sequencing. Cat reference assembly F.catus_Fca126_mat1.0 (GCF_018350155.1; BioProject: PRJNA684600; BioSample: SAMN19729387) and RefSeq Annotation 105 were used to design PCR primers using Primer3Plus (www.bioinformatics.nl/cgi-bin/primer3plus/primer3plus.cgi) to amplify both exons of FOXF1 (XM_023245078). Exon 1 was amplified using two overlapping PCR fragments. PCR and Sanger sequencing were conducted as previously described. 16 The generated sequences were aligned to the cat reference genome assembly F.catus_Fca126_mat1.0 and visually inspected for variants using Sequencher 5.1 (GeneCodes). The only variant detected within the two exons was within the tandem repeat of the glycine codon (GGC) in which the case cat was heterozygous and had 9–10 glycine repeats. This repeat occurs at approximately position E2:61213780 in transcript XM_023245078.1:c.219_221delCGG; p.Gly78del. This in-frame insertion-deletion variant is commonly identified in the 99 Lives Cat Genome Sequencing database, which includes data from 362 cats with whole genome sequence data and 52 cats with whole exome sequence data (See Table 1 in File 1 of the supplementary material). 17 Therefore, this variant is not anticipated to be pathogenic and causal for the cat’s condition.
Discussion
This case report highlights the clinical presentation, diagnostic evaluation and natural history of a cat with a histopathological diagnosis of DLD with a high probability of severe PH. Few cases of congenital lung disorders in cats are reported4 –7 and none to date had associated PH. In cats, DLDs are likely under-recognised as extensive diagnostic investigations are needed to exclude other causes, with lung tissue ultimately required for definitive diagnosis.
Although congenital or acquired heart disease would have been considered more likely with the patient’s signalment and clinical presentation, other congenital lung conditions, such as DLD, could still fit the clinical presentation in a young cat with severe respiratory signs and associated heart murmur. As is true for certain parenchymal diseases and pulmonary vascular disorders, thoracic radiographs and CT imaging lacked pathognomonic features of a specific disease, albeit were helpful in excluding other disease conditions. 18 Echocardiography was pivotal in confirming the high probability of PH and excluding congenital or acquired cardiac disease conditions that could have similarly caused PH.19 –22
PH was the main echocardiographic finding and was likely responsible for the original complaint and contributed to the subsequent clinical deterioration and death. The presence of PH is unusual in cats and little information is available for non-cardiac causes. 23 Lacking specific guidelines for a PH diagnosis and management in cats, the authors investigated cardiac, vascular and pulmonary causes, which could have been responsible for the development of PH.22 –26
The presence of left ventricular hypertrophy and systolic anterior motion were considered a consequence rather than a primary cause of PH, because of the lack of left atrial enlargement or severe hypertrophy, the magnitude of tricuspid regurgitant velocity and the absence of histopathological changes typical of hypertrophic cardiomyopathy. Although not specifically reported in cats, the development of systolic anterior motion of the mitral valve has been reported in dogs with PH or dehydration.27,28
Ante-mortem histological characterisation of the lung parenchyma by lung biopsy was necessary to reach a definitive diagnosis.18,29 –32
Further diagnostic procedures, such as endoscopy and bronchoalveolar lavage, were not performed because of their limitations in diagnosing interstitial disease, and the low probability of reaching a definitive diagnosis based on the CT scan’s findings and laboratory analysis.33 –35
In humans, pathology has a central role in establishing the World Health Organization classification scheme for PH and was considered for this condition of PH in cats.36,37
Lung biopsy allowed direct assessment of architectural changes that may be present in primary pulmonary parenchymal disease or pulmonary arterial hypertension.22,38 –40 Histopathological findings in the cat resembled forms of DLD, such as pulmonary alveolar underdevelopment described in children.41,42 Diagnosis of these conditions is challenging because of the non-specific clinical signs and the absence of pathognomonic markers on diagnostic imaging. Overall prognosis tends to be poor and PH is a predictor of mortality.1,8
Although life expectancy of the cat was markedly reduced by the disease process, at the time of the initial evaluation, the cat presented with apparent mild clinical signs, mainly exacerbated by exercise, which is consistent with reported non-specific and moderate clinical signs in cats with PH. 23 In this particular case, echocardiographic changes were supportive of PH, including the marked right-sided remodelling without obvious cardiac reasons. Throughout the disease course, the overall quality of life was perceived as acceptable. Unfortunately, once right heart failure occurred, survival was markedly reduced, and medical therapy was limited to diuretic treatment as a result of poor compliance with oral medication, a common issue in feline patients.43 –46
Some genetic variants are described in infants with DLD, including variants affecting FOXF1, which is associated with alveolar capillary dysplasia with misalignment of the pulmonary veins. 9 Genomic analysis did not identify a causal variant in FOXF1 in this cat.
