Abstract
Background:
Hypertension is a major cause of cardiovascular disease, chronic kidney disease (CKD), and death. Several studies have demonstrated the efficacy of home blood pressure telemonitoring (HBPT) for blood pressure (BP) control and outcomes, but the effects of this intervention remain unclear in patients with CKD.
Objective:
To determine the impact of HBPT on cardiovascular–related and kidney disease–related outcomes in patients with CKD.
Design:
Systematic review and meta-analysis.
Setting:
All studies that met our criteria regardless of country of origin.
Participants:
Patients with chronic kidney disease included in studies using HBPT for BP assessment and control.
Measurements:
Descriptive and quantitative analysis of our primary and secondary outcomes.
Methods:
We searched MEDLINE, Embase, CINAHL Plus, PsycINFO, Cochrane CENTRAL, Web of Science, and gray literature from inception for observational and randomized controlled studies in nondialysis (ND) CKD using HBPT for BP control. We selected studies that used HBPT as intervention (with or without a control arm) for BP control in ND-CKD populations. The primary outcome was change in mean systolic BP (SBP) and mean diastolic BP (DBP).
Results:
We selected 7 studies from 1669 articles that were initially identified. Overall, pooled estimates in the mean difference (MD) for SBP and DBP were −8.8 mm Hg; 95% confidence interval (CI): −16.2 to −1.4; P = .02 and −2.4 mm Hg; 95% CI: −3.8 to −1.0; P < .001, respectively. For studies comparing intervention with usual care (UC), pooled estimate in MD for SBP was −8.0 mm Hg (P = .02) with no significant reduction for DBP (−2.6 mm Hg; P = .18). In studies without a UC arm, both SBP and DBP were not significantly reduced (P > .05). The pooled estimate in MD for estimated glomerular filtration rate showed a significant improvement (5.4 mL/min/1.73 m2; P < .001).
Limitations:
Heterogeneity and few available studies for inclusion limited our ability to identify a robust link between HBPT use and BP and kidney function improvement.
Conclusion:
Home blood pressure telemonitoring is associated with mild lowering of BP and moderately improved kidney function in patients with CKD. However, larger studies with improved designs and prolonged interventions are still needed to assess the effects of HBPT on patients’ outcomes.
PROSPERO registration ID
Introduction
The prevalence of systemic arterial hypertension continues to rise globally, especially in developing countries. The global prevalence of hypertension (defined as systolic blood pressure [SBP] ≥140 mm Hg and/or diastolic blood pressure [DBP] ≥90 mm Hg) in adults was estimated to be 31.1% in 2010, corresponding to 1.38 billion people affected. 1 Although treatment options are available, blood pressure (BP) control remains suboptimal, particularly in low- and middle-income countries, due to poor adherence, clinical inertia, and organizational failure.1,2 A major challenge associated with hypertension care is the proportion of patients able to achieve BP control to target. One large survey involving more than 1.5 million people across 92 countries who were screened for hypertension showed that only 31.7% were controlled to <140/90 mm Hg and 23.3% had untreated or inadequately treated hypertension. 3 The consequence of poorly controlled hypertension is an increase in target organ damage including cardiovascular (CV) disease, stroke, chronic kidney disease (CKD), and increased mortality.4-8
Measurement of BP remains an important aspect of care associated with BP control. Home BP monitoring (HBPM) has been shown to be potentially more reproducible than either office BP monitoring (OBPM) or ambulatory BP monitoring and is more accurate and superior to OBPM in predicting CV events and all-cause mortality.9,10 Moreover, as OBPM does not always correctly identify patients with hypertension due to “white-coat” or “masking” effects, HBPM improves BP monitoring and provides more representative BP data and better prediction of outcomes. 11 The ability to transmit, in real time, data from HBPM device to a care giver improves the chance of better BP control and accelerated delivery of best practice. When combined with decision-making strategies, it can also reduce adverse outcomes associated with hypertension.12,13
Home BP telemonitoring (HBPT) is based on the use of electronic automated BP monitors storing BP values obtained at patient’s home and promotes a more effective link between patients and their caregivers.12,14 In the Telemonitoring and Self-Management in Hypertensions (TASMINH2) study, participants made medication changes, felt confident about self-monitoring, and felt that their home readings were more valid than office readings taken by their doctor.15,16 A following study (TASMINH4) reported that, in comparison with usual care (UC), the mean SBP difference with telemonitoring was −3.5 mm Hg. 17 Home BP telemonitoring has also been shown to be both cost effective and more effective in achieving BP control than UC. 18 In addition, when HBPT is combined with additional care (counseling, education, behavioral management, etc), there was a further increased mean change in SBP and DBP as compared with HBPT alone, suggesting that HBPT can be more efficacious when additional support is provided. 18 A number of other studies have equally demonstrated the efficacy of HBPT on BP control and outcomes.13,15-17,19,20 One systematic review that assessed the role of HBPT in patients with CKD and included only 2 studies in quantitative analysis did not find statistically significant changes in SBP, DBP, mean arterial pressure (MAP), or estimated glomerular filtration rate (eGFR) at the end of the studies from baseline values. 21 This is likely related to the small sample size and limited number of studies included. However, researchers have pointed to the role of out-of-office BP monitoring in patients with CKD, particularly in patients on hemodialysis where self-measured BP but not predialysis or postdialysis BP shows high sensitivity and specificity of >80% to make a diagnosis of hypertension. 22
Objectives
We systematically reviewed studies using HBPT in nondialysis (ND) patients with CKD to determine the impact of HBPT and management support on BP control, CV-related and kidney disease–related outcomes in this population.
