Abstract
Despite a record number of solid organs transplanted in 2023, ongoing issues with donor shortages leave thousands of people in the United States waiting. Increased utilization of donors after circulatory death (DCD) has been the primary driver of the increase in total transplants. Availability of newer technologies including normothermic machine perfusion (NMP), normothermic regional perfusion (NRP), and hypothermic machine perfusion (HMP) have been shown to attenuate ischemia-reperfusion injury and allow for graft viability assessment prior to implantation. Outcomes across all solid organs, especially in higher risk donors, have improved using machine perfusion to decrease rates of primary non-function and damage related to ischemia-reperfusion injury (IRI). Here, we present an overview of deceased donation practices across all solid organs.
Historical Evolution of Deceased Donation
Deceased organ donation has evolved through several key historical phases. Initially, organ transplantation relied exclusively on organs recovered after cardiac death (donation after circulatory death, DCD), as this was the only accepted criterion for death before the late 1960s.1,2 The development of the concept of brain death in 1968, and subsequent adoption of the Uniform Determination of Death Act (UDDA) in the United States, established irreversible loss of all brain function as a legal determination of death, enabling organ recovery from donors with ongoing circulation of absent neurologic function.2-4 This shift dramatically increased the supply and quality of transplantable organs, as organs could be procured before the onset of warm ischemia (Figure 1). Evolving Trends in the Last Decade in Deceased Donor Transplants in the United States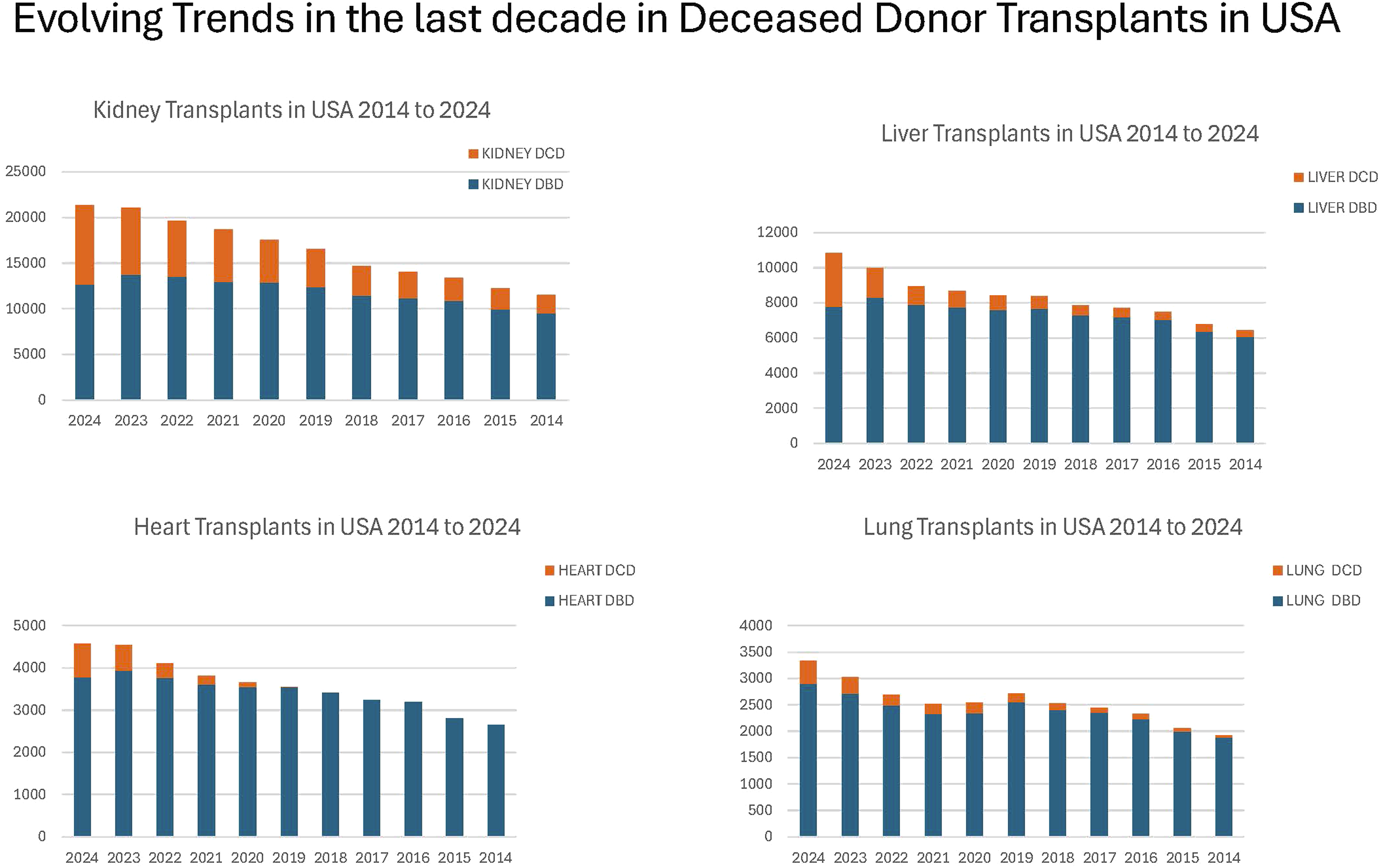
Throughout the late 20th century, most deceased organ donations were from donation after brain death (DBD) donors. Due to an increasing organ demand and changes in end-of-life care, there has been a resurgence in DCD donation since the early 2000s, with DCD now accounting for a substantial proportion of deceased donor contributions in many countries.1,2,5 Advances in organ preservation, perfusion technology, and ethical frameworks have further expanded the donor pool, including pediatric and, in some jurisdictions, donation after euthanasia.3,6
Modern practice is governed by the dead donor rule, which requires that an organ recovery only occur after death is declared by either neurologic or circulatory criteria.3,4 The American Academy of Pediatrics and other societies have published guidelines defining accurate determination of death and ethical standards for organ recovery. 3 The evolution continues, with ongoing efforts to optimize donor identification, organ preservation, and expand the types of eligible donors.2,7,8
Definitions of Brain and Circulatory Death
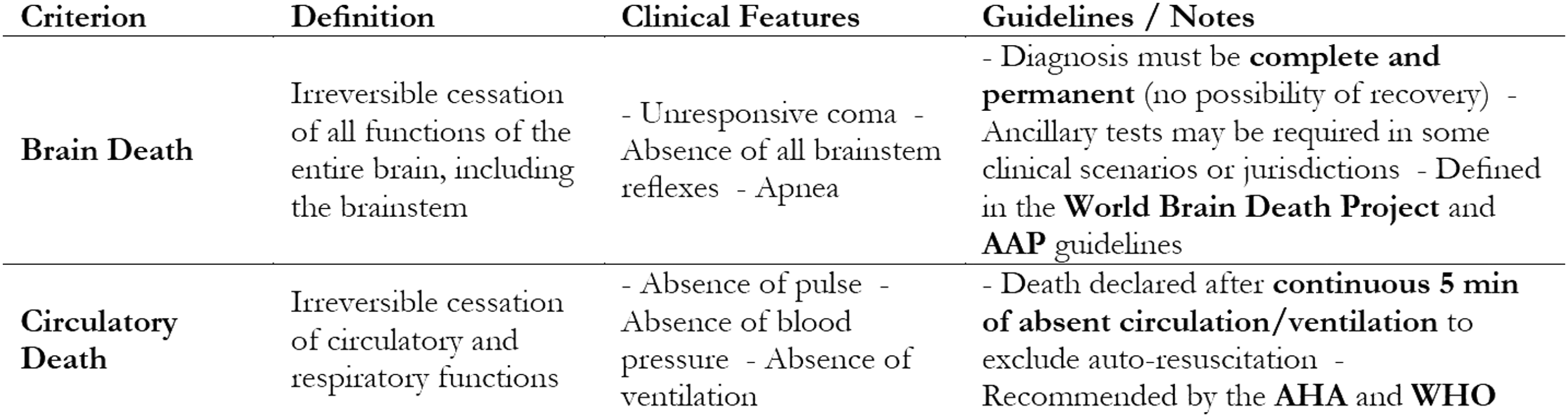
Definitions of Death in the Context of Deceased Organ Donation
