Abstract
Background:
Sodium bicarbonate (NaHCO3) is one of the promising solutions that has good safety profile and theoretical advantages regarding antimicrobial and antithrombotic properties but there are still limited reports.
Objective:
To compare the efficacy in lowering rate of catheter loss due to catheter-related thrombosis (CRT) or catheter-related blood stream infection (CRBSI) between sodium bicarbonate and heparin lock in prevalent chronic hemodialysis (HD) patients.
Design:
A multicenter, randomized, open-label study
Setting:
In a developing country, Thailand
Patients:
Chronic HD patients with tunneled central venous catheter
Measurements:
Catheter loss rate, rate of catheter-related blood stream infection, catheter-related thrombosis, and exit site or tunnel infection
Methods:
The prospective multicenter randomized controlled trial was conducted, we randomly assigned 118 patients undergoing HD with tunneled central venous catheter to receive a catheter locking solution of sodium bicarbonate or heparin. The primary outcome was a catheter loss rate due to CRT or CRBSI, while the secondary outcome was a composite outcome of CRT, CRBSI, or exit site/tunnel infection (ESI/TI).
Results:
The present study was stopped early due to an excess of catheter-related thrombosis in the sodium bicarbonate group. From the first 6 weeks of follow-up, there were no catheter losses due to CRT or CRBSI in both groups. The sodium bicarbonate group had a significantly higher rate of the secondary composite outcomes and this was entirely caused by CRT with the median time to thrombosis of 23.6 days. Every CRT event could be successfully rescued by using a single dose of recombinant tissue plasminogen activator (rt-PA).
Limitations:
Short follow-up period.
Conclusions:
In prevalent HD patients with tunneled CVCs, use of a sodium bicarbonate locking solution for prevention of CRT is inferior to heparin and is associated with a high rate of catheter-related thrombosis.
Trial registration:
The study was registered with the Thai Clinical Trials Registry TCTR 20200610003
Introduction
The current options for vascular access in chronic hemodialysis (HD) patients comprise arteriovenous fistula, arteriovenous graft, and central venous catheter (CVC). Choosing type of vascular access should be individualized for each patient to achieve his or her life goals. 1 Tunneled CVC is a selected choice in patients who fail arteriovenous access or have limited life expectancy. From a recent USRDS annual data report, 80% of patients at HD initiation and 19.6% of prevalent HD patients had been using CVC as vascular access. 2 A major concern of tunneled CVCs use is catheter patency. The main contributors to catheter loss are catheter-related thrombosis (CRT) and catheter-related blood stream infection (CRBSI). 3 Approximately 50% of CVC failed within 1 year 4 and up to two-thirds of failures were due to thrombosis. 5 The clinical impact of CRT is poorer clearance of uremic toxins, such as decreased KT/V. In addition, CRT was also associated with increased risks of missing dialysis session, hospitalization, and mortality. Besides CRT, the incidence of CRBSI had been reported from 1.1 to 5.5 episodes per 1,000 catheter days and was associated with increases in morbidity, hospitalization, and death. 6
Instillation of catheter locking solution at the end of HD session is a routine clinical practice to maintain catheter patency. Although heparin has been widely used as the standard locking solution of CVC in HD patients, it is associated with several risks, 7 including bleeding from systemic anticoagulant effect of the inadvertent leakage of heparin into the circulation.8-12 and heparin-induced thrombocytopenia (HIT) characterized as clotting and thrombocytopenia in certain HD patients.
Citrate is another recommended solution from the 2019 updated Kidney Disease Outcomes Quality Initiative (KDOQI) clinical practice guideline 1 due to non-inferior efficacy to heparin. However, the U.S. Food and Drug Administration issued an urgent warning about citrate-related life-threatening cardiac toxicity when high concentration of citrate was infused into HD patients. 13 From these reasons, initiation of another locking solution as an alternative is mandatory to circumvent heparin and citrate-related complications in such HD patients.
