Abstract
Case summary
A feral domestic shorthair cat was euthanized owing to acute onset and progression of neurological signs attributed to ethylene glycol toxicity. At post-mortem examination two nodules were identified within the fundus of the stomach. Examination of the gastric nodules revealed an intact mucosal surface, each with multiple red slender nematodes extending through an individual central pore. Histopathological evaluation of the nodules highlighted unique reactive fibroplasia, mimicking feline gastrointestinal eosinophilic sclerosing fibroplasia (FGESF), encasing numerous nematodes with females possessing gravid uteri containing abundant larvated eggs. The latter findings were highly suggestive of the Cylicospirura genus, further supported by an en face evaluation of the buccal cavity, highlighting a distinctive trifid tooth appearance. Together, these findings are consistent with Cylicospirura felineus. PCR for the COX-1 gene was unsuccessful on formalin-fixed specimens, attributed to nucleic acid and protein crosslinking.
Relevance and novel information
This represents the first documented case of Cylicospirura species in a feral domestic shorthair cat in North America. This particular cat lived in the highly urban environment of New Orleans, Louisiana. Identification of this case demonstrates the potential for feral cats to serve as reservoir hosts and ultimately support transmission of Cylicospirura species into domesticated cat populations. Gastric cylicospiruriasis may present clinically as a firm abdominal mass, potentially with a history of chronic vomiting. The latter emphasizes the importance of differentiating this condition from a neoplastic process such as alimentary lymphoma and adenocarcinoma. Histologically, the unique thick anastomosing collagenous cords encasing nematodes represent a stereotypical response observed in a broad array of gastrointestinal inflammation in felines, including intralesional bacteria, fungal hyphae, foreign bodies and, in this case, gastric nematodes that closely resemble FGESF. Additionally, these unique histological lesions have previously been misinterpreted as neoplastic conditions, including sclerosing mast cell tumor and extraosseous osteosarcoma.
Introduction
Cylicospirura is within the family Spiruroidea, which also includes Physaloptera species, Spirocerca lupi and Cyathospirura species. These all are believed to have arthropod intermediate hosts with thick-shelled larvated eggs or a paratenic vertebrate host. Owing to its unique histologic appearance, Eckstrand et al proposed the term verminous alimentary sclerosing fibroplasia (VASF) for gastrointestinal cylicospiruriasis. 1 Unlike lupi, Cylicospirura species nodules have not been shown to progress to neoplasms. In this particular case an adult neutered feral domestic shorthair feline of unknown age was humanely euthanized owing to the acute and progressive neurologic signs ultimately attributed to ethylene glycol toxicity, with gastric cylicospiruriasis representing an incidental finding. However, in wild felids, chronic vomiting, weight loss and intestinal perforation have been attributed to Cylicospirura species infection. 2
Currently, no data have been published with regard to available efficacious anthelmintic treatments for Cylicospirura species. The unique stromal reaction surrounding the gastric nematodes resembles feline gastrointestinal eosinophilic sclerosing fibroplasia (FGESF), which, owing to the presence of large numbers of mast cells and the sclerotic appearance of collagen trabeculae, may be misdiagnosed as sclerosing mast cell tumors or extraosseous osteosarcoma, respectively. In contrast to our case, FGESF typically occurs as an ulcerated intramural mass within the pyloric sphincter, ileocecocolic junction or colon, with occasional regional lymph node involvement and common intralesional bacteria (56% of overall cases, n = 25), 3 or fungal hyphae associated with the characteristic collagenous cords, although bacteria were concurrently present in this lesion. 4 Interestingly, one case report of FGESF mimicked a metastatic neoplasm with multiple nodules involving the pylorus, colon, multiple mesenteric and mediastinal lymph nodes. 5 FGESF cases typically fail to respond to antibiotic treatment; however, the longest survival curves are seen in animals receiving corticosteroids or surgical excision. 3 Eosinophils require leukotriene D4 for initiation of transforming growth factor-β production, a key fibrinogenic mediator, supporting the role of corticosteroids in medical management of these cases by inhibiting lipoxygenase and subsequent leukotriene production. Similar histologic features characterized by an anastomosing fibrous network observed with both VASF and FGESF have also been observed in cougars (Puma concolor) with intralesional intestinal porcupine quills.