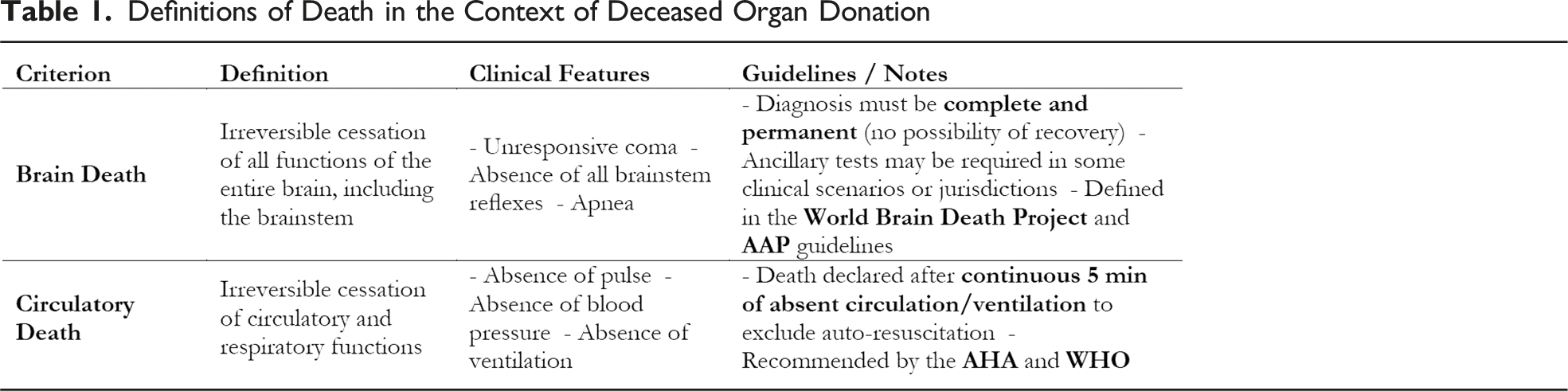
Circulatory death (also referred to as cardiocirculatory death or cardiac death) is defined as the irreversible cessation of circulatory and respiratory functions (Table 1). The American Heart Association (AHA) and the World Health Organization (WHO) recommended that death be declared after a period of continuously observed absence of pulse, blood pressure, ventilation, typically for 5 minutes, to confirm permanent loss of circulation to the brain. This period is intended to exclude the possibility of spontaneous return of circulation (autoresuscitation).10,11
Both definitions are grounded in the UDDA, which is the legal standard in the United States and aligns with international consensus.3,9
Categories of Donation after Circulatory Death (DCD)
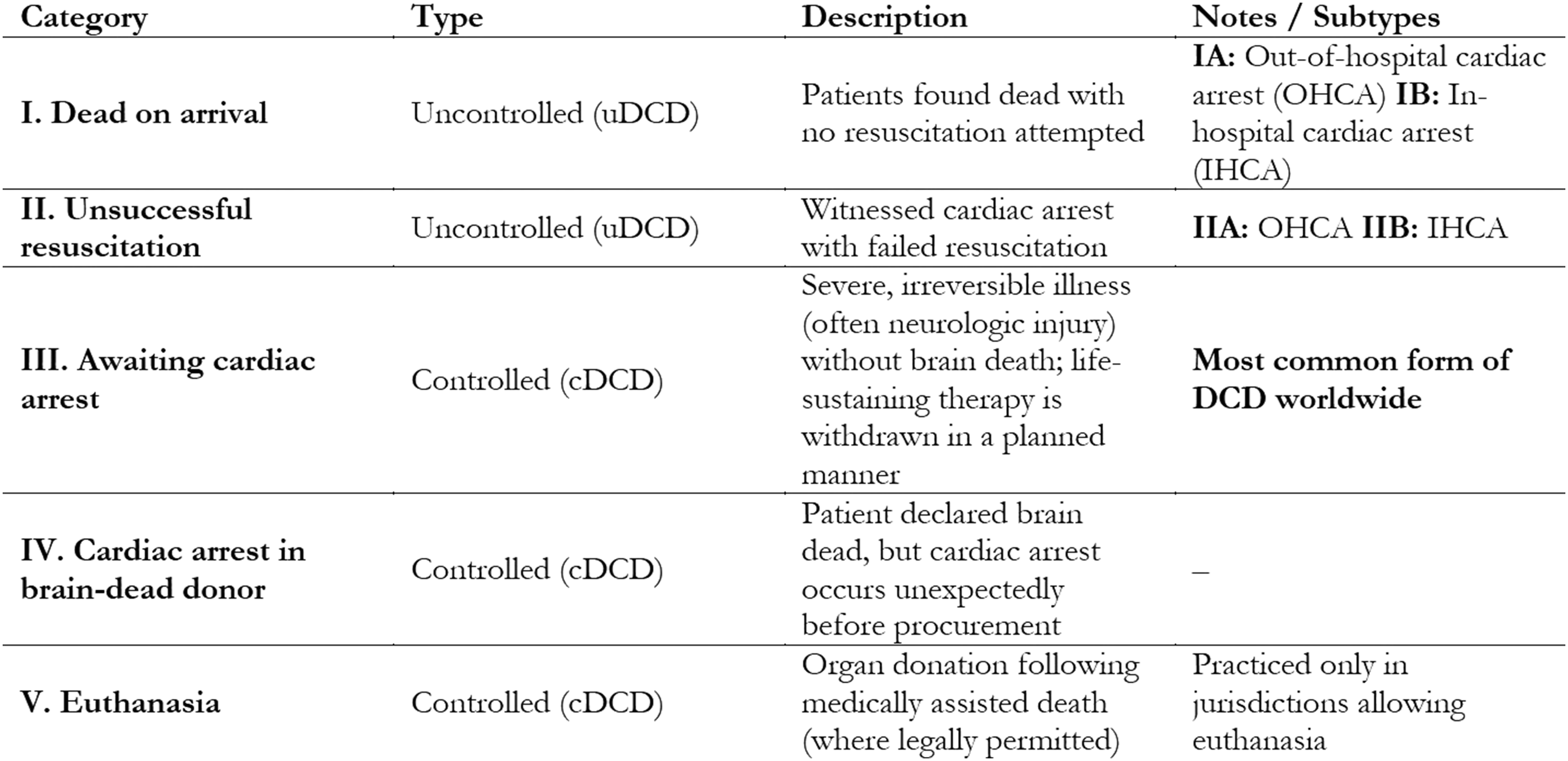
The Maastricht categories of donation after circulatory death (DCD) are an internationally recognized classification system that delineates clinical scenarios for DCD. The categories are divided into uncontrolled (uDCD) and controlled (cDCD) settings (Table 2): • Category I (dead on arrival): uncontrolled DCD; patients found dead outside the hospital with no resuscitation attempted (subdivided into IA for out-of-hospital cardiac arrest [OHCA] and IB for in-hospital cardiac arrest [IHCA]). • Category II (unsuccessful resuscitation): uncontrolled DCD; witnessed cardiac arrest with failed resuscitation efforts (IIA for OHCA, IIB for IHCA). • Category III (awaiting cardiac arrest): controlled DCD; patients with severe, irreversible illness (often neurologic injury) who do not meet brain death criteria, and for whom life-sustaining therapy is withdrawn in a planned manner. • Category IV (cardiac arrest in a brain-dead donor): controlled DCD; patients who have been declared brain dead but experience an unexpected cardiac arrest before organ procurement. • Category V (euthanasia): controlled DCD; organ donation following medically assisted death, where permitted by law. Maastricht Categories of Donation After Circulatory Death (DCD)
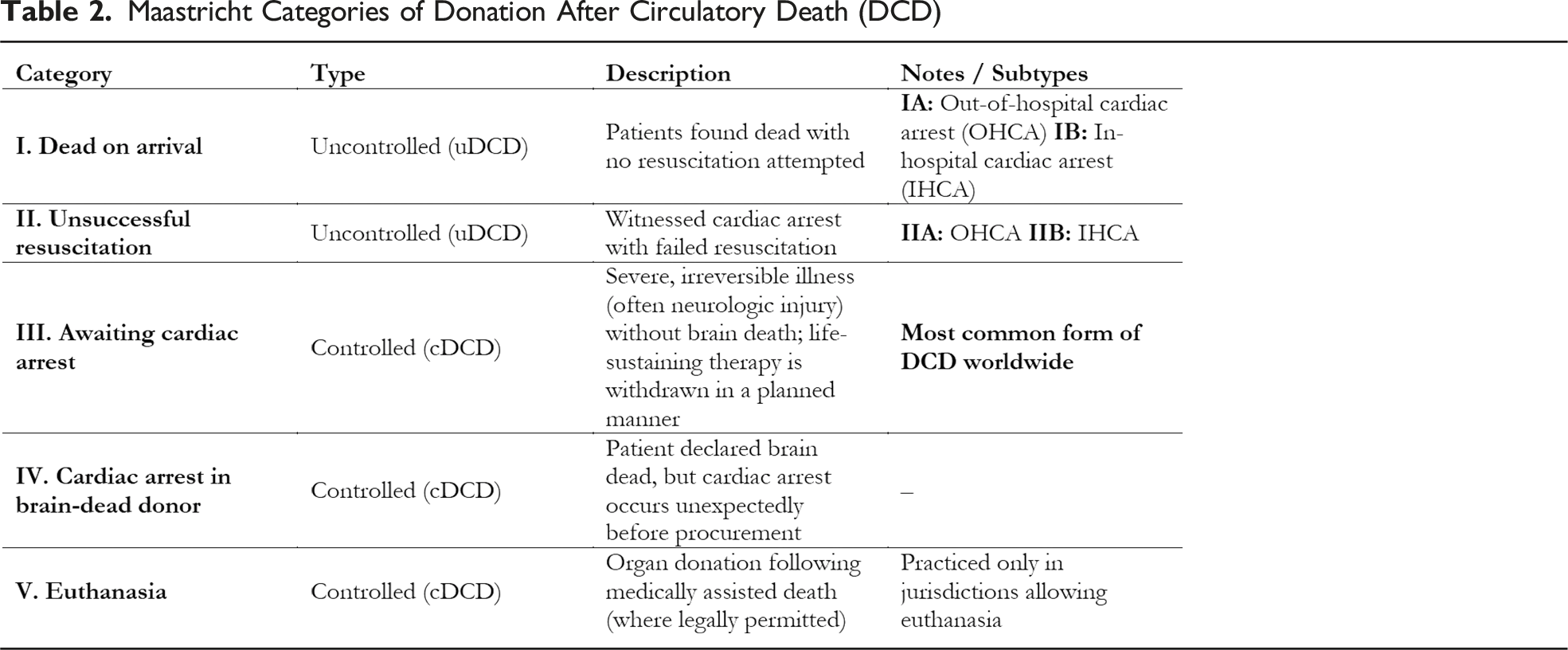
This classification system is widely used in clinical practice and is endorsed by the AHA in its scientific statement on organ donation after out-of-hospital cardiac arrest.11-13 The categories help standardize reporting and guide protocols for organ procurement in DCD.
Of these, cDCD (Category III) is the predominant form of DCD practiced globally. At the same time, uDCD is limited to a small number of countries and represents a minority of DCD activity. The AHA notes that cDCD is the most prevalent category in most countries where DCD is performed, including Australia, Canada, the United States, the United Kingdom, the Netherlands, Belgium, France, Spain, and several other countries. By contrast, uDCD programs are mainly established in Spain, France, and the Netherlands, with France and Spain