Abstract
Background:
Living donor kidney transplantation (LDKT) is the optimal treatment for eligible patients with kidney failure, although it is underutilized. Contextually tailored patient- and family-centered interventions may be effective to increase LDKT.
Objective:
We outline a protocol to test the feasibility of the Multidisciplinary Support To Access living donor Kidney Transplant (MuST AKT) intervention designed to increase LDKT.
Design:
Non-blinded single-center pilot randomized controlled trial with a qualitative interview component.
Setting:
Academic transplant referral center in Northern Alberta Region with a population of more than 2 million in its catchment area.
Patients:
English-speaking patients of the age range 18 to 75 years who are referred for kidney transplantation are eligible to participate.
Measurements:
Feasibility will be assessed by indicators of recruitment, retention, and completion rates, treatment fidelity, adherence to intervention, engagement in intervention, and acceptability.
Methods:
Participants will be randomly assigned 1:1 to either standard care (control) or the experimental group who receive standard care plus the MuST AKT intervention, a person-centered program designed to assist and enable the kidney transplant candidate to achieve what is required to receive an LDKT. The intervention consists of an introductory session and 4 intervention sessions delivered in-person or virtually.
Limitations:
Inferences cannot be drawn regarding the efficacy/effectiveness of the MuST AKT intervention. This study is non-blinded.
Conclusions:
This pilot study is the first step in our broader initiative to increase LDKT in our health care jurisdiction. The results of this study will be used to inform the development of a future definitive randomized controlled trial.
Trial registration number:
NCT04666545.
Introduction
Kidney transplantation is the optimal treatment for eligible patients with kidney failure due to its association with better quality of life and improved survival compared to dialysis,1-3 as well as being cost saving compared with dialysis. 4 Living donor kidney transplant (LDKT) offers superior outcomes for patients and graft survival, 5 eliminates long wait times, and prevents the need for dialysis when pre-emptive transplantation is feasible. However, the prevalence of kidney failure is increasing in many countries, including Canada, while the rate of LDKT remains stagnant. In Canada, between 2010 and 2019, despite a 35% increase in kidney failure, the rate of LDKT decreased by 11%.6,7 Therefore, there is an urgent need for jurisdictions to implement effective strategies to increase LDKT.
Key modifiable targets for interventions to increase LDKT include knowledge,8-12 communication,8,9,11 health-related perceptions,8,11 and social support.13,14 The most effective way to modify these targets and increase LDKT is through carefully designed and implemented individualized (one-on-one) educational interventions delivered by a social worker to both transplant candidates as well as their family and friends.15-17 This interventional approach was found to increase potential donor contacts to transplant programs by 40% to 50%, the number of donors evaluated by 25% to 47%, and the number of actual donors by 34%. 17 Another promising approach to increase LDKT is to facilitate the communication between a transplant candidate and potential donors. A prospective cohort study testing a Facebook® application (app) to enable kidney-only or liver-only transplant candidates to share their story with their social network found that those who used the Facebook® app were 6.6 times more likely to have a donor referral than controls. 18 These interventions provide promising strategies to adapt and implement in a contextually relevant manner.
We have developed the Multidisciplinary Support To Access living donor Kidney Transplant (MuST AKT) program, an individually tailored person-centered intervention designed to increase LDKT in our health care jurisdiction. Best practice guidelines for the development, evaluation, and implementation of complex behavioral interventions recommend a phased approach, whereby a pilot study is conducted prior to a definitive study and implementation as a part of standard care.19,20 As the first step in this phased approach, we propose to undertake a pilot randomized controlled trial (RCT) to evaluate the feasibility of the MuST AKT intervention using indicators of recruitment, retention, treatment fidelity, adherence to intervention, engagement in intervention, and acceptability. 21
Methods
Study Design and Setting
This is a pilot parallel RCT with a qualitative interview component, which will be conducted at a regional academic transplant referral center with a population of more than 2 million in its catchment area. The trial is registered with ClinicalTrials.gov (NCT04666545) and has been approved by the University of Alberta Health Research Ethics Board—Health Panel and Northern Alberta Clinical Trials and Research Center (Pro00097902). The Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guidelines were followed for reporting.22-24
Study Population
Patient participants
Patients with kidney failure who have been referred or approved for kidney transplantation are the target population. At our institution, a general nephrologist conducts an initial assessment to determine suitability for kidney transplantation, including review of contraindications to transplant and treatment compliance, as well as cardiac and psychological assessments. Those deemed eligible will be referred for further approval by a multidisciplinary transplant team, including further evaluation by a transplant nephrologist and a surgical team. Patients are eligible if they have been referred or approved for transplant, are aged between 18 and 75 years, speak the English language, and have completed the standard care 2-hour in-person or virtual “introduction to kidney transplant” education class, which outlines what a person goes through before and after a transplant. Patients will be excluded if they have a potential living kidney donor (LKD) who is actively being assessed for donation; have previously received an organ transplant or are a candidate for multi-organ transplant; have a Stanford Integrated Psychosocial Assessment for Transplant (SIPAT) score >20, indicating less than a good candidate for transplant 25 ; or have a Rapid Estimate of Adult Literacy in Medicine (REALM-66) score <19, indicating illiteracy in English. 26
Invitees of patient participants
All individuals from the patients’ social network (eg, friends, family members, co-workers, acquaintances) that the patients invite to a group session (session 4) will be asked to participate in a qualitative interview. Invitees are eligible if they speak English.
