Abstract

Department of Haematological Medicine, King's College Hospital; National Institute for Health Research and Wellcome King's Clinical Research Facility and King's College London, London, UK
A.G.K. has received honoraria from Agios Pharmaceuticals, Alexion AstraZeneca Rare Disease, Amgen, Celgene/Bristol Myers Squibb, Novartis, and Ra Pharma/UCB; is on the board of directors or is an advisory board member for Agios Pharmaceuticals, Alexion AstraZeneca Rare Disease, Amgen, Celgene/Bristol Myers Squibb, Geron, Novartis, F. Hoffmann-La Roche Ltd, and Ra Pharma; and has received consulting fees from Achillion, Agios Pharmaceuticals, Akari Therapeutics, Alexion AstraZeneca Rare Disease, BioCryst, Celgene/Bristol Myers Squibb, Novo Nordisk, Janssen Pharmaceuticals, F. Hoffmann-La Roche Ltd, Samsung, and Novartis.Title: Phase III COMMODORE 1 AND COMMODORE 2 Trials: Treatment Satisfaction and preference in patients with paroxysmal nocturnal hemoglobinuria (PNH) treated with Crovalimab and Eculizumab or Ravulizumab.
Email:
Fernando Ataulfo Gonzalez Fernandez, MD; Hematology Service, Hospital Clínico San Carlos, Madrid, Spain
F.A.G.F. is a consultant for Alexion AstraZeneca Rare Disease, F. Hoffmann-La Roche Ltd, Novartis, and Sobi; received honoraria from Alexion AstraZeneca Rare Disease, F. Hoffmann-La Roche Ltd, Novartis, and Sobi; and participated in speaker’s bureau for Alexion AstraZeneca Rare Disease, F. Hoffmann-La Roche Ltd, Novartis, and Sobi.
Email:
Phillip Scheinberg, MD; Division of Hematology, Hospital A Beneficência Portuguesa de São Paulo, São Paulo, Brazil
P.S. is a consultant for AbbVie, Alexion AstraZeneca Rare Disease, AstraZeneca, BioCryst, Janssen Pharmaceuticals, Novartis, Pfizer, and F. Hoffmann-La Roche Ltd; has given scientific presentations for AbbVie, Alexion AstraZeneca Rare Disease, Amgen, AstraZeneca, Bristol Myers Squibb, Janssen Pharmaceuticals, and F. Hoffmann-La Roche Ltd; received research funding from Alnylam, AstraZeneca, BioCryst, Pfizer, and Viracta; and participated in speaker’s bureau for Novartis.
Email:
Nicole Straetmans, MD PhD; Department of Haematology, University Hospital Saint-Luc, Brussels, Belgium
N.S. is a consultant for Amgen, Novartis, and Sobi.
Email:
Yasutaka Ueda, MD PhD; Department of Hematology and Oncology, Graduate School of Medicine, Faculty of Medicine, Osaka University, Suita, Japan
Y.U. is a consultant for Alexion AstraZeneca Rare Disease, Asahi Kase, Chugai Pharmaceutical, Janssen Pharmaceuticals, Novartis, Sanofi, and Sobi; is on the board of directors or is an advisory board member for Alexion AstraZeneca Rare Disease, Novartis, and Sanofi; has received honoraria from Alexion AstraZeneca Rare Disease, Chugai Pharmaceutical, and Sobi; received research funding from Chugai Pharmaceutical; and has participated in speaker’s bureau for Alexion AstraZeneca Rare Disease, Novartis, and Sanofi.
Email:
Brittany Gentile, PhD; Genentech, Inc., South San Francisco, CA, USA
B.G. is a former employee of Genentech Inc.
Email:
Jennifer Stefani, PhD; F. Hoffmann-La Roche Ltd, Basel, Switzerland
J.S. is a current employee of F. Hoffmann-La Roche Ltd.
Email:
Marianne Uguen, MS; F. Hoffmann-La Roche Ltd, Basel, Switzerland
M.U. is a current employee of F. Hoffmann-La Roche Ltd.
Email:
Alexander Röth, MD; Department of Hematology and Stem Cell Transplantation, University Hospital Essen, West German Cancer Center, University of Duisburg-Essen, Essen, Germany
A.R. is a consultant for Alexion AstraZeneca Rare Disease, Amgen, Apellis, Bioverativ, BioCryst, F. Hoffmann-La Roche Ltd, Novartis, Sanofi, and Sobi; is on the board of directors or is an advisory board member for Alexion AstraZeneca Rare Disease, Apellis, BioCryst, F. Hoffmann-La Roche Ltd, and Sanofi; and has received honoraria from Alexion AstraZeneca Rare Disease, Bioverativ, F. Hoffmann-La Roche Ltd, Grifols, Sanofi, and Sobi.