accounting for the majority of uDCD donors in Europe.10,11,14 In Europe, approximately 25% of DCD donors are uDCD (Maastrict I and II), with the remaining 75% being cDCD (Maastrict III). Most uDCD activity is concentrated in France and Spain. Outside of these countries, uDCD is rarely practiced due to logistical, ethical, and legal barriers, as well as concerns about post-transplant outcomes and the complexity of protocols required for organ preservation after unexpected cardiac arrest.10,14,15 Donation after euthanasia is only performed under highly restricted conditions given the inherent legal and ethical concerns, and only practiced in Spain, Canada, the Netherlands, and Belgium.16,17 The majority of countries with DCD programs either exclusively perform cDCD or have both cDCD and uDCD, but cDCD remains the dominant practice. 14
Liver Transplant
Liver transplant remains the gold standard therapy for end-stage liver disease. Outcomes continue to improve with patient and graft survival greater than 92% at 1 year, 80% at 5 years, and around 65% at 10 years. 18 While the number of living donor liver transplants has been growing, they still comprise only 5.7% of liver transplants performed in the United States. 18 To address the ongoing donor shortage, centers are pressed to balance the acceptance of “marginal” or “extended criteria” donors against the increased risks of inferior outcomes including primary nonfunction, graft loss, and biliary strictures.
