Abstract
Background:
Patients with diabetes mellitus (DM) have worse graft and overall survival, but recent evidence suggests that the difference is no longer significant.
Objective:
To compare the outcomes between patients with end-stage kidney disease due to DM (ESKD-DM) and ESKD due to nondiabetic etiology (ESKD-non-DM) who underwent kidney transplantation (KT) up to 10 years of follow-up.
Design:
Survival analysis of a retrospective cohort.
Setting and Patients:
All patients who underwent KT at the Hospital Universitario San Ignacio, Colombia, between 2004 and 2022.
Measurements:
Overall and graft survival in ESKD-DM and ESKD-non-DM who received KT. Patients who died with functional graft were censored for the calculation of kidney graft survival.
Methods:
Log-rank test, Cox proportional hazards model, and competing risk analysis were used to compare overall and graft survival in patients with ESKD-DM and ESKD-non-DM who underwent KT.
Results:
A total of 375 patients were included: 60 (16%) with ESKD-DM and 315 (84%) with ESKD-non-DM. Median follow-up was 83.3 months. Overall survival was lower in patients with ESKD-DM at 5 (75.0% vs 90.8%,
Limitations:
Change in diabetes treatment received over time and adherence to glycemic targets were not considered. The sample size is relatively small, which limits the precision of our estimates. The Kidney Donor Profile Index and the occurrence of treated acute rejection were not included in the regression models.
Conclusion:
Overall survival is lower in patients with diabetes, possibly due to older age and cardiovascular comorbidities. Therefore, patients with diabetes should be followed more closely to control cardiovascular risk factors. However, there is no difference in graft survival.
Introduction
Diabetes mellitus (DM) and hypertension are the major causes of advanced chronic kidney disease (CKD), accounting for approximately 80% of cases.1,2 It is estimated that more than 40% of people with diabetes will develop CKD and are at risk of progressing to end-stage renal disease (ESRD) and requiring renal replacement therapy (RRT).3,4 Elderly patients with diabetes on dialysis have up to a 3.9-fold increased risk of mortality compared with the general population. 5
Kidney transplantation (KT) is the best renal replacement therapy considering the longer survival, better quality of life, and lower cost compared with dialysis.6-8 However, patients with ESRD have significant comorbidities and are elderly, which can be associated with significant mortality rates, such as those associated with cancer and primarily cardiovascular causes, which are higher than in the general population and account for approximately one third of deaths from known causes in transplant patients.9-13 Pretransplant and posttransplant risk factors associated with graft mortality and graft survival have been described. These include, age, heart failure, severe infections, number of rejection episodes, cadaveric donor transplantation, BK virus nephropathy, acute rejection and retransplantation, graft function, and DM.10,14-16
Patients with diabetes have limited access to the benefits of this therapy due to age and comorbidities, limiting their access to the waiting list. 17 Kervinen e al 18 showed that patients with type 2 diabetes (T2DM) had a relative probability to receive KT of 0.51 compared with patients with type 1 diabetes (T1DM) and 0.59 compared with patients without diabetes, after adjustment for age, sex, laboratory values, and comorbidities.
There are significant disparities in reported post-transplant outcomes in patients with end-stage kidney disease due to DM (ESKD-DM). Previous publications show that overall and graft survival at 5 years is lower in patients with DM compared with non-DM (69% vs 93% for overall survival and 70% vs 96% for graft survival, respectively),19,20 which may be associated with older recipient age, higher body mass index (BMI), and greater cardiovascular disease. 20 However, some recent data have shown a significant reduction in 5-year mortality risk in patients with diabetes (hazard ratio [HR] = 0.88, 95% confidence interval [CI] = 0.81-0.95), with similar survival in both groups (88% DM vs 93% non-DM) at 5 years posttransplant.18,21 Another study showed similar results, where the 10-year survival of transplanted DM patients was 90.91% versus 95.34% in non-DM patients, but the graft survival was 82.95% versus 90%, respectively. 14
There are no published data in the literature evaluating the long-term outcomes of transplantation in Latin American patients with diabetes. The aim of the present study was to evaluate the differences in overall and kidney graft survival in patients with ESKD-DM compared with ESKD-non-DM (ESKD due to nondiabetic etiology) who underwent KT in a cohort of patients managed in a kidney transplant referral center in Colombia.
