Abstract
Background:
Diabetes mellitus is increasingly common among deceased donors and may signal donor-derived kidney injury that affects post-transplant outcomes.
Objective:
To evaluate whether donor proteinuria is associated with graft and patient outcomes after kidney transplantation from deceased donors with diabetes.
Design:
Retrospective cohort study using a national transplant registry.
Setting:
United States; Scientific Registry of Transplant Recipients (SRTR), February 28, 2013–2023.
Patients:
9486 kidney-alone transplant recipients from deceased donors with diabetes in whom a pre-implantation (procurement) biopsy was performed and donor proteinuria status was available.
Measurements:
Primary outcome: death-censored graft failure (DCGF). Secondary outcomes: all-cause graft failure (ACGF), death with graft function (DWGF), and delayed graft function (DGF). Exposure: donor proteinuria (present vs absent).
Methods:
Kaplan-Meier analyses and multivariable Cox models (a priori covariables from a directed acyclic graph) assessed associations between donor proteinuria and time-to-event outcomes. Because proportional hazards were violated for DCGF, analyses were performed in two periods: an “early” cohort up to 2.5 years post-transplant and a landmarked cohort of recipients with functioning grafts at 2.5 years. Logistic regression evaluated DGF. Sensitivity analyses adjusted for donor insulin dependence (proxy for diabetes severity) and recipient characteristics; exploratory effect modification by biopsy glomerulosclerosis (GS) was assessed.
Results:
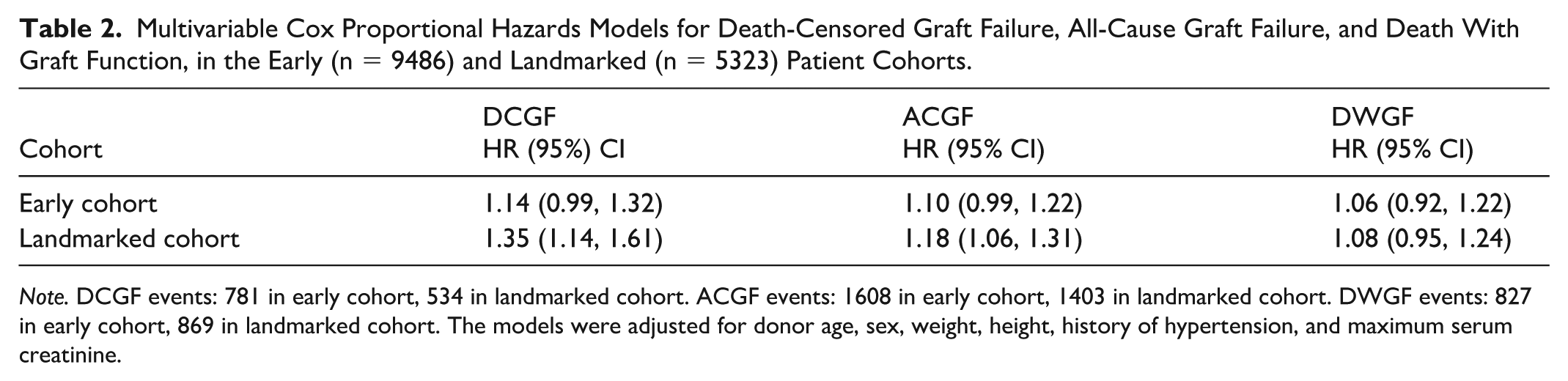
Donor proteinuria was present in 54.9% of cases. In adjusted Cox models, donor proteinuria was not associated with early DCGF (<2.5 years; HR 1.14, 95% CI: 0.99, 1.32) but was associated with increased risk of late DCGF >2.5 years post-transplant (HR 1.36, 95% CI: 1.15, 1.62), with similar findings for ACGF. No associations were observed with DWGF or DGF. Results were consistent after adjustment for donor insulin dependence as a proxy for severity and recipient factors including diabetes status. The association between proteinuria and late graft failure was more pronounced in kidneys with lower GS, suggesting proteinuria may reflect chronic injury not well-captured by biopsy.
Limitations:
Observational design with potential residual confounding. Because the cohort includes only kidneys that were actually transplanted, findings reflect outcomes among accepted organs and are not intended to guide offer acceptance or decline decisions. Donor proteinuria was recorded only as present or absent, without standardized measurement. This may have led to misclassification, prevented assessment of dose-response relationships, and likely made it harder to detect true associations. Registry constraints limited histologic detail beyond GS.
Conclusions:
Among diabetic deceased donors, the presence of proteinuria is a time-dependent marker of increased long-term graft-failure risk, complementing biopsy and clinical data. Standardized, quantitative proteinuria assessment may improve risk stratification and post-transplant management while supporting judicious utilization of diabetic donor kidneys.