Historically DCD donors were a considerable component comprising the “marginal” liver donor pool as a result of early data demonstrating disappointing outcomes including graft loss from primary nonfunction as well as ischemic cholangiopathy (IC) as compared to transplants from brain-dead donors. 19 Ischemic cholangiopathy, leading to diffuse, multifocal biliary strictures with or without intraductal cast formation, requires repeated endoscopic interventions to improve biliary drainage and often results in graft loss requiring retransplantation. The biliary tree, which relies mainly on arterial blood supply (as opposed to liver parenchyma, which derives its supply from both the hepatic artery and portal vein), is highly susceptible to ischemic injury. Grafts obtained from DCD donors experience warm ischemia (dWIT) starting from the time life-sustaining support is withdrawn from the donor until initiation of cold preservation. Numerous studies have demonstrated the relationship between longer dWIT and up to a 3-fold risk in the development of ischemic cholangiopathy.20,21 Allografts are subjected to further ischemia: cold ischemia during storage and transportation, and then a second period of warm ischemic time during the implantation (ie, when the liver is removed from cold storage and the vascular anastomoses are performed in the recipient). Most transplant centers perform portal reperfusion prior to hepatic artery reperfusion to allow the portal system to decompress. As the portal system does not meaningfully contribute to biliary perfusion, this may lead to additional warm ischemia of the biliary system, though reperfusing the artery first or performing simultaneous reperfusion has not been supported by the literature. 22
The past several years have seen technological advances in organ perfusion and preservation. Perhaps the first of these technologies to gain traction in the United States was ex-situ normothermic machine perfusion. The PROTECT trial was a multicenter trial randomizing recipients to either standard static cold storage (SCS) or the Organ Care System (OCS) liver device. 23 The device perfuses the liver through both the hepatic artery and portal vein with warm (34°C-37°C), oxygenated blood supplemented with fluids containing amino acids, dextrose, insulin, and bile salts. 23 The use of normothermic machine perfusion (NMP) decreased early allograft dysfunction (defined as aspartate aminotransferase >2000 IU/L, bilirubin ≥10 mg/dL, or international normalized ratio ≥1.6 on postoperative day 7) as well as ischemic biliary complications.
The results of the PROTECT trial were promising and prompted many centers to adopt the use of NMP in practice. Perhaps the most excitement generated around machine perfusion technology lies in the opportunity to expand use of donors previously considered “marginal” or “extended criteria” (DCDs, older, or steatotic donors) with marked decreases in major complications, early allograft dysfunction and ischemic biliary complications.24-26 NMP allows for safe preservation of the liver allograft for longer periods of time than afforded for SCS, recovery from dWIT-induced hypoxia, and a viability assessment. Many of these studies have demonstrated a reduction in central reperfusion syndrome, defined as life-threatening cardiovascular collapse upon reperfusion.23,26,27 For grafts felt to be at a higher risk of triggering postreperfusion syndrome, the use of NMP can prevent discards.
Another technological advance has been with in situ normothermic regional perfusion (NRP). NRP is a postmortem procurement technique for donors after circulatory death (DCD) wherein oxygenated blood is used to perfuse the organs intended for transplantation during the procurement process. 28 NRP is not a method of resuscitation. Preclinical studies have demonstrated that NRP attenuates and potentially reverses damage incurred during dWIT. 29 In clinical practice, this technique was first popularized in Spain for recovery of organs from uncontrolled DCD donors. 30 NRP is initiated after the withdrawal of life-sustaining therapy, declaration of the donor’s death, and a mandatory “no-touch” interval following arrest to ensure that there is no autoresuscitation. Once organ recovery is underway, the donor’s vessels (often the abdominal aorta and inferior vena cava) are cannulated. The proximal aorta or branches of the aortic arch are clamped to prevent circulation of the brain, and the circuit is run to deliver oxygenated blood to the intended organs. This can be done in the abdominal cavity alone (A-NRP) or to both the thoracic and abdominal cavities (TA-NRP). While NRP is ongoing, aminotransferases, lactate, and other markers can be taken from the circuit. Currently there is no standard criteria for acceptance, and therefore considerable variability exists in the threshold at which centers accept or decline an organ based on this viability assessment. 31