Methods
A survival analysis was performed on a retrospective cohort that included all patients above18 years of age who underwent preemptive living and deceased donor KT at the Hospital Universitario San Ignacio, Bogotá (Colombia) between December 2004 and January 2022. Those with combined pancreas and KT and with less than 3 months of follow-up were excluded because of the possibility of primary graft failure due to surgical, vascular, or immunologic causes that could affect renal graft survival. The protocol was approved by the Research and Ethics Committee of the Pontificia Universidad Javeriana and the Hospital Universitario San Ignacio in Bogotá (act number: MI 056-2022).
Data were collected in a standardized database. Information was systematically recorded at each visit. Information evaluated included recipient data: age, BMI, sex, cause of CKD, type of dialysis, date of waiting list entry, date of dialysis initiation, comorbidities, serostatus for hepatitis and cytomegalovirus (CMV); donor data: age, type (deceased or living); transplant data: date, number of transplants, ischemia time, HLA mismatch, induction therapy, BK polyomavirus infection, cellular rejection, delayed graft function; and follow-up data (date of last follow-up, date and cause of graft loss, date and cause of mortality). When missing information was identified, we consulted the institutional electronic medical record. In patients with loss of follow-up, the vital status was consulted in the electronic page of the administrator of the resources of the General System of Social Security in Health—ADRES, where the information of the affiliates to the Colombian health system is registered in a mandatory way.
The diagnosis of diabetes-related ESKD was made by the clinical nephrologist who referred the patient to the transplant group. The etiology of the kidney disease was determined from the medical records. Most patients did not have a renal biopsy. Delayed graft function was defined as acute dysfunction of the transplanted kidney requiring dialysis during the first week after transplantation. BK polyomavirus infection was excluded during the first months after transplantation and was diagnosed by molecular testing (polymerase chain reaction [PCR] in blood). All patients received CMV prophylaxis with valganciclovir for 90 days, except those at high risk, who received treatment for 180 days. The diagnosis of CMV infection was confirmed by blood or tissue PCR. Cellular rejection was diagnosed by biopsy according to the Banff criteria. 22
The sample size was calculated to detect a difference in survival functions using the log-rank test by Freedman’s method 23 with adjustment for unbalanced allocation as suggested by Hsieh. 24 Assuming an HR between diabetics and nondiabetics of 0.7 and a ratio of diabetics to nondiabetics of 1/3, with an alpha error of 0.05 and 80% power, a total sample size of 59 in the ESKD-DM group and 295 in the ESKD-non-DM group was calculated.
Statistical Analysis
Absolute and relative frequencies were used to describe categorical variables. Continuous variables were expressed as median and interquartile range because there were no variables that met the assumption of normal distribution according to the Shapiro-Wilk test. Differences between patients with ESKD-DM and ESKD-non-DM were evaluated using the chi-squared test for categorical variables and the Mann-Whitney U test for continuous variables. The Kaplan-Meier method was used to evaluate overall and graft survival functions. Patients who died with a functional graft were censored for the calculation of kidney graft survival. Curves were compared using the log-rank test.
Cox regression model was used to evaluate the effect of diabetes on overall and graft survival, controlling for variables identified as significant in univariate analysis and some that have been shown to be associated in previous studies: recipient age, donor age, sex, donor type, heart failure, coronary artery disease, delayed graft function, cold ischemia time, type of dialysis, number of HLA mismatches, and BK polyomavirus infection.10-12,15,16,25,26 Bivariate analysis was followed by multivariate analysis.
Finally, a competing risk analysis was performed to assess the effect of diabetes on overall survival, considering cardiovascular and COVID-related deaths as competing risks. The Aalen-Johansen method was used because it has been shown to be more appropriate for evaluating renal survival. 27 Statistical analyses were performed using STATA software (Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC).
Results
A total of 392 transplanted patients were included in the cohort. Seventeen patients were excluded, 9 for graft loss before 3 months posttransplant (6 for acute renal vein thrombosis, 3 for hyperacute rejection) and 8 for mortality before 3 months posttransplant (5 for sepsis, 2 for cardiovascular causes, and 1 for COVID-19). A total of 375 patients were included in the analysis. Table 1 shows the characteristics of recipients and donors.