Introduction
The rising prevalence of diabetes mellitus (DM) in the general population is mirrored in potential deceased organ donors. 1 A history of DM was reported in up to eight percent of deceased donors.2-5 Diabetic nephropathy (DN), a leading cause of kidney failure, occurs in 10-30% of patients with DM and is frequently underdiagnosed, even in advanced stages, due to clinical-histologic discordance.6,7 Few small cohort studies and case series of deceased donor kidney biopsies report variable prevalence (up to 25%) and severity of DN among kidneys ultimately transplanted, often poorly correlated with glycemic control or DM duration.5,8-11 Non-glomerular chronic injury is also frequently observed and linked to worse allograft function and survival. 8 The implications of donor DN are further complicated by interactions with recipient factors and the post-transplant course influencing the evolution of these lesions, with limited studies documenting histologic trajectories over follow-up.8-15
Kidneys from diabetic donors are inconsistently associated with worse early and late outcomes after transplantation, including higher serum creatinine and risks of delayed graft function (DGF), death-censored graft failure (DCGF), all-cause graft failure (ACGF), and mortality.3,4 Diabetes is weighted heavily as a risk factor for DCGF in the Kidney Donor Risk Index (KDRI). 16 Adverse outcomes appear to be especially pronounced with higher DN class, longer DM duration, and extended criteria donor (ECD) status.2,3,8,17 Although recipients with diabetes face higher risks of graft failure and mortality, there still appears to be a greater survival benefit receiving a diabetic donor kidney when compared to remaining on the waitlist, particularly for older recipients with longer anticipated wait times.4,10,18 Despite potential survival benefits for recipients, kidneys from diabetic donors have twofold higher odds of being discarded. 19 In 2022, as many as 52% of procured diabetic donor kidneys were ultimately discarded (vs. 21% for non-diabetic donors), with organ or donor quality concerns accounting for over 92% of declined kidney offers in the United States.20,21
Recognizing these challenges, efforts to optimize the use of diabetic donor kidneys have emphasized improved risk stratification through pre-implantation biopsies and the prudent selection of recipients most likely to yield the greatest benefit. 22 Current US guidelines recommend biopsies for donors with DM, yet practices vary widely in biopsy technique, interpretation, and reporting, leading to inconsistent association between biopsy scores and clinical outcomes.23,24 Considering these shortcomings of biopsies, clinical markers of kidney function in diabetic donors may offer additional insights into assessment of donor kidney quality. When present, elevated serum creatinine and proteinuria are associated with accelerated decline in kidney function.25-27 However, no composite scores have integrated donor DM status alongside biopsy findings and clinical characteristics. There is the need for a more nuanced approach to diabetic donor kidney evaluation which may permit increased utilization of these kidneys while optimizing post-transplant outcomes. We sought to explore the association of proteinuria status among donors with DM who had procurement biopsies, on post-transplant outcomes of graft and patient survival in a large US registry cohort of kidney-alone transplant recipients from 2013 to 2023.
Methods
Study Design and Population
This cohort study consisted of all consecutive deceased donor kidney-alone transplant recipients performed in the United States from 28th February 2013 to 2023 where there was a donor history of diabetes mellitus (DM). This study used data from the Scientific Registry of Transplant Recipients (SRTR). This study used data from the Scientific Registry of Transplant Recipients (SRTR). The SRTR data system includes data on all donor, wait-listed candidates, and transplant recipients in the US, submitted by the members of the Organ Procurement and Transplantation Network (OPTN). The Health Resources and Services Administration (HRSA), U.S. Department of Health and Human Services provides oversight to the activities of the OPTN and SRTR contractors. Data is mandatorily reported to SRTR by transplant centers at time of donation and transplantation, and with post-transplant follow-up records at six months, one year, and then annually, until the recipient is re-transplanted, dies, or is lost to follow-up.
Donor history of DM was captured at the time of donation and did not distinguish between type 1 or type 2 DM. Recipients were excluded if there was no available kidney biopsy report done from time of kidney procurement until implantation, and if there was no donor proteinuria testing result available.
Research Ethics Board approval was obtained from the University Health Network and this study was approved by the SRTR.
Outcomes
The primary outcome of interest was time to death-censored graft failure (DCGF), with patients censored at last follow-up or death with a functioning graft. Secondary outcomes were: (i) time to all-cause graft failure (ACGF), (ii) time to death with graft function (DWGF), and (iii) delayed graft function (DGF). DGF was defined as the need for dialysis within the first week after transplantation.
Donor Proteinuria Exposure
Donor proteinuria was classified as a binary exposure, present or not present, as captured in the SRTR dataset and reported by transplant centers. Proteinuria measurement methods were not available.
Statistical Analysis
Analyses were conducted using R (Version 2023). Categorical variables were presented as frequencies and percentages, while continuous variables were presented as means with standard deviations (SD). The distribution of baseline demographics, grouped by proteinuria status, were compared using standardized mean differences (SMD), with SMD > 0.1 suggesting a notable difference between groups.
We utilized the Kaplan-Meier product limit method and log-rank statistic to graphically assess the time-to-event outcomes of DCGF, ACGF, and DWGF by donor proteinuria status.