Under such situations, sodium bicarbonate has been recently used as one of the new upcoming alternative locking solutions with potential good safety profile and advantageous benefits involving antithrombotic and antimicrobial properties. Regarding blood coagulation, the chelation of calcium ions by sodium bicarbonate inhibits the conversion of fibrinogen to fibrin,14,15 resulting in decreased thrombosis. With respect to antimicrobial effects, sodium bicarbonate suppressed bacterial proliferation by lowering bacterial adherence and biofilm formation of certain bacteria, especially Gram-positive organisms, including Staphylococci. 16 Moreover, an abundant ionic factor could alter bacterial membrane permeability and structure, leading to less viability. 17 However, only few studies have evaluated clinical efficacy of the agent.
An earlier prospective randomized trial by El-Hennawy et al 18 demonstrated that sodium bicarbonate yielded a higher catheter retention rate at 6 months among chronic HD patients compared with normal saline. A following prospective cohort trial by El-Hennawy et al 19 in both acute and chronic setting of HD patients showed that sodium bicarbonate was statistically superior to normal saline in preventing HD catheter loss due to CRT and CRBSI. However, instead of heparin, normal saline solution (0.9% NaCl) was used as the control in both studies and yielded quite low rate of CRT (4.16 per 1,000 catheter), the value of which was surprisingly comparable to the efficacy of heparin in several previous studies.7,20,21
Therefore, we performed a prospective multicenter, randomized controlled trial (RCT) to compare the efficacy between 7.5% NaHCO3 and heparin 2,500 U per mL as a locking solution for tunneled CVC in HD patients. The primary aim was to determine whether 7.5% NaHCO3 locking solution could reduce the catheter loss rate due to CRT or CRBSI.
Methods
Study Population
This study was a prospective multicenter randomized control trial (RCT) which was conducted from June 1, 2020 until 30 November 2020, in 7 HD centers in Bangkok, Thailand. Due to the uncertainty in the efficacy of sodium bicarbonate as a locking solution, the study was planned to follow up for 12 weeks and a preliminary analysis at 6-week during the follow-up time was performed. Patients were eligible for enrollment in the study when they were older than 18 years and required long term twice- or thrice-a-week HD with tunneled CVC. All patients should be clinically stable within 1 month before enrollment, defined as no hospitalizations, no vascular access related infection, and no vascular access dysfunction.
We excluded patients with known coagulopathy or hemophilia, cirrhosis Child-Pugh B or C, thrombocytopenia less than 100,000 u per liter, long-term receipt of systemic anticoagulant therapy, history of HIT, and previously documented central vein stenosis. Written informed consent was obtained in all patients before enrollment. The study protocol was approved by the Ethics Research Committee, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand (IRB no. 233/63) and was registered in Thai Clinical Trial Registry (TCTR 20200610003).
Study Design
After providing written informed consent, eligible patients were randomly assigned in a 1:1 ratio by a computer-generated randomization with the use of permuted block design stratified according to frequency of HD (twice- vs thrice-a-week). Patients were assigned to one of the 2 groups: heparin LEO® (heparin sodium) pharmaceutical product (2,500 U per milliliter, full luminal volume), or 7.5% NaHCO3 (5 mL/catheter lumen) locking solution after each HD session (Figure 1). Before each HD session, the catheter site was carefully examined for signs of exit site and tunnel infection (ESI/TI) then the catheter locking solution was removed by the HD nurse. Test flow of each catheter lumen was performed to evaluate catheter dysfunction. If the flow problem occurred, bedside managements, which included repositioning the patients and performing rapid saline flushes to clear a possible clot, were performed, respectively. During HD treatment, patients in both groups received the same standard prescription: blood flow rate at least 300 mL/min and systemic heparin (loading dose 2,000 u and maintenance dose 1,000 u/hr until 1 hr before the end of dialysis session). Monitoring for complications was provided to every patient as the standard of care.

Consort flow diagram.
