Abstract
Background:
Chronic pain is a common and distressing symptom reported by patients with chronic kidney disease (CKD). Clinical practice and research in this area do not appear to be advancing sufficiently to address the issue of chronic pain management in patients with CKD.
Objectives:
To determine the prevalence and severity of chronic pain in patients with CKD.
Design:
Systematic review and meta-analysis.
Setting:
Interventional and observational studies presenting data from 2000 or later. Exclusion criteria included acute kidney injury or studies that limited the study population to a specific cause, symptom, and/or comorbidity.
Patients:
Adults with glomerular filtration rate (GFR) category 3 to 5 CKD including dialysis patients and those managed conservatively without dialysis.
Measurements:
Data extracted included title, first author, design, country, year of data collection, publication year, mean age, stage of CKD, prevalence of pain, and severity of pain.
Methods:
Databases searched included MEDLINE, CINAHL, EMBASE, and Cochrane Library, last searched on February 3, 2020. Two reviewers independently screened all titles and abstracts, assessed potentially relevant articles, and extracted data. We estimated pooled prevalence of overall chronic pain, musculoskeletal pain, bone/joint pain, muscle pain/soreness, and neuropathic pain and the I2 statistic was computed to measure heterogeneity. Random effects models were used to account for variations in study design and sample populations and a double arcsine transformation was used in the model calculations to account for potential overweighting of studies reporting either very high or very low prevalence measurements. Pain severity scores were calibrated to a score out of 10, to compare across studies. Weighted mean severity scores and 95% confidence intervals were reported.
Results:
Sixty-eight studies representing 16 558 patients from 26 countries were included. The mean prevalence of chronic pain in hemodialysis patients was 60.5%, and the mean prevalence of moderate or severe pain was 43.6%. Although limited, pain prevalence data for peritoneal dialysis patients (35.9%), those managed conservatively without dialysis (59.8%), those following withdrawal of dialysis (39.2%), and patients with earlier GFR category of CKD (61.2%) suggest similarly high prevalence rates.
Limitations:
Studies lacked a consistent approach to defining the chronicity and nature of pain. There was also variability in the measures used to determine pain severity, limiting the ability to compare findings across populations. Furthermore, most studies reported mean severity scores for the entire cohort, rather than reporting the prevalence (numerator and denominator) for each of the pain severity categories (mild, moderate, and severe). Mean severity scores for a population do not allow for “responder analyses” nor allow for an understanding of clinically relevant pain.
Conclusions:
Chronic pain is common and often severe across diverse CKD populations providing a strong imperative to establish chronic pain management as a clinical and research priority. Future research needs to move toward a better understanding of the determinants of chronic pain and to evaluating the effectiveness of pain management strategies with particular attention to the patient outcomes such as overall symptom burden, physical function, and quality of life. The current variability in the outcome measures used to assess pain limits the ability to pool data or make comparisons among studies, which will hinder future evaluations of the efficacy and effectiveness of treatments. Recommendations for measuring and reporting pain in future CKD studies are provided.
Trial registration:
PROSPERO Registration number CRD42020166965
Introduction
Patients with chronic kidney disease (CKD) experience multiple and burdensome symptoms, the number and severity of which have been described as being similar to those of cancer patients hospitalized in palliative care settings.1-9 The high symptom burden in patients with CKD negatively affects patients’ health-related quality of life (HRQL) and functional capacity. Hence, symptom management has been identified as a top priority for patients with CKD. 10 A recent scoping review conducted as part of Kidney Disease: Improving Global Outcomes’ (KDIGO) effort to develop formal international recommendations for kidney supportive care reinforced that chronic pain is a common and distressing symptom reported by patients with CKD. 11 It is often not possible to completely alleviate chronic pain. The clinical aim is to reduce pain to levels where function is not adversely affected, which is typically perceived as “mild” pain or pain rated as 0 to 3 on a 0 to 10 numerical rating scale (NRS).12,13 However, clinical practice and research in this area do not appear to be advancing sufficiently to address the issue of chronic pain management in patients with CKD. If quality person-centered care is to be delivered, assessment and treatment strategies must be developed and integrated to align care with patient preferences and treatment goals.
Our main objective was to determine the prevalence and severity of chronic pain across broad populations of patients with CKD glomerular filtration rate (GFR) categories (G) 3 to 5. We hypothesized that extensive data exists illustrating a high pain burden across CKD G3-5.
Methods
Eligibility Criteria and Search Strategy
The literature search was developed and conducted by an experienced librarian; PROSPERO Registration number CRD42020166965. Inclusion and exclusion criteria are listed in Table 1. We included all interventional and observational studies that presented original data of the prevalence and severity of chronic pain in patients with CKD G3-5. We included studies presenting data from 2000 or later, given that the CKD population, especially those starting dialysis, have become increasingly older with greater comorbidity, which may add to the burden of chronic pain. Single case studies or case series were excluded, as were studies that were presented only as abstracts, posters, or letters to the Editor. Articles published in a language other than English were translated and included. Eligible patient populations included CKD G3-5 and ≥18 years of age. Studies that only enrolled patients with a primary diagnosis of acute kidney injury or kidney transplant patients were excluded as were studies that limited the study population to a specific cause, symptom, and/or comorbidity (with the exception of chronic pain) of CKD as these studies were outside the scope of our study objectives. Dialysis patients also experience acute pain syndromes, but these are distinct entities from chronic pain with different trajectories and impact on HRQL and function. Hence, studies that were limited to acute pain or pain related to dialysis treatment were also excluded.