Multivariable Cox proportional hazards models were used to assess the association between donor proteinuria status and DCGF while adjusting for potential confounders. Covariables included in the Cox models were selected a priori and their relationships were visualized in a directed acyclic graph (DAG). [Figure 1] Candidate covariables included the following donor characteristics: age, biologic sex, height, weight, history of hypertension, donation after cardiac death (DCD) status, stroke as cause of death, and peak serum creatinine prior to donation (in umol/L). If there was <10% missingness, the mechanism was assumed to be at random and multiple imputation with chained equations (MICE) was used (10 imputations); variables with >10% missingness were omitted from models. The proportional hazards assumption was tested with the Grambsch-Therneau test and visually inspected with Schoenfeld residual plots over time. After preliminary analyses, the proportionality assumption was violated in the model for DCGF. All survival analyses were subsequently done in two parts, informed by visual inspection of the survival curve: (i) the “early cohort” analyzed outcomes up to 2.5-years post-transplant, and otherwise censored at 2.5-years, followed by (ii) analysis of a “landmarked cohort” consisting of recipients with functioning allografts landmarked at 2.5-years post-transplant. Hazard ratios (HR) were presented with their associated 95% confidence intervals.

Directed acyclic graph (DAG) showing causal relationships of exposure, outcomes, and covariables.
A multivariable logistic regression model was used to assess donor proteinuria as a predictor of DGF and covariables selected using the DAG. Logistic regression models were tested with the Hosmer-Lemeshow test for goodness of fit, variance inflation factor (VIF) for multicollinearity, and Cook’s D for influential observations. Odds ratios (OR) and their associated 95% confidence intervals were presented.
A two-sided
Sensitivity analyses
Survival analyses were repeated in recipients whose donor’s insulin-dependence status was documented at transplant, with models adjusted for insulin dependence. Assuming adequate DM treatment in donors, insulin dependence was used as a proxy for DM severity.
To account for recipient factors influencing the decision to accept a donor kidney with a history of DM, models were adjusted for recipient factors including age, biologic sex, wait time on dialysis pre-transplant, any history of diabetes mellitus, history of hypertension, prior solid organ transplant, cold ischemic time, and number of Human Leukocyte Antigen (HLA) mismatches.
Subgroup analyses
Two-way interaction between exposure and selected covariables (biologic sex, DCD status, and recipient diabetes status) were tested and excluded from multivariable models if
We also conducted an exploratory analysis for effect measure modification by amount of glomerulosclerosis (GS) on donor kidney biopsy at time of transplant, on the effect of donor proteinuria and DCGF. Univariable Cox proportional hazards models for DCGF were conducted in dichotomized subgroups of recipients based on thresholds of 5%, 10%, and 20% GS among glomeruli sampled. The ratio of hazard ratios (RHR) was reported. Assessment of other histologic lesions (vascular, interstitial fibrosis and tubular atrophy (IFTA)) were precluded by high missingness in the dataset.
Results
Study Population
The cohort included 9486 kidney transplant recipients with 33 492 person-years of follow-up (median 3.0 years; IQR 1.0 to 5.0), including 5323 recipients in the 2.5-year landmarked cohort. [Figure 2] Mean recipient age was 59.3 ± 10.9 years and 63% were male.
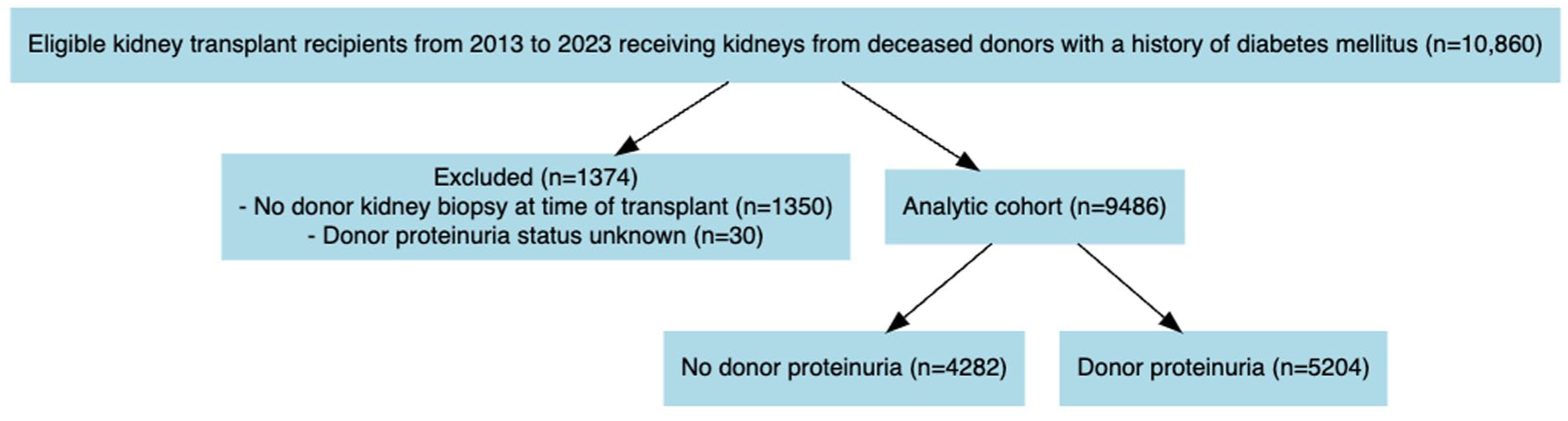
Patient flow in study.