Methods
This systematic review and meta-analysis is reported using the established framework of Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA). 23 The protocol for this review was registered in PROSPERO (CRD42020190705) and has been published. 24
Eligibility Criteria
Studies published from inception to June 30, 2020 were included, in all publication languages, if HBPT was used for BP assessment and monitoring irrespective of additional management support by physicians and/or allied health care workers in ND-CKD adults (≥18 years of age). We accepted the definition of CKD as used by the authors. We also included observational studies, randomized controlled trials (RCTs), and published abstracts that evaluated telemonitoring for BP control and reported at least 1 outcome. Studies comparing HBPT with standardized care and other interventions, such as counseling and education around BP management, use of other BP measuring devices, etc, were included, as well as HBPT studies in ND-CKD with no comparators. Studies were excluded if other forms of eHealth (eg, telemedicine, short message service [SMS]) were used for BP assessment, if a specific pharmacological management was utilized for BP control, if patients were receiving kidney replacement therapy (KRT), and if BP change was not reported or cannot be deduced. We also excluded review articles, editorials, letters to the editor, commentaries, case studies, case reports, images, and studies from which we were unable to gather relevant data even after attempts to obtain these from the authors. We defined HBPT as the use of an electronic automated BP monitor storing BP readings obtained at home and transmitted to a remote computer through a wired or wireless telephone line, a modem, or an Internet connection. 25
Outcomes
The primary outcome was defined as any change in mean SBP, mean DBP, MAP, and/or proportion of participants with controlled BP as defined by the study investigators. Secondary outcomes included clinical and biochemical factors related to kidney disease (eg changes in eGFR, proteinuria, or requirement for KRT), hospitalizations, incident fatal and nonfatal CV events, all-cause mortality, cost effectiveness, patient-reported outcome measures, and patient-reported experience measures.
Search Strategy for Identifying Relevant Studies
We searched MEDLINE (Ovid), Embase (Ovid), CINAHL Plus with Full Text (EBSCO), PsycINFO (Ovid), Cochrane CENTRAL (Cochrane Library), Web of Science, and Dissertations and Theses Global (ProQuest). Searches employed both controlled vocabularies, such as Medical Subject Headings (MeSH), and keywords representing concepts such as: (hypertension or blood pressure) AND (chronic kidney disease or chronic renal insufficiency) AND (telemedicine or virtual monitoring). The search was conducted by our medical librarian (L.H.) and the strategy was reviewed for completeness and accuracy using Peer Review of Electronic Search Strategies (PRESS) 26 by a second research librarian. No limitations or filters were used, and search strategies were adapted for each database (Supplementary Table S1).
Study Selection
Identified studies were added to the Covidence database (https://www.covidence.org/) and 2 reviewers (S.M. and M.T.) independently screened the titles and abstracts of the records retrieved by database searches. Subsequently, full texts of all potentially eligible articles were obtained and further assessed for final inclusion (Figure 1—PRISMA flow chart). Disagreements were resolved by a third reviewer (I.G.O.).