Inclusion and Exclusion Criteria.

Note. CKD = chronic kidney disease; GFR = glomerular filtration rate; CKM = conservative kidney management; eGFR = estimated glomerular filtration rate; ESKD = end-stage kidney disease.
Data Items
Outcomes of interest were prevalence and severity of chronic or persistent pain, as defined by the individual studies, recognizing that definitions of chronic pain were likely to vary. To determine prevalence, both the number of cases of pain and the total number within the cohort had to be reported. In addition, eligible studies needed to report pain as either general overall pain or pain broken down into categories of musculoskeletal pain, bone/joint pain, muscle pain/soreness, and/or neuropathic pain. This was considered important as the commonly used symptom screening tools in CKD use a combination of these categories to classify pain. 11 In cases when more than one study appeared to report on the same cohort of patients, the study with the most complete data or highest methodological quality was included.
Information Sources
Information sources included electronic databases, reference lists of relevant literature, and Web sites of relevant networks, organizations, and societies. Relevant information sources that were obtained from colleagues and stakeholders and unpublished studies were also considered for inclusion. The electronic databases searched included MEDLINE, CINAHL, EMBASE, and Cochrane Library databases. These were last searched on February 3, 2020.
Study Selection, Data Collection, and Quality Assessment
Two reviewers independently screened all titles and abstracts to identify potentially relevant articles. Full texts of potentially relevant articles were retrieved and independently assessed by 2 reviewers for possible inclusion based on the predetermined selection criteria. The reference lists of reviews, systematic reviews, and guidelines were also reviewed to ensure all relevant studies were identified. The 2 reviewers compared individually recorded decisions for inclusion and exclusion and disagreements were resolved based on discussion and consensus with a third reviewer. The research team developed a standardized data extraction table using Microsoft Excel. The 2 reviewers independently populated the table from the selected full-text articles. The data extracted from each study included year and country of study, number of study participants, patient population, age, definition of pain, pain assessment tools used, and the prevalence and severity of pain. The 2 data extraction tables were subsequently compared and cross-checked for accuracy and then merged into a single unified table for data analysis and presentation in the article. Study quality was also reviewed independently by 2 reviewers using the McMaster University Critical Review for Quantitative Studies. 14 This included assessing the study design, study sample, outcomes of interest, statistical analysis, and final conclusions.
Data Analysis
Meta-analyses of prevalence data were conducted in Microsoft R Open version 3.4.1, using package meta to estimate the pooled prevalence and 95% confidence intervals.15,16 Random effects models were used to account for variations in study design and sample populations with results plotted using forest plots. A double arcsine transformation was used in the model calculations to account for the possible overweighting of studies reporting either very high or very low prevalence measurements. 17 Heterogeneity between the estimates was assessed using I2 statistics. 18 The I2 value is the percentage of total observed variation across studies due to real heterogeneity rather than chance; a value of greater than 75% is indicative of high heterogeneity. The Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) checklist was used in the reporting of this work.
The prevalence of overall chronic pain, musculoskeletal pain, bone/joint pain, muscle pain/soreness, and neuropathic pain were estimated. Some studies presented the prevalence of pain based on severity characterized as mild, moderate, or severe, with others reporting the prevalence of moderate to severe chronic pain. For those studies that reported prevalence by pain intensity, information on clinically relevant moderate to severe and severe pain was included.
Pain severity scores were calibrated to a score out of 10, to compare across studies. We ensured all scales were oriented such that a severity score of 0 represented no pain and 10 represented the worst pain. One of the studies had its score presented in the opposite orientation, which was reversed for the sake of this analysis. 19 Pain scores of zero were assumed for patients not reporting pain. Mean pain severity scores were recalculated to reflect the severity of pain for patients reporting pain in the cases where reported severity scores included those not experiencing pain (ie, removal of scores equaling 0). Weighted mean severity scores and 95% confidence intervals were reported. 20
Meta-regressions were conducted where the number of studies was sufficiently large enough to yield robust results (ie, 10 or more).21,22 Funnel-plot asymmetry was tested using a Peters’ regression to assess the possibility of publication bias.16,23 Meta-regressions on various categorical and continuous variables were conducted, both to estimate the effect of these variables on estimated prevalence and to investigate possible sources of heterogeneity. Covariates included publication year, sample size, age, country, definition of pain, and type of measurement scale used. Bubble plots were used to illustrate the effect of continuous covariates and stratified forest plots to illustrate the effects of categorical covariates.
Results
The literature review yielded 3336 citations of which 220 were deemed eligible for full-text review. Of these, 152 studies were excluded leaving 68 studies for inclusion in the analysis.2,3,5,7,8,14,19,24-84 The flow chart in Figure 1 outlines this process, including reasons for exclusion. Supplemental Table S1 provides a list of excluded studies with reasons for exclusion.