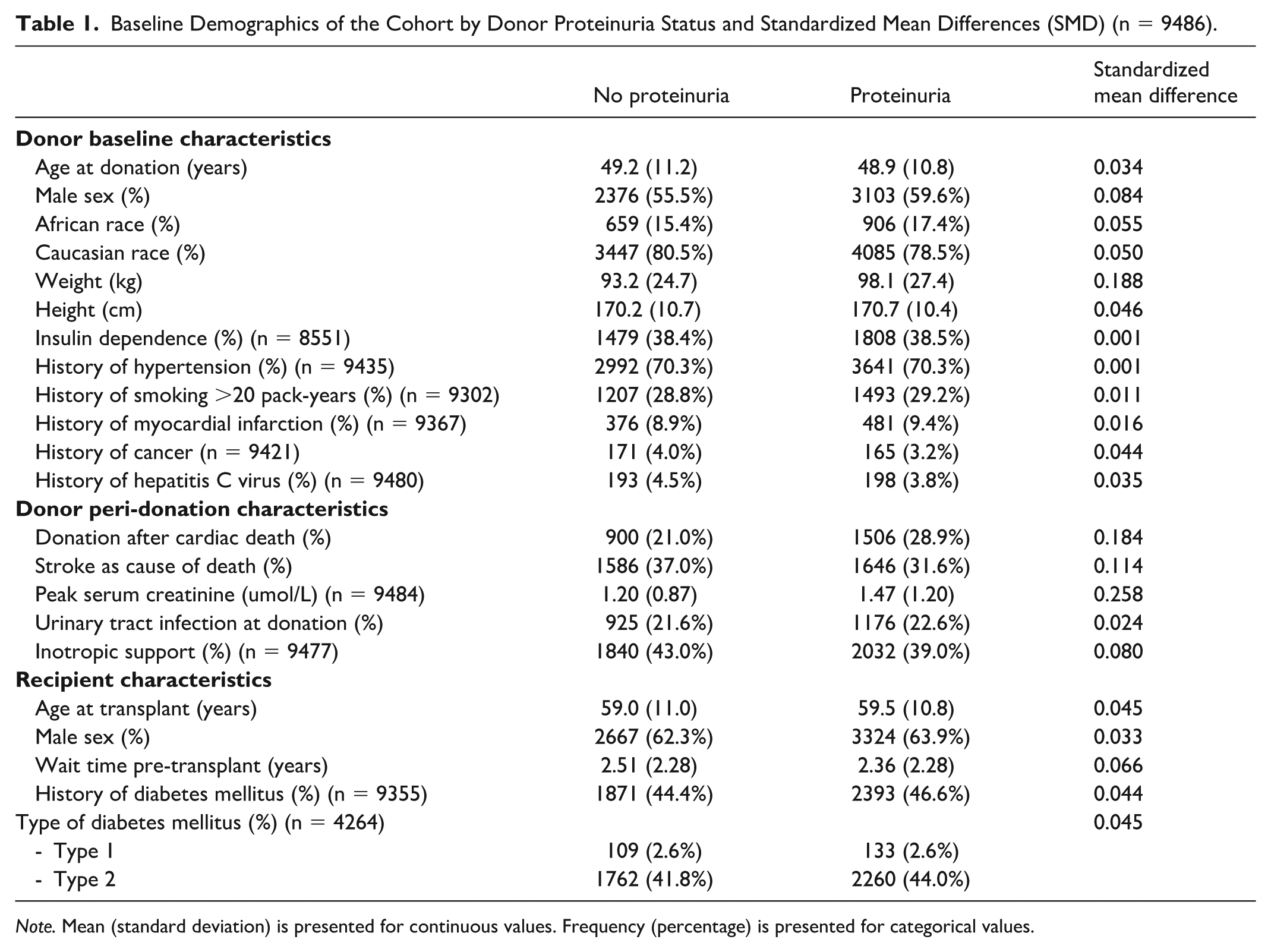
Donor proteinuria at time of transplant was present in 5204 (54.9%) of donors. Baseline demographics of the analyzed population grouped by donor proteinuria status are shown in Table 1. Donors with proteinuria were more likely to have higher peak serum creatinine (1.47 vs 1.20 umol/L), higher weight, and DCD status. There was no difference in donor insulin dependence (38%) or history of hypertension (70%), and there were no donors with diabetes duration known to exceed 10 years prior to death. Recipients were also balanced in age, sex, and diabetes status (~45%).
Baseline Demographics of the Cohort by Donor Proteinuria Status and Standardized Mean Differences (SMD) (n = 9486).
Death-Censored Graft Failure
There were 1315 DCGF events observed during the follow-up period, with 534 of these events occurring beyond 2.5 years post-transplant.