Recruitment and Procedures
A member of the patients’ clinical care team will prescreen patient medical files for preliminary eligibility (ie, transplant status, contraindications, age, living donor status, previous organ transplant, and multi-organ transplant eligibility). Patients meeting preliminary eligibility criteria will be approached by a member of the clinical care team either (1) in-person or virtually during a usual care kidney transplant education session or (2) via telephone if the patient is on the deceased donor kidney transplant wait list. Those expressing interest to know more about the study will be contacted via telephone by the project coordinator who will discuss the study in more detail and go over the informed consent document. The REDCap 27 database app will be used to distribute, collect, and store electronic informed consent documents. Paper consent and electronic consent documents will be accepted. Following written informed consent, a social worker will screen participants for remaining eligibility criteria (ie, SIPAT, REALM) and confirm other eligibility criteria have not changed since the preliminary screening. Recruitment will continue until an a priori sample size is achieved (38 total, 19 per arm: see the “Sample Size and Power Calculation” section for details). Those who decline participation in this study will be invited to take part in an interview with separate informed consent procedures to determine the reasons why they declined.
Consenting participants meeting the eligibility requirements will be contacted via telephone by the project coordinator to confirm their enrollment and complete a baseline questionnaire. Then, participants will be randomized 1:1 to either the experimental arm or the control arm while on the telephone with the project coordinator, using predetermined randomly generated permuted blocks of 4 and 6 created by a statistician in Stata/MP 17·0. 28 Assignment to study arm will be concealed by the database up until the point of assignment. Participants in the experimental arm will receive standard care plus the MuST AKT intervention, and participants in the control arm will receive standard care only. After completion of the MuST AKT intervention or standard of care, the project coordinator will contact participants over the telephone to complete a post-questionnaire. For participants in the experimental arm, the post-questionnaire will be completed within 1 week of intervention end. For participants in the control arm, the post-questionnaire will be completed 12 to 14 weeks after randomization to approximate the expected timeframe of the MuST AKT intervention.
Living donor and transplant outcomes (see the “Efficacy outcomes and measures” section for details) will be tracked for 24 months following completion of the post-questionnaire. These data will not be analyzed as part of the pilot study but will be pooled with the forthcoming definitive RCT data and evaluated then. To track the living donor and transplant outcomes, an employee of the regional health authority, who is independent of the study team, will generate a monthly report of participants’ living donor and transplant outcomes, which will be blinded to the investigators and the data-analyst.
As a part of the protocol, several qualitative interviews will be conducted. All interviews will be conducted by an experienced qualitative researcher independent of the study team and unfamiliar to the participants. The interviews will be semi-structured, conducted over the telephone, and last approximately 40 minutes. The interviews will be digitally recorded, with consent, and transcribed clean-verbatim. All participants in the experimental arm will be contacted for a telephone interview after the intervention to explore their experience of the MuST AKT intervention, and if they have not had a potential donor come forward after 12 months, they will be contacted to complete an additional interview to explore potentially contributing factors. Participants who withdraw from the study will be invited to participate in an interview with separate informed consent procedures to determine the reasons why they withdrew. All invitees of patient participants who attend the group session (session 4) will be invited at the end of the session to participate in a semi-structured interview about their experience of the session. Invitees who are interested will be contacted by a qualitative researcher independent of the study team to discuss the interview in more detail and seek informed consent prior to completing the interview. The study flow is presented in Figure 1.