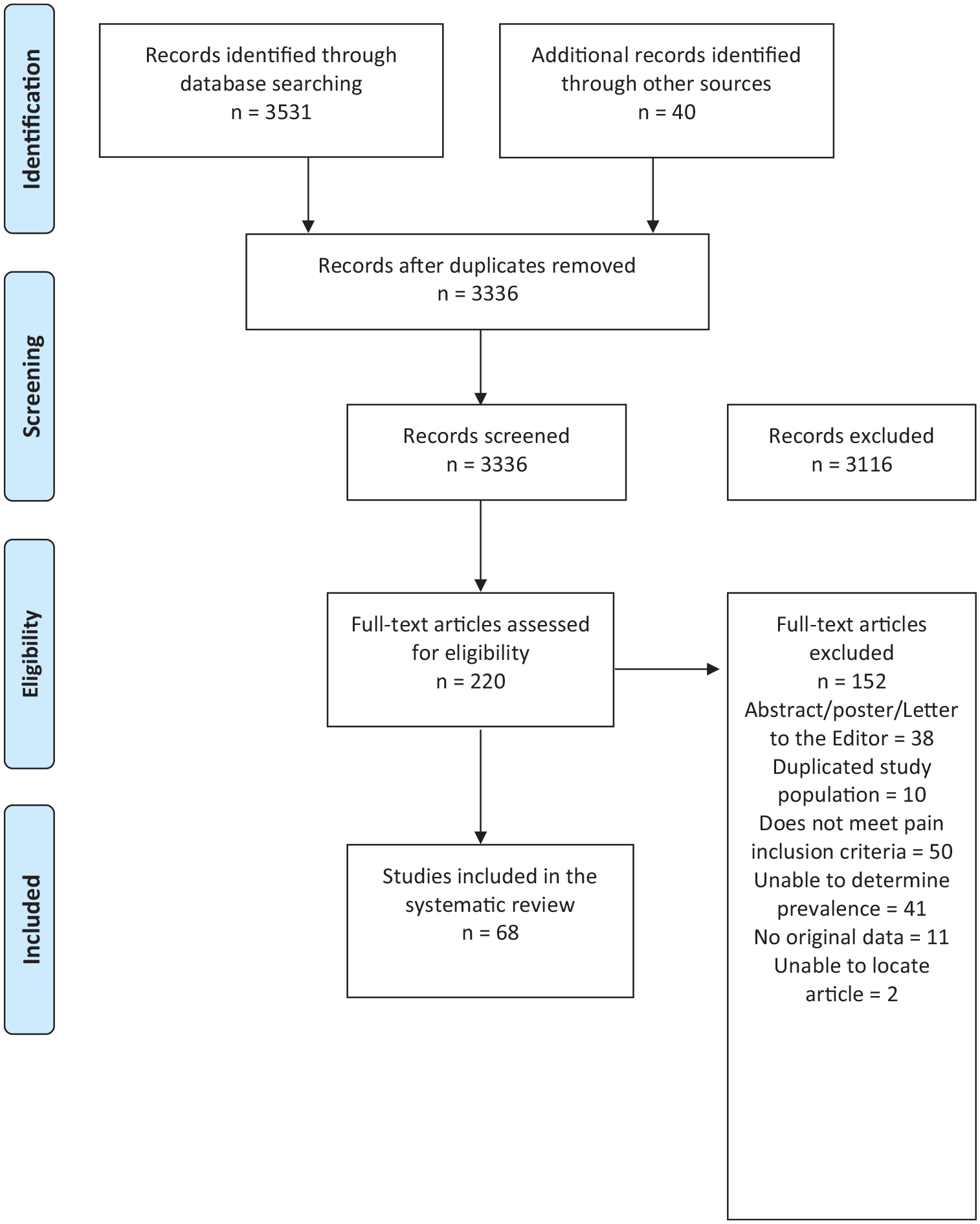
Literature search PRISMA flow diagram.
Details of Included Studies
Details of the 68 included studies are reported in Table 2 and include data from 16 558 patients from 26 countries. Forty-eight of the studies examined 8464 hemodialysis (HD) patients from 23 countries,5,7,8,14,24,28,29,32,34-41,43-46,49,51-56,58,59,61-63,65-68,70,72,73,75,76,78-84 3 studies from 3 countries included data from 679 peritoneal dialysis (PD) patients,24,49,81 and 8 studies from 6 countries reported data from 3701 patients on either HD or PD (without separating treatment groups).2,3,25,26,60,69,71,77 Two studies assessed 112 patients from Canada and the United States following the withdrawal of dialysis.19,33 Eight studies explored pain in 1361 conservative kidney management (CKM), ie, GFR category 4 (G4) and/or 5 (G5), patients from 5 countries who had chosen conservative (non-dialytic) kidney management (CKM).27,30,47,48,58,64,74,75 Nine studies from Malaysia, Saudi Arabia, Spain, Taiwan, and the United States presented data from 2241 pre-dialysis patients with various stages of CKD.24,31,42,50,57,60,71,72,78
Characteristics and Results of Included Studies.

Note. CKD = chronic kidney disease; HD = hemodialysis; PD = peritoneal dialysis; VRS = verbal rating scale; G4 = glomerular filtration rate category G4; G5ND = glomerular filtration rate category G5 not treated with maintenance dialysis; NRS = numerical rating scale; N/A = not available; CKM = conservative kidney management; VAS = visual analogue scale; VDS = verbal descriptive scale; FMS = fibromyalgia syndrome; HDF = hemodiafiltration; GFR = glomerular filtration rate; G1 = glomerular filtration rate category G1; G2 = glomerular filtration rate category G2; G3 = glomerular filtration rate category G3; NECOSAD = Netherlands Cooperative Study on the Adequacy if Dialysis.
