Abstract
Background and Objectives:
There is a renewed interest in the successful use of aminoglycosides due to increasing resistance in gram-negative infections. Few studies to date have examined the pharmacokinetics (PK) of intradialytic infusions of tobramycin. This study sought to characterize the pharmacokinetic profile of intradialytically administered tobramycin in infected patients receiving chronic intermittent hemodialysis and to determine whether it is possible to achieve favorable PK targets.
Design, Setting, Participants, and Measurements:
In this prospective pharmacokinetic study, a single dose (5 mg/kg) of tobramycin was administered intradialytically to 11 noncritically ill patients undergoing chronic intermittent hemodialysis. Blood samples were collected at selected time to determine tobramycin serum concentrations. The PK analysis was performed using Phoenix™ NLME. The efficacy exposure outcome for nonsevere gram-negative infections sensitive to tobramycin with a minimum inhibitory concentration ≤1 were maximum concentration (Cmax ≥ 10 mg/L) and area under the curve (AUC24 h > 30 mg⋅h/L). For toxicity, the goal was to identify plasma trough concentrations <2 mg/L.
Results:
Tobramycin disposition was best described by a one-compartment model using a total clearance composed of the systemic clearance and a transitory hemodialysis clearance. Tobramycin mean (SD) Cmax, trough levels, and AUC24h were 13.1 (1.3) mg/L, 1.32 (0.47) mg/L, and 61 (23) mg⋅h/L, respectively. Monte Carlo simulation run with 1000 virtual patients showed that a 5 mg/kg dose of tobramycin administered intradialytically can outperformed the usual low-dose postdialysis dosing (80% meeting all targets versus <1%, respectively).
Conclusions:
A single high dose of tobramycin can achieve favorable PK outcome when administered using intradialytic infusions in hemodialysis patients. This practical dosing regimen may represent an effective and safer alternative to the usual dosing in the treatment of nonsevere gram-negative infections.
Introduction
Infections are a significant risk of mortality and morbidity in patients with kidney failure, representing 20% of deaths in these patients.1,2 Patients undergoing hemodialysis sessions are particularly susceptible to infections due to their impaired immune system and the use of intravascular catheters, providing a direct access to bacterial pathogen into the bloodstream. 2
The increasing prevalence of bacterial resistance, while generating a growing concern over antimicrobial use, has led clinicians to seek for a wider range of therapeutic alternatives. These considerations are particularly important for vulnerable populations, such as kidney failure patients, due to their greater consumption of antibiotics. 3 Given the increasing rates of bacterial resistance, many clinicians are considering aminoglycosides as an additional treatment alternative to treat those high-risk patients. 4
Aminoglycosides have a narrow therapeutic window and are eliminated by the kidney. 5 Controversies remain regarding the optimal dosing regimen in chronically hemodialyzed patients. 4 In current clinical practice, aminoglycosides are usually administered at a low dose at the end of the hemodialysis session. For moderate to severe infections, a loading dose of 2 to 3 mg/kg is administered followed by subsequent doses of 1 to 1.5 mg/kg every 48 to 72 hours at the end of the hemodialysis session. 6 However, according to our experience and other studies, such regimen leads to suboptimal aminoglycosides pharmacokinetic with low peak and high trough serum concentration.7,8 Recently, 2 pharmacokinetic studies performed in critically ill patients showed that administration of gentamicin (6 mg/kg) 30 minutes before the hemodialysis session could allow for adequate exposure targets.9,10 However, administration of aminoglycosides 30 minutes before hemodialysis is not convenient for most outpatient clinic users and can be complicated. A similar high-dose regimen administered at the beginning of the hemodialysis could be more convenient. Whether this high-dose regimen allows for optimal pharmacokinetic targets remains, nonetheless, uncertain.
The goals of this study were to characterize the pharmacokinetics (PK) of an intradialytic infusion of tobramycin at a 5 mg/kg dose early in the hemodialysis session and to determine whether it is possible to achieve favorable pharmacokinetic exposure targets with such dosing regimen.