Case description
At post-mortem examination, the cat was in adequate body condition. Large numbers of fleas (Ctenocephalides felis) were detected on external examination. The fundus of the stomach contained two raised, well-circumscribed nodules measuring 2.5 × 1.5 × 1 cm and 2 × 2 × 1 cm, respectively (Figure 1), each with a central mucosal pore containing several red slender nematodes, approximately 2–3 cm in length. On cut surface of the nodules, the wall of the stomach was transmurally expanded by a cavernous network of white-to-gray fibrous connective tissue surrounding numerous slender red nematodes (Figure 2). The renal cortices displayed punctate fluorescence with the use of a Wood’s lamp, but were normal in size and cortical contour. Tissues were routinely fixed in neutral-buffered 10% formalin, trimmed, embedded in paraffin and sectioned for microscopic examination.

Post-mortem photograph of one of the nodules present in the fundus of the stomach; the raised nodule contains a central mucosal pore with multiple slender red nematodes extending onto the mucosal surface of the stomach. The inset highlights a larvated egg, observed upon fecal float examination; mean measurements of 10 eggs were 40 × 30 µm
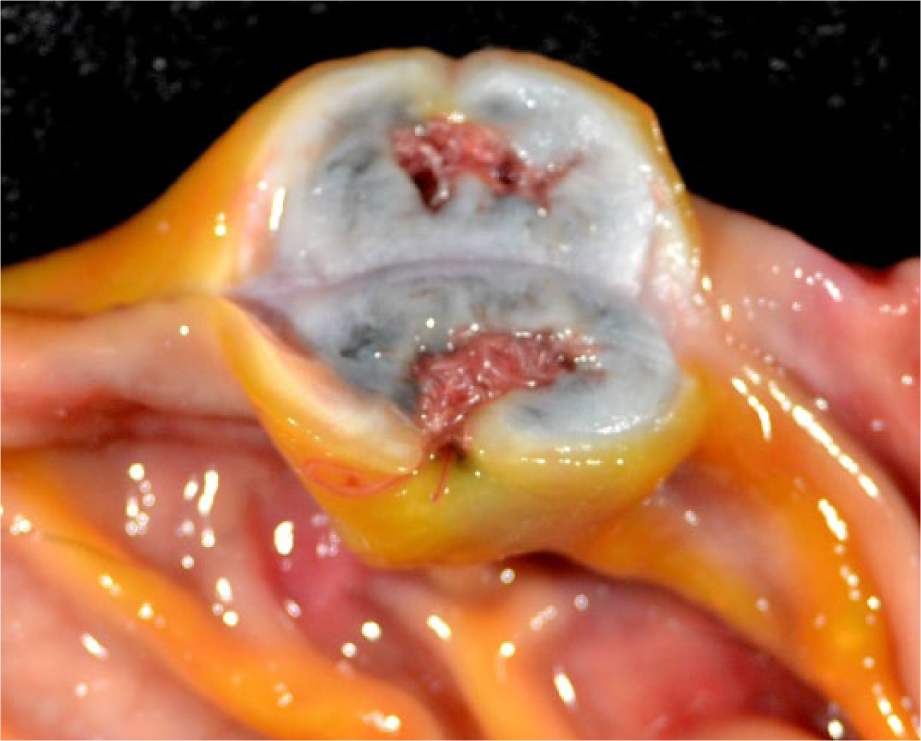
Post-mortem cross-sectional photograph of one of the two stomach nodules displaying mural expansion of the submucosa by a labyrinth of white-to-gray fibrous connective tissue surrounding numerous red nematodes
Nodules within the gastric fundus were char-acterized by marked expansion of the submucosa by thick anastomosing cords of dense sclerotic collagen interlaced by loose aggregates of spindle cells, moderate numbers of lymphocytes and plasma cells, with lesser numbers of eosinophils and mast cells. The underlying muscularis and serosa were otherwise unaffected. Within the confines of the dense connective tissue were numerous transverse and longitudinal sections of adult nematodes measuring approximately 250–450 µm in diameter, with an overlying mucosal pore. Nematodes contained an 8-µm-thick eosinophilic cuticle with fine serrated ridges, prominent compartmentalized lateral cords, coelomyarian musculature and a pseudocoelom containing amorphous basophilic material and occasional foci of yellow-to-brown granular pigment. The intestinal tract was lined by uninucleate columnar epithelium with long, fine apical projections.