Characteristics of the Donor and Recipient According to the Diagnosis of Diabetes Mellitus.

Waiting time: time on waiting list for transplant.
Sixty (16%) had ESKD-DM (82% T2DM and 18% T1DM), and the median age was higher in recipients with diabetes (60.7 vs 45.1 years,
The median time on dialysis in the ESKD-DM population was 5.6 versus 4.4 years in the ESKD-non-DM population (
The median donor age was higher in the ESKD-DM group (45 vs 39 years,
Characteristics of Kidney Transplantation and Outcomes According to the Diagnosis of Diabetes Mellitus.

Molecular testing: in the first year as screening and those with compromised renal function.
The median follow-up per patient was 84.3 months, with a minimum of 3 months and a maximum of 120 months. Kaplan-Meier curves for death-censored graft survival are shown in Figure 1. There was no significant difference between recipients with and without diabetes (log-rank test
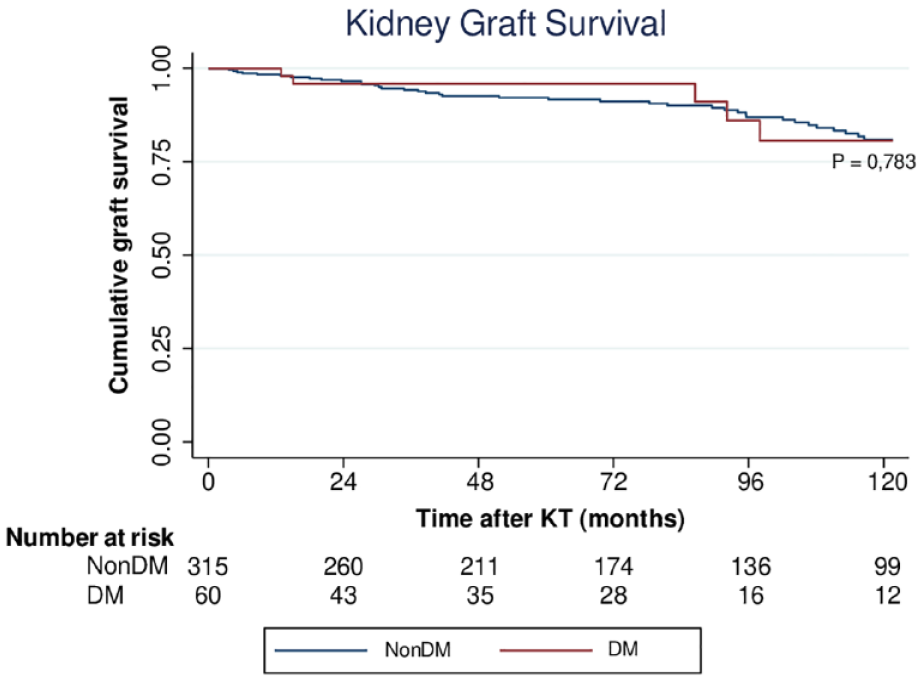
Kaplan-Meier analysis of censored graft survival.
