Abstract
Rationale:
Hypokalemia is a common finding. Typically asymptomatic presentations of neuromuscular weakness emerge at levels below 2.5 mmol/L. Causes include gastrointestinal losses, renal losses, or intracellular shift, with gastrointestinal losses and diuretics accounting for the majority. Although the cause is often apparent on clinical assessment, a systematic approach incorporating urine biochemistry can aid in narrowing the differential in obscure cases.
Presentation:
We describe a case of a previously healthy 27-year-old man who presented with acute ascending paralysis, with an associated severe hypokalemia and metabolic acidosis. There were no apparent causes on clinical assessment.
Diagnosis:
Based on analysis of urine biochemistry, we concluded that a pathologic kaluresis was present, and given his acidemia and transient pathology, we diagnosed the patient with hypokalemic paralysis secondary to toluene toxicity.
Interventions:
We provided supportive care and electrolyte repletion for our patient; no specific therapies for toluene were required. Our patient was counseled regarding appropriate protective measures when handling toluene.
Outcomes:
Complete neurologic recovery and biochemical normalization occurred within 48 hours of presentation with supportive care. He continued to use proper precautions when handling toluene, and experienced no symptom relapse, or further abnormalities on both blood and urine chemistry.
Lessons learned:
Using this case, we review an algorithmic approach incorporating urine biochemistries to aid in the workup of hypokalemia. We review toluene as a toxicologic entity and highlight its role as a cause of hypokalemia.
What was known before
Although mild hypokalemia is a common electrolyte disturbance with a broad differential diagnosis, severe hypokalemia (potassium ≤ 2.5 mmol/L) is uncommon, with malnutrition, diuretics, and prolonged gastrointestinal losses as the main causes. 1 Toluene, a common drug-of-abuse, is a known but underappreciated cause of severe hypokalemia.
What this adds
We present a case of severe hypokalemia precipitated by transdermal toluene toxicity and review an approach to severe hypokalemia.
Case Presentation
A 27-year-old man presented to the emergency department with 2 days of ascending paralysis, diffuse myalgias, vomiting, dyspnea, headache, blurred vision, and hand paresthesias. His medical history included generalized anxiety and gastroesophageal reflux. He consumed 3 to 5 alcoholic drinks each Saturday and had a remote smoking history. He worked as a mechanic and had returned from a boating trip 3 days ago.
His vital signs were within normal limits. Mental status, cardiopulmonary, and abdominal examinations were normal. Neurologic examination noted 0/5 and 1/5 strength in proximal and distal extremities, respectively. Deep tendon reflexes were absent in lower extremities and hyporeflexic in upper extremities. Sensation was normal throughout.
Table 1 displays his serum and urine laboratory data. Initial blood work was notable for severe hypokalemia with a potassium of 1.5 mmol/L, bicarbonate of 19 mmol/L, glucose of 11.9 mmol/L, magnesium of 0.59 mmol/L, phosphate of 0.11 mmol/L, creatinine of 91 µmol/L, and urea of 3.4 mmol/L. Initial venous blood gas (VBG) showed a pH of 7.34, pCO2 of 38 mmHg, and bicarbonate of 21 mmol/L; venous lactate was 3.4 mmol/L. Creatine kinase was low at 36 U/L, whereas myoglobin was normal. Thyroid stimulating hormone, parathyroid hormone, 25-hydroxyvitamin D, C3 and C4, and serum protein electrophoresis were normal. Anti-nuclear antibody and rheumatoid factor screens were negative.
Relevant Serum and Urine Data.