Materials and Methods
Study Design
A prospective open-label pharmacokinetic study was conducted at the hemodialysis clinic of Hôpital Maisonneuve-Rosemont. Recruitment occurred from February through August 2019.
Study Population
Eligible patients (inpatients or outpatients) were ≥18 years old with kidney failure, had been undergoing 3- to 4-hour hemodialysis sessions 3 times a week for at least 1 month prior to study inclusion and had a suspected or confirmed gram-negative infection treated with an antibiotic (eg, ciprofloxacin, piperacillin/tazobactam, amoxicillin clavulanate). Patients were excluded if they had any contraindications to receiving aminoglycoside agents, had prior tobramycin administration in the last month, fluctuating kidney function, were kidney transplant recipient, had vulnerability to aminoglycosides adverse events (AEs; eg, history of myasthenia gravis, Parkinson disease, documented vestibular or auditory impairment), had altered volume of distribution (V), severe burn injury (>20%), significant ascites, acute heart failure, cystic fibrosis, acute cardiovascular failure, admission to the critical care unit, or morbid obesity. Hemodynamically stable subjects were selected to ensure that the 4-hour hemodialysis session would be completed. No intensive care unit (ICU) patients were included in the study. Patients did not participate in another study. Pregnancy or actively breastfeeding were also exclusion factors.
Prohibited Drugs
Neuromuscular blocker agents (eg, succinylcholine) were prohibited throughout the study due the reported risk of neuromuscular block or respiratory paralysis following administration of an aminoglycoside. 5 Subjects regularly taking a loop diuretic were instructed to skip the morning or evening dose, if it was less than 4 hours from tobramycin administration as concomitant administration of loop diuretics may potentiate the ototoxicity of aminoglycosides.11,12
Study Procedures
Hemodialyzed patients treated with an antibiotic covering gram-negative rod-type bacteria were screened using local pharmacy distribution tool. Then, preliminary admissibility was assessed to ensure compliance with the inclusion criteria and the absence of exclusion criteria. Potentially eligible participants were met and the study was explained to them. Interested participants who gave their consent to participate to the study were given one dose of tobramycin of 5 mg/kg at their next hemodialysis session in addition to their current antibiotic.
The dose was calculated with their dosing weight based on dry weight. 13 An adjusted body weight was used when the body mass index exceeded 30 kg/m2 (see Supplementary Appendix). Marketed tobramycin sulfate (Sandoz Canada Inc., Boucherville, QC, Canada) was diluted with sodium chloride 0.9% for a total volume of 27 mL and was then infused through the hemodialysis venous line within the first 30 minutes of the hemodialysis session using a syringe driver (ESP60) at a constant rate.
The chosen tobramycin dose was based on a preliminary simulation using the Phoenix Software (build 8.003176, Ridgewood, NJ). Cmax and Ctrough were simulated using the model published by Veinstein et al. 9 The model consists of a zero-order input one-compartment model with 2 clearances (systemic and hemodialysis) and a proportional-error model. Different doses were simulated at various administration times following initiation of hemodialysis (data not shown). The dose of 5 mg/kg at the beginning of hemodialysis was selected considering that mean Cmax and C24h were similar to those obtained by Veinstein et al.
Pharmacokinetic Sampling and Tobramycin Assay
Intradialytic blood samples were obtained from the arterial line according to a predefined schedule: predrug infusion (control), 30 minutes post end of infusion, at a variable time (90, 120, 150, or 180 minutes), after the end of infusion (intradialytic sampling), at the end of hemodialysis session, and just before the next hemodialysis session (interdialytic—trough level). An optional interdialytic blood sample was also obtained at a convenient time for the patient between hemodialysis sessions using venipuncture.
Samples were collected in a 3.5 mL capacity Vacuum SST tube with gel BD Vacutainer (yellow cap) and sent to the biochemistry laboratory in less than 30 minutes. Serum tobramycin concentration was measured using a particle-enhanced turbidimetric immunoassay (PETINIA) with the Architect c16000 clinical chemistry analyzer. The lower limit of detection was 0.2 mg/L.