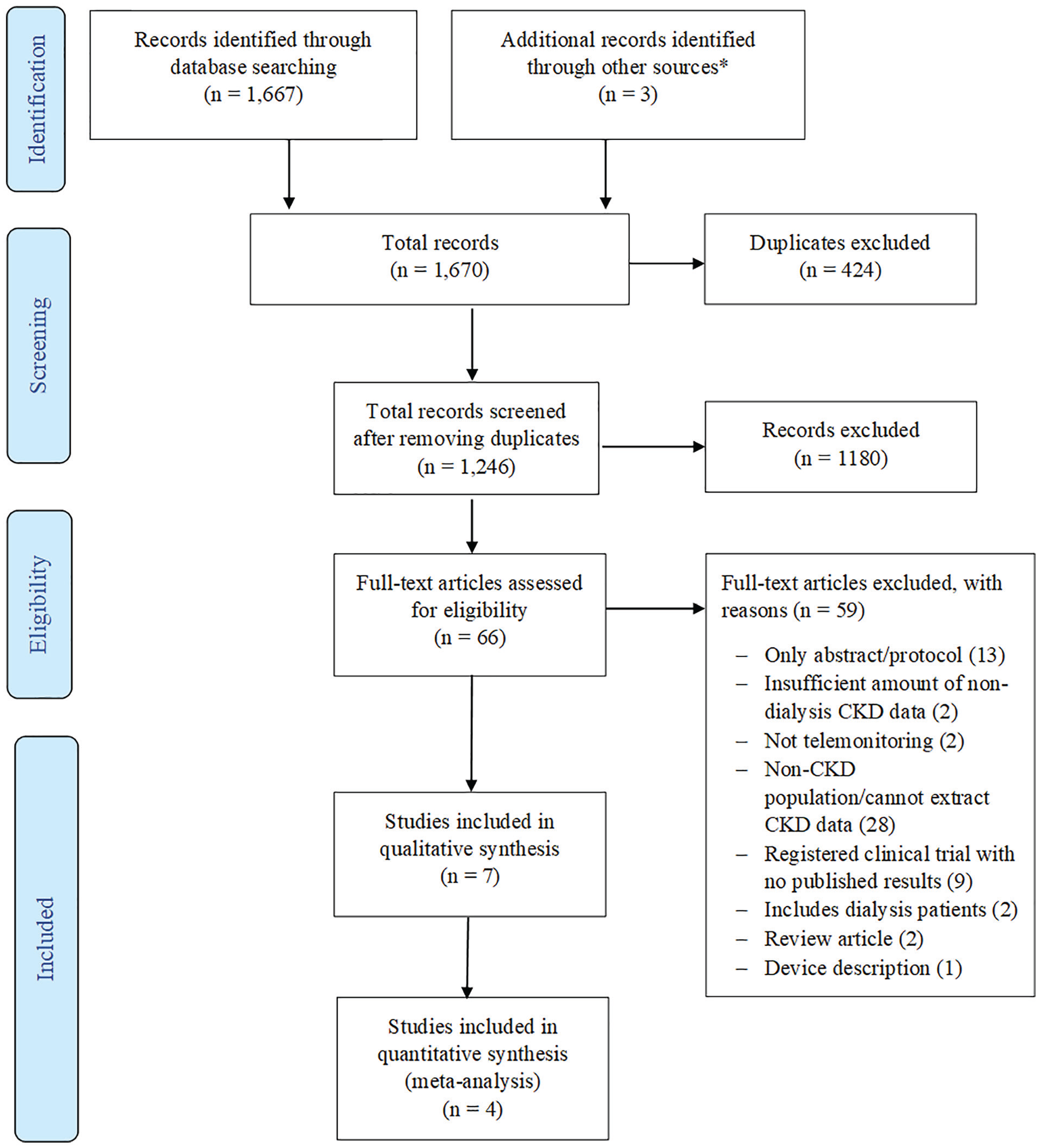
Study selection process.
Data Extraction
Data were extracted onto a predeveloped data capture sheet and reviewed by 2 investigators (D.Z. and I.G.O.) for accuracy and completeness. The following data elements were collected for each study as detailed below:
Study characteristics: Publication year, country, study design, sample size, study length, intervention length, management support type, and type of HBPT device used.
Participant characteristics: Total number of participants, total number and percent of intervention and control patients (where applicable), CKD stage or eGFR value, and comorbidities (eg, number of diabetic patients). We also collected data related to participants’ clinical and biochemical variables including antihypertensive medications, mean SBP, DBP, creatinine, and eGFR levels.