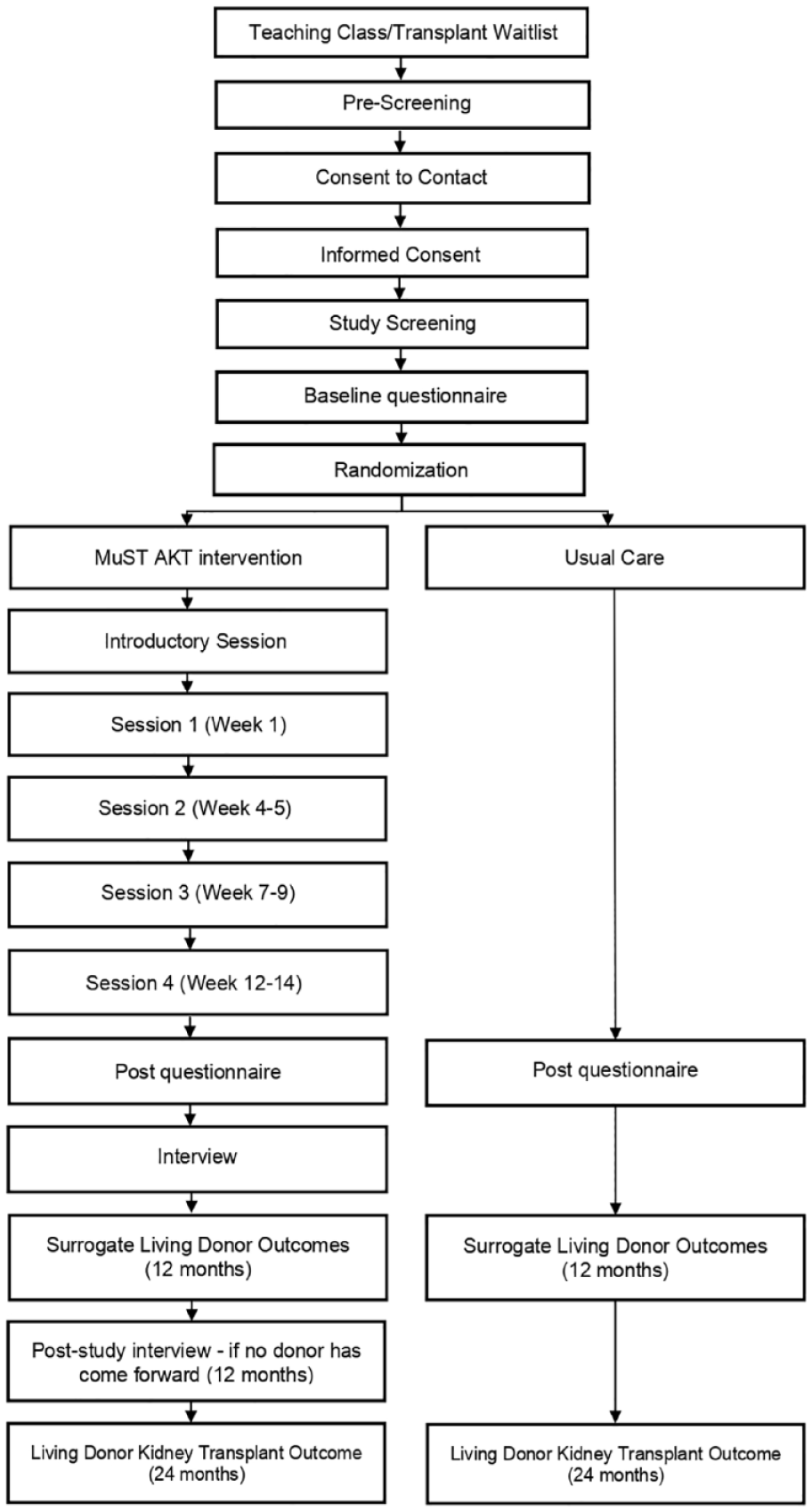
Study flow and timeline.
Blinding and avoiding bias
It is not possible to blind the participants or the research team as to which program participants will receive, although intervention allocation will not be conducted/revealed until the participant has been entered into the study and completed the baseline questionnaire. Although the clinical care team is not informed about the assignment of participants, it is possible that it is revealed to the clinical care team by the participant. Data analysis will be conducted by a statistician not involved in the conduct of the study.
Study Arms
Experimental arm
Participants in the experimental arm receive standard care plus the MuST AKT intervention. The MuST AKT intervention is an individually tailored person-centered intervention designed to “help and enable” the kidney transplant candidate to achieve what is required to receive an LDKT, not simply to “inform or ask” the individual to do it. It was developed by a team of transplant nephrologists, transplant and kidney social workers, nurses and nurse practitioners, behavioral psychology researchers, patient and family advisors, and social media experts from the Kidney Foundation of Canada—Northern and Southern Alberta branches.
