Abstract
Background:
Nephrectomy is the mainstay of treatment for many kidney cancers, but has been correlated with increased incidence of acute kidney injury (AKI) and chronic kidney disease (CKD). Recently, sodium-glucose cotransporter-2 (SGLT2) inhibition has been shown to decrease the incidence of end-stage kidney disease and death in people with type 2 diabetes mellitus (T2D). However, at present, there has been no description of the use of SGLT2 inhibition in patients with T2D and solitary kidney despite the high risk of CKD progression.
Objective:
To characterize the use of SGLT2 inhibition and kidney function in a series of patients with T2D with prior nephrectomy for renal cell carcinoma (RCC).
Design:
Retrospective case series.
Setting:
University hospital outpatient onco-nephrology clinic.
Patients:
Patients post-nephrectomy for RCC with T2D who were prescribed an SGLT2 inhibitor.
Measurements:
Serum creatinine, albumin to creatinine ratio (ACR), HgA1c, and blood pressure measurements.
Methods:
Patients post-nephrectomy with incident use of SGLT2 inhibitor were identified from an existing registry of patients followed in the Onco-Nephrology Clinic at our institution from May 2019 to March 2021. Demographics, medication use, time since nephrectomy, cancer diagnosis, serum creatinine, ACR measurements, and blood pressure measurements were extracted from electronic medical records.
Results:
Five patients were identified who had initiated SGLT2 inhibition post-nephrectomy. All patients were male, had T2D, and a prior history of hypertension. Renal cell carcinoma was the clinical indication for nephrectomy in all patients. None of patients were prescribed diuretics, and all were receiving renin-angiotensin system (RAS) inhibition therapies. The time from nephrectomy to SGLT2 inhibitor initiation ranged from 5 to 74 months. Baseline mean estimated glomerular filtration rate (eGFR) values were 49 mL/min/1.73 m2 (95% confidence interval [CI]: 31.5-66.5), and mean ACRs were 8.7 mg/mmol (95% CI: 0.6-16.9). After 6 months of SGLT2 inhibition, the mean eGFR and ACR values were 58 mL/min/1.73 m2 (95% CI: 29.7-86.2) and 23.8 mg/mmol (95% CI: 0-60), respectively. After 16 to 18 months of follow-up (4 patients), the mean eGFR was 56 mL/min/1.73 m2 (95% CI: 37.3-74.7), and mean ACR was 10.5 (95% CI: 0-30.5), similar to baseline values before SGTL2i therapy initiation. At baseline, mean systolic blood pressure was 128 mm Hg (95% CI: 118.3-140.9) and remained similar after 12 months of treatment (mean 131 mm Hg [95% CI: 112.3-149.7]). There were no adverse events related to AKI, electrolyte disturbances, ketoacidosis, or genitourinary infections during the 18-month follow-up period.
Limitations:
Small sample size, lack of a comparison group, and the variable timing of clinical data collection, including eGFR levels following initiation of SGLT2 inhibition.
Conclusions:
SGLT2 inhibition is becoming a standard component of nephrology care to reduce kidney function decline, cardiovascular risk, and mortality. To our knowledge, our report is the first to provide longitudinal data on SGLT2 inhibitor usage in patients with T2D and solitary kidneys post-nephrectomy. Larger prospective studies are needed to determine the efficacy and safety of SGLT2 inhibition strategies for kidney protection in patients post-nephrectomy.
Introduction
Nephrectomy is a mainstay of treatment for many kidney cancers, but is associated with a higher risk of acute kidney injury and chronic kidney disease (CKD) progression. Individuals with renal cell carcinoma (RCC) have a significant risk of CKD progression after partial or radical nephrectomy. 1 Despite this high risk, the optimal medical approach to safely prevent CKD progression is unclear.
Historically, the pharmacologic management of CKD post-nephrectomy is based on attenuation of hyperfiltration and proteinuria using renin-angiotensin system (RAS) inhibition strategies. 2 Unfortunately, there are limited data specific to the population post-nephrectomy, despite elevated risks of CKD progression and predisposition to develop hyperfiltration. More recently, sodium-glucose cotransporter-2 (SGLT2) inhibition has been shown to decrease the risk of end-stage kidney disease, cardiovascular disease, and mortality in people with CKD, including those with type 2 diabetes mellitus (T2D).3,4 However, it is not yet known whether these benefits of SGLT2 inhibition extend to individuals with T2D post-nephrectomy, where reducing glomerular hypertension has the potential to translate into declines in proteinuria and CKD progression. Accordingly, in this report, we characterize the use of SGLT2 inhibition in a series of patients with T2D who previously underwent nephrectomy as a treatment for RCC. We describe the trends in kidney function, albuminuria and report on general safety and tolerability.