Death-censored graft survival for patients with and without diabetes was 96.7% versus 93.3% at 5 years (
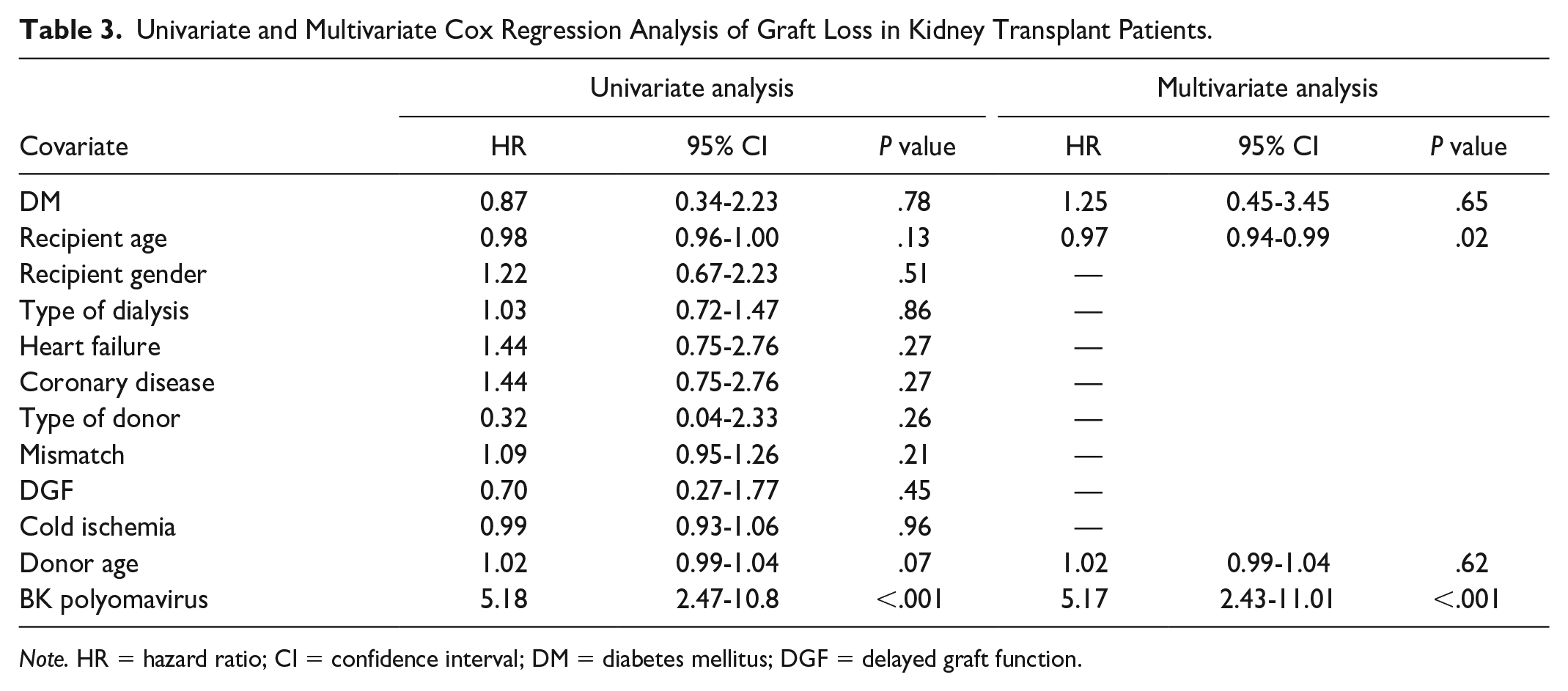
Univariate and Multivariate Cox Regression Analysis of Graft Loss in Kidney Transplant Patients.

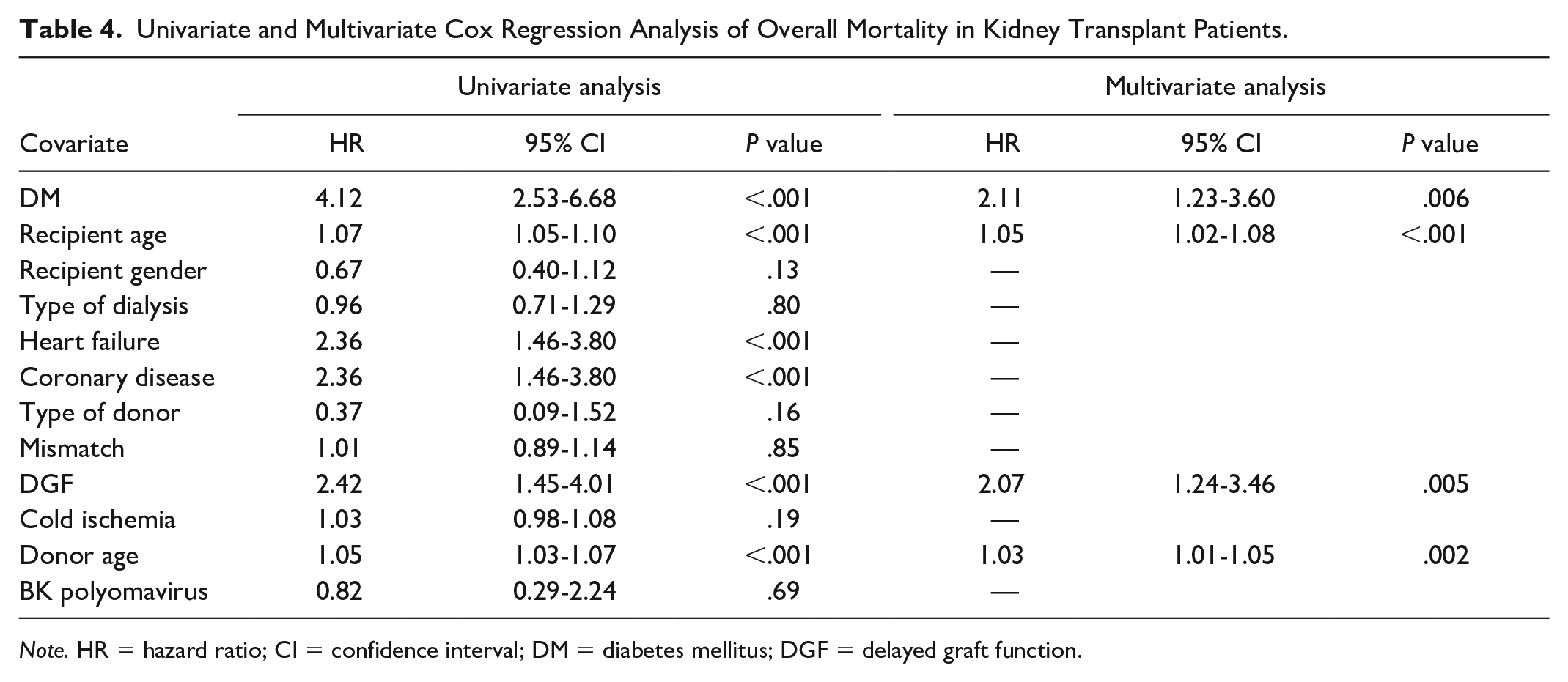
Kaplan-Meier analysis of global survival.
Overall survival was lower in patients with diabetes at 5 (75.0% vs 90.8%,
Univariate and Multivariate Cox Regression Analysis of Overall Mortality in Kidney Transplant Patients.
