Abstract
Case summary
Two castrated male cats, aged 8 months old (case 1) and 10 months old (case 2), showed a history of progressive paraparesis, an over-reaching pelvic limb gait, urinary incontinence and a palpable dermoid fistula. In case 1, the fistula was connected to the dural sac on the conus medullaris, and the tethered spinal cord was retracted caudally. In case 2, the tubular structure was connected to the dural sac on the thoracic spinal cord, and the tethered spinal cord was retracted dorsally. Tethered cord syndrome secondary to spina bifida aperta was suspected in both cats. Excision of the fistula and release of the tethered spinal cord was performed. A histopathological examination confirmed the diagnosis of a meningomyelocele in case 1 and a meningocele in case 2. Paraparesis improved postoperatively in both cats. However, urinary incontinence in case 1 remained partially unresolved.
Relevance and novel information
This is the first report to describe the imaging characteristics, surgical treatments and outcomes of two different types of tethered cord syndrome with spina bifida aperta in cats. Tethered cord syndrome with spina bifida aperta needs to be included in the differential diagnosis of slowly progressive paraparesis in younger cats with or without vesicorectal failure and a palpable dermoid fistula.
Introduction
Spina bifida is a congenital malformation of unknown aetiology in which incomplete closure or fusion of the dorsal vertebral arches occurs with or without protrusion or dysplasia of the meninges, spinal cord and cerebrospinal fluid (CSF). 1 Spina bifida aperta (SBA) is a type of spina bifida that is associated with protrusion of the meninges (a meningocele) or the spinal cord, meninges and CSF (a meningomyelocele) through a defect in the vertebral arch. 2
Tethered cord syndrome (TCS) is a neurological disorder caused by tissue attachments that result in abnormal stretching of the spinal cord. 2 Although TCS occurs secondary to SBA in humans, 3 it has not been examined in as much detail in veterinary medicine.4–7 We herein report two feline cases of TCS with SBA for which imaging characteristics, surgical treatments and outcomes are presented.
Case description
Case 1 was an 8-month-old castrated male domestic shorthair cat presented to the veterinary referral hospital with a 2 week history of progressive paraparesis and constipation. The owner also reported progressively worsening urinary incontinence when the cat was adopted at 3 months of age. A physical examination revealed a palpable dermoid fistula over the caudal vertebra. A neurological examination showed proprioceptive deficits in the hindlimbs and the absence of anal and perineal reflexes. The lesion was neuroanatomically localised to the L4–S3 spinal cord segments. A complete blood count and biochemistry panels were within normal limits. MRI (EXCELART Vantage 1.5 Tesla; Toshiba) and CT (Aquilion 64-slice scanner; Toshiba) were performed. The palpable dermoid fistula was hyperintense on T2-weighted images (T2WI) and hypointense on T1-weighted images (T1WI), and was connected to the dural sac on the conus medullaris. This fistula appeared to have retracted the spinal cord caudally (Figure 1a–d). CT images revealed the lack of the dorsal arches of S3, Cd1 and Cd2 vertebrae and ventral displacement of the caudal vertebra (Figure 1e). Severe fecal impaction was also present. Based on these imaging findings and clinical signs, caudally retracted TCS secondary to SBA was suspected.