Conclusions
DLD represents a newly identified cause of PH in the domestic cat. A lung biopsy is required for a definitive ante-mortem diagnosis. The prognosis is overall poor for diffuse disease with PH, but quality of life can be acceptable until congestive heart failure occurs. Additional genetic analyses, such as whole genome or exome sequencing, could be conducted to potentially identify a genetic cause for the disease.
Supplemental Material
sj-docx-1-jor-10.1177_20551169241249003 – Supplemental material for Developmental lung disease in a cat associated with high probability of severe pulmonary hypertension: natural history, histopathology and genetic analysis
Supplemental material, sj-docx-1-jor-10.1177_20551169241249003 for Developmental lung disease in a cat associated with high probability of severe pulmonary hypertension: natural history, histopathology and genetic analysis by Yari Nebel, Kurt Williams, Leslie A Lyons, Carol Reinero, Riccardo Ferriani, Roberto Toschi Corneliani and Ilaria Spalla in Journal of Feline Medicine and Surgery Open Reports
Supplemental Material
sj-docx-2-jor-10.1177_20551169241249003 – Supplemental material for Developmental lung disease in a cat associated with high probability of severe pulmonary hypertension: natural history, histopathology and genetic analysis
Supplemental material, sj-docx-2-jor-10.1177_20551169241249003 for Developmental lung disease in a cat associated with high probability of severe pulmonary hypertension: natural history, histopathology and genetic analysis by Yari Nebel, Kurt Williams, Leslie A Lyons, Carol Reinero, Riccardo Ferriani, Roberto Toschi Corneliani and Ilaria Spalla in Journal of Feline Medicine and Surgery Open Reports
Footnotes
Acknowledgements
The authors would like to thank Dr Isabelle Masseau, Dr Silvia Fabbri and Giovanni Pastorino for their help in radiographic and CT interpretation. The authors are also thankful for the help and support provided by Dr Maria Longeri for the DNA extraction and shipping of the samples for genetic analysis. They appreciate the technical laboratory assistance of Thomas R Juba and thank the contributors to the 99 Lives Cat Genome Sequencing Consortium for access to the variant call file for variant allelic data.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Gilbreath McLorn Endowment of the University of Missouri, College of Veterinary Medicine (LAL), which financed the execution of the genetic evaluation.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
Supplementary material
The following files are available as supplementary material:
File 1: Additional details on genomic analysis in a cat with developmental lung disease and high probability of severe pulmonary hypertension
Video 1: Two-dimensional transthoracic echocardiography, right parasternal four-chamber long-axis view optimised for the right chambers, highlighting the degree of right atrial enlargement and right ventricular hypertrophy.
Video 2: Two-dimensional transthoracic echocardiography, right parasternal four-chamber long-axis view optimised for the right chambers.
Video 3: Two-dimensional transthoracic echocardiography, right parasternal short-axis view at the level of the mitral valve leaflet.
Video 4: Two-dimensional transthoracic echocardiography, right parasternal five-chamber long-axis view with Colour Doppler.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