Donor proteinuria was associated with an increased risk of DCGF events in the landmarked cohort using an unadjusted Kaplan-Meier analysis (
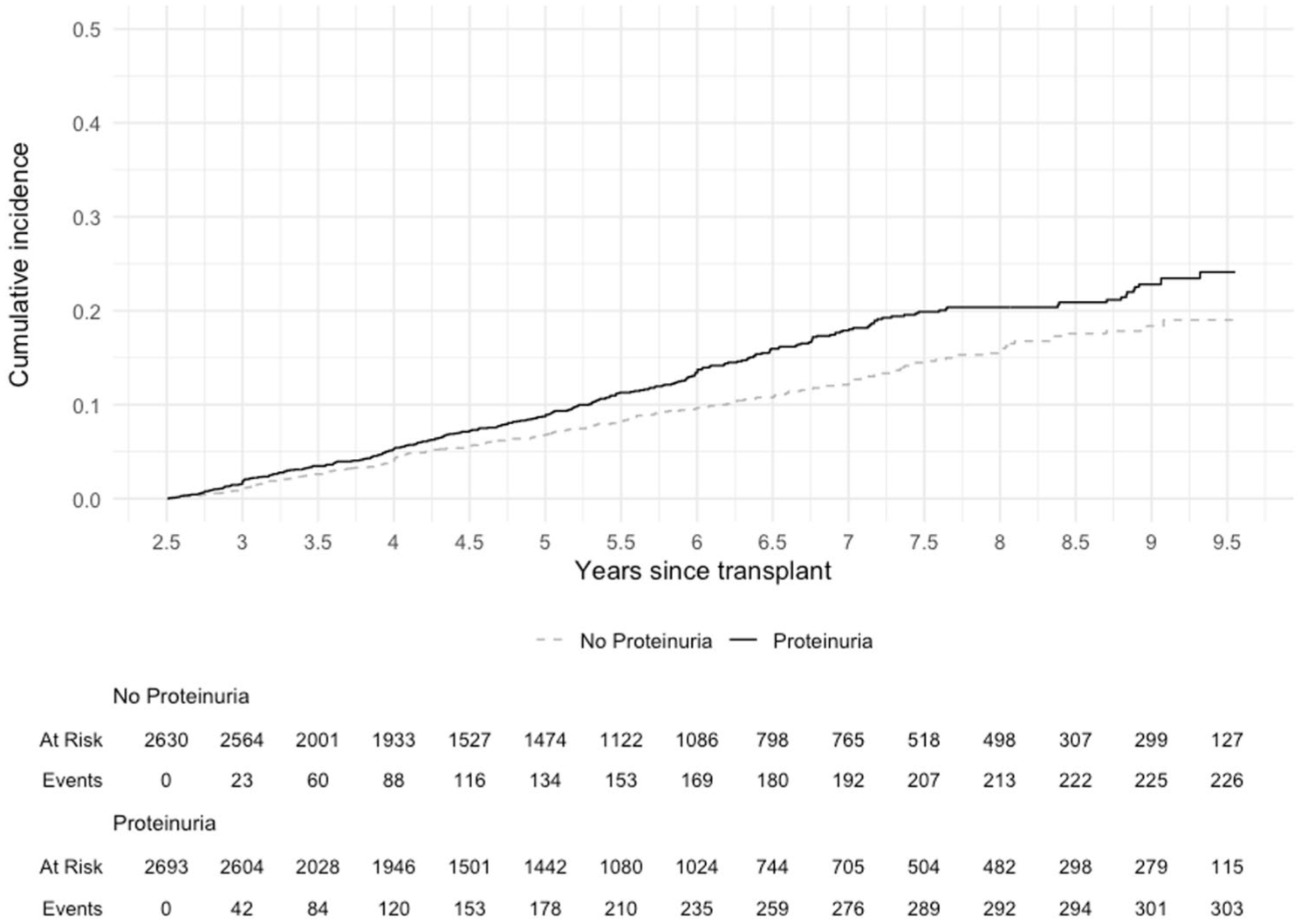
Cumulative incidence of death-censored graft failure in landmarked cohort of kidney transplant patients by donor proteinuria status (log rank
Multivariable Cox Proportional Hazards Models for Death-Censored Graft Failure, All-Cause Graft Failure, and Death With Graft Function, in the Early (n = 9486) and Landmarked (n = 5323) Patient Cohorts.
