Abstract
Background:
Point of care ultrasound (POCUS) has become increasingly integrated into routine clinical care, though its adoption in nephrology remains limited.
Objective:
This pilot study evaluated a program to train nephrology fellows to perform POCUS to detect small kidney size and hydronephrosis.
Design and Setting:
We performed a quality improvement initiative at a single academic center (St. Michael’s Hospital).
Patients:
63 patients were included.
Measurements:
Pre- and post-workshop surveys assessed trainees’ comfort level with kidney POCUS imaging. Patient satisfaction was also measured using a questionnaire. Time to kidney imaging, radiologic diagnosis and kidney POCUS diagnostic accuracy were also assessed.
Methods:
Nephrology fellows participated in two 1-hour workshops featuring didactic and hands-on training using POCUS machines, after which they scanned hospitalized patients.
Results:
Sixty-two native kidneys and 32 transplant kidneys were scanned. Patient surveys indicated high satisfaction with POCUS, with 71% preferring bedside ultrasound in future care. Trainee confidence with using POCUS improved post-workshop. Trainee-performed POCUS demonstrated a specificity of 1.00 (95% CI 0.94-1.00) and 0.96 (95% CI 0.80-1.00) for the detection of hydronephrosis in native and transplant kidneys, respectively. Nephrology trainees demonstrated a specificity of 0.75 (95% CI 0.53-0.90) [left native kidneys], 0.81 (95% CI 0.61-0.93) [right native kidneys] and 0.97 (95% CI 0.84-1.00) [transplant kidneys] for the detection of small kidney size.
Limitations:
The prevalence of hydronephrosis and small kidneys was too low in this pilot study to draw conclusions about sensitivity. Additionally, a majority of the kidneys underwent POCUS imaging after an ultrasound had been performed in the medical imaging department.
Conclusions:
Despite the increasing demand for POCUS training in nephrology, a significant gap persists in its clinical integration. Our study demonstrates that a structured workshop improves trainee confidence in kidney POCUS, with high patient satisfaction. Preliminary findings suggest that nephrology fellow-performed POCUS is feasible and promising, though further large-scale studies are needed to validate its clinical utility.
Introduction
Insonation is now considered the fifth pillar in physical examination. 1 As a bedside tool, point-of-care ultrasound (POCUS) can provide timely answers to focused clinical questions, potentially expediting patient care. Multiple studies have demonstrated the usefulness of POCUS in enhancing patient care.2-5 Performed properly, POCUS has a higher sensitivity and specificity than physical exam findings in diagnosing pleural effusions, pneumonia, pulmonary edema and bladder obstruction. 6 Within the field of nephrology, POCUS offers multiple potential benefits, including the assessment of volume status, detection of hydronephrosis, determination of chronicity of kidney injury with kidney size and cortical echogenicity, and performance of ultrasound-guided procedures such as dialysis catheter insertion and kidney biopsy.7-16
To date, however, the incorporation of POCUS training into nephrology fellowship programs remains limited. 17 Indeed, recent surveys of nephrology fellows in the United States showed that despite a high interest in learning POCUS, the majority indicated that they had not received POCUS education, and of those who did, most felt insufficiently trained.17,18 Recognizing this need, interest has grown in developing educational programs that provide concise yet comprehensive POCUS training for nephrology fellows. 19
Here, we report the development and implementation of a 2-hour workshop to train nephrology fellows to perform a focused kidney POCUS scan to answer two important questions in the workup of kidney dysfunction in hospitalized patients: (1) is there hydronephrosis, and (2) are the kidneys small?
