Abstract
Case summary
A 15-week-old kitten presented with a 1-month history of intermittent generalised tremors and abdominal distension. Hypocalcaemia associated with increased 1,25-vitamin D3 was consistent with vitamin D3-dependent rickets type II. The bone appearance on CT scan was most consistent with the changes typically seen with nutritional secondary hyperparathyroidism and less typical of the changes seen with rickets. Our patient had a positive response to high vitamin D3 therapy as it remained normocalcaemic 16 months after diagnosis, supporting the diagnosis of rickets.
Relevance and novel information
This case report is an unusual and interesting presentation of rickets in a kitten. Despite the characteristic vitamin D3 disturbance for rickets type II, the atypical radiographic changes have not been previously reported. In the literature, a positive response to treatment is not commonly seen or follow-up is short. Our case responded well to treatment and was followed for 16 months from the time of diagnosis. This emphasises that the pathophysiology of the condition is not well understood, and that different types of vitamin D3-dependent rickets type II may exist. Although the genetic defects responsible for some cases of rickets type I have been identified, this has still not been determined for rickets type II.
Keywords
Introduction
Rickets is a metabolic bone disease caused by dietary deficiency of vitamin D3 or phosphorus, or by genetic defects affecting vitamin D3 or phosphorus metabolism. 1 Vitamin D3-dependent rickets (VDDR) is a rare disease in cats causing hypocalcaemia and skeletal abnormalities. Softening and bowing of the long bones 2 is observed and so VDDR is also called osteomalacia in the literature.
The only available source of vitamin D3 in cats is obtained from their diet. However, dietary vitamin D (cholecalciferol) is not metabolically active and two enzymatic conversions are necessary to synthesise the active form of vitamin D3. 3 First, the hydroxylation of vitamin D (cholecalciferol) to 25-hydroxycholecalciferol (calcidiol) occurs in the liver and is catalysed by vitamin D 25-hydroxylase (CYP2R1). Then, the hydroxylation of calcidiol to 1,25-hydroxycholecalciferol (calcitriol) occurs in the kidneys and is catalysed by 1-alpha-hydroxylase (CYP27B1). Finally, calcitriol binds and activates the nuclear vitamin D receptor (VDR) playing a central role in calcium homeostasis. 2
The parathyroid glands produce parathyroid hormone (PTH) secondary to ionised calcium (iCa) fluctuations in the extracellular fluid. iCa is the biologically active form of calcium, readily available to cells and not bound to proteins. When iCa is low, PTH secretion increases and when iCa is high, PTH secretion decreases. PTH has an effect on the kidneys, bones and gastrointestinal tract (Figure 1). In the kidneys, PTH will increase calcium reabsorption, phosphorous excretion and activity of 1-alpha-hydroxylase leading to enhanced calcitriol production. In the gut, calcitriol increases the absorption of calcium and phosphorous. Finally, in the bone, PTH promotes osteoclastic bone resorption, increasing calcium and phosphorous levels. 1
