Abstract
Background:
Kidney transplantation is the best treatment for chronic kidney failure, but antibody-mediated rejection (AMR) is a major cause of kidney transplant loss. Human leukocyte antigen (HLA) molecular-compatibility-based organ allocation aims to reduce the risk of donor-specific antibody formation and thereby lower the risk of AMR. However, integrating molecular compatibility in organ allocation could introduce barriers to access, consequently raising ethical concerns.
Objective:
The objective of this study was to gather perspectives of HLA professionals on molecular matching in kidney transplantation.
Design:
Individual semi-structured interviews.
Setting:
Canadian HLA laboratories.
Participants:
HLA laboratory directors or HLA professionals.
Methods:
Seven participants took part in semi-structured interviews between January and June 2024. The interviews were digitally recorded, transcribed, and analyzed using the qualitative description approach.
Results:
Participants reported positive feelings regarding the current allocation system but highlighted that HLA matching could be improved. They differed on whether kidney allocation should prioritize medical utility or fairness. While acknowledging the potential benefits of precision medicine in improving transplant outcomes, experts emphasized that its implementation confronts both scientific uncertainties and practical challenges, identifying logistical, financial, technological, and occupational barriers. They expressed concerns regarding decreased access to kidney transplantation for marginalized groups, recommending the adoption of mitigation measures. Regarding the kidney-paired donation program, experts supported integrating molecular matching as an optimizing tool to complement the current algorithm. Participants recommended that future implementation of molecular matching in Canada should involve nationwide collaboration, establishing a maximum wait time and appropriate selection criteria, additional research, adequate staffing and funding for HLA laboratories, as well as education of transplant professionals and patients.
Limitations:
The major limitation of this study is the small number of participants, all of whom were Canadian HLA professionals. Consequently, results may not be generalizable to transplant contexts in other countries.
Conclusion:
This study highlights the complexities of integrating molecular matching into organ allocation, raising concerns about equity, feasibility, and implementation. While HLA experts agree on the importance of ensuring equity and timely access, their perspectives also underscore the challenges of implementation, such as the availability of timely high-resolution HLA typing, stakeholder buy-in, and the need for dedicated tools and applications. To ensure an ethical and equitable implementation, future efforts must address access disparities through targeted mitigation strategies, such as the introduction of a maximum waiting time for a molecular-matched kidney.
Introduction
Kidney transplantation is the preferred treatment for patients living with end-stage kidney failure, offering better survival odds and quality of life compared to dialysis.1-3 However, graft loss remains a significant challenge in kidney transplantation, particularly in deceased donor transplants, where the 10-year graft survival rate is 54% in Canada. 4 While multiple factors contribute to graft failure, rejection remains the leading cause, with antibody-mediated rejection (AMR) being the most severe and difficult to treat. The AMR can occur at any point in the transplant course, and its presentation ranges from the less common acute AMR, with rapid and fulminant graft injury, to the more common chronic AMR, with progressive graft injury.5-9 The AMR is associated with the development of donor-specific antibodies, which occur when there is an immune recognition by the recipient of non-self HLA of the organ donor. As there are limited effective treatments for AMR, preventive strategies, including improved donor-recipient matching, are critical.
In Canada, provincial Organ Donation Organizations (ODOs) are responsible for organ recovery and allocation, as well as transplant coordination. The ODOs try to balance equity and utility when allocating organs, based on an array of factors such as time on dialysis, medical urgency, HLA compatibility, and pediatric priority.10,11 Highly sensitized patients (ie, with a calculated panel reactive antibody or calculated panel reactive antibody [cPRA] >95%) may receive priority access to deceased donor kidneys through a national registry managed by Canadian Blood Services (CBS). 12
To improve transplant outcomes, molecular-based matching has been proposed as a novel, more precise method for assessing HLA compatibility by analyzing amino acid variations that influence cellular and/or antibody responses (T-cell and B-cell epitopes, respectively). 13 Molecular compatibility (previously referred to as epitope matching) could help reduce donor-specific antibody (DSA) formation and lower the risk of AMR. 14 Responding to these recent advancements, the Genome Canada Transplant Consortium is developing a national molecular matching program in kidney transplantation aimed at reducing the incidence of rejection. 15
Despite its potential benefits, integrating molecular matching into kidney allocation could raise ethical and logistical concerns. 16 While it may improve long-term outcomes in some kidney recipients, it could also limit access to transplants for others, such as those with rare HLA profiles and racial minorities. Given a persistent organ shortage and the complexity of organ allocation, it is important to consider how and to what extent changes in current allocation policy might impact the Canadian population.