Methods
Kidney POCUS Training Workshop
The program consisted of an initial 1-hour didactic session at Unity Health Toronto (St. Michael’s Hospital), a large tertiary care hospital located in Toronto, Canada. The didactic session was led by a POCUS-certified nephrologist (M.A.Q.) and covered the following topics: (1) principles of POCUS, (2) kidney anatomy, (3) ultrasonography findings of hydronephrosis, and (4) measurement techniques for kidney size using ultrasound. The following day, a 90-minute hands-on training session was provided, during which nephrology fellows used 4 convex POCUS probes (C3 high definition [C3HD], Clarius Mobile Health, Vancouver, British Columbia, Canada) to scan patients with or without known hydronephrosis and native kidneys of various sizes. There were 4 instructors for 5-6 fellows per session. All nephrology fellows participating in the POCUS training program completed pre- and post-workshop surveys to document their self-reported comfort level with the use of kidney POCUS to rule out hydronephrosis and measure kidney size. This quality improvement initiative was formally reviewed by institutional authorities at Unity Health Toronto and deemed to neither require Research Ethics Board approval nor written informed consent from participants.
Application of POCUS Training in Clinical Care
From November 1 to December 16, 2023, nephrology fellows offered kidney POCUS scans to all hospitalized patients whom they consulted for kidney dysfunction. POCUS scans were done in addition to a standard kidney ultrasound performed by the medical imaging department. There were no exclusion criteria. POCUS images were obtained using 2 Clarius C3 High-Definition POCUS probes that connect wirelessly with a smartphone using the freely available Clarius imaging application. An internal medicine resident with more extensive kidney POCUS experience (T.F.) was available to provide real-time feedback and to ensure maximal uptake of kidney POCUS scanning by the nephrology fellows. The resident, however, did not aid with POCUS-based diagnostic calls. Age and sex of all study subjects were recorded. The serum creatinine measured prior to the POCUS and medical imaging-based scans was also recorded. Patients were asked to complete a Likert-scale questionnaire to assess the effects of POCUS on patient satisfaction. 5
Assessing Hydronephrosis and Kidney Sizes
The date and time of each POCUS scan was recorded by the nephrology fellow. Following image acquisition, the fellow recorded the presence or absence of hydronephrosis in each native kidney or kidney allograft on their linked smartphone. They also measured maximum kidney length in the longitudinal axis, which was then classified as small (≤ 9 cm) or not small (> 9 cm). A 9 cm cut-off was chosen based on a review of normal kidney lengths across multiple populations around the world. 20 The timing of the radiologic diagnoses was recorded by the fellows. Acquired images were stored on a cloud-based interface for later review with deidentified data to maintain privacy and confidentiality.
All patients also underwent an ultrasound scan in the St. Michael’s Hospital medical imaging department by a radiology technician with scans read formally by a staff radiologist as per routine clinical care. For medical imaging-based ultrasound scans, scan time was based on the time stamps on the collected images. Medical imaging-based radiologic diagnoses of hydronephrosis and kidney size measurements were recorded and time-stamped based on final radiologist reports in the hospital’s electronic medical record. Diagnostic test characteristics (sensitivity, specificity) for nephrology fellow POCUS-based diagnoses compared to gold standard medical imaging-based diagnoses were determined using 2 × 2 tables.
Results
Kidney POCUS Training Workshop
A total of 11 nephrology fellows participated in the kidney POCUS training workshop. Prior to the workshop, only 4 of the fellows had integrated POCUS into their patient assessments. These fellows reported being moderately or extremely comfortable performing a kidney POCUS scan. In contrast, roughly half of the trainees reported being uncomfortable with the use of kidney POCUS. Post-workshop, 10 out of the 11 trainees reported some level of comfort with kidney POCUS, with a majority reporting being moderately or extremely comfortable with performing kidney POCUS to diagnose hydronephrosis and small kidney size (Figure 1).

Self-reported nephrology fellow kidney POCUS skill level. (A) Pre-workshop, (B) Post-workshop.
Application of POCUS Training in Clinical Care
A total of 63 patients were scanned, including 62 native and 32 transplant kidneys. Demographic and clinical data are provided in Table 1.
Demographic and Clinical Data of Study Subjects.

Note. Values are presented as either mean ± standard deviation or median (interquartile range).