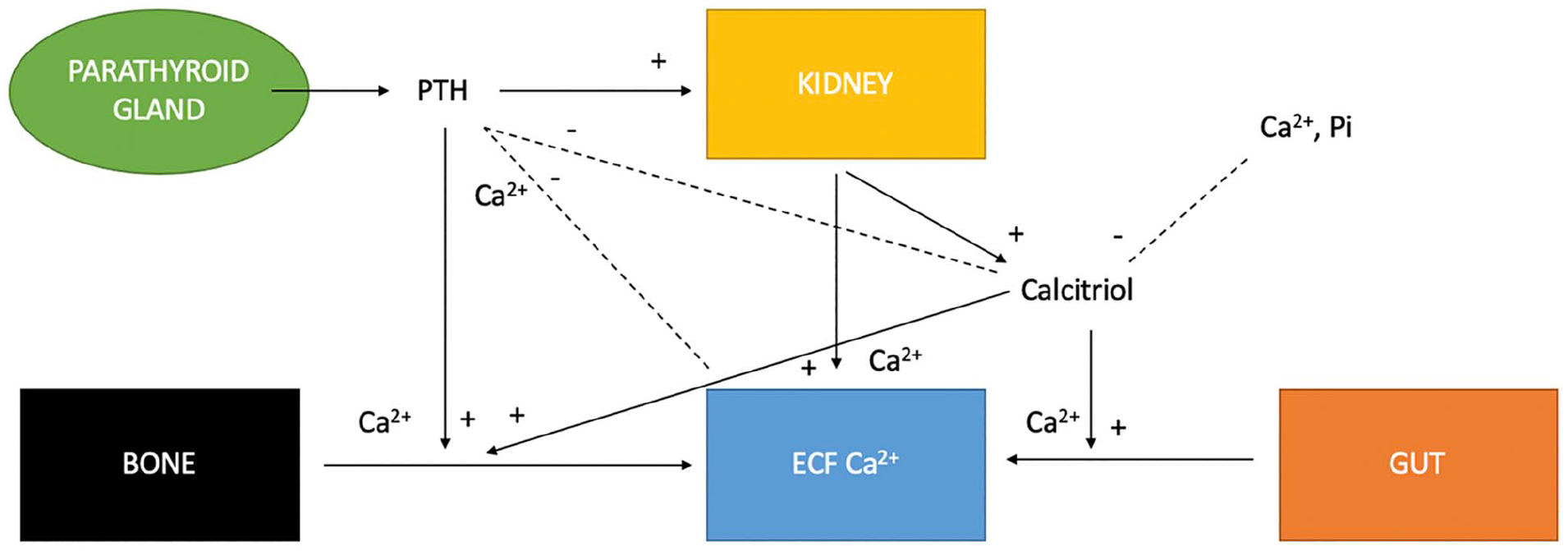
Regulation of extracellular fluid (ECF) calcium concentration by the effects of parathyroid hormone (PTH) and calcitriol on the gut, kidneys, bone and parathyroid gland (from Parker et al 4 ). Ca2+ = calcium; Pi = phosphate
Four types of rickets are described in humans: (1) nutritional rickets (nutritional vitamin D3 or phosphorous deficiency); (2) hereditary X-linked hypophosphataemia rickets; (3) VDDR type I (defect in calcitriol production, type IA: mutation in the CYP27B1 gene; or type IB: mutation in the CYP2R1 gene); and (4) VDDR type II (impaired response of target organs to calcitriol, mutation in the VDR gene).2,5
It is necessary to measure the level of calcitriol to differentiate between VDDR type I from type II: high 1,25-vitamin D3 is consistent with VDDR type II and low 1,25-vitamin D3 is consistent with VDDR type I.5,6
Normal functioning vitamin D3 receptors in the kidneys are required for feedback suppression of calcitriol synthesis. The defect of the vitamin D3 receptors in VDDR type II results in increased 1,25-vitamin D3 (calcitriol). Calcitriol is necessary for bone mineralisation during skeletal growth; thus, abnormal calcitriol metabolism results in defective ossification. 6
In the veterinary literature, there are only a few case reports of rickets in kittens unrelated to nutritional deficiency.2,5–11 Ours is an unusual presentation of rickets in a kitten with characteristic (type II) vitamin D3 disturbance (high level of calcitriol) but atypical radiographic changes and a positive response to treatment over time.
Case description
A 15-week-old, male entire, domestic longhair kitten presented with a 1-month history of intermittent generalised tremors and abdominal distension. The kitten received its first vaccination, was fully dewormed and had not travelled outside of the UK. It was fed a mixture of three different commercial diets (AVA Veterinary Approved kitten dry food, Felix kitten pouch and Whiskas kitten pouch), and ate approximately a third of each diet daily in manufacturer-recommended quantities, according to the owner. The owner stated that the diet had not changed since adoption and the cat was not given any treats. The composition and analysis of the three different diets, as declared by the manufacturer, can be found in the supplementary material. The kitten diet was formulated for growth according to FEDIAF (The European Pet Food Industry Federation) nutrition guidelines. No information was available for the other kittens from the litter.
On physical examination, the kitten was bright and responsive. It weighed 1.2 kg and was in reduced body condition (body condition score 3/9 [normal 5/9]). The patient exhibited moderate muscle wasting. Its heart rate, respiratory rate and temperature were normal. Its long bones were deformed with pronounced bowing. No pain was elicited on palpation of the bones. Marked abdominal distension was noted and abdominal palpation revealed a distended colon full of faeces. Neurological examination was unremarkable.