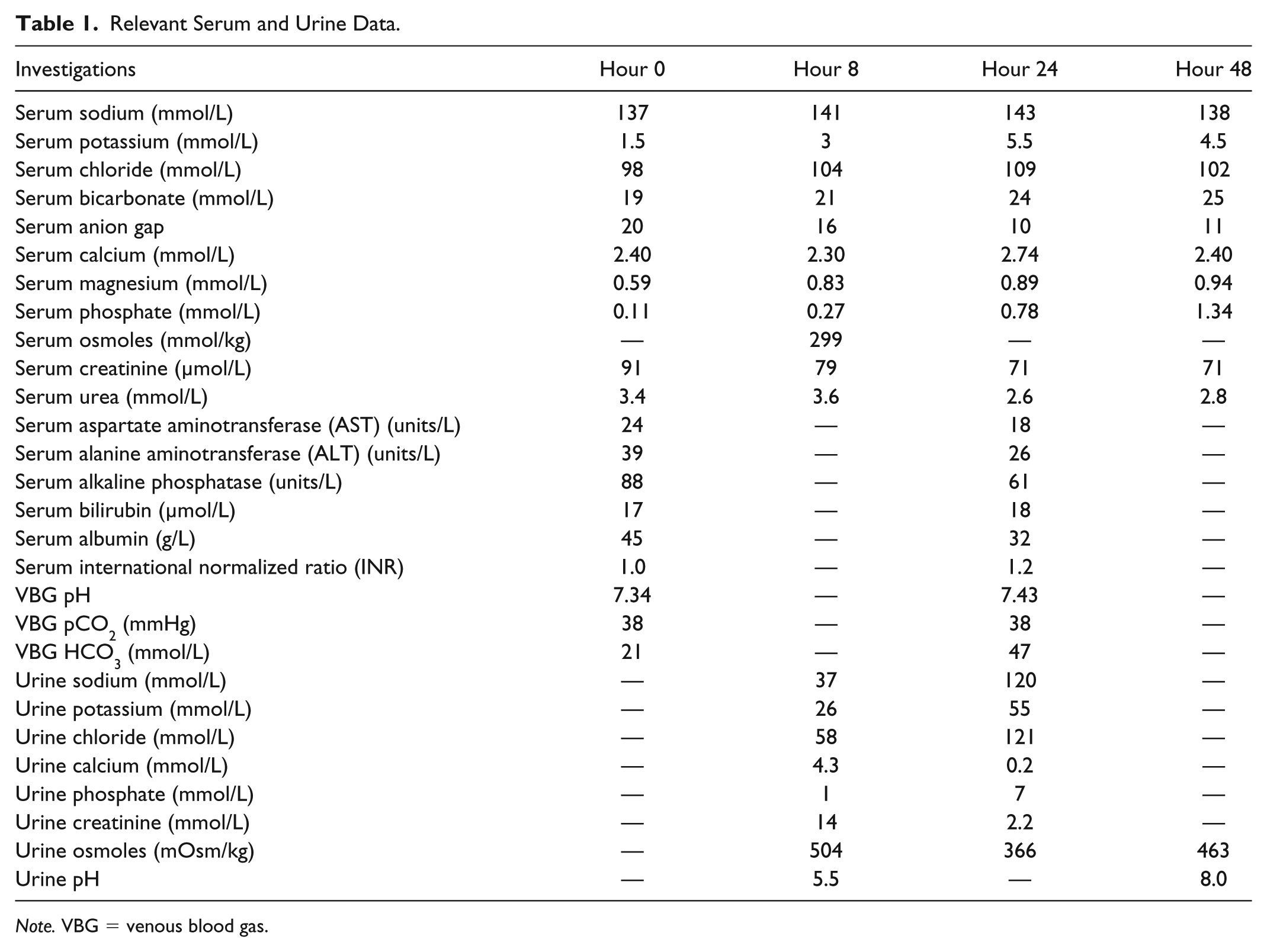
Note. VBG = venous blood gas.
No urinary testing was conducted at hour 0 in the emergency department. Hour 8 urinalysis showed only occasional hyaline casts. Repeat urine microscopy at 48 hours was bland.
Electrocardiogram demonstrated sinus rhythm, with a prolonged PR interval (154 ms), T-wave inversion, and U-waves in all leads, and a prolonged QT interval (550 ms). Respiratory muscle strength testing showed borderline reduced maximal inspiratory and expiratory pressures, at 54 and 58 cm H2O, respectively; vital capacity was normal (3.9 L).