In the competing risk analysis, DM was associated with mortality in the cardiovascular death group (SHR = 6.06, 95% CI = 1.01-36.4,
Competing Risk Regression Analysis From Subdistribution Hazard Models for Cardiac Death, COVID Death, and Overall Death in Kidney Transplant Patients.
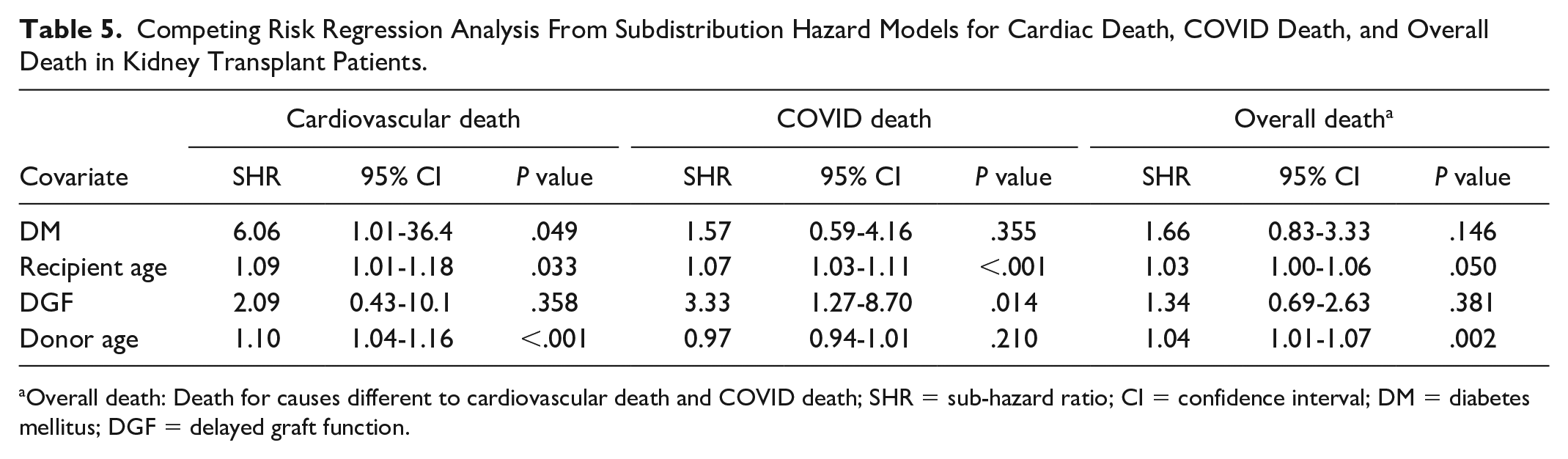
Overall death: Death for causes different to cardiovascular death and COVID death; SHR = sub-hazard ratio; CI = confidence interval; DM = diabetes mellitus; DGF = delayed graft function.
