Abstract
Background:
Acute kidney injury (AKI) is associated with increased mortality and dialysis in hospitalized patients but has been little explored in the emergency department (ED) setting.
Objective:
The objective of this study was to describe the risk factors, prevalence, management, and outcomes in the ED population, and to identify the proportion of AKI patients who were discharged home with no renal-specific follow-up.
Design:
This is a retrospective cohort study using administrative and laboratory databases.
Setting:
Two urban EDs in Vancouver, British Columbia, Canada.
Patients:
We included all unique ED patients over a 1-week period.
Methods:
All patients had their described demographics, comorbidities, medications, laboratory values, and ED treatments collected. AKI was defined pragmatically, based upon accepted guidelines. The cohort was then probabilistically linked to the provincial renal database to ascertain renal replacement (transplant or dialysis) and the provincial vital statistics database to obtain mortality. The primary outcome was the prevalence of AKI; secondary outcomes included (1) the proportion of AKI patients who were discharged home with no renal-specific follow-up and (2) the combined 30-day rate of death or renal replacement among AKI patients.
Results:
There were 1651 ED unique patients, and 840 had at least one serum creatinine (SCr) obtained. Overall, 90 patients had AKI (10.7% of ED patients with at least one SCr, 95% confidence interval [CI], 8.7%-13.1%; 5.5% of all ED patients, 95% CI, 4.4%-6.7%) with a median age of 74 and 70% male. Of the 31 (34.4%) AKI patients discharged home, 4 (12.9%) had renal-specific follow-up arranged in the ED. Among the 90 AKI patients, 11 died and none required renal replacement at 30 days, for a combined outcome of 12.2% (95% CI, 6.5%-21.2%).
Limitations:
Sample sizes may be small. Nearly half of ED patients did not obtain an SCr. Many patients did not have sequential SCr testing, and a modified definition of AKI was used.
Keywords
What was known before
There is a scarcity of data on the prevalence, management, and outcomes of emergency department patients with acute kidney injury.
What this adds
Overall, 5.5% of all emergency department patients had acute kidney injury. One-third of acute kidney injury patients were discharged home, the majority with no renal-specific follow-up.
Background
Acute kidney injury (AKI) is a reduction in kidney function that is characterized by increased serum creatinine (SCr) concentration or reduced urine output. 1 AKI is typically associated with a number of acute medical conditions such as dehydration, infection, heart failure, or nephrotoxic medication exposure, typically superimposed upon risk factors such as age, diabetes, high blood pressure, cardiovascular disease, or chronic kidney disease (CKD).1-7 In the last 2 decades, the incidence of AKI has increased from 322 to 522 per 100 000 person-years.8-11
AKI patients who survive their initial insult are at substantially elevated risk of progression to CKD, and as evidence-based care 12 can ameliorate such sequelae, appropriate identification and treatment of AKI patients is paramount. The prevalence, risk factors, and outcomes of AKI have been characterized in a number of acute care populations,13-32 and it is estimated that one-third of all AKI episodes acquired in the community may be identified in the emergency departments (EDs). 33 In ED patients who undergo contrast-enhanced computed tomography (CT), the risk of AKI may be up to 11%. 34
However, AKI has been little studied in a general ED population. The prevalence, demographics, risk factors, and ED management are unknown in this population. Given that there are 130 million annual ED visits in the United States, 35 even a low proportion of ED patients with AKI could represent a substantial opportunity to study and potentially improve overall renal care. The goals of this study were to (1) identify the proportion of ED patients with AKI, (2) characterize the comorbidities of the group, and (3) describe the ED management, including follow-up.
Methods
Study Design and Setting
This was a retrospective cohort study at 2 Canadian university–affiliated teaching EDs that share a common database. St Paul’s Hospital is an inner-city referral center with 78 000 annual ED visits during the study period; it is a provincial renal center with transplant and dialysis capabilities. The nephrology service is typically on-call for dialysis-related emergencies, and patients with stable renal issues are referred to the internal medicine service. Although the site receives penetrating trauma, patients with blunt trauma are generally not transported by ambulance to this site. Mount Saint Joseph Hospital is a community hospital with 27 000 yearly visits and has a general internal medicine ward but no dialysis; a nephrologist is available by telephone consultation. Both hospitals see primarily adult patients, and sick children are typically referred to the local specialty pediatric hospital. Patients were managed at the discretion of the emergency physician, including all investigations, therapies, and consultations. At both institutions, SCr is measured by the Roche Hitachi 917 enzymatic assay (Roche Diagnostics, Laval, Quebec, Canada). The ethics review board of Providence Health Care and the University of British Columbia approved this study.
Patient Selection
From January 1 to 7, 2014, all consecutive residents of British Columbia who attended either ED were identified by their unique personal health number and included. To satisfy the assumption of independence, only the first visit was analyzed. Since 1999, the 2 sites have shared a database, which records demographics, chief complaints, and a digital order system (Sunrise Clinical Manager, 4.0, Eclipsys Solutions/Allscripts, Chicago, Illinois). This records all diagnostic, therapeutic, and consultation orders, all laboratory investigations and results, and all hospital records.