Email:
D.M. is a consultant for Sobi Canada, Alexion AstraZeneca Rare Disease, and Hoffmann-La Roche Ltd; received research funding from Sobi Canada and Alexion AstraZeneca Rare Disease; and has participated in speaker’s bureau for Alexion AstraZeneca Rare Disease.
Email:
Submitted on behalf of Dr. Danièle Marceau by Naomi Eterman (Roche Canada)
All authors declare third-party medical writing assistance, furnished by Akshaya Srinivasan, PhD, of Nucleus Global, an Inizio Company, and funded by F. Hoffmann-La Roche Ltd.
Röth A. et al. Am J Hematol. 2024 Sep;99(9):1768-1777
Scheinberg P. et al. Am J Hematol. 2024 Sep;99(9):1757-1767.
Liu ASH 2022; #293
Evaluating the Neuroprotective Effects of Caplacizumab in Immune-mediated Thrombotic Thrombocytopenic Purpura
Lawson Health Research Institute, London, ON, Canada
This study makes use of Dr. Lee Ting-Yim’s CT perfusion protocol which is licensed to GE Healthcare.
Despite modern methods to treat immune-mediated thrombotic thrombocytopenic purpura (iTTP), patients often suffer from increased risks of cerebrovascular disease, cognitive decline, and depression. Caplacizumab has shown to be an effective treatment for iTTP by targeting bindings zones on ultra-large von Willebrand factors, preventing platelets from aggregating and forming clots. This study investigates whether the use of Caplacizumab can reduce/prevent brain tissue damage compared to standard of care treatment (SOC).
We conducted a multi-center study involving 13 iTTP patients, six of whom were refractory to SOC treatment and received Caplacizumab. Participants underwent MRI and a contrast-enhanced dynamic CT perfusion scan to assess gross pathology, blood-brain barrier (BBB) permeability, cerebral blood flow, and volume. Additionally, a comprehensive cognitive and neuropsychiatric assessment were conducted to evaluate reasoning, short-term memory, verbal memory, concentration, and depression scores. Data collection was conducted at two timepoints; baseline, 30-days post-remission, and follow-up within one year.
BBB disruption was evident in all patients regardless of treatment method. Baseline whole brain mean permeability surface (PS) product for patients receiving Caplacizumab was 0.35 +/- 0.09 mL/min/100g, compared to SOC at 0.27 +/- 0.10mL/min/100g. A significant reduction (p = 0.047) in BBB integrity to 0.24 +/- 0.08 mL/min/100g was found in the Caplacizumab group in the follow-up visit (See Figure 1). Cognitive scores remained low across all cognitive domains for both groups with persistent white matter hyperintensities observed on MRI scans. Depression assessments revealed consistent concentration difficulties and depressive symptoms across all participants.

Blood brain barrier permeability values for Patients on SOC and Caplacizumab at baseline and follow-up.
This study highlights the persistent challenges of managing iTTP post-remission, particularly regarding neurocognitive health. The compromised BBB integrity observed in patients, regardless of remission, underscores the chronic nature of iTTP's impact on the brain. Despite patients on Caplacizumab having a higher whole brain mean PS product at baseline, likely due to being refractory, follow-up scans show a significant reduction, highlighting Caplacizumab's potential neuroprotective effects. However, no improvement was seen in patient cognitive scores and depression scores. However, to demonstrate impact in the cognitive and depression function, we will need to have more patients. While Caplacizumab shows promise in reducing BBB permeability and potentially offering a neuroprotective effect, further longitudinal studies are needed to see if this translated into neurocognitive changes.

Representative blood brain barrier permeability surface product maps for each patient at baseline and follow-up.
Mapping White Matter Degeneration in iTTP Survivors: A Diffusion Tensor Imaging Study
Lawson Health Research Institute, London, ON, Canada
None
The purpose of this study is to explore whether white matter damage in immune-mediated thrombotic thrombocytopenic purpura (iTTP) survivors contributes to the cognitive decline observed in this patient population. iTTP, despite advances in treatment such as immunosuppressants and plasma exchange, leaves over 50% of survivors at increased risk of developing cognitive impairments and cerebrovascular disease. The mechanisms underlying these neurological complications remain poorly understood. We hypothesize that damage to specific white matter tracts, which play a crucial role in cognitive processing, may be a key factor contributing to the cognitive decline seen in iTTP survivors.