Data Collection
Subject characteristics, medical and medication history, information on active infections (including minimal inhibitory concentration [MIC]) and health care trajectory (inpatient or outpatient) were collected from the patient’s medical record. Other laboratory data included urea and creatinine serum concentrations before dialysis and a 24-hour urine collection for subjects with residual diuresis. Hemodialysis parameters such as dialyzer, vascular access, ultrafiltrate, dialysate flow rate, and blood flow rate were collected at the end of the hemodialysis session during which tobramycin infusion was given. When available, Kt/V (K: dialyzer clearance of urea; t: duration of hemodialysis; V: volume of distribution of urea in the body) estimated by conductivity pulse during hemodialysis (Diascan feature, Gambro Lundia AB, Lund) was collected from the same hemodialysis session as the tobramycin infusion. When unavailable, Kt/V data were collected from another hemodialysis session within a 1-week time frame. Care was taken to confirm that the dialysis session parameters such as the duration and the blood flow rate were comparable.
Pharmacokinetic Analysis
The Phoenix Software (build 8.003176) FOCE-ELS algorithm was used to determine structural and error-model parameters by fitting the simulation model to tobramycin concentrations collected from the first 10 participants. This sample size was estimated using the standard deviation of the pharmacokinetic parameter with the greatest variability in the study of Veinstein et al. 9 For an alpha error of .05 and a power of 80%, a sample size 9 to 12 subjects was found adequate to account for the variability and to obtain 40 to 60 data points for the training set. For the final model, age, sex, height, weight, creatinine clearance, 24-hour urea collection, 24-hour creatinine collection, serum urea, serum creatinine, vascular access type, dialyzer, ultrafiltrate volume and flow, Kt/V, and blood flow rate were analyzed as potential covariates. Covariate assessment was performed through a stepwise covariate search (forward inclusion P < .05 and backward exclusion P < .01). The model was used to obtain individual pharmacokinetic parameters (Cmax, transitory hemodialysis clearance [CLHD], systemic clearance [CLs], volume of distribution [V], C48h, C72h, AUC24h). Monte Carlo simulation (1000 replicates) was used to generate visual predictive check plots. The first 10 subjects were included in the pharmacokinetic simulation model. The last subject (#11) could not be used for this purpose because of a vascular access dysfunction (see results).
Pharmacokinetics and Pharmacodynamics Outcomes: Targets and Sensitivity Analysis
Minimal inhibitory concentration was unavailable for most of the patients and could not be used for the purpose of comparing PK and pharmacodynamics targets among patients. Therefore, a sensitivity analysis was conducted using Monte Carlo simulations to estimate the Cmax/MIC and AUC24h/MIC ratios with a variable MIC of 1, 2, and 4 mg/L. Targets were defined as Cmax/MIC ≥ 10, AUC24h/MIC ≥ 30, and C48h or C72h ≤ 2 mg/L.14-16 These simulations were used to define the proportion of reached targets, evaluated individually and altogether.
Safety Assessment
A specific strategy was also elaborated in the protocol to mitigate nephrotoxicity and ototoxicity. Safety was monitored through reporting of AEs during and after the first hemodialysis session by questioning the subject (see Supplementary Appendix). The subjects were asked whether there were changes in their health status. If so, any AE or serious AE occurring from the signing of the consent form up to 48 or 72 hours after the last day of blood draw (or until the participant withdraws their consent) was documented using the Common Terminology Criteria for Adverse Events (CTCAE) v.4.0 (see Supplemental Appendix).