Methods
Patients post-nephrectomy who were initiated on an SGLT2 inhibitor were identified from an existing registry of patients followed in the University Health Network Onco-Nephrology Clinic from May 2019 to March 2021. Start dates for SGLT2 inhibitor therapies were determined from the outpatient medical records. One patient had 6 months of follow-up as the start date of SGLT2 inhibitor occurred in October 2020. Clinical data such as demographics, medication use, time since nephrectomy, and cancer diagnosis are described in Figure 1A. These details together with serum creatinine and urine albumin to creatinine (ACR) ratio measurements were extracted from electronic medical records. Time 0 was considered 1 to 4 weeks before SGLT2 inhibitor therapy was initiated and included the pre-ambulatory clinic serum and urine biochemistries. The study was approved by the Research Ethics Board at the University Health Network.
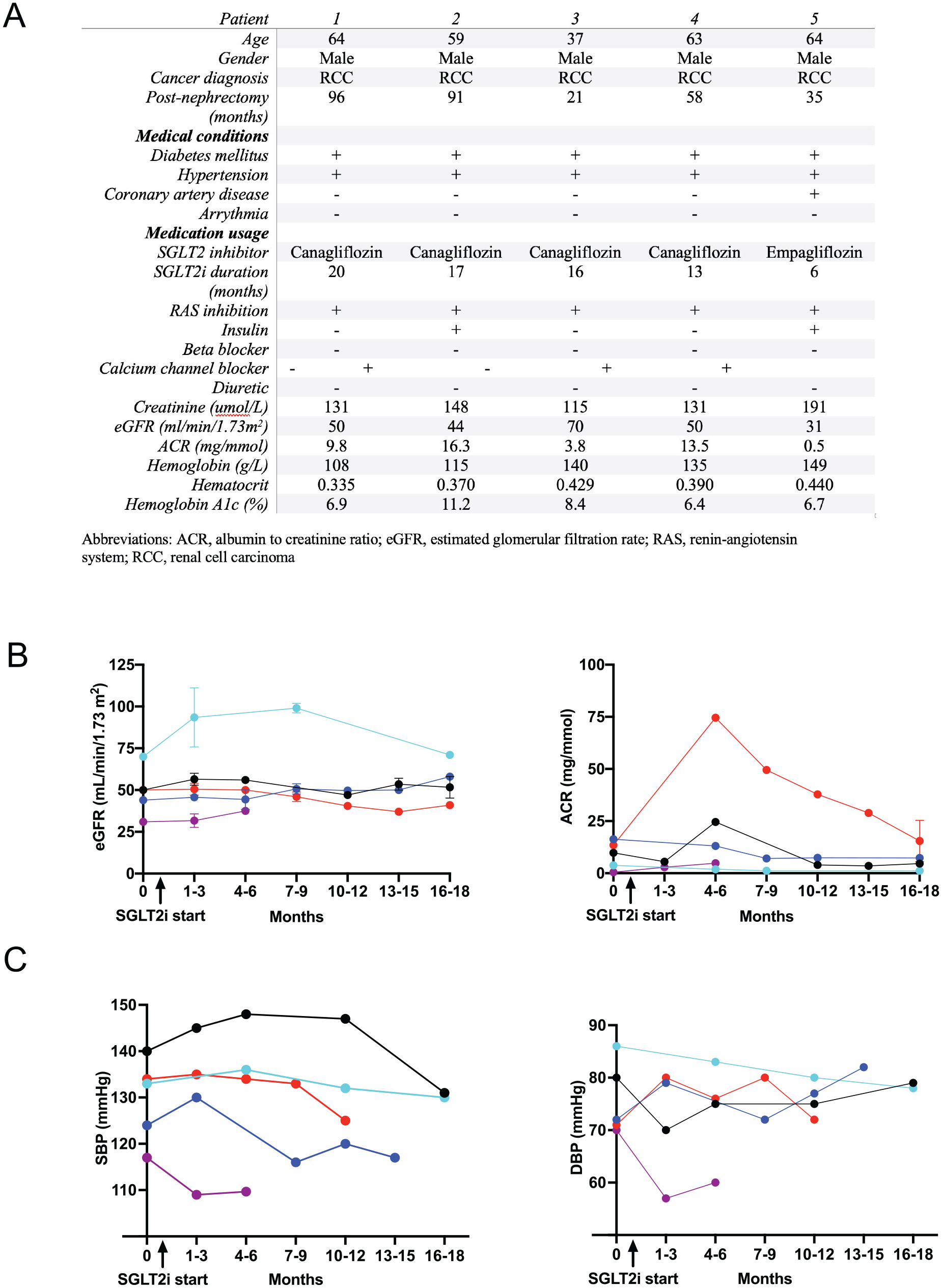
(A) Baseline characteristics of patients pre-SGLT2 inhibitor treatment. (B, C) eGFR, albumin to creatinine ratios and systolic (SBP) and diastolic blood pressures (DBP) of post-nephrectomy patients at baseline (0) and after initiation of SGLT2 inhibitors. Error bars represent standard deviation.