Patient satisfaction
A total of 51 out of 63 patients were surveyed about their experience (Figure 2). Most patients (71%) strongly agreed that POCUS improved their interaction with their doctors. A majority of patients (79%) did not find POCUS to be painful or uncomfortable. Most patients (71%) strongly agreed that POCUS improved their overall satisfaction with the care they received, and 61% strongly agreed that POCUS resulted in more efficient and faster care. The vast majority of patients (86%) expressed a strong preference for the use of bedside ultrasound in their future hospital visits.

Patient satisfaction survey.
Time to scan and time to radiologic diagnosis
Thirty-four patients had a medical imaging department scan before nephrology consultation. For the remaining 29 patients, a nephrology fellow POCUS was performed earlier than the medical imaging scan, at a median of 63 hours (IQR 41-123) for native kidneys and 19 hours (IQR 6-71) for transplant kidneys. For these patients, POCUS led to an earlier radiologic diagnosis by a median of 128 hours (IQR 24-168) for native kidneys and 82 hours (IQR 12-96) for transplant kidneys.
Kidney POCUS diagnostic accuracy—full cohort
The specificity of nephrology fellow-performed POCUS for the detection of hydronephrosis in transplant and native kidneys was 1.00 (95% CI 0.94-1.00) and 0.96 (95% CI 0.80-1.00) respectively. For the assessment of small kidney size, the specificity of POCUS was 0.75 (95% CI 0.53-0.90) [left native kidneys], 0.81 (95% CI 0.61-0.93) [right native kidneys], and 0.97 (95% CI 0.84-1.00) [transplant kidneys] (Tables 2 and 3). Given the low prevalence of hydronephrosis and small kidney size, estimates of sensitivity were lower. 21 The determination of sensitivity for small kidney size in transplant kidneys could not be reported as there were no small transplant kidneys (as defined by a length of ≤9 cm) in this study.
Sensitivity and Specificity for Detection of Hydronephrosis and Small Kidney Size in Native Kidneys. Analysis was Done per Kidney.
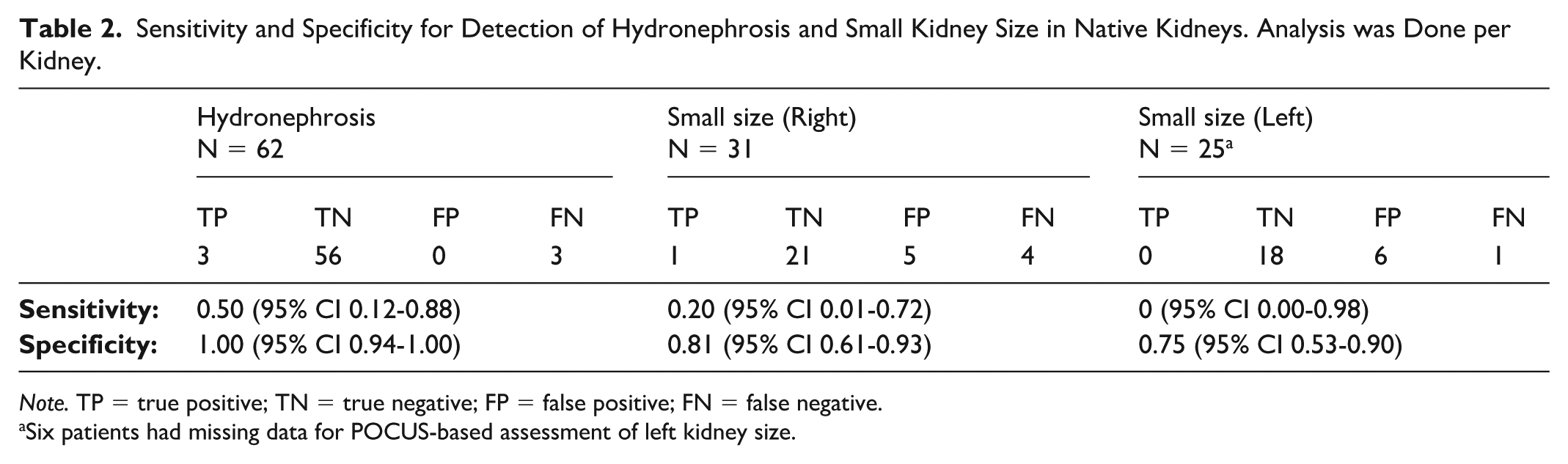
Note. TP = true positive; TN = true negative; FP = false positive; FN = false negative.