Medical Record Review
We followed accepted criteria for medical record review36-38 on patients with and without an ED SCr. Three trained staff emergency medicine reviewers and one senior medical student, who were aware of study purpose but blinded to 30-day outcomes, independently abstracted charts onto standardized electronic spreadsheets (Microsoft Excel 2011; Microsoft Corporation, Redmond, Washington) to document initial vital signs, comorbidities, and investigations. Furthermore, prior laboratory results, outpatient clinic notes, and ED and hospital discharge summaries were scrutinized. ED management was recorded, including administration of oral or intravenous fluids, and nephrotoxic agents such as contrast media, nonsteroidal anti-inflammatories, aminoglycosides, lithium, rifampin, statins, diuretics, allopurinol, and phenytoin. The nursing record provided all vital signs, as well as fluid and medication orders.
The key comorbidity of CKD, which can be difficult to ascertain in the ED, especially in early stages, was defined as having (1) a prior notation of CKD and proteinuria, albuminuria, or urinary casts, and an estimated glomerular filtration rate (eGFR) less than 90 mL/min/1.73 m2, or (2) having a prior eGFR less than 60 mL/min/1.73 m2 that did not appear to be related to a previous AKI episode.39,40 (This would have been documented during a prior admission.) Although this method is imperfect and may underestimate stage 1 and 2 CKD, it was applied consistently across the entire cohort. For patients who were not admitted to hospital, the electronic discharge summary was scrutinized for any further outpatient recommendations including medication changes, and follow-up investigations or consultations.
Reviewers were trained on the first 50 charts and submitted data at regular intervals, which were examined for errors such as single-digit SCr values. Furthermore, the primary investigator reviewed charts of all patients suspected of an AKI episode, including follow-up instructions for discharged patients. Missing or discrepant data were reconciled at regularly scheduled meetings. A second reviewer independently assessed 10% of the first reviewers’ charts, and interobserver reliability was calculated for all variables.
Outcomes
All outcomes were determined a priori. The primary outcome was the prevalence of AKI; this was obtained by dividing the number of AKI patients by the overall number of ED patients. As the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines typically define AKI via sequential SCr tests, 12 and many ED patients only obtained a single test, we relied upon an ED-specific algorithm to ascertain AKI. Briefly, we first considered the duration of the episode, and symptoms were required to start up to a week prior to ED presentation. Second, we ascertained ED SCr values and compared them with baseline levels obtained in the past year (where available) or values projected by Bellomo12,41 if those were not available.
To reduce the possibility of a CKD misclassified as an AKI episode, we reviewed all patients in whom a second SCr was obtained within 48 hours, and if the change between the 2 SCr values was at least 26.5 µmol/L, the patient was considered to have an AKI episode (see Box 1). If no second SCr was obtained in 48 hours, then the following decision tree was used: (1) If there was no prior SCr, the patient was considered to have AKI and (2) if there was a prior SCr, and the difference was greater than 26.5 µmol/L, the patient was considered to have AKI. All controversial cases were referred to 2 independent adjudicators (a nephrologist and an emergency physician) who were blinded to study hypothesis and outcomes. All AKI patients were staged: Stage 1 entailed SCr of 150% to 200% of baseline, stage 2 was 200% to 300% of baseline, and stage 3 was greater than 300% of baseline. 12
Ascertainment of Acute Kidney Injury
As a definition of AKI is a 50% increase in baseline SCr within 7 days, we required that potential ED AKI patients have their symptoms start within 7 days of presenting to the ED. (Therefore a patient with a 1-month history of weakness and an elevated SCr was not considered to have AKI; conversely, a patient with 2 days of vomiting and diarrhea with an elevated SCr was a potential AKI candidate.) Extending the above definition, all patients with symptoms less than 7 days and an ED SCr greater than 50% of their baseline were provisionally considered to have AKI. However, as very few patients would have had an SCr within 7 days prior to the ED visit, we relied upon estimates of the baseline developed by Bellomo (41 and below) and described in the KDIGO guidelines, 12 providing approximate values based upon age, sex, and ethnicity. (Please note that this had the potential to exclude some elderly patients with chronically low SCr.)

Source. Adapted from Kidney Disease: Improving Global Outcomes (KDIGO). 12
However, as there is overlap between slowly worsening CKD and an acute AKI, especially if both are subtle, we anticipated that some of these potential AKI patients would have prior CKD or a baseline different from that described by Bellomo. 41 This was handled depending upon the presence of a second SCr: (1) All patients who received a second SCr within 48 hours (including all admitted patients) and whose subsequent SCr demonstrated a minimum 26.5 µmol/L change were considered to have AKI. (2) For patients who were discharged home from the ED and thus did not receive a follow-up SCr, all prior serum SCr dating to 1999 were reviewed, with results obtained in the past year taking precedence. Of the 840 patients with an SCr obtained at the index visit, 731 (87.0%) had an SCr since 1999, and 588 (70.3%) had an SCr in the past year. This value was then taken as the baseline. For patients who had ED SCr that was at least 50% greater than an SCr obtained within 1 year, AKI was considered to have occurred. Of the 105 patients with potential AKI, 94 (89.5%) had an SCr overall, and 89 (84.8%) had an SCr in the past year. Realizing that CKD is generally a slowly progressive illness, 12 a nephrologist—blinded to study purpose, hypothesis, and outcomes—reviewed all patients who had an SCr more than a year ago to ascertain AKI status.
We anticipated that a subset of AKI patients might be candidates for safe discharge. Specific KDIGO AKI guidelines 12 may be beyond the scope of the ED, but recommendation 2.3.4 advises that AKI patients have 3-month follow-up to ascertain occult CKD development, and we felt that either would be appropriate: (1) arranging follow-up with a primary care physician, internist, nephrologist, or urologist or (2) recommending a repeat SCr. The secondary outcome was thus the proportion of AKI patients discharged home that had appropriate documented follow-up instructions.
