Abstract
Background:
There is little data modeling the impact of deemed consent legislation (eligible individuals who do not register their decision to decline to be a donor are presumed to consent after death) on outcomes for individuals with kidney failure.
Objective:
To estimate the change in life-years (LYs) and quality-adjusted life-years (QALYs) resulting from different changes in the rate of deceased donor kidney transplantation associated with deemed consent legislation and health system transformation.
Design:
Dynamic Decision Analytic Model.
Setting:
This modeling study included kidney failure patients in Atlantic Canada (all of whom receive their kidney transplants in Halifax, Nova Scotia). The adoption of deemed consent legislation was the intervention, and opt-in (the status quo) was the reference comparator.
Patients:
Prevalent kidney failure patients at the end of 2019 in all of Atlantic Canada (N = 3615) served as the starting population.
Methods:
We compared expected outcomes between the intervention and comparator. Changes in QALYs and total LYs were modeled under different changes to the proportion of patients receiving a deceased donor kidney transplant (from –10% to 20%) resulting from deemed consent relative to the status quo. Changes in QALYs and LYs were reported for 3 different time horizons (5, 10, and 30 years). Uncertainty around QALYs and total LYs was reported using 95% confidence intervals (CIs) constructed from a probabilistic sensitivity analysis using 1000 Monte Carlo Simulations.
Results:
The increase in QALYs ranged from 7 QALYs (95% CI: 5-10) with a 5% increase using a 5-year time frame to 882 QALYs (95% CI: 619-1144) with a 20% increase over a 30-year time frame. Parallel changes in total LYs were also observed. In contrast, decreases in deceased donor kidney transplantation resulted in a loss of QALYs (for example, –463 QALYs; 95% CI: –633 to –306 for a 10% decrease over a 30-year time frame). Using the most optimistic scenario (a 20% increase), there was an 18% increase in the cumulative number of deceased donor kidney transplant recipients over a 30-year observation period.
Limitations:
The results are subject to uncertainty depending on changes to the dialysis or transplant population that were not modeled and that may not be fully captured with probabilistic sensitivity analysis.
Conclusions:
Deemed consent legislation will lead to variable changes in QALYs and total LYs for the kidney failure population, depending on the degree to which deceased donor transplantation rates change and the time horizon of observation. This modeling study may serve as a baseline to monitor the future impact of deemed consent legislation.
Introduction
On January 18, 2021, Nova Scotia (NS) Canada became the first North American jurisdiction to enact deemed consent legislation for organ and tissue donation. Under this legislation, eligible individuals who do not register their decision to decline to be a donor are presumed to consent to donation after death. A recent US simulation study identified that a deemed consent policy leads to modest increases in life-years (LYs) gained for waitlisted patients. 1 Less is known about how an increase in kidney transplantation (KT) due to deemed consent will affect individuals with kidney failure. Cost-utility analyses of strategies related to increasing kidney transplantation often utilize quality-adjusted life-years (QALYs) as a measure of the impact of a policy change on health. 2 In addition, a recent study that evaluated the magnitude of the economic impact of any policy that leads to increases in deceased organ donation also used QALYs as their measure of health. 3 In this study, we estimated the change in LYs and QALYs that may result from deemed consent legislation and health system transformation in NS.
Method
A dynamic Markov model with a cycle length of 1 year served as the vehicle for the ex ante analysis. The target population was kidney failure patients in the Atlantic Canadian population. We used data from the Canadian Institute of Health Information, the Kidney Foundation of Canada, the Multi-Organ Transplant Program (MOTP) of Atlantic Canada, the Canadian Agency for Drugs and Technologies in Health (CADTH), and the literature to parameterize the model (Supplemental Table 1).4-7 Nova Scotia is the recipient transplant center for Atlantic Canada (NS, Newfoundland, New Brunswick, and Prince Edward Island). We used the prevalent population of dialysis/transplant patients at the end of 2019 as the starting point (N = 3615), acknowledging that the impact of COVID-19 may have led to changes in prevalence and incidence uncharacteristic of the norm for 2020. The intervention was deemed consent legislation. The counterfactual comparator was the opt-in model (the status quo).
We measured the treatment effect as percentage changes in the deceased donor KT probability resulting from deemed consent. We evaluated –10%, –5%, 5%, 10%, 15%, and 20% changes in deceased donor KT probability. There were 4 health states in the model: alive after a pre-emptive transplant, alive with a deceased transplant, alive after receiving a transplant from a living donor, and alive on dialysis (Supplemental Figure 1). The 2 absorbing states included “deceased while on dialysis” and “deceased after transplant.” The model consisted of an entry node, which allowed adding the annual incidence of kidney failure in Atlantic Canada to the total number of patients with kidney failure. Based on expert opinion, we assumed that all pre-emptive KTs came from living donors. If the patient was on dialysis, there was a chance that they would get a KT either from a deceased or from a living donor. After a KT, there was a chance of death or graft failure, and in the latter, the patient would return to dialysis, with a chance of getting a retransplant. The probability of graft failure depended on whether the organ came from a deceased or a living donor. For patients with functioning grafts, the annual probability of death also depended on whether the organ came from a living or a deceased donor (Table 1). We based the model on the assumption that all kidney failure patients were eligible for a transplant. The analysis involved half-cycle correction. 8 We discounted outcomes at 1.5% per annum, following CADTH’s (fourth edition) 9 recommendation.