After completing HD treatment, each catheter lumen was flushed with 10 mL of saline and checked for the residual blood in the catheter. A repeated similar dose of saline flushing was performed until the catheter lumens were apparently clear. Then, the solution was locked, depending on each group. In heparin group, we used a volume exactly equivalent to the internal volume of the lumen labeled on the catheter. In sodium bicarbonate group, 7.5% NaHCO3 5 mL was locked in each catheter lumen. In patients who were documented as CRT, a recombinant tissue plasminogen activator (rt-PA, Alteplase 2 mg), instead of the usual locking solution, was administered in each catheter lumen as a rescue therapy. The standard of care during HD was strictly followed according to the Thai Hemodialysis Clinical Practice Recommendation 2014 22 which was modified from the 2006 Kidney Disease Outcomes Quality Initiative (KDOQI) recommendation, clinical practice guidelines for vascular access. 23
Outcomes
The primary outcome was a catheter loss rate defined as the premature catheter removal from a complication of CRT or CRBSI. According to the KDOQI 2006 guidelines, 23 catheter dysfunction was diagnosed by failure to maintain an extracorporeal blood flow to 300 mL/min or greater at pre pump arterial pressure more negative than −250 mmHg. Persistent catheter dysfunction after bedside repositioning was defined as CRT. The diagnosis of CRBSI was based on the definition from The Centers for Disease Control and Prevention (CDC). 24 Patients with clinical manifestations and at least 1 positive blood culture from peripheral vein and no other apparent sources were investigated whether they were met the criteria or not. Patients with bacteremia were immediately treated by the attending nephrologists and remained in the study.
The secondary outcome was a composite outcome of CRT, CRBSI, or ESI/TI rate. ESI was defined as hyperemia, induration, and/or tenderness ≥2 cm from catheter exit site. TI was identified if the development extended along the subcutaneous tunnel. All events and adverse effects due to both locking solutions were recorded by HD nurses in each center and reported to the investigators.
Statistical Analysis
Calculation of the required sample size was based on assumption that a catheter loss rate in sodium bicarbonate was approximately 1% in 6 months. With a two-sided test, an alpha level of 0.05, and power of 80% and dropout rate at 10%. Survival curves of the primary outcome and the secondary composite outcome of CRT, CRBSI, or ESI/TI were prepared with the use of the Kaplan-Meier method and were compared with the use of a two-sided log-rank test. Cox proportional-hazards models were used to compare event-free survival in the heparin and sodium bicarbonate groups, stratified according to the HD center.
Continuous data were expressed as mean and standard deviation (SD) or median and interquartile range (IQR). Two dependent samples were compared by using paired t-test or Wilcoxon signed rank test up to distribution pattern of the data. Categorical data were expressed as percentage. P value of <.05 was accepted as statistically significant. Post-hoc analysis were performed to explore the unknown important factors that might have important effects on the interested outcomes. All statistical analyses were performed using STATA® (StataCorp. Version 16. College Station, TX)
Results
The present study was stopped early due to an excess of catheter related thrombosis in the sodium bicarbonate group. Among 205 patients for whom tunneled CVCs were used during the randomization period, 118 patients met the inclusion criteria and agreed to participate in the study (Figure 1). There were no significant differences between 2 groups regarding baseline characteristics, except for diabetes mellitus (P = .01; Table 1). Five patients in the sodium bicarbonate group (8.8%) and 1 in the heparin group (1.6%) discontinued the study before the end of the study period. The reasons for the discontinuation included patient’s concern about catheter dysfunction problem (n = 3), vascular access change to arteriovenous graft (n = 1), moving to another HD center (n = 1), and death from heart failure (n = 1).
Baseline Characteristics of Heparin and Sodium Bicarbonate Groups.

Note. *P = .01 when compared with heparin. IQR = interquartile range; HD = hemodialysis; CVC = central venous catheter.
In the preliminary analysis at 6-week follow-up, the numbers of CRT in the sodium bicarbonate group were significantly higher than the heparin group (40.4% vs 0%, P < .001; Table 2). Every CRT event could be successfully rescued by utilizing a single treatment (2 mg for each catheter lumen) of rt-PA as a locking solution instead of sodium bicarbonate. The authors decided to cease the study. In the primary outcome, there were no catheter losses due to CRT or CRBSI in both groups during the 6-week study period (Table 2).
Primary and Secondary Outcomes.