Several steps were taken to develop the intervention. First, the team used the results of a previously administered survey in our health jurisdiction to inform the LDKT barriers to be targeted in the MuST AKT intervention. The survey included questions about LDKT concerns and was administered to potential donors and recipients undergoing transplant assessment and health care providers caring for potential kidney donors and recipients. 29 The most common and impactful barriers to LDKT identified from the survey included lack of knowledge, inability to communicate, the lack of awareness of available reimbursement programs to ease the financial burden of organ donation, and lack of social support. 29
Then, on the recommendation of behavioral science researchers and social workers from the MuST AKT research group, the team selected the Capability, Opportunity, and Motivation Behaviour (COM-B) model 30 and solution-focused therapy 31 to guide the development of the MuST AKT intervention to help patients address self-identified challenges to pursuing LDKT. The COM-B model describes the essential conditions for person-level behavior change: capability (C), opportunity (O), and motivation (M) 30 and intervention strategies to facilitate change, including education, training, modeling, and enablement. For behavior change to occur, one must be physically capable of doing the behavior (C), have the social and physical opportunity to do it (O), and have the intention to carry out the targeted behavior (M). In the context of LDKT, patients must have the knowledge and ability to communicate about living kidney donation (C), a social network of support and means to communicate with their social network (O), and a desire to communicate with their social network to find a LKD (M). The COM-B model has been effectively applied to many health-enhancing behavioral interventions,32-35 and its components can be mapped onto appropriate and effective intervention strategies to address barriers for each condition. 30 Solution-focused therapy will be used as the therapeutic vehicle within the individual sessions to support participant-specific barrier identification and intervention strategy adherence. It is a strength-based therapeutic approach commonly used in our health care jurisdiction that emphasizes and applies the resources of the individual to facilitate change. 36 Solution-focused therapy has been found to be a feasible and effective therapeutic approach across diverse populations. 37
Finally, in an iterative process for over 9 months, the team mapped the previously identified barriers to LDKT in our health care jurisdiction (ie, lack of knowledge, inability to communicate, awareness of financial reimbursement programs, and social support) to appropriate COM-B intervention strategies (eg, education, training, enablement, modeling) and operationalized the strategies with accompanying session content (eg, PowerPoint presentations, educational information sheets, and activities). For example, education was the overarching intervention strategy identified for knowledge and awareness barriers. Accordingly, we developed PowerPoint slides to deliver standardized education content and educational information sheets to aid in greater learning and recall after the session. Figure 2 outlines the 4 intervention sessions, and Table 1 provides a description of the goals, details the operationalized intervention strategies from the COM-B Model, and illustrates how the intervention strategies map onto the LDKT barriers addressed in each MuST AKT session.
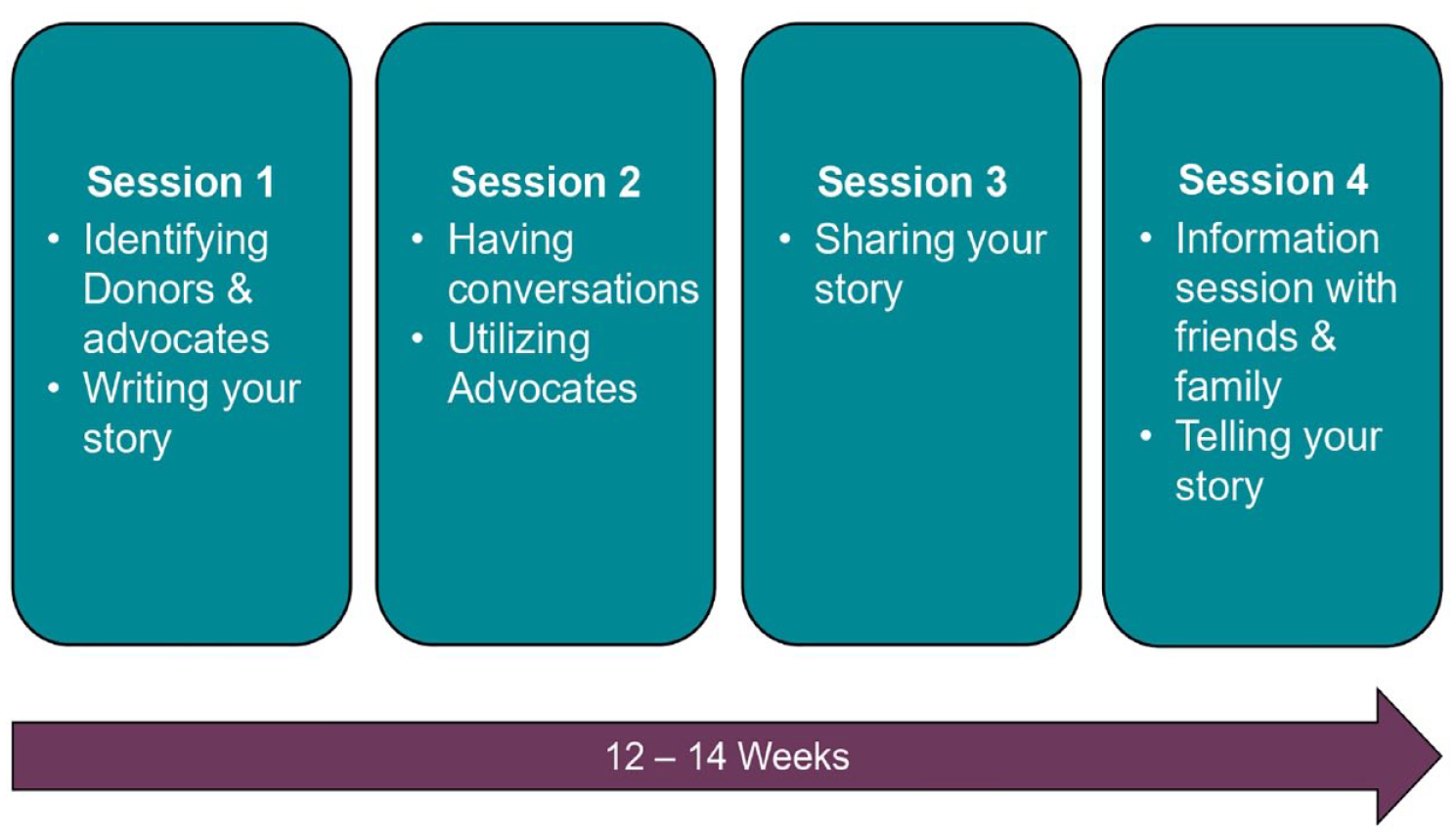
Outline of MuST AKT intervention.