Risk of Bias in Individual Studies
We adapted and used the 9-item tool developed by Hoy et al 27 and used it to assess the methodological quality of included studies (Supplementary Table S2). Studies were classified according to their overall score as high (1-3), medium (4-6), or low (7-9) quality. Three reviewers (M.T., D.Z., I.G.O.) independently assessed the quality of the studies and we assessed interrater agreement for study inclusion using the kappa (κ) coefficient. 28
Data Synthesis and Analysis
Results were pooled according to the study designs. In before-after studies (or HBPT arm-only studies), the mean difference (MD) was calculated as the follow-up mean minus the baseline mean (change from baseline). If the change from baseline SD was not available, then a within-group conservative correlation of 0.5 was assumed, as previously reported.29,30 In studies with control groups, the MD was calculated as the change from baseline in the HBPT group minus the change from baseline in the UC group. Missing SDs were calculated using 95% confidence intervals (CIs) or imputed using the interquartile range (IQR) when available.31,32 If no data on spread were reported, then missing SDs were imputed using the arithmetic mean of the SDs from the same study designs.31,32 A random-effects model was used to pool the MDs. 33 Statistical heterogeneity was tested using the χ2 test (considering a value of P <.1 to indicate heterogeneity) and quantified using the I2 statistic (I2 values of <25%, 25-50%, and >50% represent low, medium, and high heterogeneity, respectively). 29 Due to small numbers of contributing studies (<10 within study design), there was insufficient power to assess publication bias 34 and examine subgroups. Sensitivity analysis was performed using follow-up mean values only to calculate MD in the studies with control groups for meta-analysis. Data were analyzed using Stata, Version 15.1 (StataCorp LP, College Station, TX).
Patients and Public Involvement
Patients and the public were not involved.
Results
Baseline Demographic, Clinical, and Laboratory Features of Included Studies
Our search identified 1669 articles; after removing duplicates, we screened 1245 articles by titles and abstract. Following this, we reviewed the full texts of 66 articles from which 7 articles35-41 were selected for inclusion (Figure 1). The included studies were conducted across 6 countries (the United States,35,39 Belgium, 36 Taiwan, 37 Japan, 38 Canada, 40 and the United Kingdom 41 ), comprising 3 RCTs35,37,39 and 4 observational studies (2 were described as pilot implementation studies) with an overall sample size of 821 participants. The mean age of participants ranged from 58 to 75.3 years; 5 of the studies37-41 reported more male participants (55%-98.5%), and 3 studies35,39,40 reported more white participants. Physicians, general practitioners, and/or interdisciplinary teams (physicians, nurses, pharmacists, etc) provided management support in 5 (71.4%) of the studies (Table 1). Quality assessment of the studies (interrater agreement: 57.1%) revealed 5 of the studies (71.4%) to be of moderate quality,35-37,40,41 with 1 low-quality 38 and 1 high-quality 39 study. Baseline clinical and laboratory features are summarized in Table 2. Mean baseline SBP and DBP in the studies ranged from 127.8 to 152.5 mm Hg and 70.3 to 82 mm Hg, respectively. Only 2 studies35,40 reported the total number of medications taken by patients, while 5 studies35-37,39,41 reported baseline data on eGFR (range: 25.0-39.4 mL/min/1.73 m2).
Baseline Demographic Features of Studies Included in This Review.

Note. RCT = randomized controlled trial; OBS = observational study; UC = usual care; HBPT = home blood pressure telemonitoring; DM = diabetes mellitus; BP = blood pressure; CKD = chronic kidney disease; NR = not reported; IT = information technology; ESH = European Society of Hypertension.
Baseline Clinical and Laboratory Features Reported From Studies Included in This Review.

Note. SBP = systolic blood pressure; DBP = diastolic blood pressure; BP = blood pressure; eGFR = estimated glomerular filtration rate; HBPT = home blood pressure telemonitoring; UC = usual care;
Uncontrolled hypertension (>140/90 mm Hg).
Summary of Reported Outcomes and the Effects of Interventions From Individual Studies
Reported outcomes and effects of interventions from each study are summarized in Table 3. Although all the studies reported baseline BP, only 2 studies36,37 focused on improvement of BP throughout the study period, 3 studies35,40,41 focused on the acceptability of HBPT to patients with CKD, and 1 study 38 focused on the effect of seasonal temperature variation on HBPT. One study 39 that assessed a primary composite endpoint of death, hospitalization, emergency department visits, and admission to a skilled nursing facility failed to show that HBPT yields significantly better outcomes than UC.
Change in BP, Summary of Reported Outcomes, and Summary of the Effects of Interventions for Studies Included in This Review.

Note. SBP = systolic blood pressure; DBP = diastolic blood pressure; MAP = mean arterial pressure; HBPT = home blood pressure telemonitoring; UC = usual care; CKD = chronic kidney disease; ESKD = end-stage kidney disease; eGFR = estimated glomerular filtration rate; LDL-C = low density lipoprotein cholesterol; HR = hazard ratio; CI = confidence interval; IQR = interquartile range; RCT = randomized controlled trial; BP = blood pressure; BMI = body mass index; DM = diabetes mellitus.
