Abstract
This case demonstrates the value of perioperative point-of-care ultrasound for rapid bedside evaluation and treatment of pulmonary oedema in an infant. A nine-week-old male infant undergoing cleft lip repair received significant intravenous fluid resuscitation for intraoperative hypotension. After uneventful extubation, he developed increased work of breathing and a gradual decline in oxygen saturation despite supplemental oxygen by way of a facemask. Lung point-of-care ultrasound revealed confluent B-lines in multiple lung fields, consistent with pulmonary oedema, likely from fluid overload. He was treated with furosemide resulting in clinical improvement within 30 minutes.
Introduction
Pulmonary oedema is a well-recognised perioperative complication causing increased morbidity and mortality. 1 It is manifested by excessive accumulation of extravascular lung water (EVLW), which occurs due to an imbalance between net fluid filtered from the pulmonary circulation and fluid absorbed by the lymphatic system. This imbalance occurs due to a variety of contributing factors, including increased intravascular hydrostatic pressure transmitted to the pulmonary microvasculature, increased interstitial hydrostatic pressure, endothelial injury, decreased oncotic pressure, lymphatic insufficiency, and increased negative interstitial pressure. 2 The paediatric surgical population is unique in relation to the pathophysiology of pulmonary oedema. Small children have a fixed cardiac stroke volume and less compliant ventricles, thus limiting the ability to tolerate excessive preload, and are at higher risk of volume overload pulmonary oedema. 3
Pulmonary oedema is classically diagnosed using a combination of history, physical examination, chest X-ray, and/or chest computed tomography (CT), with more recent use of lung ultrasound. 1 Lung point-of-care ultrasound (POCUS) has been used perioperatively to detect atelectasis in adults and children.4,5
This case report highlights the value of lung POCUS for the diagnosis and treatment of pulmonary oedema in an infant following cleft lip repair.
Written consent and Health Insurance Portability and Accountability Act (HIPAA) authorisation were obtained from the patient’s legal guardian for the publication of this case. Case report (CARE) guidelines were used in the creation of this manuscript.
Case description
Our patient was an ex–full-term, nine-week-old, 5 kg male infant with a left, unilateral, complete cleft lip and palate. He had no other congenital abnormalities and had a normal cardiopulmonary system based on history and physical examination.
After placement of standard American Society of Anesthesiologists (ASA) monitors, the patient underwent easy inhalation induction and placement of a peripheral intravenous catheter. An oral endotracheal tube was successfully placed on the first attempt. Intraoperatively, the patient had intermittent periods of hypotension, with mean arterial blood pressure decreasing to 30–35 mmHg at times, which were responsive to intravenous fluid resuscitation. He was given a total of 28 ml/kg crystalloid fluid and 10 ml/kg of 5% albumin during the three-hour surgery. At the end of the case, neuromuscular blockade was reversed and after observation of adequate tidal volumes, minimal end-expiratory inhaled anaesthetic, purposeful movement, and facial grimacing, the patient was extubated without signs of obstruction or respiratory distress. He was observed in the operating room for approximately two minutes after extubation and continued to appear stable with an oxygen saturation of 98%–100% with supplemental oxygen by way of a facemask. The patient was then transported to the postanaesthesia care unit (PACU).
Upon arrival in the PACU, initial oxygen saturation values were 98%–100%. Clinically he demonstrated no signs of increased work of breathing, with adequate chest rise. However, his oxygen saturation gradually decreased to 87% over the next 30 minutes despite supplemental oxygen administration by facemask. An anaesthetist was called to the bedside for further evaluation, and chest auscultation revealed diffuse crackles bilaterally. Imaging was considered for further evaluation including chest X-ray, chest CT, and POCUS. Due to the immediate availability of ultrasound, POCUS was used in the PACU to elucidate aetiologies for the desaturation.
As shown in Figure 1(a), the patient’s POCUS exam revealed multiple sequential rib spaces filled with multiple B-lines, also known as confluent B-lines, consistent with alveolar interstitial syndrome as described in neonatal lung POCUS. 6 Given these findings combined with the physical examination of this patient, the diagnosis of pulmonary oedema was made, suspected from intraoperative volume overload.