This study falls under the larger CanPREVENT AMR project, which seeks to use genetic technologies to improve donor-recipient matching and reduce the risk of AMR. 15 One of the objectives of CanPREVENT AMR is to document the ethical issues associated with molecular-based matching in kidney allocation from various stakeholders’ perspectives. We have previously reported Canadian transplant professionals’ and patients’ perspectives on molecular matching in kidney transplantation.17,18 These previous studies highlighted the need to further explore the perspectives of a specific subgroup of transplant professionals—HLA professionals—whose expertise in evaluating donor-recipient compatibility and minimizing the risk of graft rejection is essential to organ allocation and precision medicine. The objective of this study is to gather HLA professionals’ perspectives on the ethical and logistical issues related to molecular matching and precision medicine in kidney transplantation.
Methods
This study was exploratory in nature and used semi-structured interviews with HLA professionals. We used the consolidated criteria for the reporting qualitative research checklist. 19 We conducted individual semi-structured interviews to gather HLA professionals’ perspectives on molecular compatibility in kidney transplantation. The Centre hospitalier de l’Université de Montréal research ethics board approved the study, and all participants provided informed consent (CE20.054, MP-02-2021-9021/F1H-105764, 2023-06-26).
Recruitment and interviews were carried out between January and June 2024. Convenience and purposive sampling 20 were used to recruit HLA professionals (immunologists working in HLA laboratories) across Canada. An invitation email was sent to 26 members of the National Human Leukocyte Antigen Advisory Committee (NHLAAC), a committee of the CBS, by the chair of the committee. In addition, one of the researchers (R.S.-P.) mentioned the study during a regular NHLAAC meeting. Three reminders were sent by email, and those interested were contacted by a research team member (F.B.) to schedule an interview date. The NHLAAC is characterized by a preponderance of male members and includes two to three representatives per province. Ten HLA professionals replied to the invitation and were interested in participating. Of those 10 experts, one had participated in a related prior study, one withdrew due to working exclusively with stem cells, and one could not be reached to schedule an interview date. Ultimately, seven HLA experts participated. Figure 1 summarizes the recruitment process.
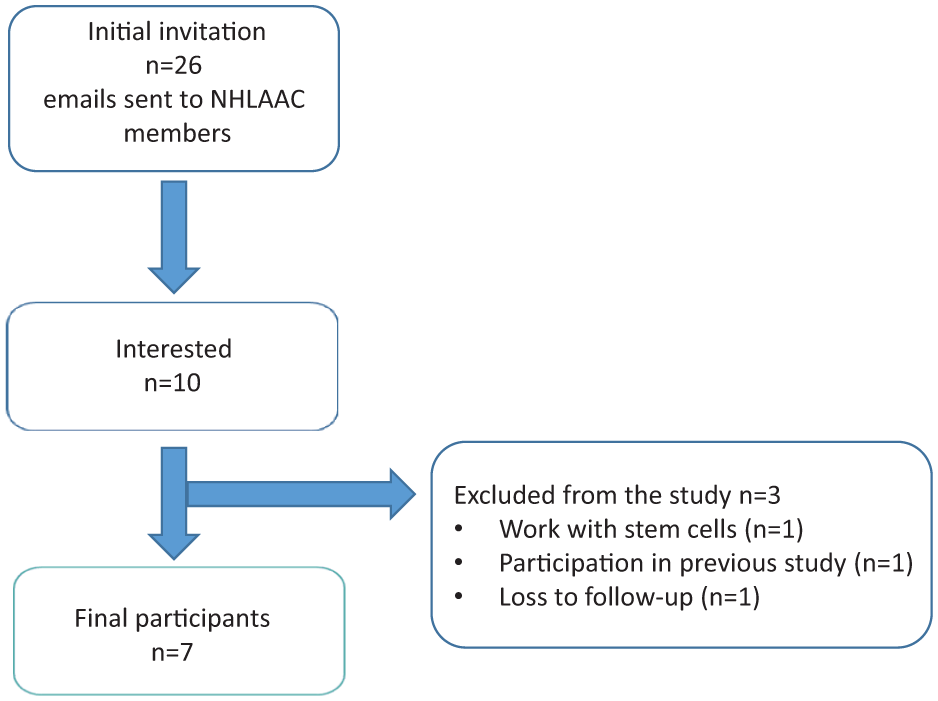
Flowchart for the recruitment process.
Seven participants took part in an individual interview. Of these, one was conducted by phone and six by videoconference; six in English and one in French. All interviews were facilitated by a male member of the research team who has more than 10 years of expertise in qualitative methods and no relationship with HLA professionals (F.B.). The interviews lasted around 44 (30-60) minutes and were digitally recorded and transcribed by a professional transcriptionist. All transcriptions were sent to participants for verification. One participant modified their transcription for clarification purposes, without changing the opinions expressed during the interview. These modifications were integrated into the final analysis.