Sample sizes varied widely from 21 to 1553 patients. Five studies31,33,46,80,83 used a yes/no categorization to determine presence of pain, one created a 28-point survey which included a pain question but was not validated, 68 one study 50 used a 10-point rating scale without further description, and one 82 study referenced a data collection sheet without further description. Of the remaining 60 studies, there was tremendous variability in the tools and severity rating scales used. A summary of the pain assessment tools used for reporting of pain prevalence and severity is presented in Table 3. Fifty-four studies used 1 of 23 different multidimensional or multi-symptom assessment tools. Most importantly, these tools used 11 different severity scales that started at either 0 or 1 with a range to 3, 4, 5, 6, 10, or 100. These scales were the NRS, the visual analogue scale (VAS), or the verbal rating scale (VRS). The VRS uses a Likert scale to ask respondents to select the verbal descriptor (eg, “mild,” “moderate,” “severe,” or “overwhelming”) that best reflects the severity of pain. A VAS consists of a horizontal line, usually 10 cm (or 100 mm) in length that is anchored with verbal descriptors. The NRS is a segmented version of the VAS in which a respondent selects a whole number that best reflects the intensity of pain, usually rated 0 for no pain to 10 for the most severe pain. Most studies characterized pain as mild when rated 1 to 3/10, moderate pain was usually defined as 4 to 6/10, and severe as 7 to 10/10. Two additional studies used a multidimensional tool with either binary yes/no or undefined responses. Fourteen studies used 1 of 7 different single-item unidimensional tools. Only 36 (53%) studies reported the prevalence of moderate and/or severe pain.2,3,8,19,25-27,29,30,32,36-42,44,45,47,48,51,52,54-57,65,67,69,72-74,77,78,84 Nine of these studies also reported mean or median severity scores.2,3,8,26,36,41,52,67,69 An additional 14 studies reported mean or median severity scores for their study cohort but without separate prevalence rates for mild, moderate, or severe pain.7,14,24,28,34,35,43,50,58-60,62,63,79
Summary of Pain Assessment Tools Used for the Reporting of Pain Prevalence and Severity.

Note. VRS = verbal rating scale; NRS = numerical rating scale; VAS = visual analogue scale; VDS = verbal descriptive scale.
The VDS is a combination of a NRS and a VRS in that each numbers has a verbal descriptor (eg, no pain, slight pain, mild pain, moderate pain, severe pain, very severe pain, the most intense pain imaginable).
There was variation and often a lack of detail regarding what constituted chronic pain. Three studies defined chronic pain as pain experienced outside of the dialysis sessions.35,41,52 Two studies defined pain at the withdrawal of dialysis, or in the last 24 hours of life following the withdrawal of dialysis.19,33 Other definitions of chronic pain ranged from pain in the past 24 hours in 1 study, 67 a duration of pain of 3 days in 1 study, 74 7 days in 20 studies,3,7,14,28,30,32,34,44,47,48,59,60,62,63,70,71,75,76,78,79 2 weeks in 1 study, 50 4 weeks in 12 studies,8,25-27,37,49,65,68,69,72,77,84 6 weeks in 1 study, 56 3 months in 11 studies,29,31,36,38-40,42,45,64,73,83 and “lasting weeks, months, or even years” in 1 study. 57 Fifteen studies did not specify a duration, despite the intent to understand chronic pain burden.2,5,24,43,46,51,53-55,58,61,66,80-82 Further details of the quality assessment for each included study are presented in Supplemental Table S2.
Prevalence and Severity of Pain
Tables 4 and 5 outline the estimated pooled prevalence of pain, and weighted mean severity of pain, for various CKD cohorts. Across the studies reporting overall pain in patients on HD, the estimated pooled prevalence was 60.5% (52.3%-68.3%) (Figure 2A). The estimated pooled prevalence of moderate or severe overall chronic pain was 43.6% (34.8%-52.7%), and the estimated pooled prevalence of severe overall chronic pain was 21.1% (12.2%-31.6%). Chronic bone/joint pain and muscle pain in patients on HD were also common with estimated pooled prevalence rates of 45.8% (35.2%-54.5%) and 44.6% (33.7%-55.7%), respectively (Figure 2B and 2C). In all cases heterogeneity was extremely high (ie, I2 > 95%). For those reporting pain, the mean severity score was 6.4 (3.7-9.0) out of 10 for overall pain, 5.9 (3.4-8.3) for bone/joint pain, and 5.3 (3.3-7.4) for muscle pain. For studies reporting median pain scores, severity of overall, bone/joint, and muscle pain were reported as 5.8, 6.0, and 2.0 out of 10, respectively. Median scores could not be adjusted for the removal of patients not experiencing pain, as such these should be interpreted with caution.
Pain Prevalence by CKD Cohort.

Note. CKD = chronic kidney disease; CI = confidence interval; HD = hemodialysis; PD = peritoneal dialysis; CKM = conservative kidney management; G3 = glomerular filtration rate category G3; G4 = glomerular filtration rate category G4; G5ND = glomerular filtration rate category G5 not treated with maintenance dialysis.
Pain Severity Synthesis by CKD Cohort.

Note. CKD = chronic kidney disease; CI = confidence interval; HD = hemodialysis; PD = peritoneal dialysis; G3 = glomerular filtration rate category G3; G4 = glomerular filtration rate category G4; G5ND = glomerular filtration rate category G5 not treated with maintenance dialysis; CKM = conservative kidney management.
Median severity may include patients who reported no pain.