Outcomes for NRP—specifically the reduction in primary nonfunction and ischemic cholangiopathy—have led to its widespread adoption throughout Europe, including some countries which have mandated its use in controlled DCD recoveries.32,33 Growth of NRP in the United States has varied at both a transplant center level as well as an organ procurement organization (OPO) level as standardized practices and protocols are still in development.28,34,35 A recent multicenter retrospective observational study of transplant centers within the United States demonstrated similar outcomes in terms of early allograft dysfunction, primary nonfunction, and ischemic cholangiopathy in cDCD-NRP livers as compared to livers recovered from brain dead donors. 36 When compared to liver transplants from cDCD recovered by standard super rapid recovery, the authors found comparable patient and graft survival, with decreased rates of EAD and IC in NRP recipients. 36
It is through the promising results generated by NMP and NRP that has led to increased utilization of cDCD liver allografts in the United States. In 2023, 1695 patients received liver transplants from DCD donors, comprising 16.7% of all adult liver transplants performed that year. 18 This represents more than a two-fold increase over the past 5 years, when DCD donors made up only 6.5% of adult liver transplants. 18 There has been a recent study comparing outcomes of liver transplants from cDCD donors on NMP vs NRP with comparable posttransplant outcomes between the 2 groups, and lower rates of ischemic cholangiopathy as compared to cDCD preserved by static cold storage. 37 A major challenge, however, lies in the rising cost of organ acquisition with these technologies, which are borne by the transplant centers.38,39 Despite these barriers, the demonstrable survival benefits and growing utilization of DCD grafts supports the case for insurance reimbursement. Although reimbursement has often been denied on the grounds that these technologies are not yet considered standard of care, emerging evidence increasingly affirms that these are essential practice in transplantation using marginal or extended criteria grafts.
Kidney Transplant
In spite of a record 28 142 kidney transplants performed in 2023, nearly 80% of eligible kidney transplant candidates remained on the wait list at the end of the year. 40 The number of wait list registrants for kidney transplant is the higher than that of other solid organ transplants, taken together with the fact that dialysis offers a way to bridge delayed graft function (DGF, often defined as need for dialysis within the first week after kidney transplant) has perhaps made kidney transplant the earliest driver of so-called “marginal” organ use. Transplants from DCD donors continued to rise, accounting for 37.3% of kidney donors in 2023, though nonuse or discard rate among DCD donors also increased to 34.8%. 40
With expanded use of extended criteria/marginal grafts, there has been an interest in preservation techniques to improve outcomes, as higher-risk donors have been associated with an increase in the risk of DGF, as well as having deleterious effects on long-term outcomes. 41 Hypothermic machine perfusion (HMP) for kidney preservation was studied as early as the 1960s but did not entire widespread clinical practice until the late 2000s due to cost and logistic barriers.42,43 Today, there are several commercially available HMP devices available for use. Following the donor procurement, the kidney is placed on a disposable circuit and continuous perfused with preservation solution at pulsatile or non-pulsatile flows, typically between 6°C and 12°C, until the time of transplant. 44 It is thought that HMP up-regulates endothelial nitric oxide production and clears toxic metabolites from microcirculation, thereby reducing ischemia-reperfusion injury.44,45 Many studies, including the international multicenter randomized Machine Preservation Trial have demonstrated a decreased incidence of DGF, with similar rates of 1-year patient- and graft-survival.46-48 The benefits of HMP appear to be most pronounced in marginal or extended criteria donors, with some studies demonstrating improved 1-year graft function in addition to reduction in rates of DGF. 49
NRP use has expanded in the United States following improved outcomes seen in liver transplant recipients. Likewise, kidneys recovered from donors using NRP have demonstrated improved short- and long-term outcomes including a reduction in DGF and increase in 1-year graft survival.50,51 Mechanistically, maintaining the kidneys at near-physiologic conditions during in situ perfusion is thought to decrease oxidative endothelial injury incurred during dWIT. 52 Reduction in DGF can increase use of higher risk kidneys while also decreasing hospital length of stay.
Evolving technology in minimally invasive surgery has impacted the ability to offer kidney transplant in high BMI index patients. Robotic-assisted kidney transplantation has gained traction over the past 5-10 years. As a minimally invasive procedure, robotic surgery requires smaller incisions, which lead to shorter recovery times and reduced postoperative pain. 53 . 54 This approach expands access to kidney transplant for patients who are morbidly obese, offering a viable option to patients who might otherwise be excluded.