As renal replacement therapy (RRT; new dialysis or kidney transplant) and death are 2 relevant AKI outcomes, 3 the tertiary outcome was the combined rate of such events at 30 days. To ascertain ED revisits, the cohort of discharged patients was linked to the Six-hospital Vancouver Coastal Health (VCH) regional ED database; to obtain new dialysis or renal transplant, the full cohort was linked to the British Columbia renal database; to determine mortality, the entire cohort was linked to the provincial vital statistics database.
Sample Size
To provide an initial estimate of ED AKI rates and study risk factors and basic epidemiology, we wished to obtain approximately 100 AKI patients. While the rate of AKI varies substantially among the population studied,13-32 the rate among general medical populations appears to be 25%.31,32 Given that the historical admission rate from the 2 study EDs is approximately 20%, we estimated that 5% of ED patients would have AKI. To obtain 100 AKI patients, we would require 2000 consecutive ED patients, and given that the 2 sites have a combined 105 000 annual patients, obtaining a single week’s worth of data would be sufficient.
Primary Data Analysis
Microsoft Excel 2011 was used for analysis. Variables were presented as means (and standard deviation) if normally distributed, and medians (with interquartile ranges) if nonnormally distributed.
Results
Study Flow
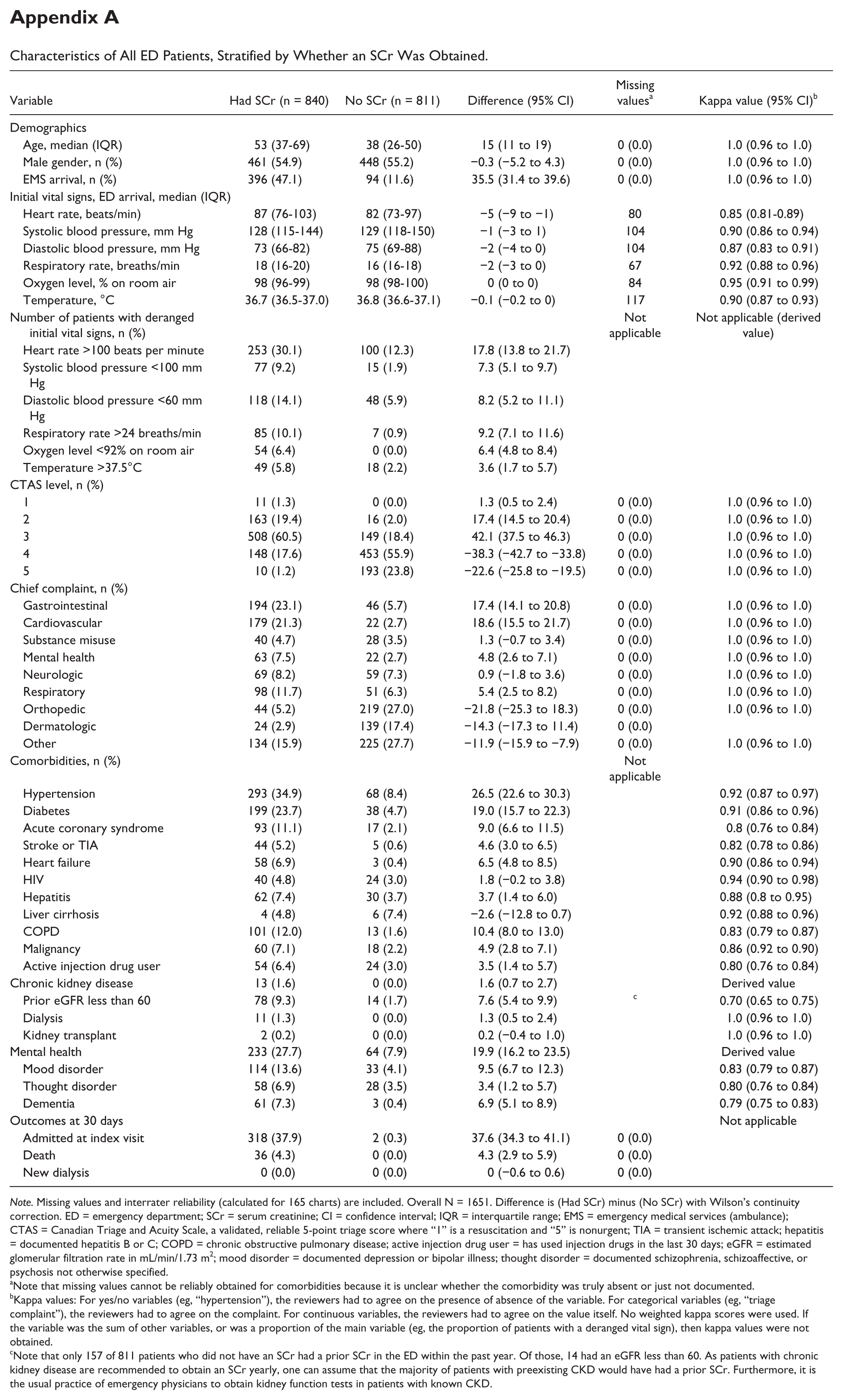
Figure 1 shows that in the 1-week study period, the 2 EDs had 1794 visits from 1651 unique patients, and 840 (50.9%) had at least one SCr in the ED. Appendix A shows baseline characteristics and 30-day outcomes for patients who had an SCr versus those who did not, along with missing values and kappa values for all variables. Patients who obtained an SCr were substantially different in almost every respect from those who did not have one. Baseline characteristics for ED patients who had at least one SCr are shown in Table 1. Five patients required adjudication, and 4 were considered to have AKI.