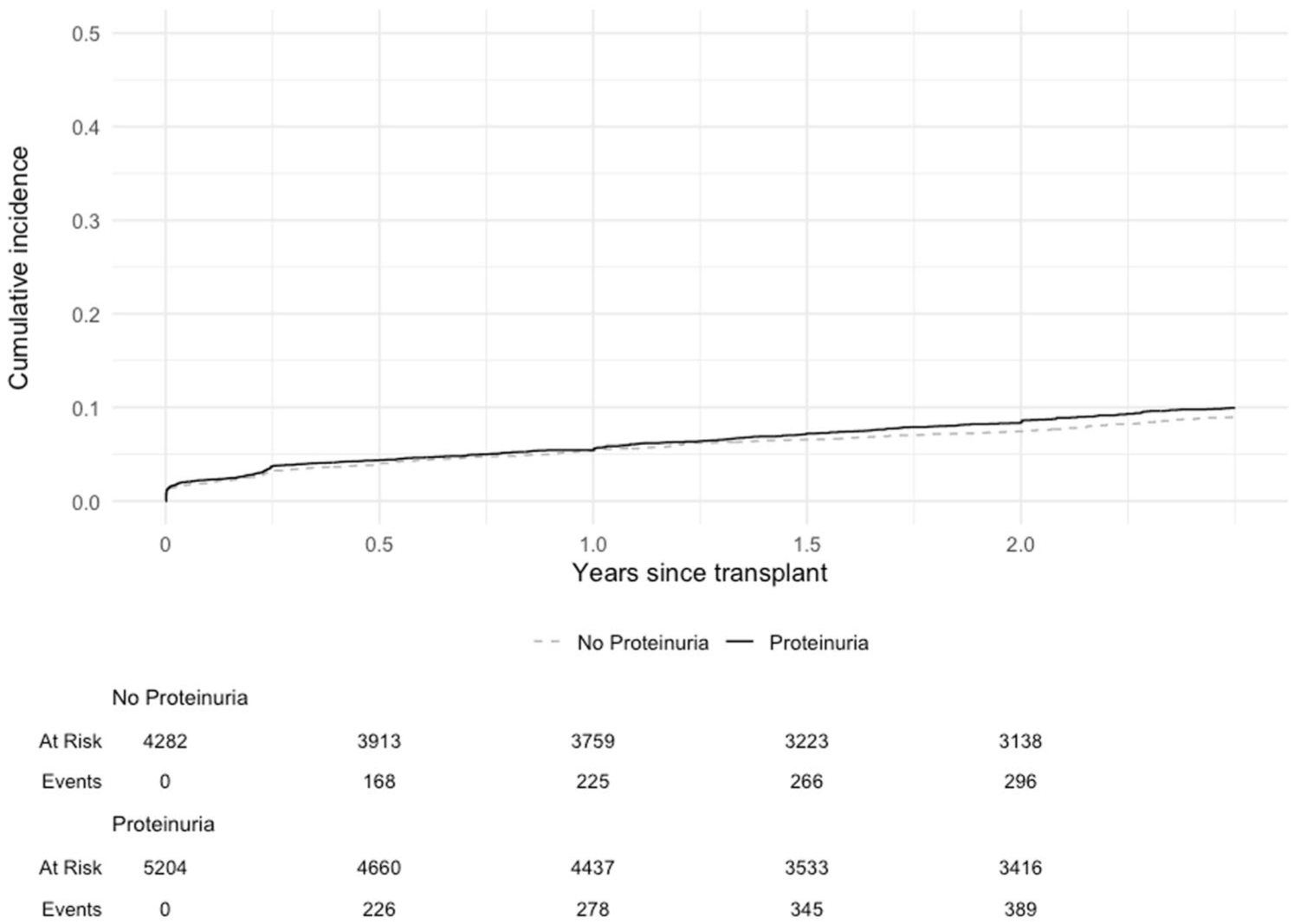
Cumulative incidence of death-censored graft failure in the early cohort of kidney transplant patients by donor proteinuria status (log rank
All-Cause Graft Failure
There were 3011 ACGF events including 1403 events in the 2.5-year landmarked cohort. Donor proteinuria was associated with an increased risk of ACGF beyond 2.5 years post-transplant (HR 1.18 [95% CI: 1.06, 1.31]), but not in the early cohort (HR 1.10 [95% CI: 0.99, 1.22]; Supplemental Table 1; Table 2).
Death With Graft Function
There were 1696 patients who died with graft function, with 869 deaths occurring more than 2.5 years post-transplant. Donor proteinuria was not associated with DWGF in the early (HR 1.06 [95% CI: 0.92, 1.22]) or landmarked (HR 1.08 [95% CI: 0.95, 1.24]) cohorts (Supplemental Table 1; Table 2).
Delayed Graft Function
DGF occurred in 33.2% (3339 of 9484) recipients and was not associated with donor proteinuria status (OR 1.08 [95% CI: 0.99, 1.18]).
Sensitivity Analyses
Survival analyses were repeated in the sub-cohort of 8551 recipients where donor history of insulin dependence was available at time of transplant; insulin dependence was reported in 38% of donors. The effect of donor proteinuria was similar to the primary analyses in a model adjusted for donor insulin dependence, and a model adjusted for recipient and donor-recipient factors (Supplemental Table 2). Insulin dependence was also independently associated with an increased risk of DCGF in both the early (HR 1.35 [95% CI: 1.16, 1.57]) and landmarked (HR 1.32 [95% CI: 1.10, 1.59]) cohorts, and ACGF in early (HR 1.19 [95% CI: 1.07, 1.33]) and landmarked (HR 1.23 [95% CI: 1.10, 1.38]) cohorts.
Primary analyses were unchanged by the addition of recipient characteristics as covariables in multivariable models (Supplemental Table 2).
Subgroup Analyses
There was no statistically significant interaction between donor proteinuria and biological sex, DCD status, or recipient diabetes status when recipient characteristics were included in the multivariable Cox models.
The effect of donor proteinuria on DCGF beyond 2.5 years post-transplant was more pronounced in recipients of donor kidneys with GS below versus above the threshold, and this was observed at all GS thresholds tested: RHR 1.35 at 5% GS, 1.45 at 10% GS, and 1.82 at 20% GS (Supplemental Table 3).
Discussion
In this large cohort of kidney transplant recipients from diabetic donors, we found that donor proteinuria was associated with worse death-censored and all-cause graft outcomes in the later post-transplant period beyond 2.5 years. The absence of significant associations with DGF or early graft failure suggests a time-dependent effect of proteinuria, potentially reflecting chronic injury pathways that progress over time. These findings were robust to adjustment for donor insulin dependence—a proxy for diabetes severity—and recipient characteristics, with no difference by recipient diabetes status.