Six patients had missing data for POCUS-based assessment of left kidney size.
Sensitivity and Specificity for Detection of Hydronephrosis and Small Kidney Size in Transplant Kidneys. Analysis was Done per Kidney.

In further analyses, we compared kidney size measurements recorded by the nephrology fellows using POCUS to those obtained by the medical imaging department ultrasonographers. Nephrology fellow size measurements were lower by 1.0 ± 0.1 cm (P = .08) for right native kidneys and 1.1 ± 0.2 cm (P = .02) for left native kidneys. For transplant kidneys, size measurements by fellows using POCUS were 1.0 ± 0.3 cm (P = .0001) lower.
Kidney POCUS diagnostic accuracy—subgroup analyses
Of the 63 patients included in the main analysis, 29 patients (45 kidneys) underwent POCUS imaging prior to an ultrasound in the medical imaging department. For these patients, the specificity of nephrology fellow-performed POCUS of kidneys for the detection of hydronephrosis was 0.98 (95% CI 0.88-1.00), and for the assessment of small kidney size was 0.79 (95% CI 0.64-0.90). For the remaining 34 patients (49 kidneys) who were scanned in the medical imaging department prior to a POCUS scan, the specificity of nephrology performed POCUS was 1.00 (95% CI 0.91-1.00) for the diagnosis of hydronephrosis, and 0.92 (95% CI 0.79-0.98) for the diagnosis of small kidneys, respectively.
As several patients had a urinary catheter inserted in the time between their POCUS and medical imaging department scans, we next examined whether catheter insertion could have affected the diagnostic accuracy of nephrology fellow POCUS. We identified 10 patients (18 kidneys) who underwent catheter insertion. For these patients, the specificity of nephrology fellow POCUS for the identification of hydronephrosis [0.91 (95% CI 0.59-1.00)] and small kidney size [0.57 (95% CI 0.29-0.82)] was reduced when compared to our results for the 53 patients (76 kidneys) who did not undergo bladder catheterization (see Supplemental Table 3).
Discussion
Ultrasound is an important diagnostic test in the investigation of kidney dysfunction, with POCUS allowing for testing at the patient’s bedside. Here we describe the development of a 2-hour workshop that teaches nephrology fellows how to perform kidney POCUS to assess hydronephrosis and small kidneys. The workshop increased nephrology fellows’ comfort levels with performing kidney POCUS and a large majority of patients had a positive experience with bedside testing. Although not powered to comprehensively analyze the diagnostic characteristics of nephrology fellow POCUS, this pilot study also suggested that nephrology fellow POCUS could be performed with reasonable specificity for the diagnosis of both hydronephrosis and small kidney size. Since the prevalence of hydronephrosis and small kidney size was low in our study cohort, our pilot study was unable to reliably assess the sensitivity of nephrology fellow POCUS for these 2 findings.