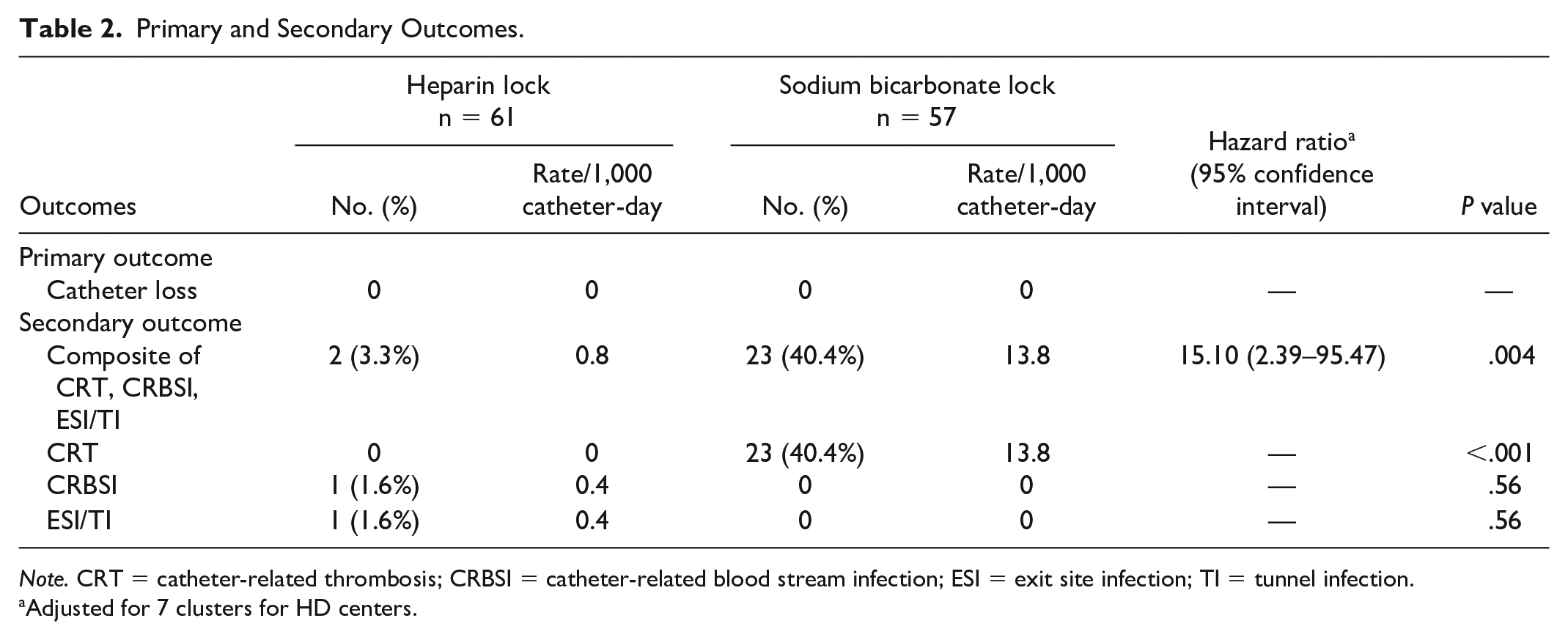
Note. CRT = catheter-related thrombosis; CRBSI = catheter-related blood stream infection; ESI = exit site infection; TI = tunnel infection.
Adjusted for 7 clusters for HD centers.
As demonstrated in Table 2, the secondary composite outcome, consisting of CRT, CRBSI, or ESI/TI, was significantly higher in the sodium bicarbonate group when compared with the heparin group (Hazard ratio 15.10; 95% CI = [2.39-95.47], P = .004; Figure 2). The results were unchanged in a model adjusted for HD centers. All events from secondary composite outcome in the sodium bicarbonate group were caused by CRT and the median time to thrombosis was 23.6 days. In the sodium bicarbonate group, there were no incidences of CRBSI and ESI/TI. In the heparin group, one patient developed CRBSI, while another patient had ESI/TI.

Kaplan–Meier plot for the time to a first episode of catheter-related event.
In post-hoc subgroup analysis, the effects of heparin on the secondary composite outcome were superior to sodium bicarbonate generally consistent across all subgroups without heterogeneity (Table 3). In addition, every 1 g/dL increase in hemoglobin level enhanced the risk of CRT by 1.23 times (Hazard ratio, 1.23; 95% CI = [1.02-1.48]; P = .03) in the sodium bicarbonate group (Supplementary Table S1).
