Abstract
Background:
Acute kidney injury (AKI) increases the risk of hospital readmission, chronic kidney disease, and death. Therefore, effective communication in discharge summaries is essential for safe transitions of care.
Objective:
The objectives of this study were to determine the quality of discharge summaries in AKI survivors and identify predictors of higher quality discharge summaries.
Design:
Retrospective chart review.
Setting:
Tertiary care academic center in Ontario, Canada.
Patients:
We examined the discharge summary quality of 300 randomly selected adult patients who survived a hospitalization with AKI at our tertiary care hospital, stratified by AKI severity. We included 150 patients each from 2015 to 2016 and 2018 to 2019, before and after introduction of a post-AKI clinic in 2017.
Measurements:
We reviewed charts for 9 elements of AKI care to create a composite score summarizing discharge summary quality.
Methods:
We used multivariable logistic regression to identify predictors of discharge summary quality.
Results:
The median discharge summary composite score was 4/9 (interquartile range, 2-6). The least frequently mentioned elements were baseline creatinine (n = 55, 18%), AKI-specific follow-up labs (n = 66, 22%), and medication recommendations (n = 80, 27%). The odds of having a higher quality discharge summary (composite score ≥4/9) was greater for every increase in baseline creatinine of 25 μmol/L (adjusted odds ratio [aOR]: 1.27; 95% confidence interval [CI]: 1.03, 1.56), intrarenal etiology (aOR: 2.32; 95% CI: 1.26, 4.27), and increased AKI severity (stage 2 aOR: 2.57; 95% CI: 1.35, 4.91 and stage 3 aOR: 3.36; 95% CI: 1.56, 7.22). There was no association between discharge summary quality and the years before and after introduction of a post-AKI clinic (aOR: 0.77; 95% CI: 0.46, 1.29).
Limitations:
The single-center study design limits generalizability.
Conclusions:
Most discharge summaries are missing key AKI elements, even in patients with severe AKI. These gaps suggest several opportunities exist to improve discharge summary communication following AKI.
Introduction
Acute kidney injury (AKI) affects approximately 20% of all hospitalized patients and is associated with increased mortality, hospital length of stay, cardiovascular disease, and progression to chronic kidney disease (CKD) or end-stage kidney disease.1-5 In addition, nearly 1 in 5 patients will be readmitted in the 30 days following discharge. 6 As a result of this high morbidity and mortality, Kidney Disease: Improving Global Outcomes (KDIGO) guidelines recommend close evaluation of patients following an episode of AKI. 7 However, less than 10% of survivors of AKI are referred to nephrologists following discharge, which places much of the responsibility for post-AKI care on primary care providers (PCPs). 8
Previous work has shown that PCPs are less likely than nephrologists to be aware of existing nephrology-related clinical practice guidelines or recognize CKD.9,10 Furthermore, PCPs have identified multiple barriers to successful post-AKI management, which include AKI complexity and poor coordination of care across organizational settings. 11 Given these challenges and the high risk of complications in patients who survive AKI, clear and comprehensive discharge communication is essential.
In the general medical and congestive heart failure populations, discharge summary availability and improved quality have been associated with a lower risk of hospital readmission.12,13 In patients with AKI, coding of AKI by PCPs following hospital discharge has also been associated with timely medication review, serum creatinine (Cr) monitoring, and patient education. 14 Therefore, higher quality discharge summaries and initiatives to improve communication from the inpatient to outpatient setting have the potential to improve outcomes in the vulnerable post-AKI population. However, few studies have investigated the quality of discharge summaries in patients who survive AKI, which have been limited by small sample size, exclusion of non-critically ill patients, or assessment of few discharge summary elements.15-18
In this study, our objectives were twofold. First, we aimed to determine the quality of discharge summaries in a randomly selected cohort of patients hospitalized with AKI at a single tertiary care academic center. Second, we sought to identify predictors of higher quality discharge summaries that may inform future educational and quality improvement (QI) initiatives. We hypothesized that most discharge summaries would be deficient in multiple elements needed for effective communication to facilitate optimal post-AKI care.