The presence of proteinuria in diabetic kidney donors was associated with worse graft survival in the later post-transplant period. Although prior studies have largely overlooked the impact of donor proteinuria on post-transplant outcomes, a recent Swiss registry study of 1725 all-comer deceased donors reported proteinuria (urine protein-to-creatinine ratio >15 mg/mmol) in three quarters of donor candidates, but no association with 1-year mortality or graft function and survival.28,29 Our cohort, which was enriched for diabetic donors more likely to have clinically significant proteinuria, found that proteinuria was a marker of late graft failure even after adjusting for clinically relevant donor characteristics.
The delayed effect on graft survival may reflect the progression of early donor DN lesions. All donor kidneys had a pre-implantation biopsy, and severe histologic lesions linked to early graft failure would have likely precluded transplantation, selecting for kidneys with less advanced lesions that may evolve over time. While case reports documented resolution of donor DN lesions in non-diabetic recipients,12-14 other small observational studies found stability or progression within one year post-transplant irrespective of recipient diabetes status or glycemic exposure.8,10,11 However, these studies were limited by short-term follow-up and lack of correlation with clinical outcomes. Although we found no interaction with recipient diabetes status, used as a surrogate for glycemic exposure post-transplant, we could not account for glycemic control, post-transplant diabetes, or diabetes-related comorbidities. This underscores the need for studies evaluating the long-term evolution of donor DN lesions. Taken together, these findings suggest that proteinuria may serve as a surrogate for donor-derived kidney disease not captured by current biopsy practices, which progresses slowly but contributes significantly to late graft failure.
All donor kidneys in this study underwent pre-implantation biopsy, yet the association of proteinuria with late graft failure highlights limitations in biopsy utility for predicting long-term outcomes. Autopsy studies suggest that proteinuria is more strongly associated with kidney function and IFTA than with classic DN lesions. 6 Donor kidneys with severe glomerular or interstitial DN lesions, more closely linked to serum creatinine, may have been excluded from transplantation based on thresholds for creatinine or GS. Although GS >20% has been inconsistently linked to worse graft survival, GS remains a key factor in kidney discard.30-33 Notably, we observed that the association between proteinuria and late graft failure was stronger in kidneys with lower levels of GS. Proteinuria in this cohort may therefore reflect alternative pathways like early DN or chronic IFTA, which could be missed on pre-implantation biopsies due to sampling error and variability across observers or biopsy techniques, or may be overlooked in clinical decisions due to scarce guidelines and poor prognostic performance of histologic scoring systems.24,34-37 This would be consistent with our findings as early or scarring-related injuries could progress over time to cause late graft failure, and IFTA would not be modified by recipient diabetes status. While current OPTN guidelines recommend standardized biopsy reporting, including percent GS and IFTA, only GS is reliably captured in the SRTR registry and was available for analyses. 38 These inconsistencies emphasize the need for standardized biopsy protocols and comprehensive reporting frameworks that incorporate DN-specific features, such as diabetic glomerulopathy and IFTA, to enhance donor kidney evaluation and selection.
Our findings suggest that proteinuria is a helpful marker of diabetic donor kidney quality, complementing traditional metrics such as donor demographics and peak serum creatinine, even in a population where pre-implantation biopsies were performed. The observed association between proteinuria and late graft failure supports its routine assessment, but given that all kidneys in this study were ultimately transplanted, applying these findings without nuance could inadvertently increase organ discard. Whether proteinuria could help guide decisions about kidneys that were previously declined remains unknown. Instead, donor proteinuria assessment may offer an opportunity to tailor post-transplant care in high-risk recipients—for example, through more intensive monitoring, risk factor management, or earlier initiation of renin-angiotensin-aldosterone-system inhibitors. Current OPTN guidelines recommend biopsies for all diabetic donors, and proteinuria measurement could similarly be standardized. 23 However, this study alone cannot guide decisions regarding the best method for measuring proteinuria. Proteinuria was reported at the discretion of centers, with minimal missing data (Figure 1), but the specific measurement methods and thresholds used to define its presence were unknown. This binary classification of proteinuria limited our ability to evaluate dose-response effects, potentially underestimating the association with outcomes by including lower, less clinically relevant levels of proteinuria. While any proteinuria was significantly associated with worse outcomes, quantitative methods such as urine albumin-to-creatinine ratio (UACR) are preferred over qualitative approaches like urinalysis, aligning with best practices in nephrology. UACR is more sensitive for detecting clinically relevant proteinuria in DN and other kidney diseases, reflecting its utility in chronic kidney disease care. 27 However, dichotomized proteinuria status may allow generalizability of these findings to resource-limited jurisdictions where proteinuria quantification is unavailable. Future studies should explore the optimal number, timing, and thresholds for proteinuria tests, as well as the integration of standardized measurements into diabetic donor kidney evaluations. Addressing these gaps could enhance donor evaluation and improve long-term outcomes without compromising organ utilization.