Subgroup Analyses for the Secondary Outcome.

Note. HD = hemodialysis.
Adverse Events
There were no serious adverse events that could be contributed to the locking solutions. In the sodium bicarbonate group, 4 patients immediately developed symptoms, mostly perioral paresthesia or metallic taste but spontaneously recovered within 1 minute. Adverse events of interest due to sodium bicarbonate related effect, including metabolic alkalosis, and symptomatic hypocalcemia were not different from the heparin group. At the end of the study, the mean serum calcium levels were 8.8 vs 8.4 mg/dL and the means of serum bicarbonate were 21.8 vs 22.1 mEq/L in heparin and sodium bicarbonate group, respectively.
Cost-Related Outcomes
In our trial, we used 3 catheter locking solutions: heparin LEO® (heparin sodium) pharmaceutical product (5,000 units/mL), 5 mL per vial, (USD5.83); 7.5% NaHCO3, 10 mL per vial (USD0.41); and rt-PA (Alteplase), 2 mg per vial (USD56.95). In the heparin group, a total of 972 treatments in 61 patients were performed. The total cost was USD3,122.98 (USD3.21/HD session) which consist of heparin locking solution (USD2,840.68) and antibiotics for managing infectious complications (USD282.30).
In the sodium bicarbonate group, a total of 898 treatments in 57 patients were prescribed. The summation cost was USD3,000.71 (USD3.34/HD session) which comprised 7.5% sodium bicarbonate locking solution (USD371.92) and rt-PA (2 mg) as rescue therapy (46 vials; USD2,628.79).
In summary, the unadjusted costs per treatment including catheter locking solution, the rescue therapy, and antibiotics for managing infectious complications over the 6-week study were USD3.21 and USD3.34 in the heparin and sodium bicarbonate groups, respectively (Supplementary Table S2).
Discussion
The present study has demonstrated that the use of 7.5% sodium bicarbonate alone as a catheter locking solution in chronic HD patients caused a significantly higher rate of CRT when compared with heparin, the standard locking solution (Table 2, Figure 2). Nonetheless, the catheter patency could be successfully maintained with rt-PA as the rescue therapy when CRT occurred. Therefore, no catheter loss, as the primary outcome, was documented. The secondary composite outcome, comprising CRT, CRBSI, and ESI/TI, was significantly greater in the sodium bicarbonate group (Table 2).
To verify the real efficacy of sodium bicarbonate as a catheter locking solution, heparin was used as the control in the present study and it appeared that sodium bicarbonate had significantly inferior antithrombotic property to heparin (Table 2, Figure 2). Of note, the rate of CRT with sodium bicarbonate in the present work was much higher than the study by El-Hennawy et al 18 (Table 2, Figure 2). The causes of this discrepancy are still unclear but might be related to several issues including differences in study population and protocol. First, approximately 70% of our patients had over 1-year duration of tunneled CVCs. Fry et al 25 illustrated that the 1-year and 3-year catheter survivals among 812 HD patients were 61% and 22%, respectively. In this regard, the commonest causes of catheter failure were thrombosis and fibrin sheath formation. Such findings may be implied to the present study that the over 1-year used CVCs might already have a heightened risk of CRT at baseline. Second, there were more diabetes mellitus patients at baseline of demographic characteristics in the sodium bicarbonate group compared with the heparin group (66.7% vs 42.6%, P = .01; Table 1). Wójtowicz et al 26 showed that diabetes mellitus was one of the poor predictors of catheter survival (HR = 0.31, 95% CI = 0.11-0.92, P = .03) due to its pro-thrombotic state. This could be explained by endothelial injury, hyperfibrinogenemia, and platelet hyperreactivity related to increased glucose levels as well as hyperinsulinemia. However, the present study demonstrated that diabetes mellitus as baseline comorbidity tended to increase the CRT event rate but no statistical significance (HR = 2.03, 95% CI = 0.92-4.48, P = .08). Finally, we used a lower concentration of sodium bicarbonate (7.5% vs 8.4%) which might result in lower ionic factors and less antithrombotic effect.