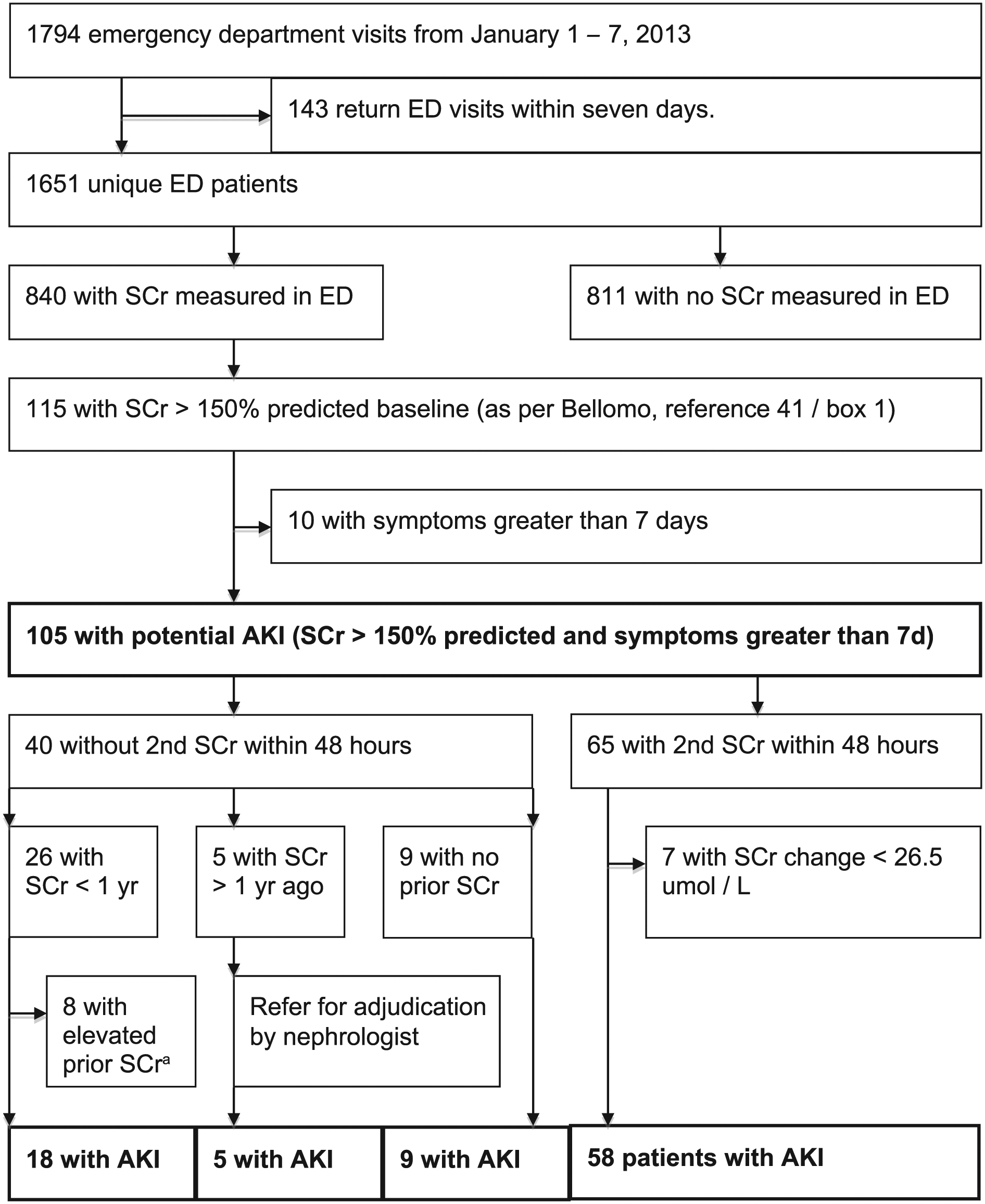
Study flow diagram.
Characteristics of Emergency Department Patients With at Least One Serum Creatinine, Stratified by Presence or Absence of AKI Episode (n = 840).