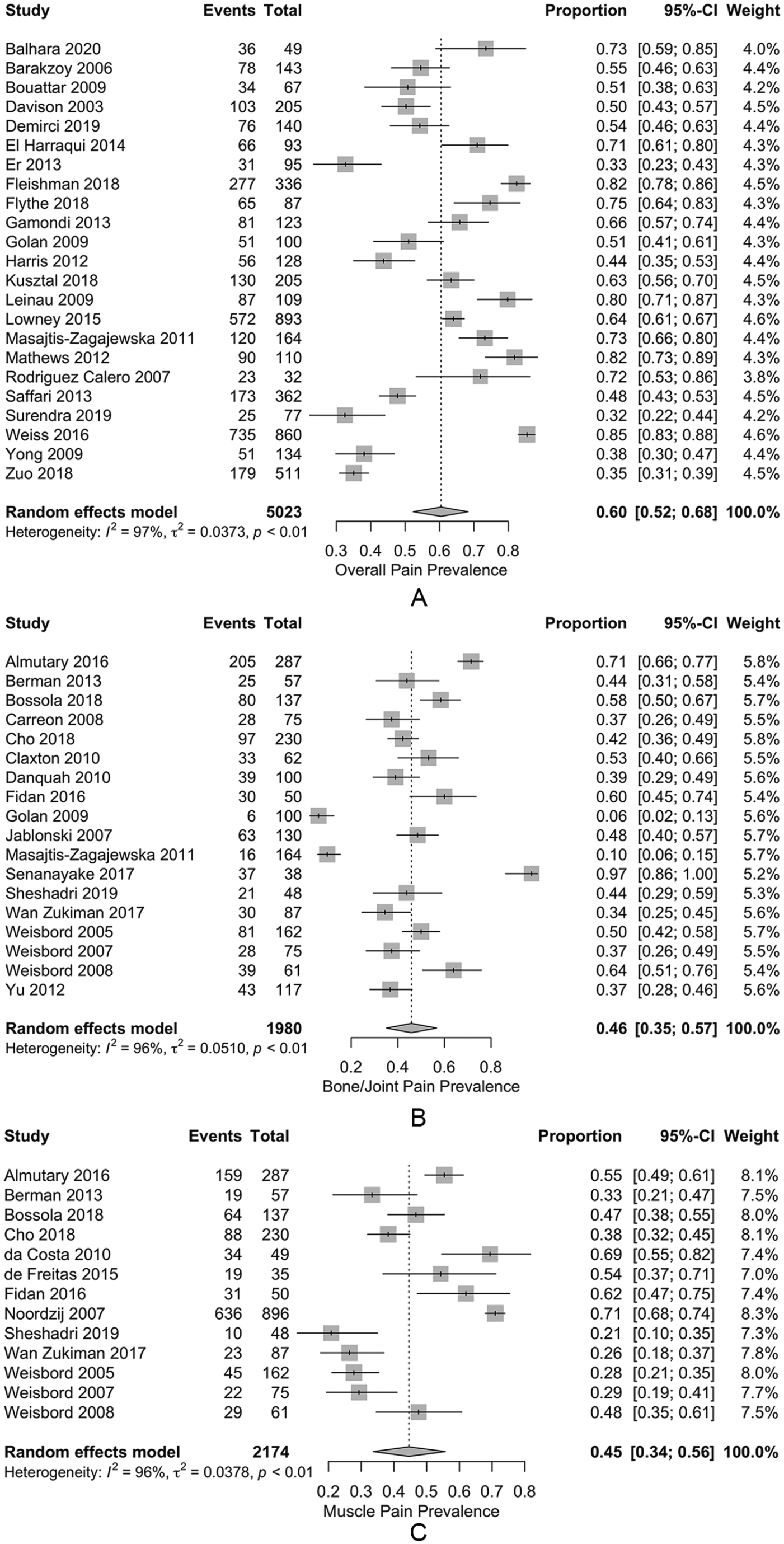
Forest plot of pooled prevalence estimates for (A) overall chronic pain, (B) bone/joint pain, and (C) muscle pain for patients on hemodialysis.
Pain prevalence rates and severity scores were similar across the other CKD cohorts. For patients on either HD or PD, the estimated pooled prevalence of overall pain was 68.3% (56.6%-78.9%), moderate to severe overall pain 40.5% (27.4%-54.3%), bone/joint pain 38.9% (29.0%-49.2%), and muscle pain 65.7% (53.9%-74.8%). Severe overall pain was not reported for this group. Heterogeneity in all groups was extremely high except for the overall pain measurement, where heterogeneity was moderate (I2 = 69.5%). Patients on PD had prevalence estimates for overall pain, bone/joint pain, and muscle pain of 35.9% (52.3%-68.3%), 50.0% (34.9%-65.2%), and 42.9% (7.4%-83.3%), respectively, although only 3 studies provided measures. Weighted mean severity scores for those reporting pain on either HD or PD were 4.4 (2.8-6.0) out of 10 for overall pain, and 5.0 (4.6-5.5) for muscle pain. Reported median severity scores for bone/joint pain and muscle pain were 5.0 and 4.0 out of 10, respectively. Peritoneal dialysis severity scores for bone/joint pain and muscle pain were reported as 3.2 (2.2-4.1) and 2.7 (2.2-3.1) out of 10, respectively.