Methods
Study Overview
We performed a single-center retrospective chart review examining the quality of discharge summary content in hospitalized patients who survived an episode of AKI at Kingston Health Sciences Centre (KHSC) in Ontario, Canada. The KHSC is a tertiary care academic center, with 440 beds and 22 000 admissions annually. The Research Ethics Board at Queen’s University approved the study, which adhered to the Declaration of Helsinki. Patient consent was waived due to the retrospective design of the study. Reporting of the study follows the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 19
Study Population
We identified all adult patients (age ≥18 years) who survived an episode of AKI at KHSC between January 1, 2015 to December 31, 2016 and January 1, 2018 to December 31, 2019. We selected these years to provide a representative sample before and after the introduction of an ambulatory post-AKI clinic in 2017.20,21 Even though the post-AKI clinic pathway did not involve any patient or health care provider interventions prior to hospital discharge (eg, education), we hypothesized that its presence could have influenced discharge summary quality through increased awareness of AKI and its long-term consequences among health care providers.
We excluded patients who required kidney replacement therapy (KRT) prior to hospitalization, kidney transplant recipients, and patients with missing pre-admission baseline serum Cr in the electronic health record (EHR). The presence and severity of AKI was defined according to the KDIGO serum Cr criteria, using all available serum Cr data during the index hospitalization in the EHR. 7 The most recent outpatient serum Cr 7-365 days prior to the admission date represented the baseline value. 22
We used a computer algorithm to identify all inpatient admissions without exclusion criteria that met KDIGO AKI serum creatinine criteria. From this subset of patients, we used a random number generator to select patients for discharge summary quality review. We included 300 total patients, with 150 patients from each time period (January 1, 2015 to December 31, 2016 and January 1, 2018 to December 31, 2019), stratified by KDIGO AKI stage. Of the 50 stage 3 AKI patients from each time period, we randomly selected 25 who received KRT during their hospitalization.
Demographic and Clinical Data
Two reviewers (CG and MN) used a standardized form for baseline data abstraction of all patients who met inclusion criteria. Data collected included patient demographics, comorbidities, intensive care unit (ICU) stay, community- versus hospital-acquired (occurring >48 hours after hospital admission) AKI, AKI etiology, nephrology consultation, baseline and peak Cr, discharge Cr percent above baseline, and relevant admission and discharge medications. We categorized AKI etiology as follows: (1) prerenal (eg, hypovolemia, cardiorenal); (2) acute tubular necrosis (ATN) and other intrarenal causes (eg, glomerulonephritis, acute interstitial nephritis, vascular); or (3) postrenal/obstructive (eg, bladder outlet obstruction). We settled any discrepancies between reviewers through involvement of a third data abstractor (SAS).
Assessment of Discharge Summary Quality
Currently, there are no discharge summary elements in AKI patients that have been validated to be associated with clinical outcomes. Therefore, we determined discharge summary quality by ascertaining the presence of key elements based on prior published work and our clinical experience.14-16,18,23-26 Overall, there were 9 key elements of AKI included (Table 1) that focus on either a description of the AKI event or follow-up and management recommendations. We calculated a composite score to act as a summary measure of overall discharge summary quality, with each element included assigned 1 point for a maximum possible score of 9 points. Two reviewers collected this information (CG and MN), with discrepancies resolved by a third reviewer (SAS).
Discharge Summary Quality Key Elements.

Statistical Analysis
We summarized baseline characteristics for the overall cohort and stratified these by AKI severity. We presented categorical variables as counts with percentages and continuous variables as means and standard deviations or medians and interquartile ranges. We assessed the underlying distributions of the continuous variables with the Shapiro-Wilk test.
We used multivariable logistic regression to assess predictors of discharge summary quality, with the cut point being the median discharge summary composite score (ie, ≥4/9). The initial univariate analysis included age, sex, comorbidities, length of stay, ICU stay, hospital-acquired AKI, AKI etiology, inpatient nephrology consult, baseline Cr, peak Cr, discharge Cr percent above baseline, and time period. We then included any significant variables (
We considered a 2-tailed
Results
Baseline Characteristics
The median age of patients admitted with AKI across all stages was 68 years (interquartile range [IQR]: 58-78), and the majority were men (n = 172, 57%) (Table 2). The most common preexisting comorbidities included hypertension (n = 191, 64%), diabetes mellitus (n = 119, 40%), and coronary artery disease (n = 100, 33%). The median baseline Cr was 82 μmol/L (IQR: 63-105) and 25% (n = 75) of patients had preexisting CKD (defined as estimated glomerular filtration rate <60 mL/min/1.73 m2). The median length of stay was 11 days (IQR: 5-21), and 173 (58%) patients received ICU-level care. The most common AKI etiology across all stages was prerenal (n = 157, 52%); however, ATN and other intrarenal etiologies were most common among patients with stage 3 AKI (n = 74, 74%). More patients with stage 3 AKI (n = 60, 60%) also had a nephrology consultation.