External haematology, performed by the referring veterinary surgeon, documented a mild leukocytosis and monocytosis (Table 1), consistent with a non-specific inflammatory response. External biochemistry documented hypocalcaemia, mildly decreased creatinine and mildly increased alkaline phosphatase (ALKP). A bile acid stimulation test and total thyroxine were normal (Table 2). The phosphorous and total protein levels were considered normal using a reference interval extrapolated from a previous study evaluating seven clinically normal kittens of a similar age. 12 The low creatinine was attributed to the moderate muscle wastage. The increased ALKP is likely due to the increased osteoblastic activity owing to the young age of the patient. The most significant abnormality was the total hypocalcaemia.
External haematology

RBCs = red blood cells; MCV = mean cell volume; MCH = mean cell haemoglobin; MCHC = mean cell haemoglobin concentration; WBCs = white blood cells
External biochemistry, bile acid stimulation test and total thyroxine

Tomsa et al (1999) data from seven healthy control kittens 4–6 months old 12
RI = reference interval
Differential diagnoses for hypocalcaemia includes rickets, nutritional secondary hyperparathyroidism, hypovitaminosis D3, primary hypoparathyroidism, pancreatitis, kidney disease, toxic ingestion, hypomagnesaemia, hypoproteinaemia and a spurious result. Kidney disease was unlikely based on the reported normal urea and decreased creatinine. There was no toxic exposure reported for this patient. Hypoproteinaemia was excluded owing to the reported normal albumin concentration. The test was repeated to ensure it was not spurious.
To further investigate the cause of hypocalcaemia, iCa, total calcium, phosphorous, PTH, magnesium, feline pancreatic lipase immunoassay (fPLI), calcidiol (25-vitamin D3) and calcitriol (1,25-vitamin D3) were measured. The iCa must be measured to determine if the total hypocalcaemia is clinically significant. The laboratory results revealed low total and iCa associated with increased PTH, low 25-vitamin D3 and increased 1,25-vitamin D3 (Table 3). The ionised hypocalcaemia was clinically significant and can explain the constipation, secondary to reduced peristalsis, and the intermittent generalised tremors. It could also explain the metabolic bone disease and the marked bowing of the long bones, although another contributing factor such as orthopaedic disease was considered possible. A congenital abnormality of the colon or rectum could also result in constipation.
Additional laboratory parameters: ionised calcium, total calcium, phosphorous, parathyroid hormone, magnesium, feline pancreatic lipase immunoassay (fPLI), calcidiol (25-vitamin D3) and calcitriol (1,25-vitamin D3)

Tomsa et al (1999) data from seven healthy control kittens 4–6 months old 12
RI = reference interval; NA = not available
To further investigate congenital or orthopaedic disease, a CT scan of the chest, abdomen and long bones was performed. Generalised marked osteopenia with abnormal metaphyseal mineralisation, normal physes and slightly distorted osseous shapes were noted; most noticeable in the right femur (Figure 2), left shoulder (Figure 3a) and left stifle (Figure 3b). Additionally, a markedly distended colon with faeces (Figure 4) and subjectively narrowed pelvic canal (Figure 2, white arrow) were identified. There was no malformation of the bones of the pelvic canal. No pathological fracture was documented.