Goals, Intervention Strategies From the COM-B Model and Barriers Addressed by Each MuST AKT Session.

Note. COM-B = Capability, Opportunity, and Motivation Behaviour; LDKT = Living donor kidney transplantation; MuST AKT = Multidisciplinary Support To Access living donor Kidney Transplant.
The intervention will be delivered by a transplant social worker, behavioral science interventionist, or a transplant coordinator registered nurse with training in solution-focused interviewing, kidney transplantation, and delivery of the MuST AKT intervention. It is designed to be delivered either in-person or virtually. There is an introductory session followed by 4 intervention sessions that will be scheduled by the study coordinator or the intervention facilitator. A one-on-one 60-minute introductory session outlines the topics and timeframe of the intervention, provides education on the benefits and risks of public solicitation of organ donors, and provides training on virtual platforms (if required). Then, 4 intervention sessions to identify and address barriers to LDKT are delivered over 12 to 14 weeks (approximately 1 session every 3 weeks). These sessions include three 90-minute one-on-one sessions and one 60-minute group session with the participant and invitees from their social network and a transplant nephrologist. The educational topics covered in the group session are outlined in Table 2.
Education Topics Discussed in Session 4 (Group Session With Friends and Family).

Note. LDKT = living donor kidney transplantation; DDKT = deceased donor kidney transplantation.
Participants will be discontinued from the intervention at their request, or if they do not respond to the study coordinator after 3 attempts. To increase attendance at the intervention sessions, participants will be given the option to receive telephone or email reminders at a frequency of their choice. Participants will continue their pre-transplant evaluation with the kidney transplant program (described in the Control arm section) and, if applicable, will not be prohibited from participating in other interventions.
Control arm
Participants assigned to the control arm will go through the current standard of care at the kidney transplant program, which involves the arrangement of the required tests and consultations (eg, cardiology, gastroenterology as indicated), medical assessment for suitability for transplantation by transplant nephrologist and surgeon. Each participant is assigned a pre-transplant coordinator nurse who provides support and will also be assessed and supported by a transplant social worker. The social worker will discuss the available social and financial support, as well as address patients’ questions or concerns about the transplant process. This control arm comparator was chosen as we seek to determine whether the new MuST AKT intervention should be the new standard of care. If the MuST AKT intervention is found to be safe, feasible, and effective for increasing living kidney donation in the definitive RCT (to be conducted later) and approved by the kidney transplant program as the “new standard of care,” then participants who completed the study but were assigned to the control arm will be contacted and given the option to participate in the MuST AKT intervention as the new standard care.
Data Collection and Management
Research participants will be assigned a unique identification code which will not be associated with any personal identifiable information. All collected study data will be stored on a password-protected computer server. Survey data will be collected orally from participants by the project coordinator and directly entered and stored into a secured REDCap database. 27 Study process data related to recruitment and retention will be directly entered into the REDCap database by the project coordinator, and study process data related to intervention delivery will be entered by the intervention facilitator. Quality assurance practices will be applied such as range restrictions for continuous data values. Living donor outcomes will be retrieved monthly until the end of the pilot RCT from ConnectCare, an electronic medical record database, by an employee of our health authority who is independent of the study. At the end of the study, electronic medical data will be de-identified, given the unique identification code matched to each participant, and linked to the survey data from REDCap.