We utilized diffusion tensor imaging (DTI), a sensitive technique for detecting microstructural changes in white matter, to evaluate 23 iTTP patients (mean age = 48 +/- 14, female = 19) at 30 days post-remission compared to 22 healthy controls (mean age = 47 +/- 15, female = 16). DTI metrics such as fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD), and radial diffusivity (RD) were measured across eight white matter tracts strongly correlated with cognitive function for a total of 25 regions of interest (ROIs) (Figure 1). Tracts were analyzed as ROIs and along 100 equidistant points along tracts. Alongside MRI scans, cognitive performance was assessed using Cambridge Brain Sciences across four cognitive domains: reasoning, short-term memory, verbal memory, and concentration.
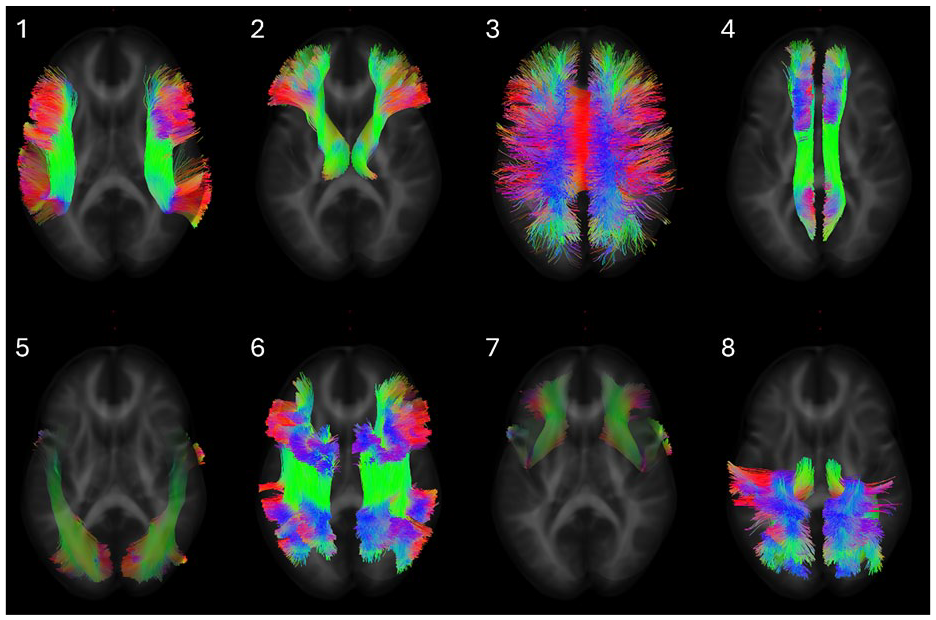
Representative diffusion tracts of eight white matter bundles: 1) arcuate fasciculus 2) anterior thalamic radiation 3) corpus callosum 4) cingulum 5) inferior longitudinal fasciculus 6) superior longitudinal fasciculus 7) uncinate fasciculus 8) thalamo-parietal. Colors indicate the direction of diffusion.
Our analysis revealed that iTTP survivors exhibited significant breakdown of brain white matter microstructure when compared to healthy controls. Specifically, FA was significantly decreased in 15 of the 25 ROIs, reflecting more isotropic water diffusion, which is indicative of axonal degeneration or demyelination. In addition, increased MD was observed in 2 of 25 ROIs, while RD was significantly elevated in 11 of 25 ROIs, suggesting possible edema, neuroinflammation, or myelin damage. These microstructural alterations were particularly pronounced in white matter tracts known to be involved in memory, such as the superior longitudinal fasciculus (Figure 2). Cognitive assessments further demonstrated that iTTP survivors had significantly reduced scores in the domains of short-term memory and concentration, which directly corresponded with the observed DTI changes (Figure 3).

Along tract analysis of the superior longitudinal fasciculus II between iTTP patients and controls. Measurements are taken at 100 equidistant points along the tract. Red-dotted lines are p < 0.05 (FWE-corrected). A) fractional anisotropy B) axial diffusivity C) radial diffusivity D) mean diffusivity.

Cambridge Brain Sciences cognitive test findings for iTTP patients and controls across four cognitive domains: reasoning, short-term memory, verbal memory, concentration. Scores are standardized to normative data. Categories with significant differences are indicated with a p-value.