Discussion
Chronic kidney disease affects more than 10% of the world’s population, 28 and diabetes is a major cause. Of all patients with DM, 40% will develop CKD. 29 In this population, the requirement for kidney replacement therapy is increasing, with an increase in the risk of mortality. 5 Kidney transplantation is the treatment of choice showing a decrease in mortality when compared with dialytic therapy.6,7,30 There are reports of worse outcomes in transplanted patients with ESKD-DM compared with ESKD-non-DM, which has led to a decrease in the number of these patients on the waiting list and receiving kidney transplants.31,32 We found significant differences in overall survival related to cardiovascular death, but not in graft survival at 10-year follow-up.
In our study, the first cause of CKD was idiopathic, followed by arterial hypertension. Diabetes mellitus was the third cause. Other comorbidities such as hypertension, congestive heart failure, and coronary artery disease were more prevalent in kidney transplanted patients with DM, with the last 2 being almost doubled compared with patients without diabetes. The systematic review by Einarson e al 33 showed that patients with DM are twice as likely to develop and die from cardiovascular disease (CVD), which occurred in 32.2% of patients with type 2 DM and was the cause of death in 9.9% (50.3% of all deaths).
Our cohort of patients with KT and DM had a higher mean age at the time of transplantation compared with non-DM (60.7 vs 45.1 years), with a longer time on dialysis and no difference in the time on the waiting list time between the 2 groups. This may be related to the late inclusion of patients with diabetes on the waiting list, probably due to higher comorbidity. A French study of 549 patients with advanced CKD documented an association between DM and lower rates of pretransplant evaluation and waitlist entry. 34 The reasons for this were the older age of the patients, the higher BMI, and the higher morbidity. Kervinen e al 18 showed that the 5-year survival of patients is not different between patients with and without diabetes, and recommends improving access to KT for these patients. They are also more likely to be inactivated from the waiting list. 35 A study of 1265 patients found that 38.8% of patients were removed from the waiting list for CVD, with diabetes being a factor associated with an increased risk of CVD (HR = 5.13). 36
In our study, overall survival is lower in transplanted patients with ESKD-DM compared with those with ESKD-non-DM at both 5 and 10 years (75% and 55%, respectively, compared with 90.8% and 86.7% in non-DM patients). Kronson e al 37 found a 5-year survival of 61% in patients with diabetes who received a kidney transplant. Rocha e al 19 studied 124 patients who received transplants, with 5-year and 10-year survival rates of 69% and 50% for patients with diabetes versus 96% and 84% without diabetes. However, more recent studies report an improvement in survival for transplanted patients with diabetes, with no difference in graft or patient survival compared with transplanted patients without diabetes, 21 likely due to the effect of new treatments that may affect cardiovascular risk and improve glycemic control. 38
Despite the decreased overall survival observed in our study, patients with ESKD-DM still have better survival compared with hemodialysis (HD) patients. In the survival analysis published by Vijayan e al, 39 patients with diabetes on hemodialysis presented a survival of 20% at 5 years, similar to that reported by the 2014 United States Renal Data system reporting a survival of 37.2% at 5 years after initiation of hemodialysis, 1 much lower than the survival achieved in our study of 75% at 5 years.