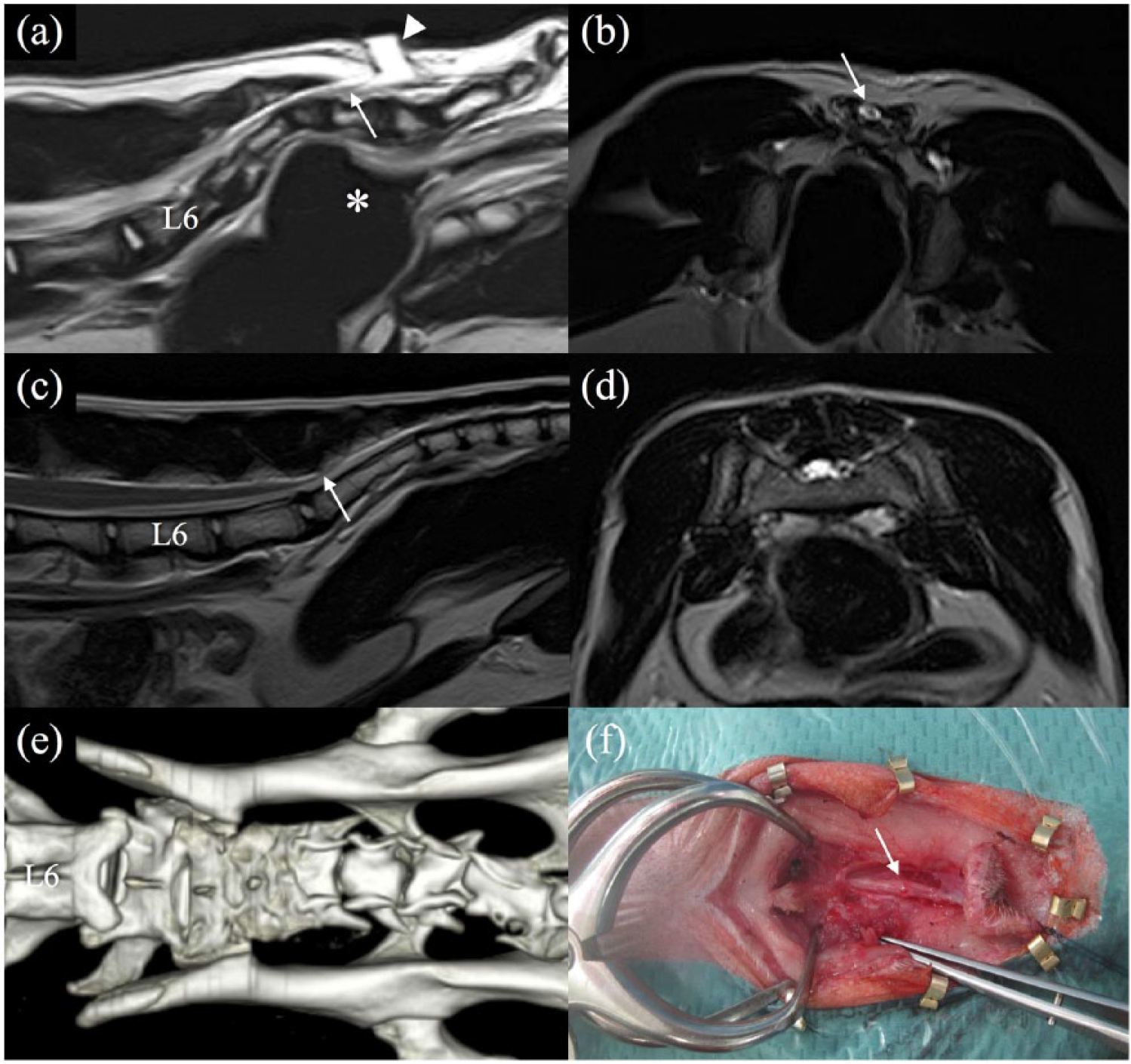
MRI, CT and intraoperative findings in case 1. (a) T2-weighted sagittal image of the lumbosacral region and (b) transverse image at the level of S3 on MRI. (a) The fistula was revealed to be hyperintense (arrowhead) and tethered the conus medullaris up to the caudal aspect of S3 (arrow). Rectal narrowing was secondary to ventral displacement of the caudal vertebra (asterisk). (b) The conus medullaris presented possible caudal displacement (arrow). (c) T2-weighted sagittal image of the lumbosacral region and (d) transverse image at the level of S1 in a 1.5-year-old male domestic shorthair cat without spinal issues. (c) The conus medullaris is the terminus of the caudal aspect of L7-S1 (arrow). (d) The dural sac does not contain the conus medullaris at the cranial aspect of S1. (e) A three-dimensional reconstruction dorsal image of the lumbosacral region on CT revealed the lack of the dorsal arches of S3, Cd1 and Cd2 vertebrae. (f) The fistula was connected to the dural sac and caused caudal traction of the conus medullaris after incision of the meninges
The excision of caudal SBA and caudectomy were performed under general anesthesia to release the tethering of the spinal cord and resolve rectal narrowing secondary to ventral displacement of the caudal vertebra. After premedication with intravenous atropine (Atropine; Tanabe) (0.05 mg/kg) and midazolam (Midazolam; Astellas) (0.2 mg/kg), general anaesthesia was induced with propofol (Propoflo 28; DS Pharma Animal Heath) (5 mg/kg, to effect IV) and maintained with isoflurane in oxygen. Fentanyl (Fentanyl; Daiichi Sankyo) (loading dose of 5 µg/kg IV followed by continuous rate infusion at 5–10 µg/kg/h IV) was administered as intraoperative analgesia. Dorsal lumbosacral laminectomy from L7 to S3 was then performed. When the vertebral canal was opened, the fistula communicating with the dural sac on the conus medullaris was observed. After the incision of the meninges, the fistula caused caudodorsal displacement and traction of the conus medullaris (Figure 1f). The fistula was completely removed, and the spinal cord was untethered. Dura regeneration matrix (expanded polytetrafluoroethylene [ePTFE] soft tissue patch; GORE-TEX) was placed over the defect of the dura prior to standard closure. The vertebra caudal to S3 was resected in order to release rectal narrowing secondary to ventral displacement. The cat was discharged on the eighth postoperative day.