Pooled Summary of Study Outcomes
BP Change
Four studies35-37,40 were included in meta-analysis for BP. Two studies35,37 with a UC arm assessed SBP and DBP changes after 6 months of follow-up in 79 participants. The baseline SBP and DBP measurements of the 2 studies were not significantly different (Supplementary Figure S1). The pooled data, however, showed a significantly lower SBP (MD: −7.98 mm Hg; 95% CI: −13.99 to −1.97; I2 = 0.0%; P = .01) and a nonsignificant difference in DBP (MD: −0.97 mm Hg; 95% CI: −10.17 to 8.23; I2 = 81.4%; P = .84; Figure 2) between HBPT and UC groups. Two other studies36,40 without a UC arm assessed BP changes after 6 months of follow-up in 51 patients. The pooled results showed no significant difference in both SBP (MD: −10.3 mm Hg; 95% CI: −24.58 to 3.99; I2 = 93.9%; P = .16) and DBP (MD: −3.61 mm Hg; 95% CI: −8.05 to 0.82; I2 = 63.8%; P = .11; Figure 2). Changes in SBP after 3 months of intervention were reported by 2 of the studies (without a UC arm in 53 participants)35,41 with pooled estimate showing a significant MD (−11.11 mm Hg; 95% CI: −15.36 to −6.85; I2 = 0.0%; P < .001; Supplementary Figure S2). The summary estimates remained essentially unchanged in sensitivity analyses using all follow-up data; however, MD in overall SBP (n = 130; MD: −8.8 mm Hg; P = .02) and DBP (n = 130, MD: −2.4 mm Hg; P < .001) were significantly reduced (Figure 3).

Forest plot and meta-analysis for the effect of HBPT on SBP, DBP, and eGFR after 6 months of follow-up.
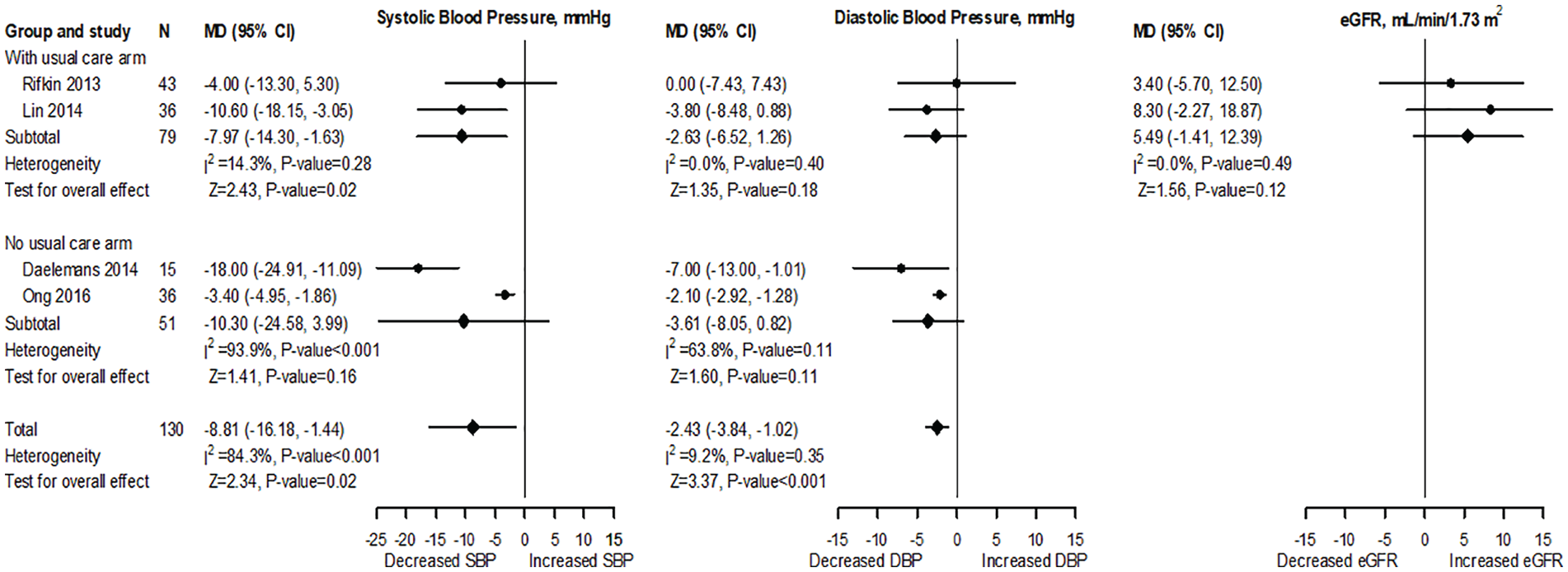
Forest plot and meta-analysis for the effect of HBPT on SBP, DBP, and eGFR using 6-month follow-up mean values (sensitivity analysis).