Patient Characteristics.

Serum Cr returned to within 50% above baseline by the time of discharge in most patients (n = 239, 80%), except for patients who received KRT (n = 23, 46%). Nearly half of the patients (n = 143, 48%) were admitted on an angiotensin-converting enzyme inhibitor, angiotensin receptor blocker, or angiotensin receptor-neprilysin inhibitor, and 38% (n = 115) of patients were discharged on these medications. At the time of admission, 52 patients (17%) were taking nonsteroidal anti-inflammatory drugs and 29 (10%) patients were discharged on them.
Discharge Summary Quality
The median discharge summary quality composite score for all patients was 4/9 (IQR: 2-6), with similar median scores before and after introduction of the post-AKI clinic (Supplementary Tables S1 and S2) (Table 3). Composite scores were lowest for stage 1 AKI (median score = 2/9, IQR: 2-6) and highest for stage 3 AKI (median score = 5/9, IQR: 2-6). Only 2 out of 300 discharge summaries contained all 9 elements, and both discharge summaries were for patients who received KRT. Few patients (n = 10, 3%) were missing all 9 elements; however, 6 of these patients had stage 2 or 3 AKI (Figure 1).
Discharge Summary Quality.

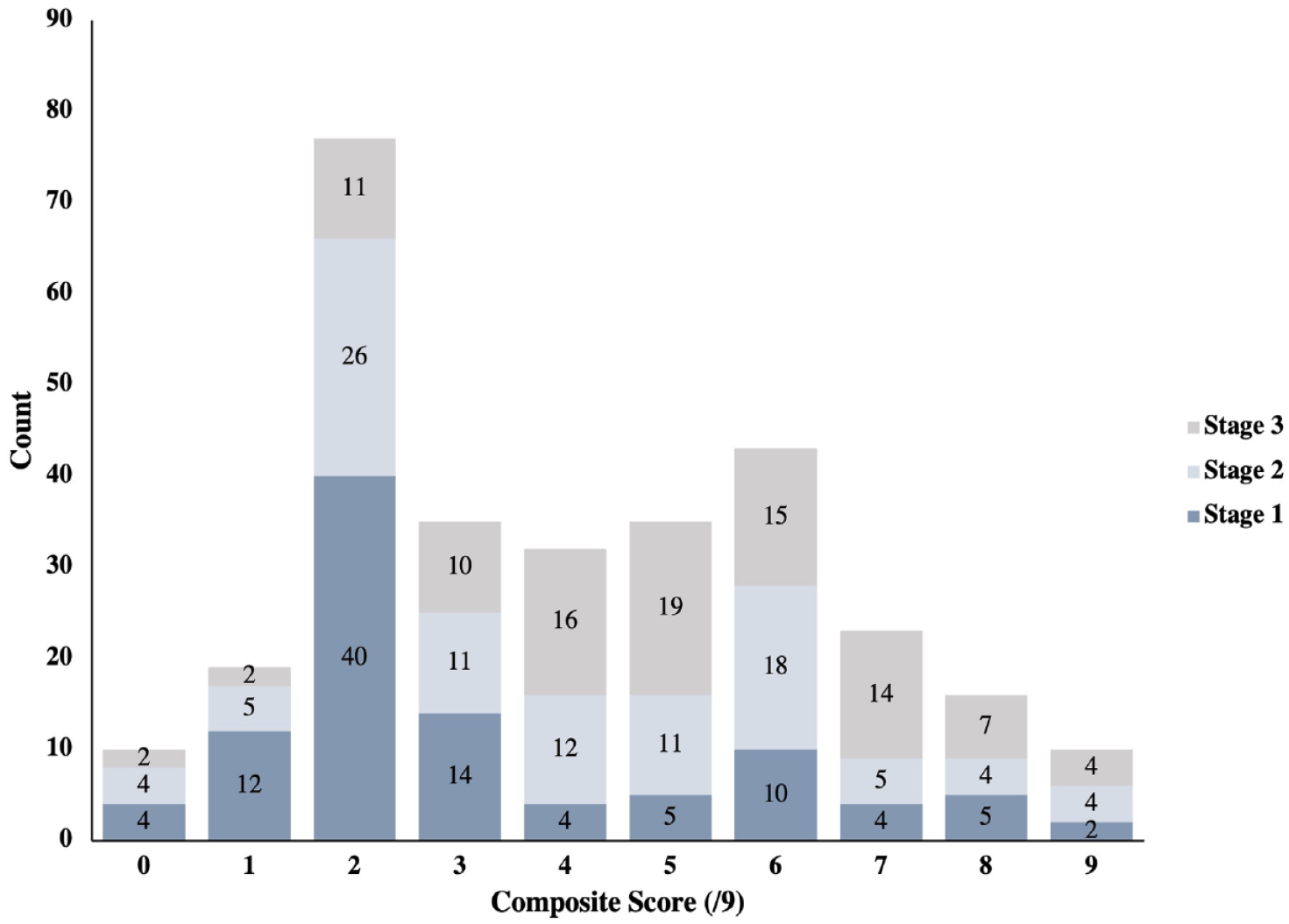
Discharge summary quality composite score distribution stratified by AKI stage.
