Abstract
Rationale:
Renal tubular acidosis (RTA) is a cause of non-anion gap metabolic acidosis (NAGMA) that is infrequently diagnosed and is due to various underlying etiologies that impair the kidney’s ability to retain bicarbonate or excrete acid. Ibuprofen is an over-the-counter non-steroidal anti-inflammatory medication that is used by patients widely for a variety of reasons. Although it is well known that ibuprofen and other non-steroidal anti-inflammatory drugs may have nephrotoxic effects, the role of ibuprofen as a cause of RTA and hypokalemia is not well recognized.
Presenting Concerns:
A 66-year-old man with chemotherapy-treated lymphoma in remission and ongoing heavy ibuprofen use for chronic pain presented to hospital with a 1-week history of increasing lethargy and otherwise unremarkable review of systems. Investigations showed acute kidney injury, hypokalemia, hyperchloremia, and NAGMA with elevated urinary pH and positive urine anion gap.
Diagnoses:
The final diagnosis of distal RTA secondary to ibuprofen was made after ruling out gastrointestinal bicarbonate loss and additional secondary causes of RTA, including other medications, autoimmune conditions, and obstructive uropathy.
Interventions:
The patient was admitted and treated with intravenous sodium bicarbonate for 24 hours with correction of hypokalemia via oral supplementation. His ibuprofen-containing medication was discontinued.
Outcomes:
His acute kidney injury and electrolyte abnormalities resolved within 48 hours of initiating treatment with concurrent resolution of his lethargy. He was discharged home and advised to stop taking ibuprofen.
Lessons Learned:
We report a case of patient with hypokalemia and NAGMA secondary to ibuprofen and highlight the importance of monitoring for this side effect in patients taking ibuprofen.
What was known before
Non-steroidal anti-inflammatory drugs such as ibuprofen have known nephrotoxic effects and have been reported to be a rare cause of renal tubular acidosis.
What this adds
We report a case of a patient with hypokalemia and acidosis secondary to heavy over-the-counter ibuprofen use and highlight the importance of monitoring for this side effect in patients taking ibuprofen.
Introduction
Non-anion gap metabolic acidosis (NAGMA) is typically due to renal or external wasting of bicarbonate. The inability to properly acidify the urine is seen in certain forms of renal tubular acidosis (RTA) with various etiologies, including medications.
Presenting Concerns
A 66-year-old man presented to hospital on day 0 (Figure 1) with a week of increasing lethargy and confusion. His medical profile was notable for diffuse large B-cell lymphoma diagnosed 18 months prior and treated with 6 cycles of rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone chemotherapy with recent imaging showing no evidence of recurrent/persistent disease. He had a history of chronic pain from lumbar degenerative disk disease.

Timeline of relevant clinical events, starting prior to patient’s presentation to hospital and ending at discharge.
The patient was admitted to hospital 5 months prior for persistent diarrhea where he was found to have mixed anion gap metabolic acidosis and NAGMA with decreased level of consciousness requiring intubation and intensive care admission. His presentation was felt to be due to starvation ketoacidemia with coexisting diarrhea-associated NAGMA, and he improved with supportive management.
Further history revealed he had been using tablets containing ibuprofen 200 mg—methocarbamol 500 mg per tablet (Robax Platinum) to treat his chronic back pain with 80 tablets used the month prior. Interestingly, his frequent use of ibuprofen-methocarbamol started just before his previous admission to hospital. His other medications were cetirizine and vitamins. He denied the use of illicit substances or herbal supplements. He denied having recent diarrhea or emesis.
Clinical Findings
Physical examination revealed blood pressure 130/70 mm Hg, heart rate 70 beats per minute, oxygen saturation 95% on room air, and respiratory rate 20 breaths per minute. He was afebrile, alert, oriented, but appeared lethargic. There was no evidence of pedal edema, and his mucosal membranes were dry. The remainder of his cardiovascular, abdominal, and skin examinations were unremarkable.
Diagnostic Focus and Assessment
Most recent outpatient bloodwork from 3 months prior showed normal electrolytes and renal function (serum creatinine 91 μmol/L with estimated glomerular filtration rate 76 mL/min/1.73 m2). He now had hypokalemia (3.2 mmol/L), hyperchloremia (119 mmol/L), acidemia with bicarbonate of 9 mmol/L, and venous blood gas showing pH of 6.97. His anion gap was calculated to be 15 (with serum albumin 38 g/L) with a delta-delta ratio ([anion gap-12]/[24-serum bicarbonate]) of 0.2, suggesting coexisting NAGMA. His serum beta-hydroxybutyrate was mildly elevated (0.4 mmol/L) and urinalysis revealed urine pH of 5.5 with 1.0 proteins but otherwise unremarkable. His serum creatinine was elevated at 144 μmol/L from baseline.