A 1 month postoperative examination showed improvements in paraparesis and constipation symptoms. A follow-up MRI revealed the release of the conus medullaris tethering and an improvement in rectal narrowing (Figure 2a). Urinary incontinence remained partially unresolved. The histopathological examination showed that the resected fistula was consistent with a segment of the spinal cord parenchyma that contained remnants of the central canal lined by cuboidal-to-columnar cells, resembling ependymal cells. This parenchyma was surrounded by a thin layer of fibrous tissue resembling the meninges. Multiple small capillaries, glial cells and occasional neurons were observed within the spinal cord parenchyma. Based on these findings, the lesion was diagnosed as a meningomyelocele (Figure 2b).

(a) Follow-up MRI 1 month after surgery in case 1. The conus medullaris is observed to release the tethering and terminates at the caudal aspect of L7 (arrow). Rectal narrowing is also improved (asterisk). (b) Histopathology; excised tissues showed a segment of a neuropil in the dermis. The space within the spinal cord parenchyma (asterisk), including multiple small capillaries, glial cells and occasional neurons, is the central canal lined by ependymal cells. Haematoxylin and eosin staining
Case 2 was a 10-month-old castrated male domestic shorthair cat referred to the veterinary referral hospital for a 6 month history of progressive paraparesis and an over-reaching pelvic limb gait. A physical examination revealed a palpable dermoid fistula at the level of T5. A neurological examination showed proprioceptive deficits and upper motor neuron signs in the hindlimbs, which suggested a T3–L3 spinal cord lesion. The palpable dermoid fistula showed hypointensity on T2WI and T1WI, and was detected as a tubular structure connected to the dural sac on the thoracic spinal cord. The tubular structure caused dorsal traction of the spinal cord. CT revealed a dorsal lamina defect between the spinous processes of T5 and T6 (Figure 3a,b). Dorsally retracted TCS secondary to SBA was suspected.

MRI, CT and intraoperative findings in case 2. (a) T2-weighted sagittal image on MRI. The dermoid fistula was hypointense and detected as a tubular structure (arrowheads), which was connected to the dural sac, and the tethered spinal cord was retracted dorsally (arrow). (b) A three-dimensional reconstruction dorsal image on CT revealed a dorsal laminar defect between the spinous processes of T5 and T6. (c) The tubular structure was connected to the dural sac (arrow)
The excision of SBA was performed to untether the spinal cord under general anaesthesia. The same perioperative analgesics as case 1 were administered. The cat was positioned in sternal recumbency and standard dorsal laminectomy of T5 and T6 were performed. The tubular structure connected to the dural sac was observed (Figure 3c). After the incision of the meninges, the tubular structure was completely removed and the spinal cord was untethered. The meninges were then sutured (CV-6 ePTFE Suture; GORE-TEX). A postoperative MRI examination confirmed the removal of the tubular structure and untethering of the spinal cord (Figure 4a). The cat was discharged 7 days postoperatively.

Case 2. (a) A postoperative MRI examination revealed the removal of the tubular structure and release from tethering of the spinal cord. (b) Gross and (c,d) histopathological findings. A histopathological evaluation showed meningeal-like cells surrounded by well-differentiated collagen fibres. (c) Haematoxylin and eosin staining. (d) Immunohistochemically, these meningeal-like cells were positive for anti-E-cadherin
In 1 week and 1 month follow-up neurological examinations the cat showed continuous improvements; however, the over-reaching pelvic limb gait persisted. In the 12 month follow-up, other than pelvic limb ataxia, the cat was able to jump and run freely. Histologically, the tubular structure revealed spindle cells arranged in interlacing fascicles (meningeal-like cells) surrounded by well-differentiated collagen fibres. Immunohistochemically, these meningeal-like cells were positive for E-cadherin. The tubular structure did not contain any neurons and/or glial cells. This confirmed the diagnosis of a meningocele (Figure 4b–d).