He was admitted to hospital, and over the next 24 hours received a total of 3.8 L of normal saline, 180 mmol of intravenous potassium (as potassium chloride and potassium phosphate), 2 g of intravenous magnesium, and 60 mmol of intravenous phosphate (as potassium phosphate); 120 mmol of potassium was given, followed by magnesium, then the remaining potassium. Symptoms rapidly improved over the first 24 hours of treatment, and the patient returned to baseline function by 48 hours with a normal neurologic exam (Figure 1).

Case timeline.
Clinical Reasoning
This case of severe hypokalemia, without preceding gastrointestinal losses or evidence of malnutrition, was suspected to be mainly from kaluresis. Our approach is outlined in Figure 2.

We determined that inappropriate kaluresis was present during hypokalemia based on 2 measures. The first was the transtubular potassium concentration gradient (TTKG), with values over 3 supporting inappropriate renal potassium excretion. 4 Our patient’s hour 8 TTKG was 5.14, (UK/PK)/(UOsm/POsm) = (26/3)/(504/299) = 5.14, suggesting inappropriate kaluresis. We also examined the urine potassium-to-creatinine ratio (UK/UCr), where ratios below 1.5 suggest minimal kaluresis and above 1.5 suggest significant kaluresis. 3 Our patient’s UK/UCr was 1.86, suggesting inappropriate renal potassium handling. Given his initial metabolic acidosis, we narrowed our differential to a distal renal tubular acidosis (RTA) or hippuric acidosis (ie, toluene toxicity). The high urine osmolality may represent nonreabsorbed anions like hippurate (although this could not be confirmed as urine ketones were not tested), raising toluene as a leading differential. We felt this was likely as the hour 8 urinary osmolal gap, while incalculable due to missing urine urea and glucose, would suggest that only 126 of 504 mOsm/kg of urinary osmoles could be accounted for by sodium and potassium, with 378 mOsm/kg being either urea, glucose, ammonia, or other osmoles (eg, hippurate and benzoate). Further review noted large acute exposures to toluene (he cleaned his hands with toluene-containing fluids on his trip). We thus attributed his hypokalemia to acute transdermal toluene toxicity.
Limitations
Our principle limitation was missing data. As we lacked time 0 urinary testing and could only reason the above based on hour 8 data (after potassium therapy had begun), we cannot definitively say kaluresis was the inciting pathology. However, the patient’s UK/UCr was elevated despite profound systemic hypokalemia, suggesting kaluresis was the underlying pathophysiology.
The TTKG and UK/UCr have limited uses. Although the requirements in urine osmolality and sodium, and serum osmolality were met here, 5 its clinical validity has been questioned.6,7 Although potentially flawed, given its discriminatory value in cohort studies, 4 we suggest it can be integrated into a larger picture. The UK/UCr is preferable over the TTKG, though there is uncertainty regarding a diagnostic threshold of 1.53,6 versus 2.5.4,7 In this case, the hour 8 UK/UCr of 1.86 may reflect mild ongoing kaluresis, which would be inappropriate given the systemic hypokalemia, but also show initial recovery in appropriate renal function; we were unable to verify this, owing to lacking hour 0 urinary data.
Missing data also made ruling out a distal RTA difficult, as we could not calculate a urine anion gap (UAG) at hour 0. However, given the rationale above, as well as the clinical history, we feel that toluene exposure is the most unifying diagnosis. Furthermore, as the abnormalities resolved with time and supportive therapy, it suggests a transient pathology like a toxin, rather than chronic pathology like an RTA. It is also notable that a positive UAG, which normally indicates decreased urinary ammonia, can instead reflect increased unmeasured urinary anions, like hippurate and benzoate, which are excreted in toluene toxicity. 7 The hour 8 urinary pH below 5.5, and the initial anion gap acidosis are incongruent with a distal RTA.