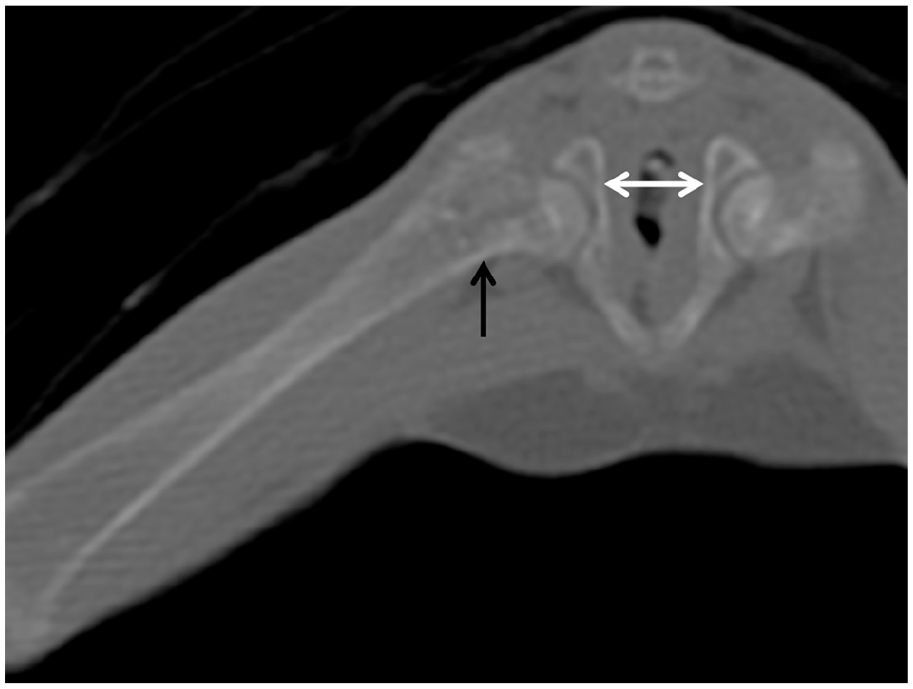
CT scan of the right femur showing marked osteopenia with abnormal metaphyseal mineralisation (black arrow), slightly distorted osseous shape and narrowed pelvic canal (white arrow)

CT scan of the (a) left shoulder and (b) left stifle showing marked osteopenia with normal physes (white arrows)

CT scan of the abdomen showing markedly distended colon with faeces (white arrows)
This patient had ionised hypocalcaemia and investigations to identify the underlying cause. Increased 1,25-vitamin D3 in association with hypocalcaemia and increased PTH can be seen with rickets (VDDR type II) and secondary nutritional hyperparathyroidism. 13 However, dietary analysis excluded secondary nutritional hyperparathyroidism. Hypovitaminosis D3 was excluded as a cause of hypocalcaemia as 1,25-vitamin D3 was increased. We suspected that low 25-vitamin D3 was secondary to the increased conversion of calcidiol to calcitriol owing to the lack of inhibition from abnormal vitamin D3 receptors. Hypoparathyroidism was excluded as PTH was increased. The increased PTH was considered an appropriate physiological response to the hypocalcaemia. Hypomagnesaemia was excluded as a cause of hypocalcaemia as total magnesium was normal. Pancreatitis was unlikely as fPLI was normal and the patient did not display any other clinical signs of pancreatitis. There was no toxic exposure reported for this patient.
Interestingly, the bone appearance on CT scan (generalised marked osteopenia with abnormal metaphyseal mineralisation, normal physes and slightly distorted osseous shapes) was most consistent with changes seen with nutritional secondary hyperparathyroidism. Marked colonic distension with faeces was noted, as reported on physical examination. The subjectively narrowed pelvic canal could be a consequence of abnormal vitamin D3 metabolism, leading to abnormal bone development. The VDR promotes osteoclastogenesis and endochondral bone formation. Vitamin receptor signalling in chondrocytes is required for timely osteoclast formation during bone development. Altered intestinal motility owing to the hypocalcaemia could also have contributed to the constipation observed.
Thus, the final diagnosis for this patient was VDDR type II presenting with generalised tremors, constipation and metabolic bone disease.
Initially, the patient received calcium carbonate 130 mg/day of elemental calcium divided into several meals and alfacalcidol (active form of vitamin D3) 60 ng/day. Its diet remained unchanged.
The patient responded slowly to treatment and both oral calcium and alfacalcidol were gradually increased up to 200 mg/day and 80 ng/day, respectively (in March 2018). At that time, the cat started responding to therapy as recorded in Table 4. Its calcium level progressively improved and became normal 7 months after diagnosis (in July 2018). Calcium supplementation was slowly discontinued and alfacalcidol was decreased at 60 ng/day. The patient significantly gained in weight from the time of diagnosis (1.6 kg over the last 16 months) but remained a small and short cat.
Total calcium after treatment with oral calcium and vitamin D3

RI = reference interval; NA = not available
Discussion
Our case report describes an interesting presentation of rickets. Hypocalcaemia associated with an increased level of 1,25-vitamin D3 is consistent with VDDR type II; reflecting malfunction of vitamin D3 receptors essential for feedback control of calcitriol. Increased calcitriol is not consistent with hypovitaminosis D or VDDR type I.5,6,11 The patient was not hypophosphataemic and so hereditary X-linked hypophosphataemia rickets was excluded.