Discussion
SBA has rarely been diagnosed in veterinary medicine, and is mainly reported in lumbo-sacral spinal cord segments.6–8 Teratogenic, nutritional and environmental factors have been suggested to contribute to the occurrence of SBA.9,10 In cats, the experimental administration of griseofulvin during pregnancy and other toxic compounds, such as methylmercury and ethylenethiourea, induced SBA in kittens. 11 Manx cats are known for congenital abnormalities in the lower spine and spinal cord. The autosomal dominant trait in Manx cats results not only in the absence of a tail, but also in the presence of severe malformations, including sacrocaudal meningocele, meningomyelocele and myelodysplasia.6,12,13 SBA in dogs may also have a breed predisposition; the incidence of spina bifida is higher in English Bulldogs than in any other canine breeds. 5 In humans, the prevalence of SBA is 3.2 per 10,000 live births. 14 The aetiology of SBA in humans currently remains unclear, but may be related to various factors including maternal nutrition, environment and genetics. For example, folic acid supplementation during pregnancy prevents spina bifida.15–17
In avian and mammalian embryos, neurulation occurs along the axis through two consecutive processes involving radically different morphogenetic events and is referred to as primary and secondary neurulation, respectively. The primary neural tube forms rostrally by rolling, folding or bending of the neural plate, which is a flat epithelial sheet. However, the secondary neural tube is generated caudally by elongation, epithelialization and cavitation of the tail bud, a rod-shaped mesenchyme. 18 Spina bifida occurs as a result of a disturbance in neurulation. Spina bifida has been classified as SBA, in which neural tissue is exposed, and ‘spina bifida occulta (SBO)’, in which neural tissue is covered by tissue. SBO includes various closed spinal defects such as diplomyelia, dorsal dermal sinus and spinal lipoma, and is caused by defects in secondary neurulation. By contrast, SBA such as that in cases 1 and 2 is caused by a failure in primary neurulation. 19
TCS in humans commonly occurs in the growing phase secondary to spina bifida. 3 In veterinary medicine, TCS has been reported in conjunction with intradural lipoma, meningomyeloceles, spina bifida, myeloschisis and split cord malformation.4–7,20,21 In humans, the early symptoms of TCS include ataxia in the lower extremities, muscle weakness and vesicorectal failure (including urinary urgency and urinary and faecal incontinence). 22 TCS in case 1 occurred in the lumbosacral region and appeared with similar clinical signs as those in humans. Urinary incontinence and dyschezia in case 1 were assumed to be caused by damage to the pudendal nerve, which controls the external urethral sphincter and external anal sphincter, and pelvic and/or autonomic nerves, which control rectal wall function. Alternatively, ventral displacement of the caudal vertebra may contribute to dyschezia. However, TCS in the thoracic region in case 2 is extremely rare. The fasciculus gracilis in the thoracolumbar spine contains neurons that mainly transmit information on general proprioception travelling to the somatosensory cortex. Damage to the fasciculus gracilis may have caused the over-reaching pelvic gait observed in case 2.
Four case reports have documented the surgical treatment of TCS in the lumbar-sacral region in a cat and dogs.5–7,20 All four cases had a history of paraparesis, and 2/4 cases had dysuria and dyschezia. All cases with a history of paraparesis only improved postoperatively, whereas none with dysuria and dyschezia improved completely. The outcomes in these reports were similar to that of case 1. In humans, Garcés-Ambrossi et al reported that the symptoms of TCS that improved after surgical treatment were painful dysesthesias (79%), lower extremity weakness (69%) and urinary symptoms (47%). 23 This study concluded that the rates of improvements in pain resolution and motor symptoms were greater than that in urinary symptoms. Kondo et al found that patients with sensory motor paralysis of the bladder or symptoms with a duration of more than 3 years did not benefit from surgery. 24 In cats, as with the symptoms reported in humans, improvements in sensory motor paralysis of the bladder may be more difficult to achieve than those in paraparesis. Only one surgical case of TCS in the thoracic region in a cat was reported prior to our case 2. 4 Both cases showed similar preoperative clinical signs and postoperative improvements. We suggest that paraparesis caused by TCS improves regardless of the location of the lesion or direction of cord traction if the spinal cord is properly released surgically.
Conclusions
This case report presents the imaging characteristics, surgical treatments and outcomes of two different types of TCS with SBA in cats. There have been few reports of TCS with SBA in cats, and it needs to be included in the differential diagnosis of slowly progressive paraparesis in younger cats with or without vesicorectal failure and a palpable dermoid fistula. Its diagnosis is challenging; however, proper surgical treatment is key for an optimal outcome. TCS with SBA in cats may be successfully treated surgically if paraparesis is the only neurological sign. Functional improvements may not be significant if vesicorectal failure presents simultaneously.
Footnotes
Acknowledgements
We acknowledge the contributions of Daisuke Hasegawa for his useful discussions and Lauren Rockey for her English proofreading of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