Results
Participant Characteristics
Ninety-eight patients were screened and 11 patients out of the 12 targeted for the study were enrolled and completed the study. Baseline characteristics of subjects and hemodialysis parameters are shown in Table 1. Two high flux dialyzers were used: polysulfone (Optiflux F250 NR; Fresenius Medical Care AG & Co, Bad Homburg) and polyphenylene (Phylther HF 22SD; Medtronic CryoCath LP, Pointe-Claire, QC, Canada). All hemodialysis machines used were Gambro Artis (Gambro Inc., Deerfield, IL). The most common type of hemodialysis access was the central venous catheter. Two subjects had an arteriovenous fistula. Blood flow rates ranged from 300 to 380 mL/min and dialysate flow rates were constant for all participants at 500 mL/min. The Kt/V was unavailable for 3 patients and the Kt/V for the previous hemodialysis session was used (the dialysis session parameters were comparable).
Subject Characteristics and Hemodialysis Parameters.
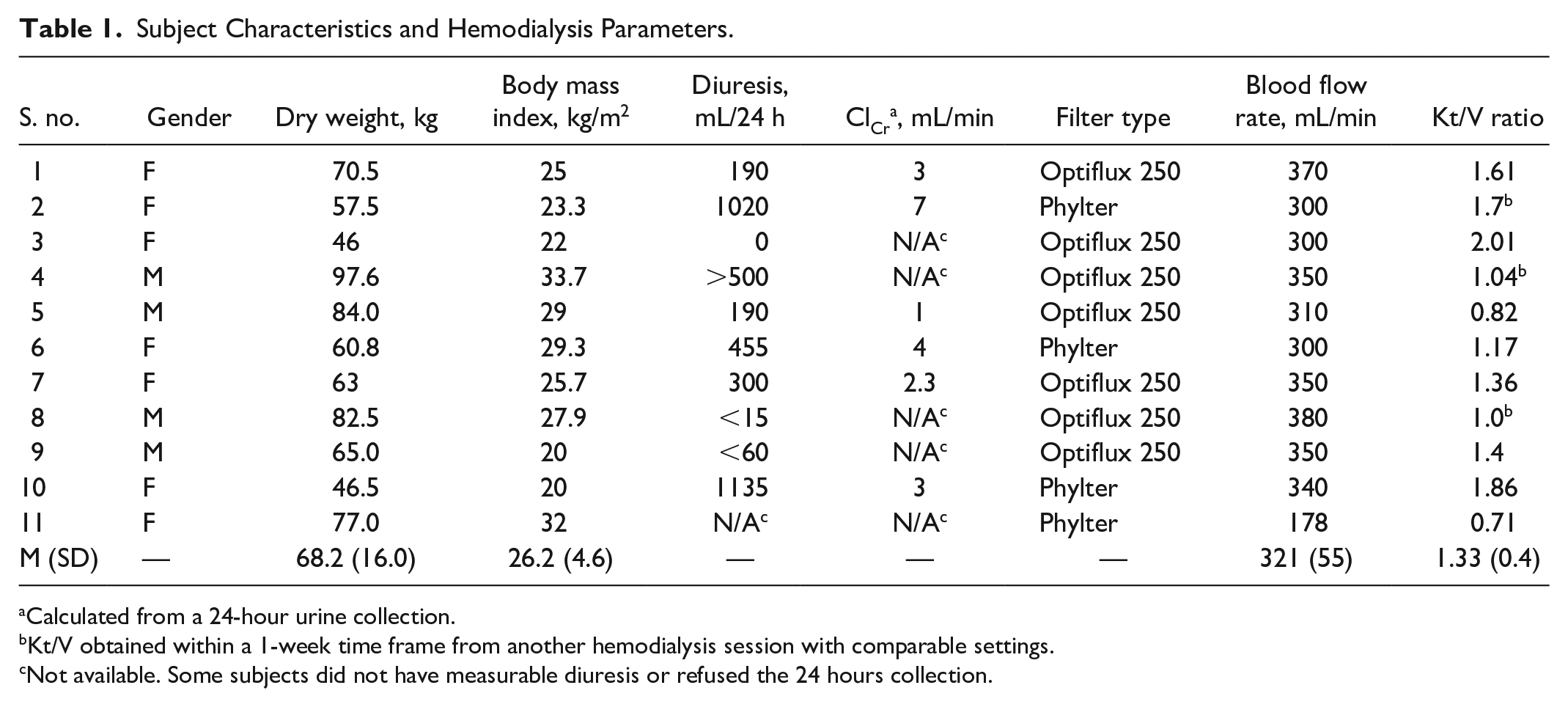
Calculated from a 24-hour urine collection.