Results
After retrospective review, 5 patients were identified who had initiated SGLT2 inhibition post-nephrectomy. All patients were male, had T2D and a prior history of hypertension (Figure 1A). Renal cell carcinoma was the clinical indication for nephrectomy in all patients. None of patients were prescribed diuretics, and all were receiving RAS inhibition therapies. One of the 5 patients reviewed was receiving insulin therapy in addition to oral hypoglycemic agents. The time from nephrectomy to SGLT2 inhibitor initiation ranged from 5 to 74 months. Baseline mean eGFR values were 49 mL/min/1.73 m2 (95% confidence interval [CI]: 31.5-66.5), and mean urine ACR was 8.7 mg/mmol (95% CI: 0.6-16.9) (Figure 1B). After 6 months of SGLT2 inhibitor treatment, the mean eGFR and ACR values were 58 mL/min/1.73 m2 (95% CI: 29.7-86.2) and 23.8 mg/mmol (95% CI: 0-60), respectively (Figure 1). After 16 to 18 months of follow-up (4 patients), the mean eGFR was 56 mL/min/1.7 3m2(95% CI: 37.3-74.7), and ACR was 10.5 mg/mmol (95% CI: 0-30.5), similar to baseline values before SGLT2 inhibition.
At baseline, mean systolic blood pressure was 129.6 mm Hg (95% CI: 118.3-140.9) and remained similar after 12 months of treatment (mean 131.0 mm Hg [95% CI: 112.3-149.7]) (Figure 1C). There were no adverse events documented in the medical records related to acute kidney injury, electrolyte disturbances, ketoacidosis, or genitourinary infections during the 18-month follow-up period.
Discussion
SGLT2 inhibition is becoming a standard component of nephrology care to reduce kidney function decline, cardiovascular risk, and mortality. Kidney outcome trials with canagliflozin and dapagliflozin have not explicitly excluded patients with a solitary kidney or previous nephrectomy. 3 However, the 5 patients in this series would have been excluded because of their history of malignancy. To our knowledge, our report is the first to provide longitudinal data on SGLT2 inhibitor usage in patients with T2D post-nephrectomy.
Glomerular hyperfiltration is a characteristic pathway leading to kidney injury in the setting of T2D. 5 Mechanistic studies in people with type 1 diabetes have demonstrated an attenuation of hyperfiltration with SGLT2 inhibition—an effect that mirrors observations in experimental models of early type 1 diabetes. 6 Similarly, we postulate that SGLT2 inhibition may yield analogous changes in glomerular hemodynamics in patients with T2D post-nephrectomy. Importantly, while we did not observe any specific issues related to safety, we also recognize that this analysis was limited by the small sample size, the lack of a comparison group, and the lack of uniform collection of clinical data, including eGFR levels soon after initiation of SGLT2 inhibition.
Conclusions
Larger prospective studies are needed to determine the efficacy and safety of SGLT2 inhibition strategies for kidney protection in patients post-nephrectomy.
Footnotes
Ethics Approval and Consent to Participate
This study was approved by the Research ethics board of the University Health Network.
Consent for Publication
All authors read and approved the final version of this manuscript.
Availability of Data and Materials
The data in this study cannot be shared due to restrictions on confidential patient-level data. Questions about the data can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: David Z.I. Cherney has received consulting fees or speaking honorarium, or both, from Bristol Myers Squibb, Novo Nordisk, Mitsubishis-Tanabe, MAZE, Janssen, Bayer, Boehringer Ingelheim-Eli Lilly, AstraZeneca, Merck & Co., Inc., Prometic, and Sanofi, and has received operating funds from Janssen, Boehringer Ingelheim-Eli Lilly, Sanofi, AstraZeneca, Novo Nordisk, and Merck & Co., Inc. Marko Škrtić is supported by un-restricted support from Amgen for Onco-Nephrology fellowship.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