Outcomes
The RE-AIM (reach, effectiveness/efficacy, adoption, implementation, and maintenance) planning and evaluation framework 38 was applied to operationalize outcome evaluation for this pilot study and throughout our initiative. RE-AIM is an implementation framework used frequently in public health, health behavior change research, and clinical settings, which focuses on challenges, steps in the design, dissemination, and implementation process that can facilitate or hinder success in achieving broad population-based or clinical outcomes. 39 Additional feasibility outcomes for this pilot study were selected, which align with the guidelines outlined by Teresi and colleagues. 21
Primary outcomes and measures
The primary outcome for this study is the feasibility of the MuST AKT intervention. We will consider feasibility a composite outcome, as measured by:
Consent/recruitment: measured by logs tracking the acceptance and decline of consent of eligible participants, logs tracking the number of family members and friends who attended session 4 of MuST AKT, as well as structured interviews/surveys of reasons for participant declining to consent (reach).
Retention: measured by logs tracking withdrawal of consent (overall dropout) with structured interviews/surveys to ascertain reasons (reach).
Adherence to study protocol/treatment fidelity: measured by logs tracking the duration of the MuST AKT intervention period (from session 1 to 4) (implementation).
Adherence to intervention: measured by logs tracking attendance at MuST AKT sessions, as well as the number of MuST AKT sessions postponed and not completed, and structured surveys of reasons for MuST AKT session postponement (implementation).
Engagement in intervention: measured by logs tracking completion of each step/component of the MuST AKT intervention, the number of participants who wrote their stories at the end of MuST AKT session 1, the number of potential donors and advocates identified during session 1 of the MuST AKT intervention, the number of participants who found at least one potential advocate at the end of the MuST AKT intervention (12-14 weeks), and the number of participants who started conversation with at least one potential donor by the end of the last MuST AKT session (implementation).
Acceptability: measured by semi-structured qualitative interviews using a qualitative description 40 approach with (1) participants who are allocated to the experimental arm to explore their perspectives on the MuST AKT intervention overall, and with each session, and to receive recommendations for improvement of the intervention, as well as (2) friends and family who attended session 4 of the MuST AKT intervention to explore their experience of this session and to receive recommendations for improvement. Acceptability will also be measured by a post-study survey of participant recommendation of the MuST AKT intervention, perception of MuST AKT intervention effectiveness, and self-efficacy.
Efficacy outcomes and measures
The following efficacy outcomes will be collected as part of the pilot RCT. However, during the conduct of this study but prior to completion, we decided to postpone the analysis of these outcomes and pool the data with the definitive RCT data. This decision was made because we secured additional funding for a definitive RCT, making a larger analysis possible, and because of the small sample size of this pilot study.
Proportion of participants with at least one potential donor who starts evaluation for living kidney donation over 12 months after completion of the study arms (efficacy, maintenance).
Proportion of living kidney transplantations over 24 months after the completion of the study arms (efficacy, maintenance).
Proportion of participants with at least one potential donor who contacted Living Donor Services over 12 months after the completion of the study arms (efficacy, maintenance).
Proportion of participants with at least one potential donor who completed the evaluation over 12 months after the completion of the study arms (efficacy, maintenance).
Demographics, social, and clinical characteristics
Participant sociodemographic variables including age, ethnic background, gender, education, income, employment status, marital status, and disease characteristics (estimated glomerular filtration rate, CKD Epidemiology Collaboration [CKD EPI], 41 dialysis [yes/no], and comorbid disease diagnoses) will be collected from electronic medical databases or via participant survey.
Sample Size and Power Calculation
A sample size of 38 participants (19 per group) was chosen prior to the decision to postpone the analysis of the efficacy outcomes in this pilot study and pool the data with the definitive RCT data, which will accrue 64 participants (32 per group). We decided to pool the unexamined data to allow for the detection of a smaller difference in LDKT proportions. A pooled sample size of 102 participants (approximately 46 participants per group after a 10% loss to follow-up) was chosen to provide 80% power (with a 5% type 1 error rate) to detect an absolute increase of 30% (from 20% to 50%, a relative increase of 150%) in LDKTs after 2 years between the pooled experimental and control groups. Our academic transplant center performs close to 40 single-kidney transplants in adult recipients out of about 200 wait-listed adults 42 each year. Similar studies15,16,43 found relative increases ranging from 10% to 300%. These calculations were performed using PASS 16 (www.ncss.com).
Data Analysis
Quantitative analysis
All analyses will be completed in Stata/MP 17.0 (www.stata.com), following an intention-to-treat approach. We will use the Fisher’s exact tests (dichotomous outcomes) and the t tests or Kruskal-Wallis tests (continuous outcomes data), as appropriate. Unadjusted percentages, means, and their differences along with 95% confidence intervals will be reported. P < .05 will be considered statistically significant for all quantitative outcomes.
Qualitative design and analysis
Qualitative description was the methodological approach used.40,44 This approach aims to describe the perception and experience of individuals and is well suited for medical and mixed-method research.40,44 The COnsolidated criteria for REporting Qualitative research (COREQ) checklist will be used as the reporting guideline. 45 Data will be analyzed in NVivo-12 by experienced qualitative researchers independent of the study, using deductive and inductive thematic analysis. 46 A coding framework will be established based on the interview guides and updated to include additional themes that emerge from the data. The coding framework will be validated by two coders, and any disagreements will be resolved through discussion. Codes will be revised and reviewed for each interview and grouped into common themes. Themes will be compared across the interviews.