The most commonly missing items were the baseline Cr and AKI-specific medication and lab recommendations. Among patients with stage 3 AKI (both with and without KRT), fewer than 50% of discharge summaries included information on baseline, peak, and discharge Cr, and AKI-specific medication and lab recommendations.
Predictors of Discharge Summary Quality
In the univariate analysis, elements associated with a discharge summary composite score ≥4/9 included stage 2 (odds ratio [OR]: 2.74; 95% confidence interval [CI]: 1.53, 4.90) and stage 3 (OR: 7.00; 95% CI: 3.76, 13.05) AKI relative to stage 1 AKI; intrarenal (OR: 4.61; 95% CI: 2.78, 7.66) and postrenal/obstructive (OR: 2.92; 95% CI: 1.01, 8.45) etiologies relative to prerenal etiologies; inpatient nephrology consultation (OR: 8.15; 95% CI: 3.98, 16.72); increased baseline Cr per 25 μmol/L (OR: 1.28; 95% CI: 1.09, 1.50); and discharge Cr 50% to <100% (OR: 2.19; 95% CI: 1.02, 4.72) and ≥100% (OR: 5.04; 95% CI: 1.86, 13.71) above baseline relative to discharge Cr <50% above baseline (Table 4). Hospital-acquired AKI was associated with a lower discharge summary composite score compared with community-acquired AKI (OR: 0.50; 95% CI: 0.29, 0.85). There was no association between discharge summary quality and the years before (2015-2016, referent) and after (2018-2019, OR: 0.92; 95% CI: 0.59, 1.45) introduction of a post-AKI clinic in 2017.
Univariate and Multivariable Logistic Regression for Predictors of Discharge Summary Quality Score ≥4/9.

Removed from multivariable model due to significant collinearity with AKI stage.
After multivariable adjustment, the elements associated with a discharge summary composite score ≥4/9 included stage 2 (aOR: 2.57; 95% CI: 1.35, 4.91) and stage 3 (aOR: 3.36; 95% CI: 1.56, 7.22) AKI relative to stage 1 AKI; intrarenal etiologies (aOR: 2.32; 95% CI: 1.26, 4.27) relative to prerenal etiologies; and increased baseline Cr per 25 μmol/L (aOR: 1.27; 95% CI: 1.03, 1.56). We excluded inpatient nephrology consultation from the multivariable analysis due to substantial collinearity with AKI stage (Pearson χ2 = 110.3,
Removing patients who received KRT produced similar results for AKI stage, intrarenal etiologies and baseline Cr (Supplementary Table S3), with a discharge Cr 50% to <100% above baseline now associated with a composite score ≥4/9 (aOR: 2.98; 95% CI: 1.09, 8.15). In this model, male sex was also associated with a lower composite score relative to female sex (aOR: 0.53; 95% CI: 0.28, 0.99).