Kt/V obtained within a 1-week time frame from another hemodialysis session with comparable settings.
Not available. Some subjects did not have measurable diuresis or refused the 24 hours collection.
Measured Tobramycin Concentrations
Measured serum tobramycin levels can be found in the Table A1 (see Supplementary Appendix). A total of 43 data points obtained from 10 subjects were available to build the PK model.
Population Pharmacokinetic (for More Details in Supplementary Appendix)
The final structural model was a zero-order, one-compartment model using a total clearance composed of the CLs and CLHD. It was used to examine the effects of different subjects and dialysis covariates on the estimated PK model parameters. A multiplicative error model was selected due to superior goodness-of-fit plots. The goodness-of-fit plots of the final population PK model are shown in Figure 1. The data suggest an adequate prediction and the suitability of the error model for the study population. Only 2 variables were found to have a statistically significant impact on pharmacokinetic parameters: height (for V) and Kt/V (for CLHD). The final population pharmacokinetic model parameters were estimated with acceptable precision (see Table 2). Individual pharmacokinetic parameters are shown in Table 3.

Goodness-of-fit plots for the population pharmacokinetic model: (A) Observed versus individual predicted tobramycin concentrations, (B) observed versus population predicted tobramycin concentrations, (C) Conditional Weighted Residuals (CWRES) versus population predicted tobramycin concentrations, (D) CWRES versus time after dose, and (E) individual weighted residuals versus individual predicted tobramycin concentrations.
Population Pharmacokinetic Parameters.

Note. Vd = Volume of distribution; ClS = systemic clearance or interdialytic; ClHD = hemodialysis clearance or intradialytic; SE = standard error.
Individual Pharmacokinetic Parameters After Receiving 5 mg/kg of Tobramycin Infused in 30 Minutes at the Beginning of the Hemodialysis Session.

Note. AUC = area under the curve; CLHD = hemodialysis clearance; CLs = systemic clearance or interdialytic.
This subject had interruptions during the hemodialysis session, but still completed a full cumulative 4-hour session.
Comparison of Chosen Dosing Regimen and Traditional Dosing Regimen
Proportions of Monte Carlo–simulated concentrations meeting efficacy and security targets can be found in Table 4. The chosen dosing regimen of tobramycin 5 mg/kg at the beginning of hemodialysis yielded mean Cmax concentrations above 10 mg/L in Monte Carlo simulations, whereas the mean Cmax obtained with the standard dosing regimen of 2 mg/kg infused at the end of hemodialysis was of 4.8 mg/L (see Table 4). With the standard dosing, less than 1% of simulation reach security target of Ctrough < 2.0 mg/L compared with 93% of the simulation with the high dose at the beginning of hemodialysis. The target AUC24h > 30 mg⋅h/L is reached with the standard regimen in 100% of simulations for bacteria with MIC of 1 or 2 mg/L. With the high-dose regimen, AUC24h > 30 mg⋅h/L is reached in 96% of simulations for bacteria with an MIC of 1 mg/L and in 42% for bacteria with an MIC of 2 mg/L. None of the Monte Carlo simulations with the 2 mg/kg dose at the end of the dialysis reached the combination of all targets (Cmax/MIC > 10, AUC24h/MIC > 30 and trough level < 2 mg/L), compared with 80% of the simulations with 5 mg/kg at the beginning of the hemodialysis session for bacteria with a MIC ≤ 1 mg/L. Simulations using our patient population suggest that little accumulation of tobramycin is expected following a 5 mg/kg dose of tobramycin 3 times a week (see Figure A4 in Supplementary Appendix).