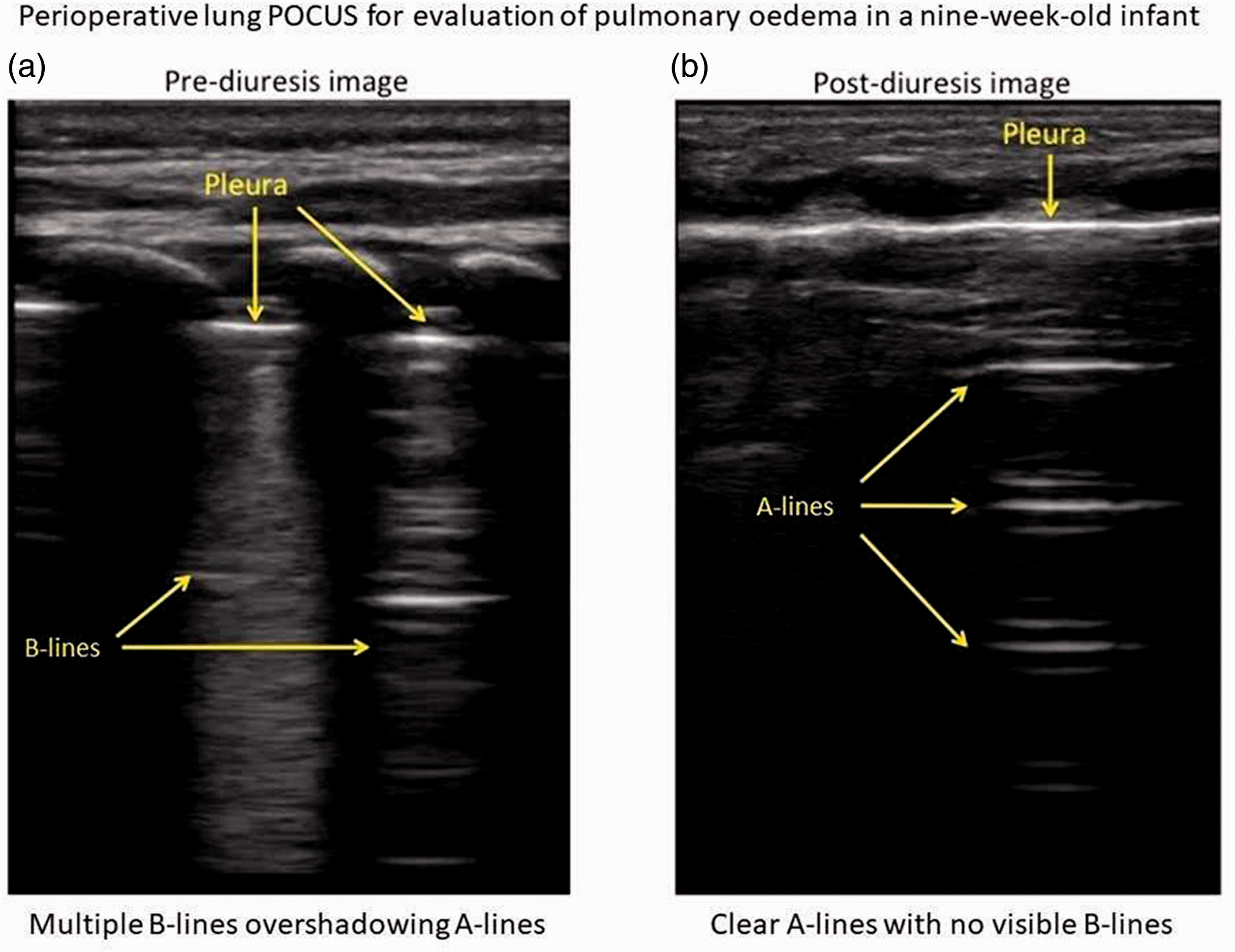
Perioperative lung point-of-care ultrasound (POCUS) for evaluation of pulmonary oedema in a nine-week-old infant. (a) Multiple B-lines overshadowing A-lines; (b) clear A-lines with no visible B-lines.
Intravenous furosemide 0.5 mg/kg was given with clinical improvement in respiratory status within 30 minutes. He was subsequently able to be weaned off oxygen without desaturation in the PACU. The patient was then admitted to a monitored bed, as was planned preoperatively. Follow-up lung POCUS evaluation (Figure 1(b)) on postoperative day one showed clear A-lines and no visible B-lines. The disappearance of B-lines and the re-emergence of normal A-lines confirmed resolution of the pulmonary oedema. He remained clinically well and required no further supplemental oxygen. He was discharged on postoperative day one.