Discussion
Our study found that the quality of discharge summaries in patients hospitalized with AKI is generally poor, with 57% of discharge summaries deficient in more than half of interrogated AKI care elements. This quality of care gap persisted even after introduction of a post-AKI clinic in 2017. We also identified several factors in our multivariable model that predicted higher quality discharge summaries in AKI survivors, including baseline serum Cr, AKI severity, and ATN and other intrarenal etiologies for AKI. These results highlight that there is a major opportunity to improve discharge communication after an episode of AKI and to evaluate whether better communication during transitions of care can improve clinical and patient-centered outcomes.
Our findings are consistent with other studies limited by smaller sample size or restricted to critically ill patients.15-17 Greer and colleagues randomly reviewed 75 discharge summaries, finding over 50% lacked information on AKI occurrence, etiology, severity, and specific follow-up recommendations for investigations or management. 15 In their multivariable analysis, only stage 3 AKI and nephrology consultation were associated with completion of more discharge summary elements. Allen and colleagues reviewed discharge summaries in a cohort of 50 patients referred to the inpatient nephrology service at a tertiary care center in the United Kingdom. 16 While AKI was mentioned in most discharge summaries, less than 50% included information on baseline and discharge Cr, AKI severity and etiology, or follow-up recommendations. Choon and colleagues reviewed the discharge summaries of 91 patients who were critically ill and received KRT, noting 32% of discharge summaries contained all 4 prespecified elements (occurrence of AKI, receipt of KRT, recommendation to monitor kidney function, and recommendation to refer for nephrology follow-up). 17 They also identified that higher discharge Cr and dialysis dependence at discharge were associated with complete discharge summaries. Therefore, our data reinforce these gaps in discharge summary communication in a larger and more representative population of patients with AKI across different AKI stages, time periods, and hospital settings.
We showed that the gap in serum Cr reporting on discharge summaries applied even to patients with severe stage 3 AKI and after introduction of a post-AKI clinic that may have increased awareness of AKI and its long-term consequences among hospital providers. This suggests suboptimal discharge summaries are not only related to underrecognition of AKI but also possibly system factors, such as AKI often not being the primary reason for admission and lack of formal education on AKI discharge summary composition. Furthermore, our predictive model found that baseline kidney function, AKI severity, and etiology of AKI are associated with discharge summary quality, along with inpatient nephrology consultation in the univariate analysis. Along with these factors, discharge Cr 50% to <100% above baseline was associated with discharge summary quality when patients receiving KRT were removed from the cohort. A common thread between baseline CKD, AKI severity, higher discharge Cr, and intrarenal AKI etiology is that they increase either the absolute value of serum Cr and its likelihood of recognition or the frequency of nephrology consultation. For example, patients with baseline CKD have been shown to be less likely to have a missed AKI diagnosis and more likely to have nephrology involvement for AKI management.27,28 Similarly, patients with intrarenal AKI diagnoses have less AKI unrecognized in the hospital than patients with prerenal or postrenal AKI (missed diagnosis rate of 27% versus 86% and 43%, respectively). 27 As inpatient nephrology consultation is infrequent for most patients with AKI, our data emphasize that increased attention is particularly needed to improve discharge summary quality for high-risk patients with less severe or non-intrarenal AKI, such as patients with acute kidney disease or heart failure.29,30
As gaps in post-AKI discharge summaries have now been found in Canada, the United Kingdom, and the United States, evidence-based QI strategies are clearly needed to address the system-based issues that contribute to suboptimal discharge communication in this patient population. For example, Nye and colleagues found that poor communication in AKI discharge summaries was related to inadequate time for completion, lack of understanding of the prognostic significance of AKI, and underrecognition that most post-AKI care is provided by primary care. 24 Accordingly, they complemented physician education with AKI drug chart stickers, automated reminder messages, and AKI discharge summary templates. These interventions led to a 24% increase (11%-35%) in completion of the AKI section of discharge summaries. Similarly, Reschen and colleagues found that education events, AKI alerts with an auto-populated AKI template in the discharge summary, and a forcing function that mandated completion of the AKI template for discharge summary submission improved AKI aftercare information from 22% to 92%. 31 Of note, the largest increase in completion (40%-92%) only occurred once the AKI template and forcing function were added. Our data suggest that these QI efforts are particularly needed for patients with normal baseline kidney function who are not seen by nephrology during their acute hospitalization, as many of these discharge summaries either do not mention the occurrence of AKI or lack direction on post-AKI care.