Cardiovascular disease is the leading cause of death in the transplant population with diabetes.20,38 Similarly, in our study, cardiovascular disease accounted for 27.27% of all causes of death in patients with diabetes compared with 8% without diabetes, and our competing risks analysis showed that ESKD-DM was associated with the worst survival functions only in the cardiovascular death group. However, in our study, infection was the leading cause of death in both diabetic (30.3%) and nondiabetic (32.6%) patients, probably related to the increased mortality associated with COVID during the pandemic. A study analyzed the mortality of COVID-19 infection in chronic kidney disease, evaluating patients in hemodialysis and with renal transplant compared with patients without kidney disease. The adjusted mortality in the CKD group and HD groups were significantly higher (HR = 2.88, 95% CI = 1.52-5.44,
In multivariate analysis, the predictors of all-cause mortality were DM, recipient age, delayed graft function, and donor age. These results confirm diabetes as an independent risk factor for mortality in this population. This is explained by the cardiovascular risk that diabetes confers by presenting endothelial damage, cellular dysfunction, increased oxidation, mitochondrial dysfunction, neurohormonal activation with increased fibrosis, and cardiomyocyte hypertrophy. 43
Regarding graft survival censored by death, no difference was found between the 2 groups at either 5 or 10 years after transplantation. In univariate and multivariate analysis, BK polyomavirus infection was the only factor associated with graft loss. Other studies have reported similar results, finding no effect of diabetes on graft survival.19,38 This may be related to new immunosuppressive regimens that reduce the likelihood of rejection, the first reported cause of graft loss in the kidney transplant population. 44 However, Tsai e al 45 found increased IFTA in renal biopsies associated with increased graft failure in patients with diabetes. In our population, no differences were found between the causes of graft loss between patients in both groups, including IFTA, BK polyomavirus, and rejection.
There are some limitations in our study. We did not collect information on changes in diabetes treatment over time or adherence to glycemic control goals, which could explain the differences seen in cardiovascular mortality in patients with diabetes. New prospective studies will be needed to evaluate whether changes in these variables over time may explain the differences observed compared with other transplant groups.
Another limitation is the relatively small sample size, which limits the precision of our estimates, especially in the competing risks analysis, where the number of cardiovascular death events was limited. However, our data are consistent with recently published evidence and are also congruent with previous studies conducted in the United States.20,46
A potential limitation of our study was the different sample sizes between groups, as the smaller group may not accurately represent the characteristics or distribution of the population studied, which may limit the generalizability of the findings and make it difficult to apply the results to the larger population. However, the characteristics of our patients, in terms of older age and higher body mass index of the patients with diabetes compared with the general population, are similar to those of the Latin American and Caribbean diabetic population reported in previous studies. 47 To mitigate this risk, we performed a differential sample calculation, and the analysis was performed by subgroups, independently analyzing the different causes of mortality in the population with the competing risk analysis.
Another limitation is the probability of residual confounding associated with the unbalanced presence of some factors between the 2 groups, some of which were not ultimately included in the final model; in addition, the Kidney Donor Profile Index and the occurrence of treated acute rejections were not included in the regression models. New studies with larger sample sizes and inclusion of additional risk factors should be performed to confirm our findings.
Conclusion
Death-censored kidney graft survival was similar in patients with and without DM. Overall survival was lower in patients with diabetes and was associated with higher cardiovascular death, older mean age, and higher BMI. However, survival is better compared with patients on dialysis. It is recommended that the management of comorbidities such as hypertension, dyslipidemia, and coronary artery disease be considered to facilitate access of patients with diabetes to the kidney transplant waiting list.
Footnotes
Ethics Approval and Consent to Participate
This study was reviewed and approved by the Research and Ethics Committee of the Pontificia Universidad Javeriana and Hospital Universitario San Ignacio (No: MI 056-2022).
Consent for Publication
All authors reviewed and approved the final version of this manuscript and provided consent for publication.
Availability of Data and Materials
Patient medical record data were used in this article. The source of the data used was indicated in the article. Requests for access to anonymous aggregated data must be approved by the Ethics and Research Committee of the Hospital Universitario San Ignacio. Requests can be made by contacting the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