Management
Hypokalemia management revolves around potassium replacement. Severe hypokalemia requires aggressive replacement with intravenous formulations. Symptomatic patients require supportive care for respiratory muscle failure, cardiac arrhythmias, and rhabdomyolysis that may emerge, including mechanical ventilation, defibrillation, or antiarrhythmics. Hypomagnesemia should be corrected early, as hypokalemia is often refractory to therapy unless magnesium is replaced. 8 Normal magnesium is also cardioprotective in hypokalemia. 9
Toluene has no antidote. Management is supportive with electrolyte correction in monitored settings. Epinephrine should be avoided due to arrhythmogenic properties from toluene-induced myocardial catecholamine sensitization. 10 Acute toxicity is self-limiting—neuromuscular symptoms resolve with potassium repletion, whereas the toxicity itself resolves in 48 to 72 hours postexposure. 11 Prolonged severe hypokalemia should be evaluated for alternate causes. Prognosis is favorable, with a low mortality in case series.11-13
Toluene Review
Toluene is a hydrophobic solvent found in paint thinners, cleaning agents, adhesives, and gasoline,11,12,14-16 and is the most commonly abused volatile substance worldwide, owing to its euphoric effects.11,13,15,16 Absorption occurs from inhalation, ingestion, or transdermal exposure, and can be detected in blood within minutes. 14 There are no reported cases of transdermal toxicity, but its transdermal absorption rate is comparable with known toxins, 17 and several-minute transdermal exposures yield similar absorption as 8 hours of inhaled exposure. 17 Toluene is hepatically metabolized by cytochrome P450 to benzoic acid, then hippuric acid; these are renally cleared. 14 Toluene is mostly excreted within 24 hours, although some amount may remain in fat until 72 hours11,14; excretion rates are similar for all absorption routes. 14
Neuromuscular paralyses of varying severities are the main presentation of toluene toxicity,11,13 with severity proportional to the degree of hypokalemia.13,15 Explanations for toluene-induced hypokalemia include renal tubular acidosis, 16 independent urinary potassium wasting, 12 and kaluresis as a conjugate cation for hippurate. 18 Toluene is associated with distal and proximal RTA’s,16,19 proteinuria, and Fanconi syndrome. 11 A metabolic acidosis may present, with both anion gap and nonanion gap acidoses noted.11,12 Hypotheses regarding pathogenesis of the former revolve around hippuric and benzoic acidosis, 18 whereas the latter is thought to be due to impaired distal tubular ammoniagenesis or chronic interstitial abuse in chronic toluene exposure.18,19 Hypophosphatemia is common via an unknown mechanism postulated as either hyperphosphaturia or phosphate redistribution11-13; our patient had a fractional excretion of phosphate (FePO4) of 2.09% at hour 8, calculated as FePO4 = (UPO4/SPO4)/(UCr/SCr) = (1/0.27)/(14/79) = 2.09, suggesting appropriately decreased phosphate excretion. Rhabdomyolysis is common and may be due to direct muscle injury. 13 Various arrhythmias are possible. 11
Toluene toxicity is a clinical diagnosis, although serum toluene and urine hippuric acid have been described as diagnostic measures.16,20 In our case, diagnostic delay and improper sample handling rendered these tests inconclusive. Therefore, awareness should be lent to toluene as a cause in severe hypokalemia.
Outcomes
Our patient rapidly recovered with potassium and magnesium replacement. He was advised to use proper protective equipment when handling toluene. He has had no symptom relapses and has had repeat blood work and urine studies demonstrating no abnormalities.
Footnotes
Ethics Approval and Consent to Participate
Consent was obtained from the patient for publication of their case in the medical literature.
Consent for Publication
Consent was obtained from all participating authors for publication of this manuscript in the medical literature.
Availability of Data and Materials
Relevant data is provided in the article. Additional primary data is available on inquiry from the patient’s chart.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