Study Monitoring
Quarterly and annual reporting will be conducted. An advisory and steering committee will be assembled to support the conduct of this research. The advisory committee will include representatives from a patient and family advisory group within our regional health authority who have direct experience with kidney transplantation (as donors or recipients); leaders within our regional health authority that are involved in the care of patients pursuing kidney transplantation or the management of this research initiative; academics from the University of Alberta and the University of Calgary who specialize in nephrology, kidney transplantation, or psychology; and community partners, including the Kidney Foundation of Canada (Northern and Southern Alberta branches) and Canadian Blood Services. The steering committee will include the leadership team (primary investigator, co-investigators, research associate, department manager), a statistician, academics from the University of Alberta and the University of Calgary who have expertise in the methods and/or the patient population of this study, and experts in implementation science and behavioral interventions to increase living kidney donation (as required). The advisory committee will provide strategic and operational oversight of the MuST AKT pilot project, and the steering committee will oversee the scientific conduct of the MuST AKT pilot project. No interim analyses are planned, and a data-monitoring committee will not be formed for this pilot project as the trial duration is short and minimal risks are anticipated. Serious adverse events will be tracked and immediately reported to the research ethics boards. Protocol modifications will be submitted for approval to research ethics boards and clinicaltrials.gov and reported to the advisory and steering committees.
Dissemination Policy
Our dissemination plan is informed by the Canadian Institutes of Health Research Knowledge Translation (CIHR KT) Planning Guide. 47 Our study results will be of interest to both transplant programs in the province. Strong linkages with partner organizations will facilitate the conduct, dissemination, and uptake of our research. We used an iKT plan in the proposal development; stakeholders have been engaged from its conception and will be involved throughout. We will develop manuscripts, which will be published in open-access and peer-reviewed journals.
Discussion
Interventions to improve LDKT are needed, given that the prevalence of kidney failure has increased, while the rate of LDKT has remained the same or decreased.6,7 This pilot study is part of a broader, long-term initiative to increase LDKT in our jurisdiction. We use the Knowledge-to-Action framework, 48 which is an implementation process model, to guide this initiative, including intervention development, implementation, and evaluation. Following this framework, the problem was identified through empirical research and reviews,15-17,43,49 barriers and facilitators were assessed through empirical research,8-11,50-52 knowledge was adapted and tailored for the local context through empirical research and stakeholder participation in intervention development, and a plan for outcome evaluation was outlined.38,39 The results of this pilot study will inform a definitive RCT for the next phase of our initiative, followed by implementation into standard care within a continuous quality improvement framework. 53 First, this pilot study will provide the required metrics on feasibility, including recruitment, retention, treatment fidelity, adherence to intervention, engagement in intervention, and acceptability, which are all essential to optimize the “MuST AKT” intervention for a definitive RCT. Second, we will collect outcomes to evaluate the efficacy of the MuST AKT intervention in a future definitive RCT.
We considered many factors in our decisions regarding the pilot study design. We decided to apply stringent enrollment criteria to evaluate our intervention under ideal circumstances. This approach will be re-evaluated as we progress to implementation. First, we will enroll patients that are likely to be approved for kidney transplantation, if not already approved, to ensure that we obtain an adequate sample size to assess the primary and secondary outcomes (ie, outcomes related to LDKT). Second, we will enroll patients without a previous kidney transplant, to ensure that the patients’ potential donors have not been exhausted and our outcomes are more attainable. Finally, we decided to enroll participants that speak the English language due to feasibility within the research team, but if the MuST AKT intervention is implemented as the new standard of care, there will be provisions to expand to non–English-speaking participants.
We used the RE-AIM planning and evaluation framework38,39 to guide the selection of study outcomes because it encompasses the evaluation of individual benefits (ie, effectiveness/efficacy and maintenance dimensions), feasibility (ie, reach and implementation dimensions), and study processes (ie, implementation and adoption dimensions). The study feasibility and process outcomes will provide a comprehensive understanding of how the intervention is delivered and received by participants, which will allow us to make informed improvements to the MuST AKT intervention for a future definitive RCT. At this time, we decided not to operationalize outcomes from the “adoption” dimension given that the pilot study will be delivered by the research team and not the health care staff directly involved with usual patient care.