Following its introduction into medicine in the 1940s, ultrasound rapidly evolved into an important diagnostic tool used by clinicians in nearly all fields of medicine, with billions of scans now performed worldwide every year in both inpatient and outpatient settings.1,6 The advantages of ultrasound are manifold, including its rapid and non-invasive nature, and the fact that it uses no ionizing radiation. As such, it has become the preferred modality for the assessment of many clinically important problems. However, the popularity of ultrasound has meant increasing demand for scans.3,22 Unfortunately, medical imaging departments have not been able to increase imaging capacity to meet this burgeoning demand, due in part to financial constraints, the large size and high cost of conventional cart-based ultrasound systems, and a lack of highly skilled staff to operate these machines. This supply-demand imbalance has increased wait times, ranging in recent reports between 1 and 2 days for hospitalized patients, even at large academic centers.3,4,22 Importantly, this delay in obtaining ultrasound scans is associated with longer hospital stays and worse outcomes in general.5,7,8
POCUS technology offers an exciting opportunity to increase imaging capacity to address this growing supply-demand imbalance. Indeed, the lower cost of POCUS systems has enabled non-radiology clinical divisions, and even individual physicians, to purchase these machines and begin imaging patients during clinical encounters. This ability to perform POCUS imaging at the time of consultation creates potential time savings by expediting the diagnostic process. Several studies have examined the effects of POCUS technology on clinical care. For example, a prospective observational study showed that utilizing POCUS in an intensive care unit (ICU) was associated with a shorter duration of mechanical ventilation and reduced ICU length of stay. 3 Similarly, the use of POCUS in hospitalized patients with chest pain or dyspnea shortened the time to appropriate management. 4 Finally, patients on kidney replacement therapy with lung congestion had improved symptoms guided by point-of-care lung ultrasound, although no improvement in a composite end point of all-cause death, non-fatal myocardial infarction, and decompensated heart failure was observed. 23 In the current study, we demonstrated that for patients who had not yet undergone ultrasound scanning at the time of nephrology consultation, the availability of nephrology fellow POCUS shortened the time to a kidney ultrasound and led to faster assessment of hydronephrosis and kidney size. This highlights the opportunity for nephrology fellow-performed POCUS to accelerate the diagnostic process and potentially lead to earlier intervention. Although beyond the scope of the current study, future work will need to explore whether this time savings results in improved outcomes such as shortened length of stay and reduced treatment costs, as has been shown in other settings.
Over the last decade, the nephrology community has seen a growing interest in the use of POCUS technology given its many potential uses (e.g. guidance for biopsy and vascular access procedures, as well as diagnostic kidney, lung, cardiac, and access imaging). Despite this interest, widespread routine adoption of POCUS by nephrologists has been slow.15,16 A key barrier to POCUS uptake has been the lack of formal training for nephrology fellows and appropriate supervision. 19 Although several comprehensive POCUS training programs have been developed, they are still relatively rare and time-intensive. Our results demonstrate that a simple workshop can be quickly and easily provided to nephrology fellows to train them on a specific POCUS skill set, with satisfactory results. These findings are in line with other reports, which have demonstrated the ability to train kidney POCUS to clinicians outside of nephrology.9-14,17,24
It is worthwhile noting that nephrology trainees tended to underestimate kidney size, particularly for left native kidneys, sometimes leading to misdiagnosis of a small kidney. Imaging the left kidney is more challenging due to its more posterior and cranial position, often requiring views through the lower ribs. Thus, future efforts should invest additional time in capturing true long-axis views, especially for the left kidney.
An important limitation of our study is that 34 patients underwent kidney ultrasound in the medical imaging department prior to POCUS by the nephrology fellows. As the fellows were not blinded to the results of the medical imaging scans, for these patients it is possible that the fellows’ interpretation of the POCUS images may have been influenced by prior knowledge of the radiologist-read ultrasounds. However, subgroup analysis of the 29 patients who underwent POCUS imaging prior to a medical imaging ultrasound demonstrated similar results to the 34 patients who had a prior radiologist-read scan, suggesting that nephrology fellow POCUS was not affected by prior scans done in the medical imaging department (Supplemental Table 2). Furthermore, during the time between a radiologist-read ultrasound and nephrology fellow POCUS, patients may have had changes in their clinical status that might have affected the radiologic findings. We explored the most likely possibility—a urinary catheter insertion resulting in the resolution of hydronephrosis—by analyzing the 10 patients (18 kidneys) who had a urinary catheter inserted in between their POCUS and medical imaging department scans separately from the remaining 53 patients (76 kidneys) who did not receive a urinary catheter during this time. As expected, the diagnostic specificity of nephrology fellow POCUS for hydronephrosis was lower for the 10 patients who underwent bladder catheterization (Supplemental Table 3). More broadly, this pilot study was underpowered to definitively assess the diagnostic accuracy of nephrology fellow POCUS, especially given the low incidence of hydronephrosis and small kidney size in our study. Thus, in the future, larger studies with POCUS done uniformly prior to a scan in the medical imaging department, and with careful recording of potential intervening interventions that might change the radiologic diagnosis, will be required to better evaluate the accuracy of nephrology fellow kidney POCUS imaging.