Overall pain prevalence remained high in patients following withdrawal from dialysis (54.6%; 37.3%-71.3%), even in the last 24 hours of life (32.6%; 15.1%-52.8%). For patients with G4-5 CKD not on dialysis, the estimated pooled prevalence of overall pain and moderate to severe pain was 56.4% (43.0%-69.3%) and 27.3% (16.2%-39.9%), respectively. Cohorts specifying only G5 CKD patients managed conservatively had higher estimated pooled prevalence of overall pain and moderate to severe pain at 60.4% (27.7%-88.8%) and 35.0% (27.6%-42.7%), respectively. Heterogeneity was extremely high for overall pain reporting, but negligible in the studies reporting moderate to severe pain (I2 = 0%). The reported mean severity for these patients was 4.2 (3.5-4.9) out of 10.
Data were limited for the prevalence of pain in patients with earlier stages of CKD. While there was some variability in the reported prevalences, the combining of CKD G category in some studies and separation in others made the data difficult to interpret. In one small study, there were no statistically or clinically significant differences in mean overall pain severity scores between CKD G3 and G4-5, which ranged from 5.4 to 5.7 out of 10. 50
Meta-regressions were completed for pain prevalence in HD patients reporting overall pain, moderate to severe and severe overall pain, bone/joint pain, and muscle pain. No evidence was found for a publication bias in any of the above measures (P = .61, .89, .64, .62, and .10, respectively).
None of the meta-regressions returned evidence suggesting a difference in prevalence by either publication year or scale type. While there was evidence that muscle pain prevalence increased with larger sample sizes (P = .03), this appears to be the result of one very large sample 49 influencing results (Figure 3). There was also evidence that overall pain prevalence reports increase with cohort average age (P = .02, Figure 4). In both cases, heterogeneity was only marginally reduced (I2 = 86.7% and 94.4%, respectively).

Bubble plot of transformed prevalence of muscle pain against sample size for patients on hemodialysis.
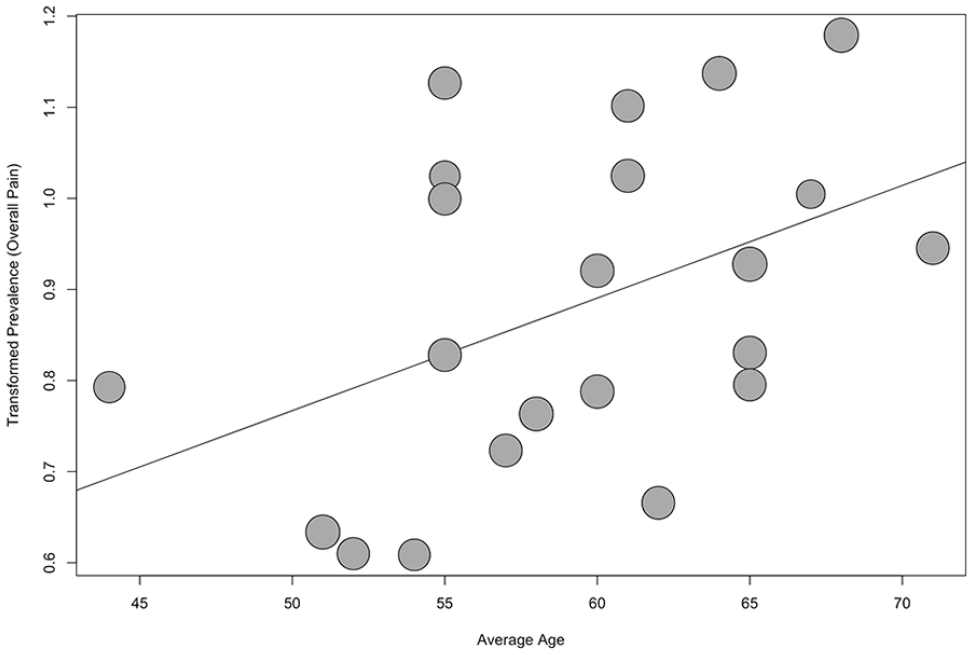
Bubble plot of transformed prevalence of overall pain against average age for patients on hemodialysis.
There was strong evidence to suggest an effect of both country and pain definition on bone/joint pain as well as muscle pain (P < .001 in all cases). Supplemental Figures S1-S4 illustrate the model results in stratified forest plots. Stratifying by country significantly reduced heterogeneity in both cases: residual I2 was 15.3% in bone/joint pain prevalence and approximately 0% in muscle pain prevalence. In both groups, there was a small cluster of Italian studies with negligible heterogeneity (I2 = 0% in both cases), at or above the ungrouped estimated pooled prevalence estimates (stratified estimates for bone/joint pain were 60.1% [53.2%-66.8%] and 47.0% [40.0%-54.0%] for muscle pain), and a larger cluster of studies out of the United States with negligible or low heterogeneity (I2 = 0% and 21.6%, respectively), estimating stratified pooled prevalence at or below the ungrouped estimates (44.6% [40.4%-48.8%] for bone/joint pain and 27.9% [23.3%-32.9%] for muscle pain). One additional cluster of 2 studies out of Brazil was present in the muscle pain model, which had a high estimated pooled prevalence and moderate heterogeneity (62.7% [47.5%-76.7%], I2 = 48.5%). The remaining countries (Malaysia, Netherlands, Saudi Arabia, South Korea, Turkey, Poland, Israel, Sri Lanka, and Taiwan) were present in one or both of the models, each with only one study and large variations of reported prevalence, with ranges of 6.0% to 97.4% for bone/joint pain and 26.4% to 71.0% for muscle pain.