His urine electrolytes drawn before any management showed urine sodium of 33 mmol/L, potassium 37.5 mmol/L, and chloride 51 mmol/L with urine anion gap of 20 mmol/L. With these values and presumed negligible urine glucose given his urinalysis, his calculated urine osmolality was 459 mmol/kg. Using a measured urine osmolality of 401 mmol/kg, his urine osmolar gap was 58 mmol/kg. Using a measured serum and urine osmolality of 312 and 401 mmol/kg, respectively, his transtubular potassium gradient (TTKG) was 9. His urine potassium-creatinine ratio was 5.0. Overall, these studies suggested impairment in renal ammonium excretion and renal potassium wasting. We do acknowledge that recent literature has been contentious regarding the use of TTKG. 1 A comprehensive list of his initial laboratory measurements is given in Figure 2.
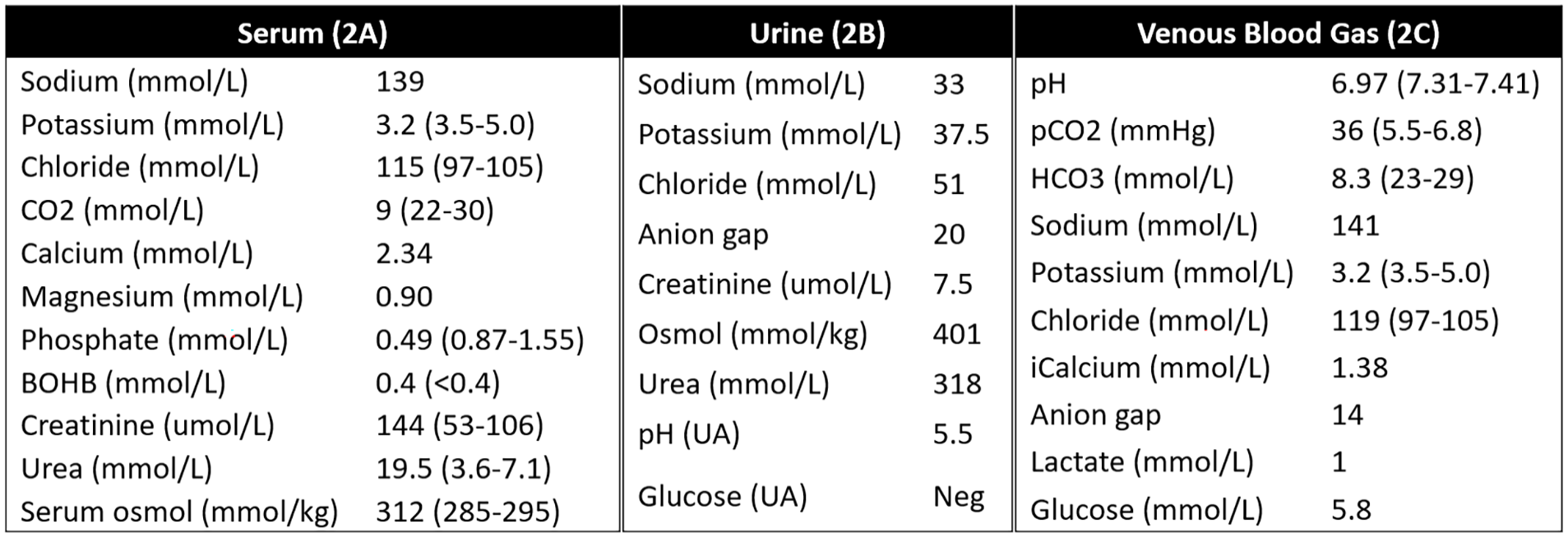
Laboratory values at presentation.
Workup for other potential causes of RTA included extractable nuclear antigen antibody panel, anti-nuclear antibody panel, serum electrophoresis, and serum calcium levels, which were benign. Thorough medication review did not reveal any other potential culprit agents for RTA. Although lymphoma may invade the kidneys and cause proximal tubular dysfunction, our patient did not have acidemia prior to ibuprofen use and was stable from a lymphoma perspective since his initial treatment. While cyclophosphamide can also cause proximal tubular dysfunction, 1 the lack of glucosuria and normal serum urate (355 μmol/L) suggested otherwise. He had no recent diarrhea. Given his hypokalemic NAGMA in the setting of heavy ibuprofen-methocarbamol use with no other identifiable causes, we felt his NAGMA was most likely secondary to ibuprofen use.
Therapeutic Focus and Assessment
He was administered an intravenous infusion of 150 mEq sodium bicarbonate in 1 L of D5W at 100 mL/h for 24 to 48 hours with 40 mEq of oral potassium chloride daily for hypokalemia. His ibuprofen-methocarbamol was discontinued.
Follow-up and Outcomes
His acute kidney injury resolved within 24 hours with serum creatinine returning to 96 mmol/L. Within 48 hours, his serum bicarbonate normalized to 22 mmol/L and his hypokalemia resolved. He was monitored in hospital for another 2 days with no recurrence of his hypokalemia or acidemia. On discharge, he was advised to stop taking ibuprofen going forward. Four months after discharge, his lab work demonstrated resolution of the acidemia and hyperkalemia with sodium of 144 mmol/L, potassium of 4.7 mmol/L, and bicarbonate of 22 mmol/L.