Prior to the interviews, participants were briefed on the study objectives and given the opportunity to ask questions. The issues covered during the interviews were outlined in an interview guide comprising open-ended questions that was developed by the research team based on their previous work on the perspectives of kidney transplant professionals on molecular-based matching and kidney allocation. 18 The interview guide previously designed for health care professionals was adapted to include more precise questions pertaining to the expertise of HLA professionals. Questions addressed the following topics: (1) perspectives on the current organ allocation system; (2) role of molecular-based matching in deceased and living organ donations; (3) implementation and challenges of precision medicine; and (4) recommendations for improving the allocation process. Participants were also asked to complete a separate online questionnaire designed to collect basic socio-demographic data (sex, age, race, type of job, province of practice, and years of experience). Consistent with qualitative methodology, the interview guide was modified during the study as new themes emerged from the interviews. 20 The finalized interview guide can be found in the Supplemental Material.
A qualitative description approach was employed to summarize the perspectives of HLA professionals on organ allocation and molecular matching.21,22 The goal of this pragmatic approach was to stay close to the data and provide a comprehensive summary of the topic studied, 21 using thematic analysis. 20 The latest version of NVivo 15.0.1 (Lumivero) software was used to facilitate coding and analysis. Prior to coding verbatim transcripts, the research team created an initial coding frame based on the interview guide and a review of the literature. New codes were added to the initial coding frame as new themes emerged from interview content. The research team met frequently to discuss the coding frame and data analysis.
A female research team member trained in qualitative methodology (L.A.) coded all interviews, and frequent team discussion ensured consistency in the coding process. A female independent researcher with more than 10 years of experience in qualitative methods (A.A.) coded 75% of the raw data. Discrepancies were resolved through discussion. The coded data were organized into themes and subthemes to provide a comprehensive summary of findings.
Results
Participants’ Characteristics
Of the seven participants, six were male and aged between 41 and 61. They worked in five Canadian provinces. All except one were HLA lab directors. Two described themselves as white, three as Asian or South Asian, and two as other categories. Table 1 summarizes participants’ characteristics.
Participants’ Characteristics.

Perspectives on the Current Organ Allocation System and Allocation Criteria
Participants expressed generally positive feelings about the current allocation system but noted that improvements are needed, notably in terms of HLA matching (Box 1). Participants believed that HLA compatibility, maximizing graft survival, waiting time, medical urgency, age, race, and ABO blood group should be taken into account in a fair allocation algorithm. They used the term “race” to refer to the probability of having common ancestry, which they thought was an important consideration in organ allocation given that the frequency of HLA profiles varies across ancestry groups, which may make it difficult to find compatible organs for transplant candidates with other than European origins. In addition, experts suggest adjusting the allocation system to account for differences in access based on ABO blood group, as individuals with rare blood types may face more limited donor organ availability. In their opinion, adherence, having dependents, lifestyle habits, and needing a second transplant should not be considered as criteria.
Perspectives on the Current Organ Allocation System.

When asked whether organ allocation should prioritize medical utility or fairness, participants expressed varying opinions. For one participant, maximizing graft survival should be prioritized since it could be influenced by medical management and post-transplant care, whereas access to transplant is unpredictable and more difficult to control. By contrast, another participant mentioned that the duration of graft survival should not be the sole measure of transplant success.
Advantages and Challenges of Precision Medicine in Kidney Allocation
Participants had mixed opinions on the potential role of precision medicine in the current organ transplant allocation system (Box 2). They acknowledged that it could improve graft survival and help monitor immunosuppression based on molecular matching. However, they also highlighted uncertainties arising from the current state of knowledge. For instance, one participant stated that the role of molecular mismatch in graft rejection is unproven. Another participant mentioned the need to develop standardized cut-offs in molecular matching to guide clinical care. Finally, a participant mentioned that there are probably plenty of epitopes that have not yet been discovered. Participants were also concerned about decreasing access to transplant for all patients, as they might have to wait longer for a better match. This delay may be exacerbated for patients from certain marginalized groups, such as those who are homozygous for certain HLA alleles or belong to underrepresented ethnic communities.
Advantages and Challenges of Precision Medicine in Kidney Allocation.

Participants also mentioned logistical challenges associated with precision medicine and molecular matching. Moving forward with molecular matching will entail more working hours for HLA staff, acquiring technology that allows for high-resolution typing and additional funding to implement this new technology.