When stratified by pain definition, bone/joint pain prevalence heterogeneity improved marginally, but remained high (residual I2 = 87.8). Groups included pain lasting more than 3 months, pain lasting 7 days, pain between dialysis, and no definition. The 3-month group had significantly lower scores than the rest, with an estimated pooled prevalence of 8.2% (5.0%-12.0%) and had low heterogeneity (I2 = 6.3%). Both the 7-day group and the no definition group had high pooled prevalence estimates and high heterogeneity (50.2% [41.5%-58.9%] with I2 = 88.2%, and 60.4% [44.0%-75.7%] with I2 = 90.2%, respectively). The between dialysis group only contained one study (with a reported prevalence of 39.0%). Stratification of muscle pain prevalence by pain definition only decreased heterogeneity to a moderate level (residual I2 = 66.8%). Three clusters of pain definitions in the model were pain lasting 4 weeks, pain lasting 7 days, and no definition. The stratified pooled prevalence estimates for each group were 67.6% (58.8%-75.8%), 33.9% (27.7%-40.4%), and 56.4% (51.0%-61.7%), with subgroup heterogeneity scores of I2 = 52.4%, 72.2%, and 0%, respectively.
Discussion
This systematic review contributes to the overall aim to address gaps in current knowledge around effective approaches to the evaluation and management of chronic pain for patients with CKD. The findings illustrate that chronic pain is extremely common and often severe across diverse CKD populations. Most patients who report pain rate their pain as either moderate (typically defined as 4-6 out of 10) or severe (7-10 out of 10) in severity. Data on PD patients and those cared for conservatively without dialysis are more limited, as are studies involving patients with CKD G3-5 not yet requiring renal replacement therapy, although the pain prevalence rates appear similar. The lowest reported prevalence of severe pain was in patients managed conservatively; this finding may reflect active pain management in CKM. Prevalence rates in patients with earlier stages of CKD were also high and did not appear to change with the severity of their CKD. This may reflect the fact that much of the pain in CKD is associated with the burden of comorbidity.
A recent qualitative systematic review explored prevalence and severity of pain in HD patients. 85 The 2 distinct syndromes of acute and chronic pain were synthesized together and no quantitative analyses or meta-analyses were conducted. However, the main message of the review that pain is common in patients with CKD and is typically perceived as moderate or severe in intensity is consistent with our results.
These findings have clinical implications, particularly given that symptom management is a top priority for patients with CKD. 10 Routine screening for pain in all patients with CKD should be integrated into nephrology care. This is consistent with KDIGO recommendations that state “Symptom assessment and management is an integral component of quality care for patients with advanced CKD.” Regular global symptom screening using validated tools such as the Edmonton Symptom Assessment System-revised: Renal (ESAS-r: Renal) and Palliative Care Outcome Scale-Renal (POS-renal) should be incorporated into routine clinical practice. 86 This systematic review suggests that routine symptom assessment should extend to patients with earlier GFR categories of CKD as well. The ESAS-r:Renal3,9,87 and the POS-renal74,88 are simple assessment tools that screen for several common symptoms experienced by patients with CKD. Both tools have been translated into several languages, are appropriate for screening patients even when they are pre-terminal, and perhaps, most importantly, provide the opportunity to redirect care toward a more patient-centered model. More comprehensive pain assessment tools with evidence for validity in patients with CKD are also available. 11 The VAS, VRS, and the NRS are all valid, reliable, and appropriate for use in clinical practice, although the VAS tends to be more difficult to use than the other two. 89 The NRS is often recommended as it has good sensitivity and generates data that can be more easily analyzed for research and audit purposes. 89
Many health care providers have limited expertise and feel unprepared to pursue effective treatment options for chronic pain. Some feel that it is not their responsibility to treat symptoms that are not directly related to CKD or dialysis and are therefore reluctant to prescribe and monitor analgesics. 90 Many of these barriers result from inadequate training in the basic principles of palliative care such as symptom and pain management. Several surveys of renal fellows reported that they receive little education in palliative and end-of-life care; only 44% of fellows in 2013 reported being explicitly taught how to treat dialysis patients’ pain 91 (although this was an increase from 30% in 2003) 92 and only 9.4% felt very comfortable treating pain in patients with advanced CKD. 93 However, nearly all the fellows thought that it was important to receive education on appropriate palliative care. Enhanced education in pain management will be required to address the burden of pain experienced by patients with CKD. 86
These findings also have research implications. Developing and evaluating the relative effectiveness of pain management strategies should be assessed with particular attention to the impact on patient outcomes such as overall symptom burden, physical function, and HRQL. Most treatment recommendations have been extrapolated from treatments used successfully in the general population, with special considerations made for the selection of various analgesics based on their different pharmacokinetic properties in renal failure. In addition, future studies should be more inclusive across CKD G3-5 populations and renal replacement modalities, including patients cared for with CKM, to ensure appropriate strategies are in place for the monitoring and management of pain for all patients in need.
Several limitations of the studies included in this review were identified. If these limitations are not addressed in future studies, this will introduce bias, limit our ability to interpret the data, and ultimately compromise our ability to improve pain management. First, studies lacked a consistent approach to determining or reporting the chronicity of pain. Dialysis patients also experience recurring episodes of acute pain such as intra-dialytic headaches and cramps. This acute pain is often associated with tissue damage but typically has no progressive pattern, lasts a predictable period, subsides as healing occurs, and is episodic with periods of no pain. In contrast, chronic pain is more likely to result in functional impairment and disability, psychological distress (eg, anxiety or depression), sleep deprivation, disruption of activities of daily living, and poor HRQL as it is present for long periods of time and is often out of proportion with the extent of pain from the originating injury. Chronic pain is most commonly defined as any painful condition that persists for greater than 3 months. 94 Studies that report pain should make a clear and consistent distinction between these 2 different pain syndromes. Given the variability in the reporting and defining of chronic pain in these studies, patients with acute pain may also have been included, falsely elevating the prevalence rates of true chronic pain.