As documented in clinicaltrials.gov, we made adaptations to the protocol during the course of the study but prior to study completion, which involve the outcomes and analysis. When the protocol was developed, we had funding to conduct the pilot RCT only, and as such, we planned to evaluate the efficacy and feasibility of the MuST AKT intervention simultaneously. However, we received funding to conduct a definitive RCT, which will allow us to evaluate the efficacy of the MuST intervention in a larger, more appropriately powered study. As such, it was decided to collect the efficacy outcomes as part of the pilot RCT but delay the analysis of such outcomes until the pilot data can be pooled with the definitive RCT data. As a result, we changed the timeframe for collecting the outcome “living donor kidney transplantations” in the pilot study from 12 months to 24 months to match the timeframe for data collection in the definitive RCT. This update to the protocol is consistent with current guidelines for conducting pilot studies 21 and will allow for a more robust determination of the efficacy of the MuST AKT intervention in the definitive RCT. Thus, a limitation of this pilot study is that we will be unable to draw inferences regarding the efficacy/effectiveness of the MuST AKT intervention. An additional limitation is that this study is non-blinded.
The MuST AKT intervention is a complex behavioral intervention, which is defined by having several interacting components, several behaviors required by those delivering or receiving the intervention, multiple groups or organizational levels targeted, several outcomes, and a high degree of tailoring required. 54 However, through the development of intervention training seminars and manuals for practitioners that outline the approach, steps, materials, and resources, our aim is to make this intervention simple for social workers and other qualified health care practitioners to conduct as part of their standard practice. A strength of this initiative is that we have incorporated team members that are directly involved with the care of patients with kidney failure and are involving stakeholders that are internal and external to our health care jurisdiction throughout the planning, evaluating, and implementation phases of our work.
Conclusion
This pilot study is an essential part in our broader initiative to increase LDKT in our health care jurisdiction. The results of this study will be used to inform the development of a future definitive RCT, before implementation into standard care. In addition, the findings will provide information about strategies to reduce barriers to LDKT and their effectiveness at increasing LDKT rates.
Footnotes
Acknowledgements
The authors thank the members of the Multidisciplinary Support To Access living donor Kidney Transplant (MuST AKT) Research Group that were active during the development and conduct of this research, who, in addition to the authors are Branko Braam, Ross Bennett, Sandra Cockfield, Aman Dhaliwal, Carol Easton, Manuel Escoto, Dyan Franco, Jill Goth, Michelle Hofer, Kailash Jindal, Shayroz Khosla, Lana Marchinko, Julia Moore, Louise Morrin, Dan Muruve, Nairne Scott-Douglas, Kyle Nash, Neesh Pannu, Deanna Paulson, Kara Penny, Flavia Robles, Mike Simoens, Lee Ann Tibbles, Marta Tsoukalas, Sandi Vanderzee, Joyce Van Deurzen, and Kathy Yetzer. The authors thank the Medicine Strategic Clinical NetworkTM of Alberta Health Services for their support and contributions to help make this study possible. The authors also thank the Kidney Foundation of Canada—Northern and Southern Alberta Branches for their support and contributions to the study.
Ethics Approval and Consent to Participate
All participants will provide informed consent to participate prior to enrollment. The study was approved by the University of Alberta Health Research Ethics Board - Health Panel and Northern Alberta Clinical Trials and Research Centre (Pro00097902). The study is registered at clinicaltrials.gov (NCT04666545).
Consent for Publication
All authors have reviewed the manuscript and consent to its publication.
Availability of Data and Materials
Data sharing is not applicable to this article as no data sets were generated or analyzed during the current study. Future data sets used and/or analyzed will be available from the corresponding author on reasonable request.
Author Contributions
Anne-Marie Selzler: Conceptualization, Methodology, Software, Writing—original draft, Visualization, Supervision, Project Administration. Paris Davoodi: Conceptualization, Methodology, Resources, Writing—review & editing, Funding. Scott Klarenbach: Conceptualization, Methodology, Resources, Writing—review & editing, Funding. Ngan Lam: Conceptualization, Methodology, Resources, Writing—review & editing, Funding. Terry Smith: Conceptualization, Writing—review & editing, Supervision, Project administration. Abigail Ackroyd: Conceptualization, Writing—original draft, Visualization. Natasha Wiebe: Methodology, Software, Resources, Writing—review & editing. Michelle Stiphout: Methodology, Software, Resources, Writing—review & editing. Bonnie Corradetti: Conceptualization, Writing—review & editing. Sharron Ferdinand: Conceptualization, Writing—review & editing. Dorothy Iyekekpolor: Conceptualization, Writing—review & editing. Gordon Smith: Conceptualization, Writing—review & editing. Nancy Verdin: Conceptualization, Writing—review & editing. Aminu Bello: Methodology, Writing—review & editing. Kevin Wen: Methodology, Writing—review & editing. Soroush Shojai: Conceptualization, Methodology, Resources, Writing—original draft, Supervision, Project administration, Funding acquisition.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University Hospital Foundation-Kaye Competition grant (grant number: RES0046891) and Alberta Innovates—Partnership for Research and Innovation in the Health System (PRIHS) (grant number: G2020000182).