Discussion
We report a case of a patient who developed impairment in renal acidification in the context of significant ibuprofen use. Further workup revealed he had developed a hypokalemic RTA without any evidence of gastrointestinal bicarbonate loss or another cause of RTA. This case supports a diagnosis of RTA due to ibuprofen-induced carbonic anhydrase II (CAII) inhibition.
Broadly speaking, RTA can be divided into proximal (type II) versus distal RTA (type I) versus hypoaldosteronism (type IV). Proximal RTA results from bicarbonate wasting in proximal tubule and can be due to Fanconi syndrome, carbonic anhydrase inhibitors such as acetazolamide, and genetic bicarbonate reabsorption abnormalities among other etiologies. 2 Distal RTA is the result of impaired distal acidification of urine and is suggested by the presence of NAGMA with concurrent positive urine anion gap and low (<150 mosm/kg) urine osmolar gap, which suggests an abnormally low urine ammonium. The urine anion gap and osmolar gap indirectly measure the urine ammonium. During acidemia, urinary ammonium should be elevated as the acid load is being renally excreted. Etiologies for distal RTA are diverse and can be mechanistically distinguished based on whether they involve the apical epithelial sodium channel (eNAC) transporter (eg, dehydration), distal alpha intercalated cells (eg, Sjogren’s syndrome), tubulotoxicity (eg, liposomal amphotericin-B), or impairment of carbonic anhydrase.3-7 Type IV RTA is associated with hypoaldosteronism and is seen with diabetes mellitus (hyporeninemic hypoaldosteronism) or primary hyperaldosteronism usually in children.4,5
Renal tubular acidosis can also be subclassified based on etiologies that present with hypokalemia versus hyperkalemia. Proximal RTA occurs with hypokalemia and can be due to decreased sodium absorption in the proximal tubules4,5 leading to enhanced effects of aldosterone in the distal tubule to reabsorb sodium and excrete potassium. Type IV RTA is associated with hyperkalemia due to hypoaldosteronism. 4 Distal RTA can be associated with hyperkalemia or hypokalemia. When the eNAC channel is inhibited from creating a transcellular voltage gradient, such as with lithium use, obstructive uropathy, 7 and lupus nephritis, 8 hyperkalemia ensues from impairment in distal tubular potassium excretion. Hypokalemia is seen with other subtypes of distal RTA. A more comprehensive list of causes of RTA can be found in Table 1.
Causes of Renal Tubular Acidosis as Stratified by Pathophysiology and Etiologies.

Note. RTA = renal tubular acidosis; NSAIDs = non-steroidal anti-inflammatory drugs; SLE = systemic lupus erythematosus.
Our patient presented with hypokalemic RTA which narrowed the differential into proximal or certain subtypes of distal RTA. Calculation of the TTKG 9 can help assess whether the kidneys are implicated. In this case, the TTKG was greater than 3, which, in the context of hypokalemia, suggests renal potassium wasting and RTA pathophysiology.
Ibuprofen has been previously linked to hypokalemia and RTA.10-15 A recent systematic review 12 found 50 reported cases of patients with profound hypokalemia and/or acidosis after ibuprofen use and most cases had evidence of impaired urinary acidification or renal potassium wasting. Notably, this review found 3 lethal cases and that hypokalemia and acidemia were also seen at doses generally regarded as safe (<40 mg/kg of body weight). The mechanism of ibuprofen-induced RTA relates to inhibition of CAII.16-20 Carbonic anhydrase II is an enzyme found in both proximal and distal tubules and catalyzes the conversion of carbon dioxide and water into bicarbonate which is then retained in the respective site. 18 Thus, inhibition of CAII may lead to both proximal and distal RTA or a combination of both known as type III RTA. 19 However, here, there was significant acidosis with initial bicarbonate of 8. The patient also had a relatively low urine osmolar gap (58 mmol/kg) and positive urine osmolar gap (20 mmol/L), both of which suggest impaired ammonium excretion in the distal nephron. Overall, we believe that this presentation was more consistent with distal RTA, although we cannot rule out coexisting CAII inhibition in the proximal tubules.
Ibuprofen is one of the most frequently used non-steroidal anti-inflammatory drugs (NSAIDs)—a class of medications which sees widespread use in both prescribed and over-the-counter settings. Nephrotoxic side effects of NSAID use are well known, but the association between ibuprofen and hypokalemic RTA is much less reported despite the mechanistic link, raising the possibility that cases may be underrecognized.
In summary, we report a case of RTA and hypokalemia due to ibuprofen use and emphasize the following takeaway points:
Given the prevalence of and over-the-counter access to NSAIDs, it should be explored in the differential for RTA.
Monitoring for acidemia and electrolyte abnormalities may be warranted for chronic, heavy NSAID use.
Footnotes
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Written informed consent was obtained from the patient featured in this report.
Availability of Data and Materials
Data available upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