Role of Precision Medicine in Living Kidney Transplantation
Participants had a positive perspective on the use of precision medicine in kidney-paired donation (KPD) (Box 3). They saw value in incorporating molecular matching into the existing algorithm to help optimize donor-recipient compatibility, as long as this does not disadvantage certain patients. Since KPD operates on a national scale with predefined matching criteria, some participants suggested that molecular-based matching be integrated into the existing scoring system in a similar way to how HLA matching is currently used.
Role of Precision Medicine in Living Kidney Transplantation.

That being said, participants acknowledged that living donor transplants offer superior outcomes compared to deceased donor transplants, raising the question of how much additional benefit molecular-based matching would provide in this situation. They mention that, in practice, an available but mismatched living donor would often offer a better outcome than waiting for a better-matched deceased donor. Decisions also depend on how long a patient is willing or able to wait for an optimal match.
In addition, participants noted that those entering the KPD program often do so because their initial intended donor was incompatible, and they join the pool in the hope of finding a better match. Some suggest that molecular-based matching should be used as an optimizing tool but emphasize the need to remain flexible in its application.
Recommendations for the Implementation of Precision Medicine in Kidney Allocation
One of the recommendations made by HLA professionals was to set a maximum waiting time for a molecularly matched kidney (Box 4). They did not specify how long this maximum waiting time should be, but they said that it should be adapted to each patient’s situation and should not exceed the average waiting time. If a patient waits longer than the maximum wait time that has been set, they should have access to the regular organ donation pool.
Recommendations for the Implementation of Precision Medicine in Kidney Allocation.

For HLA professionals who participated in this study, it appears of paramount importance to revisit the policy or program regularly and to conduct research. One participant mentioned that future policy should be reassessed to ensure that there are no unintended consequences or disadvantage to any group of patients. In addition, mitigation strategies could be implemented through a modified point system for patients from such populations. Implementing molecular matching will also require nationwide collaboration and allocation, standardization amongst HLA laboratories, and education of physicians and patients. There is also a need for further research on molecular-based matching, epitope matching, and outcomes.
When asked whether we should shift from a provincial to a national kidney allocation system with the implementation of molecular-based matching, participants’ opinions were divided. Many viewed a national allocation system as a way to improve access to molecular-matched kidneys. By contrast, some participants raised potential logistical challenges arising from geographical distance in Canada and possible inequities for smaller provinces.
Discussion
This is the first study to report HLA professionals’ perspectives on precision medicine application to improve molecular compatibility in kidney transplantation. The HLA professionals identified benefits of integrating molecular matching for improving graft survival and reducing the incidence of kidney rejection. There are many similarities between HLA professionals’ perspectives and those of patients and transplant professionals that we have previously reported. Patients as well as transplant and HLA professionals all expressed concerns about the risk of decreasing access to donor kidneys for certain groups of patients, and they all suggested setting a minimal waiting time for a molecular-matched kidney.17,18
Disadvantaging certain groups of patients is a common concern for patients and for transplant and HLA professionals. With current HLA matching, there are existing disparities whereby white transplant candidates have a higher chance of finding a zero mismatch HLA-A,B,DR donor than black or Asian recipients. 23 It is currently uncertain if this concern is founded in the case of molecular-based matching. Recently, Mangiola et al 24 showed that that there is a relatively uniform distribution of HLA eplets across diverse population groups. Another study has shown that giving priority to low HLA-DR and/or HLA-DQ eplet mismatch would not increase disparities in access to deceased donor transplantation for black and Latino transplant recipients. 23 Future prospective studies are needed to document whether molecular-based matching impacts access to transplantation for some ethnic groups.
In contrast to the participants of our previous studies, HLA professionals were particularly concerned about knowledge gaps around molecular-based matching. Their concerns concur with the knowledge gaps identified with the Sensitization in Transplantation: Assessment of Risk workgroup. 25 This working group is a collaborative effort between the American Society of Transplantation and the American Society for Histocompatibility and Immunogenetics. Its objective is to identify knowledge gaps, establish research priorities, and provide guidance in the field of histocompatibility matching and organ transplantation. 25 This could be explained by their awareness of recent developments in the field. Moreover, there is current evidence of a relationship between molecular incompatibilities, the development of de novo DSA, and graft loss. However, this evidence is based on retrospective studies, and there are no established clear cut-offs of molecular mismatch that would contraindicate kidney transplantation.26-29
The HLA professionals highlighted the technical and logistical challenges associated with the implementation of molecular-based matching. Molecular-based matching requires high-resolution HLA genotyping, which is not universally available, is costly, and needs to be performed in a timely manner for deceased donor transplantation. 30 Currently, low- or intermediate-resolution molecular HLA genotyping is the standard method in deceased kidney transplantation because of the required speed of testing. 31 In addition, implementing molecular-based matching will require increased resources in terms of funding and staffing in HLA laboratories. Future health economics studies are needed to assess the costs of molecular-based matching and the costs of graft loss and AMR for health care systems.