In a prior kidney POCUS training session provided to the nephrology fellows at our hospital, the need for an experienced trainer was highlighted. 25 Thus, in this iteration, support was provided by an internal medicine resident with more extensive kidney POCUS experience. The resident provided help with the use of the POCUS probe and real-time feedback on imaging technique, but did not provide support for diagnostic calls. It is nonetheless possible that this assistance may have improved the diagnostic characteristics of nephrology fellow POCUS. Future studies will need to explore whether this added support is indeed important to help fellows consolidate their knowledge.
Taken together, our results demonstrate that a short kidney POCUS workshop might represent a potential “first step” for programs seeking to introduce POCUS training and could represent one of several educational modules that could be combined into a full-scale educational curriculum. Importantly, multiple studies have demonstrated that short courses such as what we employed in our kidney POCUS workshop can successfully teach focused point-of-care ultrasound skills to medical students, medical residents, physicians, and nurse practitioners. 16 ,24-27
Conclusions
In summary, this study describes the development and benefits of a focused workshop to train nephrology fellows on how to perform kidney POCUS. Our results highlight a potential approach to POCUS training for the nephrology community to improve patient care. Future work is needed to assess methods to maximize skill maintenance and the next steps for curriculum development.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251378793 – Supplemental material for Development and Evaluation of a Kidney Point-Of-Care Ultrasound (POCUS) Training Program for Nephrology Fellows: A Quality Improvement Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581251378793 for Development and Evaluation of a Kidney Point-Of-Care Ultrasound (POCUS) Training Program for Nephrology Fellows: A Quality Improvement Study by Tony Fang, Mohammad Azfar Qureshi, Sara S. Jdiaa, Abdelhamid Aboghanem, Klement Yeung, Mohamed Saad, Muhammad Abdur Razzak, Arifuddin Saad Mohammed, Raheel Ahmed, Almouhannad Alkurdi, Kendrix Kek, Eno Hysi, Alireza Zahirieh, Darren A. Yuen and Ann Young in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
Not applicable.
List of Abbreviations
C3HD, C3 high definition; ED, emergency department; POCUS; point of care ultrasound.
Authors’ Note
A.Y. is supported by a KRESCENT New Investigator award. E.H. is the Canada Research Chair (Tier II) in Quantitative Ultrasound and Photoacoustic Imaging. D.A.Y. is a Canada Research Chair (Tier II) in Fibrotic Injury.
Author Contributions
T.F., M.A.Q, S.S.J., K.K., A.Z., E.H., A.Y., and D.A.Y. contributed to the development of the project. T.F., M.A.Q, S.S.J., A.Y., and D.A.Y. contributed significantly to the delivery, data collection, analysis, writing, and revisions of the manuscript. A.A., K.Y., M.S., M.A.R., S.A., and A.A. contributed to collection of data. T.F., M.A.Q, S.S.J., A.Y., and D.A.Y. read and approved the final manuscript.
Ethical Considerations
This study was formally reviewed through a Review of Quality Improvement Studies (ReQuIST) process at Unity Health Toronto, which is overseen by the Vice-President (Care Experience and Equity) of our institution. It was deemed to neither require Research Ethics Board approval nor written informed consent from participants (ie, the need for ethics approval was waived).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by an Alternative Funding Program (AFP) Innovation Grant from the St. Michael’s Physicians’ Association. E.H. is the Canada Research Chair (Tier II) in Quantitative Ultrasound and Photoacoustic Imaging. D.A.Y. is the Canada Research Chair (Tier II) in Fibrotic Injury. A.Y. is supported by a KRESCENT New Investigator award.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to our institution’s privacy policies but are available from the corresponding author on reasonable request. The SQUIRE 2.0 Guideline was used in the preparation of the manuscript. Efforts were made to include all relevant items.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