eGFR Change
Two studies35,37 with a UC arm and baseline GFR values that were not significantly different (Supplementary Figure S1) evaluated changes of eGFR after 6 months of follow-up in 79 participants. The pooled results showed a significant difference (MD: 5.35 mL/min/1.73 m2; 95% CI: 2.49-8.21; I2 = 0.0%; P < .001; Figure 2). However, sensitivity analysis using all the follow-up measurements showed that there was a nonsignificant difference (MD: 5.49 mL/min/1.73 m2; 95% CI: −1.41 to 12.39; I2 = 0.0%; P = .12; Figure 3).
Antihypertensive Medication Change
Only 1 study 35 reported the changes in number of antihypertensive medications after 6 months of follow-up (MD: 0.10; 95% CI: −1.57 to 1.77).
Discussion
Systemic arterial hypertension is a public health problem that continues to increase globally and remains a major cause of CV disease, CKD, and death.1,4,5,8 The aim of this study was to assess the impact of HBPT in patients with CKD and our results have demonstrated a statistically significant impact of HBPT on overall SBP and DBP reduction as well as some improvement of kidney function (eGFR); however, no significant changes in DBP (within 3 months and when studies were separated into groups—with UC vs without UC) or number of antihypertensive medications required by patients with CKD were observed. Qualitative analysis of our study showed that HBPT was acceptable to patients with CKD with no significant effect on patient hospitalization, readmission, and mortality.
The MD for BP was only significantly reduced for SBP in studies comparing HBPT and UC (−7.9 mm Hg; P = .02), whereas the MD was not significantly reduced for DBP (−2.6 mm Hg; P = .18) and for both SBP and DBP in studies without UC (−10.3 mm Hg; P = .16 and −3.61 mm Hg; P = .11), respectively (Figure 3). Luo et al 21 had also assessed the effects of telehealth on BP management in ND-CKD (stages 3-5 only) but did not identify significant reductions in SBP and DBP. They suggested 2 main reasons for this: (1) lack of significance due to statistically low sample size of telehealth studies in patients with CKD and improper study designs of those studies and (2) telehealth for BP management in CKD may be clinically ineffective as it fails to meet the demands of clinical practice. 21 Based on the results of our data, we think both reasons could apply as the increased sampling in our study was able to detect a significant reduction in SBP in studies comparing HBPT with UC and that the studies we included used different numbers of BP transmission, duration of telemonitoring, and management support for BP evaluation and control in their patients (Tables 2 and 3).
Other studies have suggested that a clinically significant BP-lowering effect of home (self) BP monitoring is proportional to the intensity of co-interventions administered as well as use of a multidisciplinary team.21,35,42 One systematic review that included 25 RCTs reported that effect of self-monitoring of BP was strongly influenced by the intensity of co-intervention ranging from no effect with self-monitoring alone (−1.0 mm Hg; 95% CI: −3.3 to 1.2) to a reduction when monitoring was combined with intensive support (6.1 mm Hg; 95% CI: −9.0 to −3.2). 42 Also, a cluster randomized study that included of a pharmacist for management support in the HBPT group reported significant BP reductions at 12 months that persisted during 6 months of postintervention follow-up. 19 How management support was utilized in the included studies may have influenced the extent of pooled BP reduction observed in our study. Although modest, with most BP reductions not statistically significant, they are, however, clinically important reductions as several studies have already shown the benefits of even small reductions in SBP and DBP for primary prevention with up to 10% reduction in CV mortality and up to 20% to 30% reduction in major CV events.43-45 Additional factors that influence changes in BP when using HBPT have been reported to include the duration of the intervention, the frequency of remote transmission of BP data, additional interventions, and the intervention pathway (mobile phone/web, telephone-linked computer system, or telephone) on HBPT. 46 Although the differences in these factors across studies we included could have contributed to the measure of change in both SBP and DBP in our pooled analysis, the availability of only a few studies using HBPT in patients with CKD may have had more significant effects on our findings.