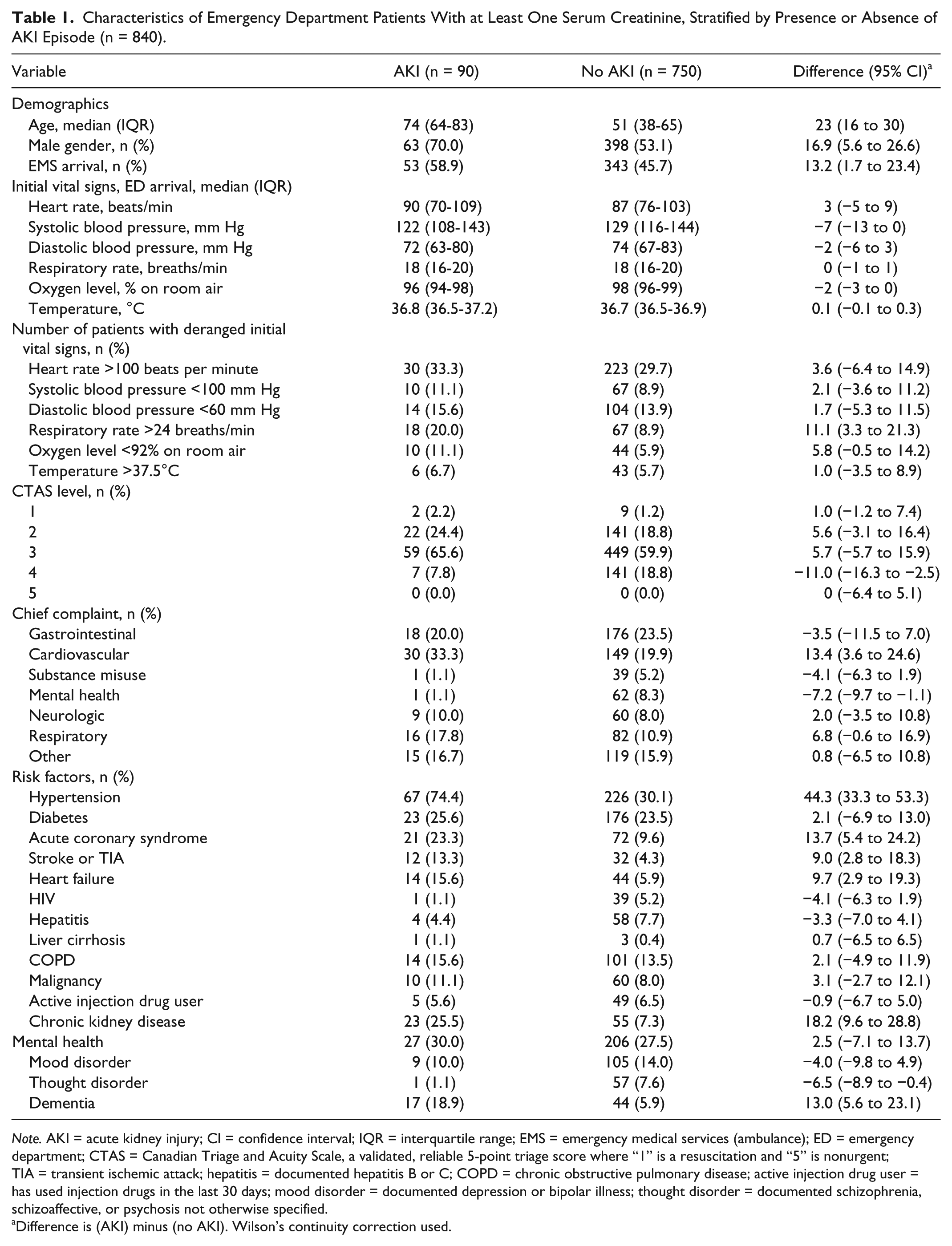
Note. AKI = acute kidney injury; CI = confidence interval; IQR = interquartile range; EMS = emergency medical services (ambulance); ED = emergency department; CTAS = Canadian Triage and Acuity Scale, a validated, reliable 5-point triage score where “1” is a resuscitation and “5” is nonurgent; TIA = transient ischemic attack; hepatitis = documented hepatitis B or C; COPD = chronic obstructive pulmonary disease; active injection drug user = has used injection drugs in the last 30 days; mood disorder = documented depression or bipolar illness; thought disorder = documented schizophrenia, schizoaffective, or psychosis not otherwise specified.
Difference is (AKI) minus (no AKI). Wilson’s continuity correction used.
Main ED-Based Results
Overall, 90 patients had an AKI, for a prevalence of 5.5%. (95% confidence interval [CI], 4.4%-6.7%). (The prevalence among the 840 patients who had obtained at least one SCr was 10.7% [95% CI, 8.7%-13.1%].) AKI patients had a median age of 74, versus 51 for non-AKI patients; initial vitals and triage complaints were similar. AKI patients appeared to have a significantly higher proportion of hypertension, coronary artery disease, prior stroke or transient ischemic attack (TIA), heart failure, CKD, and dementia. Of the AKI patients, 52 (57.8%) were stage 1, 29 (32.2%) were stage 2, and the remaining 9 (10.0%) were stage 3.
ED management was as follows: 86 patients received crystalloid, and 6 received colloids. No patients received nephrotoxic antibiotics, and none underwent CT with contrast agents. Seven patient received aspirin, and 8 received loop diuretics for presumed heart failure. To illustrate the ED epidemiology of AKI, ED diagnoses are provided in Table 2.
Emergency Department Diagnoses of Patients With Acute Kidney Injury.

Note. There is potential for overlap among diagnoses. Only the primary diagnosis was used.
For the 31 (34.5%) AKI patients who were discharged home, 20 (67.7%) were male and the median age was 68; 29 (93.5%) had stage 1 AKI. One patient had an SCr level rechecked in the ED. One patient was advised to recheck their SCr in the following week, 2 patients were advised to see their family doctor with no time frame given, and one patient was referred to a urologist; therefore, 4 patients (12.9%) were discharged home with appropriate follow-up. In the following 30 days, 6 patients (19.4%) revisited a regional ED with one patient being admitted to hospital. To further assist in visualizing this cohort, Appendix B details each AKI patient—clinical presentation, medical history, ED treatment and diagnosis, and follow-up—who was discharged home, including requirement for nephrologist adjudication.