There was also variability in the measures used to determine pain severity that differed in range and format (including numerical, visual, or verbal scales). Hence, a recalibration of different scales was required to compare different studies which may have introduced bias in the results of the meta-analysis. While each of these approaches has evidence for validity, they may be interpreted differently by patients, limiting the ability to compare findings across populations. There are data around what constitutes clinically significant pain and what constitutes clinically important differences in pain relief based on 0 to 10 scales and the consensus recommendation from the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) to use a 0 to 10 NRS in pain studies. 95
Substantial variability in the reported prevalence of pain was present in nearly all of the pooled groups, yielding very high heterogeneity measurements. As such the estimates should be interpreted with caution and may not reflect the true prevalence of pain. However, stratification by country and pain definition in some cases decreased the I2 substantially, which suggests that at least some of this variability may be explained by regional practices and differences in what constitutes chronic pain.
Another limitation was that these studies reported mean severity scores for the entire cohort. The reporting of average severity scores is problematic as the distribution of pain tends to be “U-shaped” rather than bell shaped. This highly skewed distribution has the maximum frequencies at the 2 extremes of the range of variables, ie, patients with no pain and patients with severe pain, or patients having good pain relief or poor pain relief. If few patients are “average,” the use of average values is misleading. To better understand patterns of pain, it is important to determine the prevalence of clinically significant pain (such as moderate and severe pain) and for those with pain to report its severity. Finally, we did not reach out to primary authors for additional information. Recommendations for future studies that explore pain prevalence and severity are outlined in Table 6 and are in keeping with international recommendations for the reporting of pain in clinical studies.
Recommendations for Assessing and Reporting the Presence and Severity of Chronic Pain.
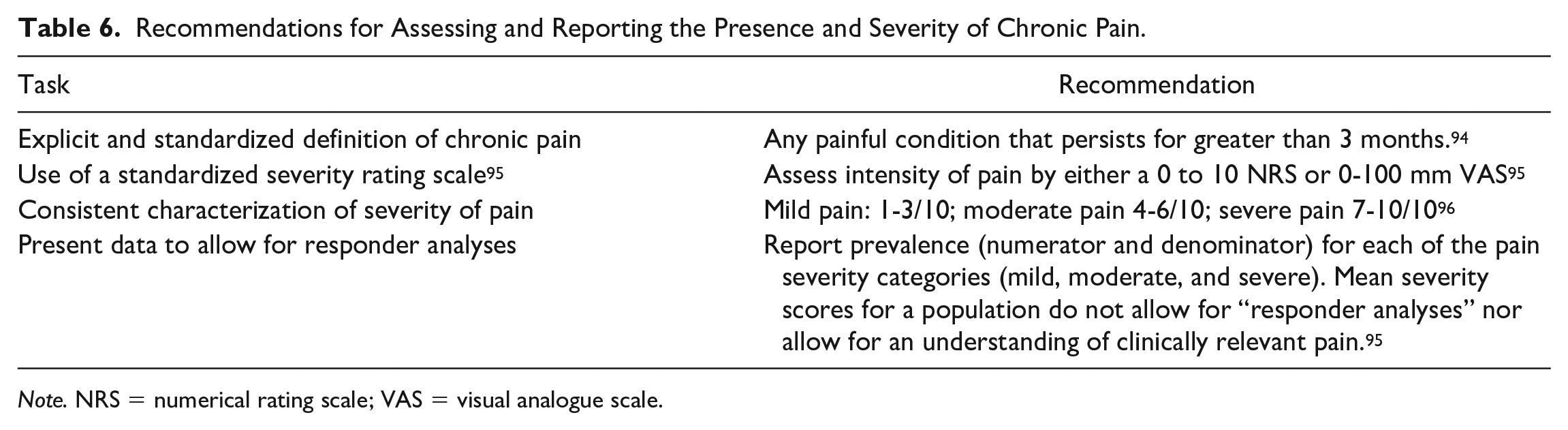
Note. NRS = numerical rating scale; VAS = visual analogue scale.
Conclusions
This review illustrates that chronic pain is extremely common and often severe across diverse CKD populations. Routine symptom assessment, therefore, should extend to patients across GFR categories of CKD. Current data provide a strong imperative to establish pain management as educational, clinical, and research priorities in nephrology. Future research needs to move beyond describing the problem to evaluating the efficacy of pain management strategies. Establishing consistent standards for measuring presence, chronicity, type, and severity of pain is needed to appropriately conduct and interpret clinical evaluation and clinical trials and determine the impact of pain management strategies on patients’ lives.
Supplemental Material
sj-docx-1-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-docx-2-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-1-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-1-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-2-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-2-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-3-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-3-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-jpg-4-cjk-10.1177_2054358121993995 – Supplemental material for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-jpg-4-cjk-10.1177_2054358121993995 for The Prevalence and Severity of Chronic Pain in Patients With Chronic Kidney Disease: A Systematic Review and Meta-Analysis by Sara N. Davison, Sarah Rathwell, Sunita Ghosh, Chelsy George, Ted Pfister and Liz Dennett in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
Not applicable
Consent for Publication
Not applicable
Availability of Data and Materials
Not applicable
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