Hypertension is associated with more rapid decline of eGFR and there is evidence that BP treatment in patients with CKD may attenuate the decline in eGFR.47,48 Two of the studies included in this meta-analysis35,37 reported nonsignificantly improved eGFR in the intervention group at 6 months. Rifkin et al showed that the eGFR in the HBPT group was maintained at the end of the study (0.6 mm Hg; IQR: −3.4 to 1.8) with a decline in the UC group (−3.69 mm Hg; IQR: −6.2 to 0; P = .14). Although the pooled effect of BP reduction between HBPT and UC on eGFR from both studies demonstrated a significant increase (5.35 ml/min/1.73 m2; 95% CI: 2.49-8.21; P =<.001), sensitivity analyses using all follow-up data showed a nonsignificant increase in eGFR (5.66 ml/min/1.73 m2; 95% CI: −1.51 to 12.83; P = .12) suggesting the initial analysis to be an overestimation of the true effects of HBPT on eGFR (Figures 2 and 3). Unfortunately, the lack of clinical trials that have evaluated the effects of HBPT for BP control on kidney function could not allow for a robust assessment of the effects of using this intervention on kidney function.15,17,19,49 We think that the lack of clinical trials may be related to the high cost associated with using HBPT, especially given the high cost associated with CKD treatment. Stoddart et al 50 found HBPT costs to be significantly higher than UC (P < .001) with the increased costs related to telemonitoring services, patient training, and additional general practitioner and nurse consultations. Despite the associated cost, other studies have shown HBPT to be more cost-effective than home monitoring alone or UC. 51 Another reason for the lack of clinical trials might also be the assumption that eHealth interventions in patients with CKD lack efficacy. A large systematic review designed to evaluate the benefits and harms of using eHealth interventions in people with CKD found eHealth interventions to only improve the management of dietary sodium intake and fluid management (out of 98 outcomes categorized into 9 domains). 52 This was reported to be due to the low quality of studies with uncertain effects and due to methodological limitations and heterogeneity of eHealth modalities and intervention types. More RCTs are clearly needed to assess the role of HBPT on kidney function both in patients with and without CKD.
Finally, our study showed an increased MD in number of antihypertensives prescribed to patients at 6 months from baseline, and qualitative analysis showed that patients found the intervention to be acceptable with no significant effects of HBPT on patient hospitalization, readmission, and mortality. The increased number of antihypertensives highlights one of the advantages of using HBPT for BP assessments as dose and/or number of medications can be adjusted by the intervention team to improve BP control as well as limit medications side effects. 40 This is corroborated by other studies17,19 that have shown that intervention patients had greater antihypertensive medication intensification and better self-reported adherence to antihypertensive medications than UC patients. Some studies have also shown improved adherence to lifestyle measures (eg, salt restriction, increased physical activity, and reduced consumption of alcohol), as an intervention to improve BP in the intervention group than the UC group, suggesting the additive effects of co-interventions in BP reduction. 19 The impact of HBPT on patient’s attitude was not consistently reported in this study as only 3 of the included studies assessed the acceptability of HBPT.35,40,41 However, in all 3 studies, patients found this method of BP assessment to be acceptable. Although the cost of equipment and nonadherence to BP measurements could likely contribute to the acceptability and persistence of telemonitoring techniques, Warner et al 41 showed high levels of acceptability of HBPT technique in their study at 3 months as they reported 92% acceptability for participants providing >65% BP readings. Adherence was also high at 1 year in the TASMINH4 study, suggesting high acceptability of the technique. Similarly, in the TASMINH2 study, patients found the intervention to be acceptable which increased their confidence in taking control of their own care by self-titrating medications when required. 16 Further, age and frailty could influence the associations of HBPT and related outcomes. A study by Takahashi et al 53 that assessed changes in hospitalizations and emergency visits among older adults (patients’ mean age 80.3 ± 8.2 years, 20.5% with CKD) showed that telemonitoring did not lead to reduced hospitalizations or ED visits. These findings as reported by Takahashi et al could be explained by the select nature of their cohort and baseline comorbidity burden. 53 Thus, the results of ongoing larger pragmatic RCTs utilizing HBPT in patients with CKD, such as the eNephro study 54 (NCT02082093) and one by our group, the telemonitoring of hypertensive patients with CKD (Telemonitoring for Improved Kidney Outcomes [TIKO]) study (NCT04098354), are likely to improve our understanding of the efficacy, cost utility, and acceptability of HBPT interventions in patients with CKD.