Lung Transplant
As with other solid organs, the demand for lung transplants exceeds the organ supply in spite of an increase in number of lung transplants performed each year. 55 Expanded use of DCD donors for other solid organs led to renewed interest for lung transplant. Lungs from DBD donors have historically been preferred, as the severity of ischemia-reperfusion injury—often worse for DCD organs—has been shown to result in structural damage to the graft, inflammatory necrosis and apoptosis which can lead to edema, further exacerbating function.56,57
This has led to similar trials being conducted with NMP for thoracic organs. The multi-institutional randomized, prospective INSPIRE trial demonstrated noninferior outcomes in lung transplants following NMP (also referred to as ex vivo lung perfusion [EVLP]) vs SCS. 58 Investigators also demonstrated a reduction in primary graft dysfunction at 72 hours posttransplant. The EXPAND trial was a multi-center international study evaluating machine perfusion for extended criteria or marginal lung donors. Investigators found that the use of NMP allowed higher-risk donors to be used for transplant without sacrificing long-term outcomes, though they did find a higher rate of early primary graft dysfunction in the trial arm. 59
Thoracoabdominal NRP (TA-NRP) has evolved along with A-NRP to allow perfusion of thoracic organs in addition to abdominal for recovery, though widespread adoption has been cautious given concerns regarding ischemic damage following withdrawal of life support on the donor exacerbated low perfusion during recovery. 60 Nonetheless, early results following TA-NRP have been optimistic, with no difference in outcomes compared to DCD-NMP lung transplants. 61 Park et al recently published a large single-center series demonstrating comparable outcomes following TA-NRP as compared to DBD donors. 62
Heart Transplant
While the world’s first heart transplant was performed using a DCD donor, the next several decades saw nearly exclusively DBD donors utilized. Procurement from a DBD donor allows assessment of cardiac function in situ. Additionally, dWIT impacts the myocardium directly and causes severe pulmonary vasoconstriction leading to right ventricular strain. 63 Availability of perfusion technologies discussed in earlier sections has led to a renewed interest in heart transplants from DCD donors, which comprised a paltry 3.4% of hearts recovered for transplant in 2020. 64 This grew dramatically to 14.0% in 2023 and is thought to have the potential to increase the donor pool by up to 30%. 65
Early results from DCD heart transplant following NMP cited high rates of severe primary graft dysfunction requiring mechanical circulatory support. 66 Experience with the technology and nuances of NMP management as well as a focus on minimizing dWIT have demonstrated a reduction in severe graft dysfunction. A recent multicenter, randomized controlled trial comparing cDCD NMP hearts to those recovered from DBD donors demonstrated no difference in patient or graft survival at 30 days and 1 year in spite of higher rates of early graft dysfunction. 67
Much of the data on NMP heart transplants come from Australia and the United Kingdom. Use of TA-NRP for heart transplant has been explored at high volume centers in the United States. Outcomes following heart transplant for DCD donors using both NMP and NRP have been shown to be non-inferior to those from DBD donors.67,68
Conclusion
Organ transplantation in the United States has steadily expanded over the years. Deceased donation has remained the primary source of organs, with both brain-dead and cardiac-dead donors now accepted as standard. More recently, a paradigm shift has occurred with the growing use of donation after circulatory death (DCD), supported by advances in regional perfusion and ex vivo organ preservation technologies.
At present, challenges remain—particularly related to organ acquisition costs, insurance coverage, and logistical barriers—that may limit further expansion of DCD utilization. Nonetheless, the quality of DCD organs has become increasingly comparable to those from brain-dead donors. Living donor transplantation, while widely practiced in Asia, has been slower to gain momentum in the United States. However, kidney transplantation has shown growth due to initiatives such as the National Kidney Registry and paired kidney exchange programs. Thoracic organ transplantation has also benefited from the acceptance of cardiac-dead donors, expanding the donor pool.
Looking to the future, the greatest opportunities may lie in xenotransplantation and artificial organ development. Despite incremental increases in organ donation, the disparity between organ supply and recipient need remains profound. Early clinical trials in xenotransplantation have demonstrated encouraging results, suggesting that what was once a distant goal may become a clinical reality sooner than anticipated.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.