Thirty-Day Outcomes
For the 811 patients who did not have an SCr measured, 2 patients were admitted at the index ED visit, while none died or required RRT at 30 days. Patients who had an SCr measured, but did not have AKI, had a 34.5% (259 of 750) admission rate. Twenty-five patients died and none had RRT, for a 30-day outcome of 3.3%. Overall, patients who did not have AKI had a mortality rate of 1.6%.
Of the 90 AKI patients, 59 (65.5%) were admitted to hospital. At 30 days, 11 died (none of whom were discharged at the index ED visit) and none required RRT, for a composite outcome of 12.2% (95% CI, 6.6%-21.2%).
Discussion
In this review of 1651 unique consecutive patients presenting to 2 urban EDs, the prevalence of AKI was 5.5%. Approximately one-third of AKI patients were discharged home, but emergency physicians provided the recommended renal-specific follow-up instructions to only 12.9% of discharged patients. In our cohort, although no patients required RRT, the 30-day mortality rate of AKI patients was 12.2%, a sevenfold increase over patients who did not have AKI.
AKI patients were a median 23 years older than non-AKI patients, more likely to be male, and more likely to arrive by ambulance. Unsurprisingly, comorbidities associated with AKI included hypertension, coronary artery disease, heart failure, prior stroke or TIA, CKD, and dementia. 12
These findings assist clinicians by demonstrating that (1) approximately 5.5% of an undifferentiated ED population may have AKI; (2) older ED patients and those with cardiovascular or chronic kidney disease appear to be at higher risk of AKI; and (3) although some AKI patients may be suitable for discharge home, emergency physicians typically do not provide kidney-specific follow-up instructions. The prevalence of AKI was similar at both hospitals, perhaps indicating that this illness may occur at a similar rate in various settings.
AKI has been characterized in many inpatient populations, including trauma,13-15 sepsis,16-24 burns,25,26 and postcardiac-27-29 and noncardiac30-32 surgery. In these cohorts, AKI is associated with higher inpatient and 30-day mortality, as well as increased dialysis rates and longer hospitalizations; even subtle SCr changes are associated with increased risk of subsequent CKD.42,43
Jones and coworkers described an 11% incidence of AKI in a cohort of ED patients who underwent contrast-enhanced CT, and the 1-year risk-adjusted incidence of adverse cardiovascular events doubled in the AKI group. 34 However, AKI has not been described in an unselected ED population, and the prevalence, demographics, risk factors, and outcomes are unknown in this cohort. In 2010, there were 130 million ED visits in the United States, 35 and even at the lower confidence boundary of our prevalence, 5.7 million annual AKI episodes can be estimated. Even if the vast majority of these patients quickly regain full renal function, this still leaves a large cohort of patients who may be vulnerable to major adverse events including progression to CKD.
Although no specific ED-based AKI guidelines exist, emergency physicians should at least be aware of recommendations based upon the KDIGO standards. 12 First, AKI requires ED recognition and evaluation of cause. 44 Resuscitation should be undertaken with crystalloids but not colloids, 45 although fluid overload can be deleterious. 46 Physicians must realize that AKI is associated with sepsis 47 and that all electrolyte or acid-base derangements should be promptly corrected. 12 Finally, nephrotoxic agents such as aminoglycoside antibiotics or contrast media should be avoided. 48
Currently, automatic hospitalization of AKI patients is not recommended, and many patients can likely be discharged home safely. Our physicians likely made the clinically appropriate decisions when discharging patients but in many cases did not ensure appropriate guideline-based follow-up within 3 months. The may be due to several reasons: Physicians may not have identified an AKI episode, or were unsure how to proceed, or simply failed to document verbal instructions. Progression to CKD typically takes months or years and depends on many factors beyond the scope of the ED, such as regular and appropriate medical follow-up, control of hypertension and diabetes, and adherence to medications. Unfortunately, up to one-third of ED patients may not have a regular primary care provider, 49 and emphasizing kidney-specific follow-up for stable AKI patients is an important task for ED physicians.
Limitations
The goal of this study was to ascertain prevalence of AKI in ED patients, and this was derived in a retrospective cohort at 2 urban Canadian centers that do not typically receive blunt trauma; patient distribution, admission rates, recognition and risk tolerance for AKI, and discharge instructions may vary in other settings. Ideally, this study would be repeated using a larger and more comprehensive ED sample, and over a longer time period, including seasonal variations. Physician decisions to order investigations (including SCr), provide management, or consult specialists were individualized, and uncollected variables may have influenced this. Given that there were less than 100 AKI patients, conclusions regarding epidemiology, ED management, and follow-up must be regarded as exploratory. Approximately half of our patients did not have an SCr obtained, and such patients may have had occult AKI episodes that went uncounted, but given their young age and lack of comorbidities, this number is likely very low.
Current AKI definitions were developed for hospitalized patients receiving sequential renal investigations—not ED patients—and require a change in SCr >26.5 µmol/L (>0.3 mg/dL) over a 48-hour period, or else monitoring of urine output over 6 to 24 hours, and these methods may be more sensitive at ascertaining AKI. 12 Unfortunately, many patients, typically those discharged home, received only a single SCr, and above definitions could not be strictly used; thus, ascertaining AKI, especially subtle episodes, may be difficult. In particular, patients with a baseline SCr lower than predicted by Bellomo12,41 might be misclassified as a non-AKI episode, when in fact they had AKI. However, only few potential AKI patients lacked baseline SCr and 5 cases required adjudication. We feel that our strategy to identify AKI, although varying from recommendations and having potential for misclassification, is clinically sensible given the inherent limitations of ED data. In addition, we have provided explanations of all discharged AKI patients (Appendix B). Because very few ED patients underwent urine dipstick testing, the prevalence of subtle early CKD may have been underestimated.