There were some limitations in our study including heterogeneity across studies which may be explained by differences in clinical settings, telemonitoring technologies, timing of self-monitoring, number of antihypertensive medications, number of BP readings/transmissions, differences in BP targets, and the features of the comparative group. Also, due to heterogeneity with CKD definitions and lack of eGFR stratification, we were unable to conduct subgroup analyses by stage of CKD. One study 38 assessed the effect of seasonal temperature variation on BP values without providing further patient details and was not included in the meta-analysis. Finally, the low sample size of included studies (eg, the sample size of all included RCTs was only 683 individuals)35,37,39 may have contributed to limiting the magnitude of the observed effectiveness of HBPT on outcomes and could be due to low availability of studies using telemonitoring technologies for BP management in CKD populations. Despite these limitations, as we have shown a significant effect of HBPT on SBP and kidney function as well as an increased acceptance of this technology, this may support limited use of HBPT in patients with CKD. Although this study exposes the scant availability of properly designed trials with adequate sample size to address the issue of efficacy of HBPT in patients with CKD, it however reveals the need for more RCTs evaluating the effects of HBPT on kidney function. One such RCT is the ongoing Canadian study—TIKO study (www.clinicaltrials.gov registration number: NCT04098354). 55 This study will assess mean change in SBP, kidney outcomes, and acceptability of HBPT at 12 months, in patients with CKD randomized to HBPT + UC versus HBPT + case manager. 55 Furthermore, through the delivery of the knowledge translation process developed for this study (Supplementary Appendix), the benefits of HBPT can be extended and broadly implemented by other knowledge users.
Conclusion
This systematic review and meta-analysis show that HBPT is associated with mild reductions of SBP and moderately improved kidney function in patients with CKD, likely modulated by HBPT effects on SBP. Although our study shows that this intervention is acceptable to patients with CKD, our results should be taken cautiously given the identified limitations including small sample size of included studies and differences in HBPT technologies utilized across different studies. We hope that future studies as well as ongoing RCTs with larger sample sizes and prolonged intervention duration may be able to provide more robust evidence of the usefulness of HBPT on BP, kidney, and CV outcomes in patients with CKD.
Supplemental Material
sj-pdf-1-cjk-10.1177_20543581221106248 – Supplemental material for Impact of Home Telemonitoring and Management Support on Blood Pressure Control in Nondialysis CKD: A Systematic Review and Meta-Analysis
Supplemental material, sj-pdf-1-cjk-10.1177_20543581221106248 for Impact of Home Telemonitoring and Management Support on Blood Pressure Control in Nondialysis CKD: A Systematic Review and Meta-Analysis by Shezel Muneer, Ikechi G. Okpechi, Feng Ye, Deenaz Zaidi, Mohammed M. Tinwala, Laura N. Hamonic, Anukul Ghimire, Naima Sultana, Dan Slabu, Maryam Khan, Branko Braam, Kailash Jindal, Scott Klarenbach, Raj Padwal, Jennifer Ringrose, Nairne Scott-Douglas, Soroush Shojai, Stephanie Thompson and Aminu K. Bello in Canadian Journal of Kidney Health and Disease
Footnotes
Author Contributions
Conceptualization: I.G.O. and A.K.B. were responsible for the conception and design of the work.
Funding acquisition: A.K.B. was responsible for funding acquisition.
Methodology: S.M., I.G.O., F.Y., D.Z., M.M.T., L.N.H., and A.K.B. participated in the design of the study methodology.
Data analysis: F.Y. was responsible for the data analysis for this study.
Project administration: I.G.O. and A.K.B. were responsible for this project’s administration.
Writing—original draft: I.G.O. and A.K.B. were responsible for the original draft of this work.
Writing—review and editing: S.M., I.G.O., F.Y., D.Z., M.M.T., L.N.H., A.G., N.S., D.S., M.K., B.B., K.J., S.K., R.P., J.R., N.S.-D., S.S., S.T., and A.K.B. participated in review, editing, and rewriting of the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.P. is CEO of mmHg Inc, a digital health company creating guideline-concordant innovations to improve the efficiency of remote patient monitoring. All other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided through the Alberta Kidney Care—North (formerly, Northern Alberta Renal Program)/AMGEN incorporated Grant: Telemonitoring and Case Management for Hypertensive Remote-Dwelling Patients with CKD (Telemonitoring for Improved Kidney Outcomes; TIKO). “Award/Grant number is not applicable.”
Patient Consent
Not required.
Previous Presentation
Abstract of this work was accepted at the World Congress of Nephrology (Montreal Canada, 2021) and Hypertension Canada (2021); otherwise, it has never been submitted or presented elsewhere.
Supplemental Material
Supplemental material for this article is available online.
Data Sharing Statement
All data relevant to the study are included in the article or as supplementary information. All included studies are published. We will consider sharing data upon reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