The findings of this study suggest that iTTP survivors experience significant white matter damage, particularly in tracts that are critical for cognitive function. The observed decreases in FA and increases in RD in these regions indicate that iTTP may lead to axonal degeneration, demyelination, and possible inflammatory processes, contributing to cognitive decline. The fact that these structural changes are mirrored by deficits in short-term memory and concentration provides compelling evidence of a link between white matter damage and cognitive impairment in iTTP survivors. This study highlights the importance of long-term neurological follow-up for iTTP survivors. Further exploration into the role of inflammation and microvascular injury as potential contributors to white matter damage will be critical for developing effective therapeutic strategies.
Role of the C5b-9 complex in kidney disease
1Department of Microbiology, Infectiology and Immunology, Faculty of Medicine, University of Montreal, Montreal, Quebec, Canada
2Immunology and Cancer axis, CHU Sainte Justine Research Centre, Montreal, Quebec, Canada
3Pathology Department, CHU Sainte Justine, Montreal, Quebec, Canada
4Division of Nephrology, Hôpital du Sacré-Coeur-de-Montréal, Montreal, Quebec, Canada
5Division of Nephrology, Hôtel-Dieu de St-Jérôme, St-Jerome, Montreal, Quebec, Canada
6Division of Hematology, CHU Sainte Justine, Montreal, Quebec, Canada
7Division of Nephrology, CHU Sainte Justine, Montreal, Quebec, Canada
None

Expression of C5b-9 on the surface of HUVECs after stimulation or not with serum from TMA patients or controls. Dapi in blue, C5b-9 in red. Objective x20.
Efficacy and safety of iptacopan in patients with C3G glomerulopathy: 12 months results from the phase III APPEAR C3G
1The University of Iowa Roy and Lucille Carver College of Medicine Iowa City,IA, US
2Newcastle University, Newcastle upon Tyne, United Kingdom
3National Renal Complement Therapeutics Center Royal Victoria Infirmary Newcastle upon Tyne, UK
4Ospedate Pediatrico Bambino Gesu, Roma, Italy
5The University of Iowa stead family Children's Hospital, Iowa City, IA, US
6IRCCS Istituto Di Ricerche Farmacologiche Mario Negri Centro Anna Maria Astori, Bergamo, Lombardia, Italy
7Peking Union Medical College Hospital, Beijing, China
8Novartis Pharmaceutical Corporation, East Hanover, NJ, US
9Novartis Pharma AG, Basel, Basel-Stat, Switzerland
10Novartis Pharma Canada inc. Montreal, Canada
11Columbia University Vagelos College of Physicians and Surgeons, New York, NY, US