This study provides valuable insights into the underexplored and underutilized population of diabetic donor kidneys, leveraging a large, contemporary national registry cohort spanning 10 years. Nearly all diabetic donors underwent pre-implantation biopsy, consistent with current practices and OPTN guidelines recommending biopsies for donors with diabetes or a single elevated HbA1c (≥6.5%). 23 A survey of 95 US transplant centers further highlighted that 51% consider donor diabetes, especially durations of 5–10 years, a criterion for biopsy. 35 By focusing on this cohort, our study addresses a gap in transplant research and provides a foundation for understanding long-term outcomes of diabetic donor kidneys. The findings were adjusted for key donor characteristics, including insulin dependence as a proxy for diabetes severity (including type 1 diabetes or insulin-dependent type 2 diabetes), and recipient factors, offering a comprehensive assessment of proteinuria’s role in post-transplant outcomes. Subgroup analyses accounted for recipient diabetes status as a surrogate for ongoing glycemic exposure, though we could not capture the post-transplant course. These strengths enhance the generalizability of our findings and underscore the importance of evaluating diabetic donor kidney function.
However, as an observational study, our findings are subject to residual and unmeasured confounding. Selection bias may also be present, as only kidneys deemed suitable for transplantation were included, likely excluding diabetic donor kidneys with more severe dysfunction or significant histologic findings identified on pre-implantation biopsies. The exclusion of kidneys expected to confer poor post-transplant outcomes therefore leaves a cohort of eligible diabetic donor kidneys where there is uncertainty about how best they may be utilized, which is a key population of interest. Additionally, the use of registry data introduced limitations in the consistency and granularity of reported outcomes.39-41 Donor proteinuria exposure was susceptible to measurement error, including in the presence of acute kidney injury, potentially underestimating its true impact by including lower levels of proteinuria with less clinical relevance. False-positive proteinuria, from factors such as hematuria, urinary tract infections, or foley catheters in deceased donors, may have also biased the results toward the null, though this ultimately strengthens conclusions that donor proteinuria captured here was a negative prognostic marker when present.42,43 Nonetheless, the diabetic donor cohort was enriched for true proteinuria related to kidney disease, supporting the clinical relevance of these findings. Future studies with standardized and quantified proteinuria measurement with detailed histologic data are essential to better assess diabetic donor kidneys and improve graft survival outcomes.
These findings emphasize the need for a more nuanced evaluation of diabetic donor kidneys and suggest that proteinuria, while not an immediate risk factor for graft loss, may herald a risk for cumulative injury that manifests in the long-term post-transplant period. Incorporating standardized and quantitative proteinuria measurements into routine donor assessments, alongside comprehensive histologic evaluation and clinical data, could improve risk stratification and enhance the utility of diabetic donor kidneys. Future studies should explore how integrating these markers into clinical decision-making might optimize outcomes while reducing kidney discard, ultimately improving access to transplantation for patients on the waitlist.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581261424568 – Supplemental material for Proteinuria in Deceased Diabetic Donors and Kidney Transplant Outcomes
Supplemental material, sj-docx-1-cjk-10.1177_20543581261424568 for Proteinuria in Deceased Diabetic Donors and Kidney Transplant Outcomes by Christie Rampersad and S. Joseph Kim in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The data reported here have been supplied by the Hennepin Healthcare Research Institute (HHRI) as the contractor for the Scientific Registry of Transplant Recipients (SRTR). The interpretation and reporting of these data are the responsibility of the author(s) and in no way should be seen as an official policy of or interpretation by the SRTR or the US Government.
List of Abbreviations
ACGF, all-cause graft failure; DAG, directed acyclic graph; DCD, donation after cardiac death; DCGF, death-censored graft failure; DGF, delayed graft function; DM, diabetes mellitus; DN, diabetic nephropathy; DWGF, death with graft function; ECD, extended criteria donor; GS, glomerulosclerosis; HR, hazard ratio; HRSA, health resources and services administration; IFTA, interstitial fibrosis and tubular atrophy; KDRI, kidney donor risk index; OPTN, organ procurement and transplantation network; OR, odds ratio; RHR, ratio of hazard ratios; SD, standard deviation; SMD, standardized mean difference; SRTR, scientific registry of transplant recipients; UACR, urine albumin-to-creatinine ratio; US, United States; VIF, variance inflation factor.
Ethical Considerations
Research Ethics Board approval was obtained from the University Health Network, and this study was approved by the SRTR.
Consent to Participate
The requirement for informed consent to participate has been waived by the relevant Ethics Committee.
Consent for Publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CR is supported by a Kidney Research Scientist Core Education and National Training Program (KRESCENT) salary award co-funded by the Kidney Foundation of Canada, the Canadian Society of Nephrology, and the Canadian Institutes of Health Research (CIHR).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CR is supported by a Kidney Research Scientist Core Education and National Training Program (KRESCENT) salary award co-funded by the Kidney Foundation of Canada, the Canadian Society of Nephrology, and the Canadian Institutes of Health Research (CIHR). SJK is a DSMB member for Phase 2 trial of transplant therapeutic (Eledon Pharmaceuticals). The authors report no conflicts of interest.
Data Availability Statement
The data supporting the findings of this study were obtained from the Scientific Registry of Transplant Recipients (SRTR) dataset and are not able to be shared directly by the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