Participants expressed mixed views on the role of molecular matching in living donor transplantation, particularly within KPD programs. They acknowledged that molecular-based matching could theoretically improve the success of living donor kidney transplants by optimizing donor-recipient compatibility. However, they also noted that living donor transplants already offer superior outcomes compared to deceased donor transplants, raising the question of whether molecular-based matching would provide significant additional benefits in this setting, particularly if it delayed access to living donor transplantation. To improve molecular matching in KPD, it is critical to have a sufficient number of pairs registered. It could also involve recruiting compatible pairs to participate in KPD in order to improve their molecular match.30,32 Recently, the National Kidney Registry in the United States integrated molecular-based matching as a tie breaker in the allocation of living donor kidneys through voucher donation. 33 Future studies are needed to document the positive impacts of molecular-based matching in living donor kidney transplantation.
The limitations of our study include its small sample size, which means that results may not reflect the perspectives of other HLA professionals. However, Canadian HLA professionals are a small community, and we were able to gather the views of seven of the 26 members of the National Human Leukocyte Antigen Advisory Committee. Also, most participants were male, which could be explained by the overrepresentation of men in the NHLAAC. In addition, they were highly educated on the technical aspects of molecular matching, which could have influenced their views on the ethical issues related to this topic.
Conclusion
This is the first study to report HLA professionals’ perspectives on molecular-based matching in kidney allocation. Their perspectives enrich our previous work with patients and transplant professionals. The HLA professionals shared some of the concerns expressed by patients and transplant professionals, such as the uncertainties related to this type of matching and the risk of disadvantaging some populations in accessing kidney transplant. However, given their expertise in the field of HLA, they additionally emphasized knowledge gaps and logistical challenges related to molecular-based matching. In the process of integrating molecular compatibility into kidney allocation in Canada, documenting HLA professionals’ perspectives will contribute to the development of an evidence-based and ethical framework for organ allocation.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251412195 – Supplemental material for HLA Experts’ Perspectives on Precision Medicine and Molecular Matching in Kidney Transplantation: A Qualitative Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581251412195 for HLA Experts’ Perspectives on Precision Medicine and Molecular Matching in Kidney Transplantation: A Qualitative Study by Lucy An, Aliya Affdal, Fabian Ballesteros, Marie-Françoise Malo, Savannah-Lou Cochran-Mavrikakis, Carina Sancho, Noémi Tousignant, Stirling Bryan, Paul Keown, Ruth Sapir-Pichhadze and Marie-Chantal Fortin in Canadian Journal of Kidney Health and Disease
Footnotes
Ethical Considerations
The Centre hospitalier de l’Université de Montréal research ethics board approved the study, and all participants provided informed consent (CE20.054, MP-02-2021-9021/F1H-105764, 2023-06-26). This research was conducted according to the World Medical Association Declaration of Helsinki.
Author Contributions
L.A. participated in the data analysis and in the writing of the manuscript.
A.A. participated in the conduct of the research and the data analysis and in the writing of the manuscript.
F.B. participated in the conduct of the research and the data analysis and in the writing of the manuscript.
M.-F.M. participated in the writing of the manuscript.
S.-L.C.-M. participated in the data analysis and in the writing of the manuscript.
C.S. participated in the data analysis and in the writing of the manuscript.
N.T. participated in the writing of the manuscript.
S.B. participated in the research design and in the writing of the manuscript.
P.K. obtained the research funding and participated in the research design and in the writing of the manuscript.
R.S.-P. obtained research funding and participated in the research design, conduct of research, and in the writing of the manuscript.
M.-C.F. participated in the research design, in the conduct of the research and the data analysis, and in the writing of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was funded by the Genome Canada Large Scale Applied Research Program Award “Precision Medicine CanPREVENT AMR,” funded by Génome Québec, Genome British Columbia, Genome Alberta, and the Canadian Institutes of Health Research. Drs R.S.-P. and M.-C.F. are Fonds de recherche du Québec—Santé research scholars. They are also Canadian Donation and Transplantation Research Program investigators. We want to thank all those who took the time to participate in this study.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.B., A.A., M.I., M.-F.M., C.S., S.-L.C.-M., S.B., P.K., R.S.-P., and M.-C.F. have no conflicts of interest to declare.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