Natacha Bastien, Yaqin Wang, Angelo J Trapani, Induja Krishnan Nicholas J Webb and Mathias Meier are Novartis employes
Novartis Pharma AG
Update to the long-term safety and efficacy of iptacopan in C3G: 33-month extension study data from patients enrolled in a Phase 2 study
1Stead Family Children’s Hospital, University of Iowa, US
2Department of Nephrology, University Hospital Essen, University of Duisburg-Essen, Germany
3Hôpital Européen Georges Pompidou, France
4Service de Néphrologie et Transplantation Rénale, Centre Hospitalier Universitaire de Montpellier Montpellier, France
5Centre for Inflammatory Disease, Department of Immunology and Inflammation, Imperial College London, UK
6Imperial College Renal and Transplant Centre, Imperial College Healthcare NHS Trust, Hammersmith Hospital, UK
7Department of Medicine, Complutense, University. Spain
8Istituto di Ricerche Farmacologiche Mario Negri IRCCS, Italy
9Nephrology Department, Hospital Universitari, Spain
10Novartis Pharmaceuticals Corporation, US
11Novartis Pharma AG, Basel, Switzerland
12Novartis Inc. Canada
13National Renal Complement Therapeutics Centre, UK
14Newcastle University, UK
Junhao Liu, Matthias Meier are Novartis employes
Deciphering Complement Activation Mechanisms in Childhood IgA Nephropathy
1University of Montreal, Faculty of Medicine, Montreal, Canada
2CHU Sainte Justine Research Centre, Immunology and Cancer Axis, Montreal, Canada
3CHU Sainte Justine, Pathology Department, Montreal, Canada
4CHU Sainte Justine, Hematology Department, Montreal, Canada
5Sacré-Cœur Hospital of Montreal, Department of Medicine, Montreal, Canada
6CHU Sainte Justine, Nephrology Department, Montreal, Canada
None
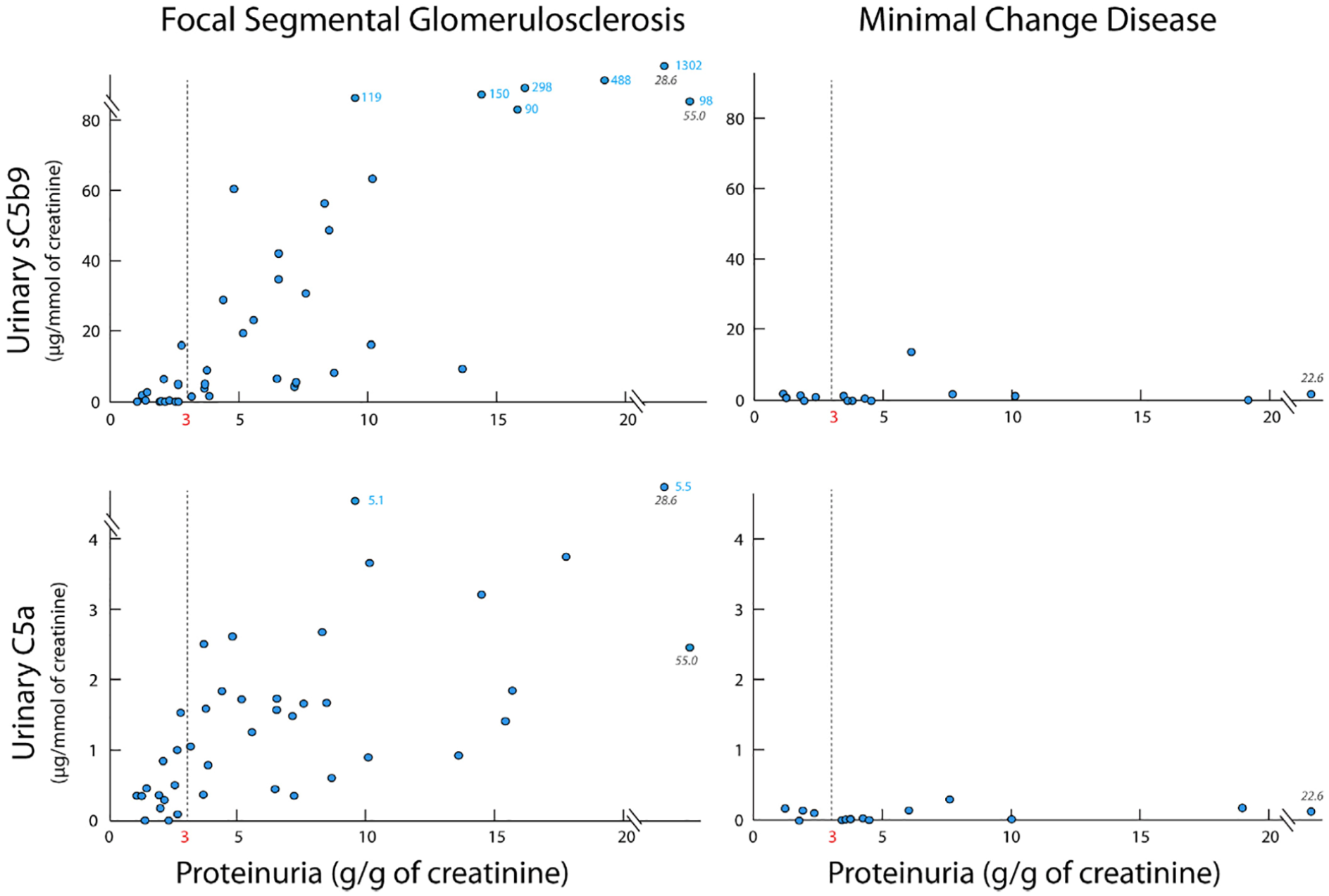
Complement Activation Distinguishes Focal Segmental Glomerulosclerosis from Minimal Change Disease
1Division of Nephrology, Centre Hospitalier Universitaire Ste-Justine, University of Montreal, Quebec, Canada
2Pathology Department, Centre Hospitalier Universitaire Ste-Justine, University of Montreal, Quebec, Canada
3Pathology Department, Hôpital Maisonneuve-Rosemont, Quebec, Canada
4Pathology Department, Centre Hospitalier de l’Université de Montréal,Quebec, Canada
5Division of Nephrology, Hôpital du Sacré-Coeur-de-Montréal, Quebec, Canada
6Nephrology Division, Centre Hospitalier de l’Université de Montréal, Quebec, Canada
7Division of Hematology, Centre Hospitalier Universitaire Sainte- Justine, University of Montreal, Quebec, Canada
8Division of Nephrology, Hôtel-Dieu de St-Jérôme, St-Jerome, Quebec, Canada
None