Mean and Incremental LYs and QALYs Gained by Various Changes in Deceased Donor Kidney Transplantation Over Time.
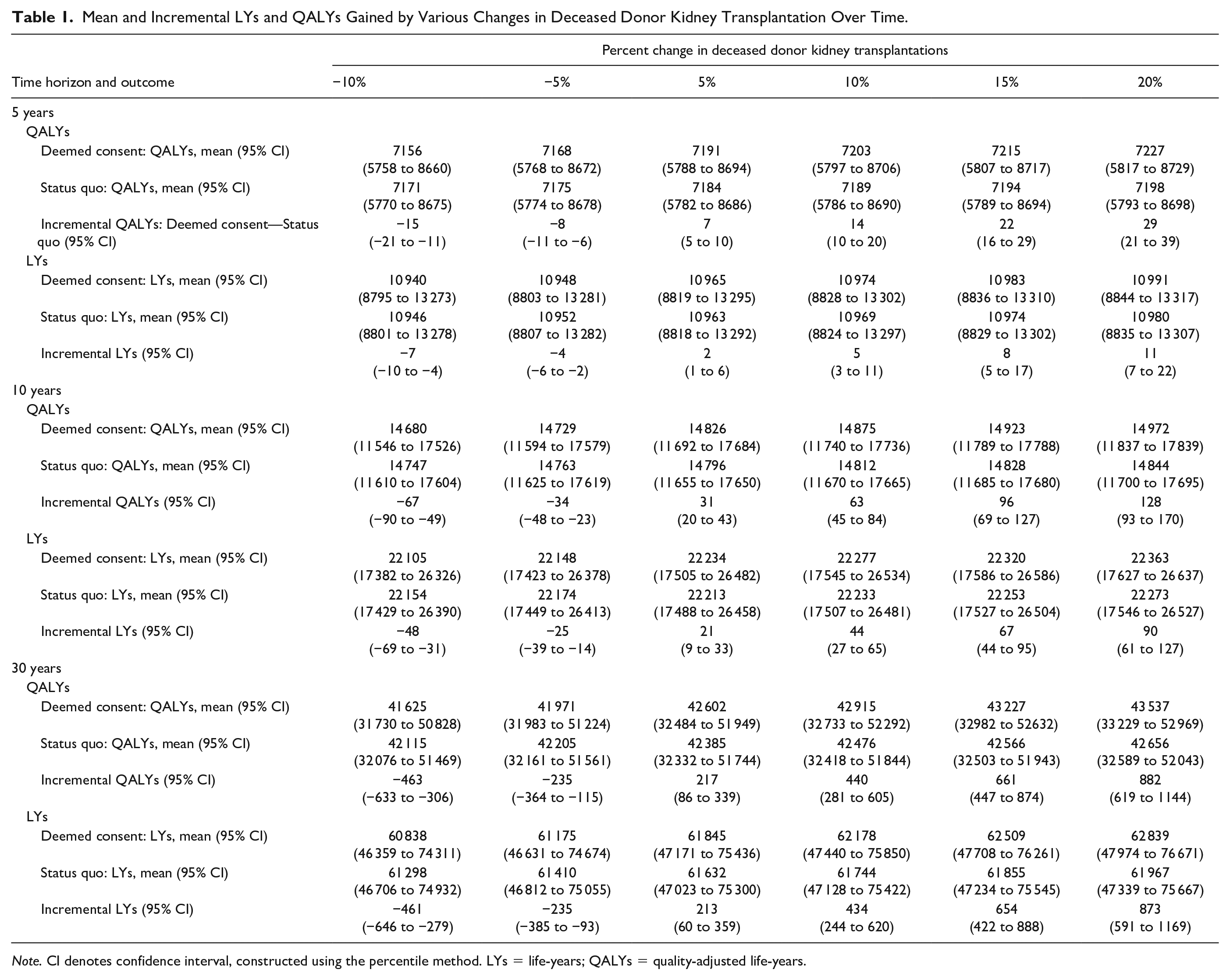
We quantified the uncertainty around the estimates using the 95% confidence interval (CI) constructed from a probabilistic sensitivity analysis (PSA) using 1000 Monte Carlo simulations. In the PSA, we fitted key parameters in the model to distributions (Supplemental Table 1). We validated the model following established procedures. 10
Results
The changes in total LYs and QALYs gained or lost following deemed consent legislation are noted in Table 1 and Figure 1A. The expected gain in QALYs ranged from 7 QALYs (95% CI: 5-10) with a 5% increase using a 5-year time frame to 882 QALYs (95% CI: 619-1144) with a 20% increase over a 30-year time frame. In contrast, decreases in deceased donor kidney transplantation resulting from deemed consent legislation led to a loss of both QALYs and total LYs (Table 1). Using the most optimistic scenario (a 20% increase), there was an 18% increase in the cumulative number of deceased donor kidney transplant recipients (Figure 1B) comparing the deemed consent policy arm with control. Similarly, the number of patients on dialysis (assuming a 20% increase in deceased donor KT probability) was lower in the policy arm (Supplemental Figure 2).