The outcome of “kidney-specific follow-up within 3 months” is a nongraded KDIGO recommendation but is both clinically relevant and easy to apply in the ED. Some physicians may have given detailed verbal advice, but providing written instructions is the accepted standard of arranging follow-up. Outcomes such as death and RRT are often dependent on many non-ED factors; considering our goal was to merely provide a point estimate of AKI prevalence and 30-day outcomes in ED patients, results were unadjusted. Finally, although we describe clinical epidemiology of ED AKI, study design does not provide insight into individual patient care.
Conclusion
The prevalence of AKI among ED patients was 5.5%. Although one-third of AKI patients were discharged home, most did not have kidney-specific follow-up.
Footnotes
Appendix A
Characteristics of All ED Patients, Stratified by Whether an SCr Was Obtained.
| Variable | Had SCr (n = 840) | No SCr (n = 811) | Difference (95% CI) | Missing values a | Kappa value (95% CI) b |
|---|---|---|---|---|---|
| Demographics | |||||
| Age, median (IQR) | 53 (37-69) | 38 (26-50) | 15 (11 to 19) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Male gender, n (%) | 461 (54.9) | 448 (55.2) | −0.3 (−5.2 to 4.3) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| EMS arrival, n (%) | 396 (47.1) | 94 (11.6) | 35.5 (31.4 to 39.6) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Initial vital signs, ED arrival, median (IQR) | |||||
| Heart rate, beats/min) | 87 (76-103) | 82 (73-97) | −5 (−9 to −1) | 80 | 0.85 (0.81-0.89) |
| Systolic blood pressure, mm Hg | 128 (115-144) | 129 (118-150) | −1 (−3 to 1) | 104 | 0.90 (0.86 to 0.94) |
| Diastolic blood pressure, mm Hg | 73 (66-82) | 75 (69-88) | −2 (−4 to 0) | 104 | 0.87 (0.83 to 0.91) |
| Respiratory rate, breaths/min | 18 (16-20) | 16 (16-18) | −2 (−3 to 0) | 67 | 0.92 (0.88 to 0.96) |
| Oxygen level, % on room air | 98 (96-99) | 98 (98-100) | 0 (0 to 0) | 84 | 0.95 (0.91 to 0.99) |
| Temperature, °C | 36.7 (36.5-37.0) | 36.8 (36.6-37.1) | −0.1 (−0.2 to 0) | 117 | 0.90 (0.87 to 0.93) |
| Number of patients with deranged initial vital signs, n (%) | Not applicable | Not applicable (derived value) | |||
| Heart rate >100 beats per minute | 253 (30.1) | 100 (12.3) | 17.8 (13.8 to 21.7) | ||
| Systolic blood pressure <100 mm Hg | 77 (9.2) | 15 (1.9) | 7.3 (5.1 to 9.7) | ||
| Diastolic blood pressure <60 mm Hg | 118 (14.1) | 48 (5.9) | 8.2 (5.2 to 11.1) | ||
| Respiratory rate >24 breaths/min | 85 (10.1) | 7 (0.9) | 9.2 (7.1 to 11.6) | ||
| Oxygen level <92% on room air | 54 (6.4) | 0 (0.0) | 6.4 (4.8 to 8.4) | ||
| Temperature >37.5°C | 49 (5.8) | 18 (2.2) | 3.6 (1.7 to 5.7) | ||
| CTAS level, n (%) | |||||
| 1 | 11 (1.3) | 0 (0.0) | 1.3 (0.5 to 2.4) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| 2 | 163 (19.4) | 16 (2.0) | 17.4 (14.5 to 20.4) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| 3 | 508 (60.5) | 149 (18.4) | 42.1 (37.5 to 46.3) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| 4 | 148 (17.6) | 453 (55.9) | −38.3 (−42.7 to −33.8) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| 5 | 10 (1.2) | 193 (23.8) | −22.6 (−25.8 to −19.5) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Chief complaint, n (%) | |||||
| Gastrointestinal | 194 (23.1) | 46 (5.7) | 17.4 (14.1 to 20.8) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Cardiovascular | 179 (21.3) | 22 (2.7) | 18.6 (15.5 to 21.7) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Substance misuse | 40 (4.7) | 28 (3.5) | 1.3 (−0.7 to 3.4) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Mental health | 63 (7.5) | 22 (2.7) | 4.8 (2.6 to 7.1) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Neurologic | 69 (8.2) | 59 (7.3) | 0.9 (−1.8 to 3.6) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Respiratory | 98 (11.7) | 51 (6.3) | 5.4 (2.5 to 8.2) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Orthopedic | 44 (5.2) | 219 (27.0) | −21.8 (−25.3 to 18.3) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Dermatologic | 24 (2.9) | 139 (17.4) | −14.3 (−17.3 to 11.4) | 0 (0.0) | |
| Other | 134 (15.9) | 225 (27.7) | −11.9 (−15.9 to −7.9) | 0 (0.0) | 1.0 (0.96 to 1.0) |
| Comorbidities, n (%) | Not applicable | ||||
| Hypertension | 293 (34.9) | 68 (8.4) | 26.5 (22.6 to 30.3) | 0.92 (0.87 to 0.97) | |
| Diabetes | 199 (23.7) | 38 (4.7) | 19.0 (15.7 to 22.3) | 0.91 (0.86 to 0.96) | |