Proportion of Monte Carlo (n = 1000) Simulations Meeting Pharmacokinetic Target.

Note. CI = confidence interval; HD = hemodialysis; AUC = area under the curve; MIC = minimal inhibitory concentration.
Ctrough measured before the next dialysis (48 or 72 h). Data were pooled to simplify analysis.
Safety Assessment
No subject withdrew from the trial due to AEs. Two subjects complained of fatigue and weakness. Hypotension, weakness, and dysarthria were reported for one subject and another subject had hypoglycemic episodes during the hemodialysis session up until 24 hours post study drug administration. These events were deemed unlikely to be related to the investigated drug and they disappeared before the next dialysis.
Discussion
To our knowledge, this is the first study evaluating high-dose tobramycin administered to infected patients with kidney failure directly at the beginning of their hemodialysis session. A 5 mg/kg dose was selected from preliminary pharmacokinetic simulations to provide a Cmax/MIC ratio ≥ 10 for MIC values ≤ 1 mg/L.14,15 As expected, a 3- to 4-hour hemodialysis session provided a sufficient replacement for normal kidney function in terms of tobramycin clearance and a 5 mg/kg dose was adequate to obtain favorable tobramycin exposure.
Treatment of nonsevere gram-negative infection with low bacterial burden is optimal clinically when Cmax/MIC ratio ≥ 10 and when an AUC24h/MIC ratio range between 30 and 50.14-16 When a 5 mg/kg dose of tobramycin was administered to infected patients, calculated Cmax and AUC24h met all targets with an average ±SD value being, respectively, 13.1 ± 1.31 mg/L and 61 ± 23 mg⋅h/L. In contrast, a retrospective chart review of 107 patients treated with an aminoglycoside dose ranging from 1 to 2 mg/kg after the dialysis found only 2.9% of treatments were able to reach dose-estimated Cmax greater than 8 mg/L. 8 These results mirror those of Teigen and Mohamed where these target levels could not be reached with a 1 to 2 mg/kg aminoglycoside dose given in a similar setting.7,17 Although Veinstein et al 9 published higher Cmax and AUC24h values when gentamicin was administered prior to the hemodialysis session at 6 mg/kg dose to critically ill subjects with acute kidney injury, the larger exposure of aminoglycoside required for high-risk population such as ICU patients is not necessary to treat low-bacterial-burden infections in an outpatient population. 16
Nephrotoxicity and ototoxicity are observed with aminoglycosides and usually feared by clinicians. Although nephrotoxicity is less of a concern in kidney failure patients receiving chronic hemodialysis, ototoxicity may bring an additional burden to these patients. This limits their utilization. Such AEs occur more frequently when trough levels exceed 2 mg/L. 5 In a retrospective chart review in patients treated with an aminoglycoside dose of 1 to 2 mg/kg after the hemodialysis, the trough levels were measured in 55 treatments. The average (range) concentration was 2.78 (0.5-6.2) mg/L. 8 This suggests that even at such low dose, it is difficult to obtain trough levels lower than 2 mg/L. When a 5 mg/kg dose of tobramycin was administered to infected patients, measured or calculated trough concentrations (48 or 72 h) were all below 2 mg/L when the hemodialysis session was completed without interruption (see Supplemental Appendix). One subject (1/11) had vascular access issues that led to a number of interruptions of his hemodialysis session which resulted in tobramycin levels greater than 2 mg/L (2.3 mg/L after 48 h). Simulations with our PK model confirmed that 93% of the simulated C48h and C72h would reach adequate trough levels if a 5 mg/kg tobramycin dose was administered at the beginning of full session of hemodialysis (see Table 4). A high-dose regimen at the beginning of hemodialysis may be a safer alternative to traditional low-dose end-of-dialysis dosing.