Intriguingly, the reported vitamin D3 distribution can also be seen with rickets type II and secondary nutritional hyperparathyroidism. FEDIAF standards for kitten growth stipulates that growing kittens should be fed a diet with adequate vitamin D3 (28 IU/100 g of dry matter) and calcium (1 g/100 g of dry matter). The fact that these three diets all provide more than adequate calcium and cholecalciferol levels excludes secondary nutritional hyperparathyroidism as a cause of the clinical signs.
Calcitriol is required for bone mineralisation during skeletal growth. Interestingly, our patient had generalised marked osteopenia with abnormal metaphyseal mineralisation, normal physes and a slightly distorted osseous shape, which is most consistent with secondary nutritional hyperparathyroidism. One would have expected an increased diameter of the growth plate and metaphysis with rickets. 10
In the veterinary literature, there are only a few case reports of rickets in kittens that are unrelated to nutritional deficiency. Our case report can be compared to the case report from Schreiner and Nagode, 11 who described a 4-month-old kitten with VDDR type II (high 1,25-vitamin D3) and a positive response to vitamin D3 supplementation. However, this patient did not have radiographic bone abnormalities. 11 Our patient can also be compared to the case reports from Godfrey et al 8 and Tanner and Langley-Hobbs, 6 who described two 4-month-old kittens with VDDR type II (high 1,25-vitamin D3) and typical radiographic bony changes but that were non-responsive to vitamin D3 supplementation. Interestingly, these reports do not state the composition or the analysis of the diet that the kittens were fed and only one report measured 25-vitamin D3, which was within the normal limits. 6 These three cases of VDDR type II, including our case report, suggest that the disease has a diverse presentation that is not yet fully understood.
Our patient had a positive response to high levels of vitamin D3 supplementation and no change in diet. It remained normocalcaemic 16 months after diagnosis, supporting the diagnosis of rickets type II. This is most likely due to ineffective binding of calcitriol by the vitamin D3 receptors and only a partial response in target organs. Some patients do not respond to administration of high dosages of calcitriol (likely due to the inability of calcitriol to bind to the vitamin D3 receptors at all) and these patients often die soon after diagnosis. 11
Ideally, we would have performed radiography of the bones, to assess the bone density, prior to decreasing the vitamin D3 dosage. The dose would only have been reduced after ensuring the patient had normal bone density. Radiographs were unfortunately declined owing to financial restrains.
Ultimately, in human medicine, vitamin D3 receptor function can be tested by skin fibroblast culture and measuring activity and binding capacity of the vitamin D3 receptors. This has only been performed in one veterinary study to confirm the diagnosis. 9
Conclusions
This case report is an unusual presentation of rickets in a kitten. Despite characteristic vitamin D3 disturbance, this patient had atypical radiographic changes, which have not been previously reported and a positive response to treatment. This emphasises that the pathophysiology of this condition is not well understood, and that different types of VDDR type II may exist in veterinary patients. We suspect a partial response by the vitamin D3 receptors in this patient rather than complete absence given the clinical improvement to high doses of calcitriol supplementation. The genetic defects for some cases of feline rickets type I have been identified but are not known for type II.7,9,11 Further studies are necessary to fully characterise rickets in feline patients.
Supplemental Material
Supplementary_file – Supplemental material for Unusual presentation of vitamin D3-dependent rickets type II in a kitten
Supplemental material, Supplementary_file for Unusual presentation of vitamin D3-dependent rickets type II in a kitten by Florent Duplan and Christina Maunder in Journal of Feline Medicine and Surgery Open Reports
Footnotes
Acknowledgements
The authors would like to thank Dr Lizzie Reeve for her help providing CT scan images for publication.
Supplementary material
The following file is available online: Composition and analysis of the kitten’s diets (AVA Veterinary Approved kitten dry food, Felix kitten pouch and Whiskas kitten pouch) as stated by the manufacturers
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (including owned or unowned animals and data from prospective or retrospective studies). Established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care were followed. Ethical approval from a committee was therefore not necessarily required.
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (either experimental or non-experimental animals) for the procedure(s) undertaken (either prospective or retrospective studies). No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