Strengths of our work include its large sample size relative to other discharge summary studies and the purposeful sampling of patients across all stages of AKI. We also included a broad population of patients across all levels of care, including the ward and ICU. Baseline kidney function was available for all patients, and chart audits were completed by multiple reviewers to improve reproducibility. Last, we were able to assess the quality of discharge summaries in an environment that prioritizes post-AKI care, with comparisons before and after introduction of a post-AKI clinic.
There are some limitations to our study. We did not have outpatient follow-up data available to determine if there was any association between the quality of discharge summaries and relevant processes and outcomes, such as follow-up Cr testing, nephrotoxin avoidance or rehospitalizations. We did not assess the impact of admitting specialty on discharge summary quality, as most discharge summaries at our institution are written by junior trainees who are often rotating through specialties different from their primary area of training, which limits the utility of distinguishing between medical and surgical specialties. Also, the discharge summary elements included in our composite score have not been formally validated by nephrologists or PCPs nor shown to be predictors of short-term and long-term outcomes; however, the items selected are consistent with previous literature examining discharge summary quality in both AKI and non-AKI populations.13,15-18,23,32-37 The single-center study design limits generalizability, as other centers may have unique discharge summary tools (eg, electronic templates) that allow easier inclusion of AKI-relevant elements (eg., baseline serum Cr). However, our results are consistent with other literature from the United Kingdom and the United States, suggesting low discharge summary quality in patients with AKI persists across different health care settings and infrastructures.15-17
We have demonstrated that among 300 survivors of AKI, more than half of discharge summaries were missing information on AKI etiology and severity, baseline and discharge Cr, and recommendations for AKI-specific follow-up labs and medication management. While baseline kidney function, ATN and other intrarenal etiologies, and stage 2-3 AKI were associated with higher quality discharge summaries, more than 50% of discharge summaries in patients who received KRT were still missing fundamental clinical information vital for postdischarge AKI care. Since most discharge summaries for patients with AKI list the PCP to follow-up with patients, QI strategies are needed to improve discharge communication post-AKI, particularly in high-risk patients with less severe AKI who are more likely to have missing discharge summary AKI elements. Furthermore, feedback from outpatient providers is also critical to optimize inpatient-to-outpatient transitions of care and related communications. Once these goals are achieved, subsequent work should determine whether higher quality discharge summaries are associated with improved clinical and patient-centered outcomes following AKI.
Supplemental Material
sj-doc-1-cjk-10.1177_20543581231199018 – Supplemental material for The Quality of Discharge Summaries After Acute Kidney Injury
Supplemental material, sj-doc-1-cjk-10.1177_20543581231199018 for The Quality of Discharge Summaries After Acute Kidney Injury by Cameron Giles, Milica Novakovic, Wilma Hopman, Erin F. Barreto, William Beaubien-Souligny, Peter Birks, Javier A. Neyra, Ron Wald and Samuel A. Silver in Canadian Journal of Kidney Health and Disease
Footnotes
Author Contributions
Study concept and design: CG, SAS; acquisition, analysis, or interpretation of data: all authors; drafting of the manuscript: CG, MN, SAS; critical revision of the manuscript for important intellectual content: all authors; statistical analysis: CG, WH, SAS; study supervision: SAS. All authors approved the final version of the submitted manuscript. We certify that this manuscript nor one with substantially similar content has been published or is being considered for publication elsewhere, except in abstract form.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form at ![]() . SAS has received speaking fees from Baxter. JAN has received support from Baxter USA/Mexico and Leadiant Biosciences. RW has received unrestricted grant support and speaking fees from Baxter. The remaining authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work.
. SAS has received speaking fees from Baxter. JAN has received support from Baxter USA/Mexico and Leadiant Biosciences. RW has received unrestricted grant support and speaking fees from Baxter. The remaining authors declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SAS and WBS were supported by a Kidney Research Scientist Core Education and National Training (KRESCENT) Program New Investigator Award (co-funded by the Kidney Foundation of Canada, Canadian Society of Nephrology, and Canadian Institutes of Health Research). EFB is supported by grants from the NIH (K23AI143882) and AHRQ (R03HS028060). JAN is supported by grants from NIDDK (R01DK128208, U01DK129989, and P30 DK079337). WBS is supported by the Fonds de Recherche du Québec en Santé. The funding sources had no role in study design; data collection, analysis, or interpretation; writing the report; or the decision to submit the report for publication. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