When compared with other PK studies with aminoglycosides,10,17,18 the V and the CLs (interdialytic) were comparable. As expected, the CLs of tobramycin were much lower than the CLs of tobramycin in individuals with kidney function. On the contrary, hemodialysis appears highly effective in removing tobramycin. The estimated CLHD was higher than those reported earlier7,17,19,20 and was strongly influenced by the Kt/V, a marker for hemodialysis efficiency.21-23 The removal of tobramycin by hemodialysis is variable, depending on patient characteristics, dialyzers used, length of dialysis sessions, and hemodialysis operating system. The specific pharmacokinetic behavior found with tobramycin in this study highlights the importance of adjusting the antibiotic dosing regimen with a specific population or with a specific hemodialysis setup.
An important strength of our study was the use PK modeling and simulation to select the right dose and the right minimal blood sampling strategy to create a structurally sound model using our own data set. The model developed provided adequate characterization of the pharmacokinetic parameters without requiring a large amount of subjects and allowed us to perform simulations. Using Monte Carlo simulations, we showed that all safety and efficacy PK targets for the treatment of mild to moderate infection could be reached for more than 80% of the patients with that new regimen compared with less than 1% of population with the traditional low-dose end-of-hemodialysis approach (see Table 4).
This study has some limitations. First, no clinical outcomes were evaluated as the aim of the study was purely pharmacokinetic. Indeed, although simulations using our patient population suggest little accumulation of tobramycin using a 3 times a week regimen, a single dose of tobramycin is not sufficient to provide insights on a full course of treatment. Second, the optimal exposure levels have not been determined for patients with kidney failure. It is unknown if the targets we have selected from the non-kidney failure patients can effectively be used in this specific population. Third, one has to be careful when using this dosing regimen and intervals because it assumes that the subject will be able to complete a full cumulative 3- to 4-hour session with comparable hemodialysis settings. If the hemodialysis session has to be prematurely stopped, an additional hemodialysis session might have to be performed to avoid sustained exposure to high tobramycin concentrations. Finally, the predictive performance of our model has yet to be confirmed as only an internal validation using bootstrap has been used so far. Upon external validation, this model may be used in our population to perform simulations and therapeutic drug monitoring.
Conclusions
In summary, this study provides strong PK support for dosing of aminoglycosides in patients in the first 30 minutes of their hemodialysis session. Compared with traditional low-dose end-of-hemodialysis dosing, the combined higher Cmax and lower trough levels found with a high dose administered at the beginning of the dialysis may help achieving the right efficacy and toxicity targets to treat low-bacterial-burden gram-negative infection. Additional studies are required to validate whether this approach can reach appropriate clinical outcomes with a full course of antibiotic treatment.
Supplemental Material
sj-pdf-1-cjk-10.1177_2054358120987061 – Supplemental material for Pharmacokinetics of Tobramycin Administered at the Beginning of Intermittent Hemodialysis Session (ESRD Study)
Supplemental material, sj-pdf-1-cjk-10.1177_2054358120987061 for Pharmacokinetics of Tobramycin Administered at the Beginning of Intermittent Hemodialysis Session (ESRD Study) by Marjolaine Giroux, Nicolas Bouchard, Anik Henderson, Lesly Lam, Van Anh Sylvie Tran, Denis Projean, Jean-François Tessier, Laurence Lepage, Paul Gavra, Georges Ouellet, Michel Vallée and Jean-Philippe Lafrance in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We acknowledge Yannick Emond and Katherine Desforges for assistance in the recruitment process and France Varin and Robert Bell for their contribution.
Ethics Approval and Consent to Participate
The study protocol was approved by the local ethics committee and a nonobjection letter was issued by Health Canada. All protocol amendments were reviewed by the local ethics committee. The study was conducted according to the Declaration of Helsinki and its amendments. Verbal and written informed consent were obtained for each subject before enrollment (ClinicalTrials.gov Identifier: NCT03904836).
Consent for Publication
All authors revised the final manuscript and have given their consent for publication.
Availability of Data and Materials
Study data are held by the Research Center at Hôpital Maisonneuve-Rosemont.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