| Acute coronary syndrome | 93 (11.1) | 17 (2.1) | 9.0 (6.6 to 11.5) | 0.8 (0.76 to 0.84) | |
| Stroke or TIA | 44 (5.2) | 5 (0.6) | 4.6 (3.0 to 6.5) | 0.82 (0.78 to 0.86) | |
| Heart failure | 58 (6.9) | 3 (0.4) | 6.5 (4.8 to 8.5) | 0.90 (0.86 to 0.94) | |
| HIV | 40 (4.8) | 24 (3.0) | 1.8 (−0.2 to 3.8) | 0.94 (0.90 to 0.98) | |
| Hepatitis | 62 (7.4) | 30 (3.7) | 3.7 (1.4 to 6.0) | 0.88 (0.8 to 0.95) | |
| Liver cirrhosis | 4 (4.8) | 6 (7.4) | −2.6 (−12.8 to 0.7) | 0.92 (0.88 to 0.96) | |
| COPD | 101 (12.0) | 13 (1.6) | 10.4 (8.0 to 13.0) | 0.83 (0.79 to 0.87) | |
| Malignancy | 60 (7.1) | 18 (2.2) | 4.9 (2.8 to 7.1) | 0.86 (0.92 to 0.90) | |
| Active injection drug user | 54 (6.4) | 24 (3.0) | 3.5 (1.4 to 5.7) | 0.80 (0.76 to 0.84) | |
| Chronic kidney disease | 13 (1.6) | 0 (0.0) | 1.6 (0.7 to 2.7) | Derived value | |
| Prior eGFR less than 60 | 78 (9.3) | 14 (1.7) | 7.6 (5.4 to 9.9) | c | 0.70 (0.65 to 0.75) |
| Dialysis | 11 (1.3) | 0 (0.0) | 1.3 (0.5 to 2.4) | 1.0 (0.96 to 1.0) | |
| Kidney transplant | 2 (0.2) | 0 (0.0) | 0.2 (−0.4 to 1.0) | 1.0 (0.96 to 1.0) | |
| Mental health | 233 (27.7) | 64 (7.9) | 19.9 (16.2 to 23.5) | Derived value | |
| Mood disorder | 114 (13.6) | 33 (4.1) | 9.5 (6.7 to 12.3) | 0.83 (0.79 to 0.87) | |
| Thought disorder | 58 (6.9) | 28 (3.5) | 3.4 (1.2 to 5.7) | 0.80 (0.76 to 0.84) | |
| Dementia | 61 (7.3) | 3 (0.4) | 6.9 (5.1 to 8.9) | 0.79 (0.75 to 0.83) | |
| Outcomes at 30 days | Not applicable | ||||
| Admitted at index visit | 318 (37.9) | 2 (0.3) | 37.6 (34.3 to 41.1) | 0 (0.0) | |
| Death | 36 (4.3) | 0 (0.0) | 4.3 (2.9 to 5.9) | 0 (0.0) | |
| New dialysis | 0 (0.0) | 0 (0.0) | 0 (−0.6 to 0.6) | 0 (0.0) | |
Note. Missing values and interrater reliability (calculated for 165 charts) are included. Overall N = 1651. Difference is (Had SCr) minus (No SCr) with Wilson’s continuity correction. ED = emergency department; SCr = serum creatinine; CI = confidence interval; IQR = interquartile range; EMS = emergency medical services (ambulance); CTAS = Canadian Triage and Acuity Scale, a validated, reliable 5-point triage score where “1” is a resuscitation and “5” is nonurgent; TIA = transient ischemic attack; hepatitis = documented hepatitis B or C; COPD = chronic obstructive pulmonary disease; active injection drug user = has used injection drugs in the last 30 days; eGFR = estimated glomerular filtration rate in mL/min/1.73 m2; mood disorder = documented depression or bipolar illness; thought disorder = documented schizophrenia, schizoaffective, or psychosis not otherwise specified.
Note that missing values cannot be reliably obtained for comorbidities because it is unclear whether the comorbidity was truly absent or just not documented.
Kappa values: For yes/no variables (eg, “hypertension”), the reviewers had to agree on the presence of absence of the variable. For categorical variables (eg, “triage complaint”), the reviewers had to agree on the complaint. For continuous variables, the reviewers had to agree on the value itself. No weighted kappa scores were used. If the variable was the sum of other variables, or was a proportion of the main variable (eg, the proportion of patients with a deranged vital sign), then kappa values were not obtained.
Note that only 157 of 811 patients who did not have an SCr had a prior SCr in the ED within the past year. Of those, 14 had an eGFR less than 60. As patients with chronic kidney disease are recommended to obtain an SCr yearly, one can assume that the majority of patients with preexisting CKD would have had a prior SCr. Furthermore, it is the usual practice of emergency physicians to obtain kidney function tests in patients with known CKD.
Appendix B
Ethics Approval and Consent to Participate
This study was approved by the Research Ethics Board of Providence Health Care and a waiver of consent was accepted.
Consent for Publication
All of the authors have read and provide consent for the publication of this work.
Availability of Data and Materials
Anonymized raw data is available from the corresponding author upon request.
Author Contributions
FS conceived and designed the study, with AL providing substantial input. EG obtained the patient database. FS, JC, BG, and AB conducted the chart review, with AL and EG providing blinded adjudication. FS performed statistical analysis. FS drafted the manuscript, and all authors contributed to its revision, especially AL. FS takes responsibility for the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