(A) QALYs gained over time with each percent increase in deceased kidney transplants over a short (5 years), medium (10 years), and long (30 years) time horizon. The health outcome was the expected QALYs gained, calculated as the difference in expected QALYs between the treatment (deemed consent) and the control arms (status quo). (B) Cumulative number of deceased donor kidney transplantations, assuming a 20% increase (over a 30-year time).
Discussion
Nova Scotia has taken a bold step in implementing deemed consent legislation. Under most circumstances, an increase in available kidneys will positively impact those requiring kidney replacement therapy. There is unlikely to be a significant downside to recipients, provided organ quality is not significantly compromised. Also, studies have shown that kidney transplantation is associated with decreases in mean annual health care costs compared with dialysis. 11 This emphasizes that there are likely cost savings that accompany policy changes that result in a greater number of dialysis patients receiving a deceased donor kidney transplant. In contrast, if the policy leads to a reduction in the deceased donor kidney transplant rate, it will expectedly lead to a decrease in both QALYs and total LYs. What remains to be seen is how adoption of deemed consent will impact the donor pool. Some studies have shown increases in kidney transplantation following adoption of deemed consent legislation. 12 In contrast, deemed consent legislation has not been successful in improving donation rates (and hence transplant rates) in all countries. 12 Whether there will be a net positive or net negative change to recipient QALYs or total LYs may also hinge on whether or not system changes occur in addition to changing the legislation. Finally, if deemed consent legislation leads to a reciprocal reduction in the rate of live donor transplantation 13 (because of the diversion of resources toward deceased donor KT), it will also mitigate the potential benefits. Therefore, monitoring live donor KT rate changes will be important after implementing deemed consent legislation. We do acknowledge that there are limitations to this study that should be considered when interpreting the results. The analysis is subject to uncertainty depending on changes to the dialysis or transplant population that were not modeled and that may not be fully captured with PSA. In addition, population-derived QALY calculations may be valuable in assessing the impact of health system change, but cannot be used to counsel individual patients. Notwithstanding, Canada has a lower donation rate compared with many countries, and if there is a significant increase in transplantation following legislation, it may encourage Canadian policymakers to consider the importance of integrating deemed consent into future donation reforms nationwide.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581221139039 – Supplemental material for Estimated Impact of Deemed Consent Legislation for Organ Donation on Individuals With Kidney Failure: A Dynamic Decision Analytic Model
Supplemental material, sj-docx-1-cjk-10.1177_20543581221139039 for Estimated Impact of Deemed Consent Legislation for Organ Donation on Individuals With Kidney Failure: A Dynamic Decision Analytic Model by Koto P., Vinson A. J., Kiberd B. A., Beed S., Krmpotic K., Dirk J., Weiss M. J. and Karthik K. Tennankore in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
This study was approved by the Nova Scotia Health Research Ethics Board (REB Number: 1026155). As this was a modeling study using publicly available data, patient consent was not required.
Consent for Publication
All authors agreed to the publication of this manuscript.
Availability of Data and Materials
The data for this study is publicly available and the models used for the analysis are available upon request.
Author Contributions
K.K.T. conceived the original idea. K.P. and K.K.T. drafted the first version of the manuscript. K.P. did the primary analysis. All authors contributed to the research idea/conception and reviewed/revised the original draft. All authors provided final approval for the manuscript in its current form.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by Health Canada as part of the Legislative Evaluation: Assessment of Deceased Donation Reform project, which is a partnership between the Nova Scotia Health Authority, the Nova Scotia Department of Health and Wellness, the Canadian Donation and Transplantation Research Program, Canadian Blood Services, and Transplant Québec. The opinions reflected in this paper are those of the author and do not necessarily reflect the views of Health Canada, Nova Scotia Health Authority, the Nova Scotia Department of Health and Wellness, the Canadian Donation and Transplantation Research Program and Canadian Blood Services or Transplant Québec.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