Discussion
The differential diagnosis of perioperative pulmonary oedema may vary depending on pre-existing cardiopulmonary disease, clinical scenario, medications, and fluid administration. Our patient had no known cardiac disease to suggest cardiogenic pulmonary oedema. He received a significant amount of intravenous fluid during the case which led to the suspicion of volume overload pulmonary oedema. However, negative pressure pulmonary oedema from covert upper airway obstruction due to residual neuromuscular blockade or upper airway oedema, as well as neostigmine-induced pulmonary oedema, 7 were considered as alternative diagnoses.
On auscultation of the chest, the finding of fine inspiratory crackles suggested pulmonary oedema. These high-pitched, adventitial sounds result from the sudden opening of small airways that have collapsed during expiration from peribronchial oedema. However, there are numerous limitations to auscultation including wide inter-observer variability, failure to detect EVLW accumulation in early phases, and difficulty with monitoring progression of disease. 1
On chest X-ray, pulmonary vascular congestion and interstitial oedema are characterised by cephalisation, septal (Kerley) lines, indistinct vessels, peribronchial cuffing, ground glass opacification, and airspace opacities. While chest radiography may aid in diagnosis, the imaging modality has limitations with regard to radiation exposure, inter-observer variability, poor image quality of portable radiographs, a time lag between clinical manifestations and radiographic evidence of pathology, and difficulty in monitoring the progression of disease. 1 Pulmonary oedema is demonstrated on chest CT with certain features: ground glass opacification, bronchovascular bundle thickening, and interlobular septal thickening. 8 CT imaging limitations include significant radiation exposure, especially in the paediatric population, and the need for patient transportation.
Lung POCUS is becoming widely accepted in diagnosing and treating pulmonary oedema in the critical care setting, 1 but it is still underutilised in the perioperative setting. Lung POCUS offers several advantages compared to the aforementioned diagnostic tools including lack of radiation, portability, and ability to assess disease progression. On lung POCUS, normal lungs are characterised by A-lines, which are reverberation artefacts pleura that indicate dry interlobular septa. Pulmonary oedema is characterised by B-lines and a loss of A-lines. B-lines also arise from the pleural line, and they are an acoustic reverberation artefact caused by an ultrasound wave encountering the alveolar gas–liquid interface, and they are the sonographic correlate to thickened interlobular septae on chest CT. The B-line predominance may indicate interstitial oedema. In adults, three or more B-lines in a single view characterise pulmonary oedema. 9 Confluent B-lines fill an entire intercostal space, whereby individual B-lines are fused together and become difficult to distinguish and count.
Lung POCUS for pulmonary oedema shows superior sensitivity and a higher negative predictive value than chest X-ray in adult congestive heart failure patients with fluid overload. 10 Furthermore, lung POCUS has been used to document real-time improvement of pulmonary oedema in fluid-overloaded paediatric dialysis patients. 11 In addition to pulmonary oedema, other common perioperative pulmonary pathologies can be identified by anaesthetists using lung POCUS, including pneumothorax, endobronchial intubation, bronchospasm, atelectasis, pleural effusion, and pulmonary embolism. In a previous study published in 2018, the overall diagnostic accuracy for cardiogenic pulmonary oedema, chronic obstructive pulmonary disease/asthma, pulmonary embolism, pneumothorax, and pneumonia is 90.5%. 12 However, routine use by anaesthetists is rare. 13
The diagnostic accuracy, portability, lack of radiation, and ability to provide real-time assessment make lung POCUS attractive in the paediatric perioperative setting. Limitations to this modality include variability of practitioner knowledge and skill, and lack of standardised training. Currently, there is no national board certification for POCUS, and there is not an accredited anaesthesia Fellowship for POCUS. Hospital credentialling and clinical privileges are institution dependent. Therefore, through multidisciplinary collaboration with cardiology, radiology, emergency medicine, and critical care may help with accessible education programmes and widespread integration into anaesthesia training programmes.
In conclusion, as demonstrated in this case report, POCUS use by paediatric anaesthetists in the perioperative setting can be valuable in assisting with the diagnosis and treatment of pulmonary oedema.
Footnotes
Finance
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contribution(s)
Declaration of conflicting interests
The author(s) have no conflicts of interest to declare.