The vast majority of nematodes were female containing gravid uteri with large numbers of larvated eggs (Figure 3, inset). Wet mount microscopic examination of a dissected gravid female identified larvated eggs measuring 40 × 30 µm (mean measurement of 10 eggs). For identification, nematodes were mounted onto glass slides for en face examination of longitudinal ribs, identifying terminal trifid projections (Figure 4).

Photomicrograph of the gastric parasitic nodule displayed in Figures 1 and 2. Numerous transverse and longitudinal sections of nematodes (indicated by arrows) are surrounded by concentric anastomosing dense fibrous bands of sclerotic collagen (indicated by stars). The inset highlights nematodes containing a thick eosinophilic cuticle and paired gravid uteri containing numerous larvated eggs (hematoxylin and eosin)

Photomicrograph of the buccal cavity en face displays a distinctive trifid appearance of the anterior segments of longitudinal ribs resembling a crown (bar = 50 μm)
Renal tubules, most often within the cortex, contained abundant translucent, pale yellow, anisotropic, birefringent crystals arranged in sheaves and prisms (calcium oxalate). Tubular epithelial cells were frequently degenerate or necrotic, as evidenced by abundant vacuolated eosinophilic cytoplasm or hypereosinophilia and shrinkage with nuclear pyknosis, respectively. These renal changes were consistent with ethylene glycol toxicity.
Discussion
To the best of our knowledge this is the first case report of Cylicospirura species in a feral domestic shorthair cat (Felis catus) within North America, although this organism is frequently observed in multiple North American wild felids, including the bobcat (Lynx rufus), lynx (Lynx lynx) and cougar (P concolor). 2 This species has been previously reported from domestic cats in Asia, Africa and Australia.4–8 Other species infecting domestic cats include Cylicospirura subaequalis and Cylicospirura (Petrowospirura) barusi in Asia, Cylicospirura heydoni in Australia, Cylicospirura advena in New Zealand and an unidentified Cylicospirura species in Italy.9–12
Species can be differentiated based on morphological differences of tooth morphology within the buccal cavity, location of the vulva in comparison with the esophageal intestinal junction or DNA sequence of the mitochondrial cytochrome c oxidase subunit 1 gene (cox1). In this particular case, specimens were identified as Cylicospirura felineus based on the distinctive trifid morphology of the anterior portion of the longitudinal ribs (Figure 4). There are three other species with distinct trifid dentation: Cylicospirura crocutae from hyenas in Africa, Cylicospirura pardalis from leopards in Africa and Cylicospirura skrjabini from foxes in Siberia. The shape of the cusps in teeth of C skrajabini are conical, whereas those in C pardalis are claw-like in appearance. 7 Both C crocutae and C felineus have rounded cusps, but C felineus has a 50–70% shorter esophagus than C crocutae. 13 We previously distinguished C felineus from C subaequalis using differences in cox1. 2 We attempted multiple times to obtain the cox1 sequence from the several of the worms in the present study using the same primer sequences that amplify approximately 700 base pairs, but were unsuccessful. Nevertheless, given the worm morphology, host and geographic location, we conclude that the infection presented here was caused by C felineus.
Conclusions
This case represents the first documented report of Cylicospirura species in a feral domestic shorthair cat in North America. This animal originated from a highly urban environment in New Orleans, Louisiana, where feral cats flourish. Although a single isolated case such as this cannot definitely support an established burden of gastric cylicospiruriasis in feral cats, animals such as this could serve as a reservoir for spillover into domestic cats, stressing the importance of monitoring feral cat populations. The lack of previous identification in domesticated cats within North America may suggest decreased consumption of intermediate and/or paratenic hosts. Additionally, standard anthelmintic medications frequently administered to domesticated cats could potentially be highly efficacious against Cylicospirura species. These findings continue to support a predictable fibrosing process within the gastrointestinal tract of felines in response to a variety of foreign objects, including nematodes, bacteria, fungal hyphae and foreign bodies (porcupine needles). These unique stromal changes may be misdiagnosed as sclerosing mast cell tumors or extraosseous osteosarcomas. Although uncommon, Cylicospirura species can result in chronic vomiting when associated with the pyloric outflow tract.
Footnotes
Funding
The authors received no specific grant from any funding agency in the public, commercial or not-for-profit sectors for the preparation of this case report.
Conflict of interest
The authors do not have any potential conflicts of interest to declare.