As the present study included patients with twice-a-week HD which had a longer time of inter-dialytic period than thrice-a-week HD, one might consider that the properties of abundant ionic factors and alkaline pH might not be optimally preserved over the time in twice-a-week HD. To solve this question, we performed post hoc subgroup analysis and the result showed no difference between patients with twice-a-week and thrice-a-week HD (P value for interaction = .946; Table 3).
Intraluminal administration of rt-PA as a thrombolytic agent is currently recommended not only to dissolve CRT but also to be used once weekly as a prophylactic therapy in HD patients with high risk CRBSI. 1 In our trial, rt-PA was used as a rescue therapy in patients with CRT event. Owing to the great efficacy, all events of CRT could be rescued by using only a single treatment of rt-PA. As the median time to the first CRT event in the sodium bicarbonate group in the present study was 23.6 days.
The unadjusted costs per treatment due to catheter locking solution and rt-PA over the 6-week study was USD2.93 and USD3.34 in the heparin and sodium bicarbonate groups, respectively. However, if we calculated a total cost of catheter care including the cost of managing infectious complications (CRBSI and ESI/TI), the cost effectiveness between the 2 groups was comparable (USD3.21 vs USD3.34/HD session in the heparin and sodium bicarbonate group, respectively).
As shown in Supplementary Table S1, one of the most important risks of CRT from post-hoc subgroup analysis was a higher hemoglobin level. Every 1 g/dL increase in hemoglobin level potentiated the risk of CRT by 1.23 times (hazard ratio = 1.23; 95% CI = 1.02-1.48; P = .03) in the sodium bicarbonate group. As the number of the patient was quite small, the CRT-potentiating role of the heightened hemoglobin level needs further investigations. Other baseline factors, HD vintage, antiplatelet drug use, or statin use were not associated with the CRT outcome.
The strength of the present study is that it is the first prospective multicenter RCT to compare the efficacy in reducing catheter loss rate due to CRT and CRBSI between sodium bicarbonate and heparin among prevalent HD patients. Due to the obviously higher rate of CRT events in sodium bicarbonate group from the preliminary analysis, the present study was ceased after 6-week study duration. Admittedly, this might reduce the power of the primary and some secondary outcomes. In the present study, the long-term catheter patency could not be determined.
Conclusions
In prevalent HD patients with tunneled CVCs, use of a sodium bicarbonate locking solution for prevention of CRT is inferior to heparin and is associated with a high rate of catheter-related thrombosis.
Supplemental Material
sj-pdf-1-cjk-10.1177_20543581211046077 – Supplemental material for A Randomized Controlled Trial of Comparative Efficacy between Sodium Bicarbonate and Heparin as A Locking Solution for Tunneled Central Venous Catheters Among Patients Requiring Maintenance Hemodialysis
Supplemental material, sj-pdf-1-cjk-10.1177_20543581211046077 for A Randomized Controlled Trial of Comparative Efficacy between Sodium Bicarbonate and Heparin as A Locking Solution for Tunneled Central Venous Catheters Among Patients Requiring Maintenance Hemodialysis by Wannasit Wathanavasin, Jeerath Phannajit, Manorom Poosoonthornsri, Songkiat Lewsuwan, Patchara Tanateerapong, Kamonrat Chongthanakorn, Kullaya Takkavatakarn, Pisut Katavetin, Khajohn Tiranathanagul, Somchai Eiam-ong and Paweena Susantitaphong in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-pdf-2-cjk-10.1177_20543581211046077 – Supplemental material for A Randomized Controlled Trial of Comparative Efficacy between Sodium Bicarbonate and Heparin as A Locking Solution for Tunneled Central Venous Catheters Among Patients Requiring Maintenance Hemodialysis
Supplemental material, sj-pdf-2-cjk-10.1177_20543581211046077 for A Randomized Controlled Trial of Comparative Efficacy between Sodium Bicarbonate and Heparin as A Locking Solution for Tunneled Central Venous Catheters Among Patients Requiring Maintenance Hemodialysis by Wannasit Wathanavasin, Jeerath Phannajit, Manorom Poosoonthornsri, Songkiat Lewsuwan, Patchara Tanateerapong, Kamonrat Chongthanakorn, Kullaya Takkavatakarn, Pisut Katavetin, Khajohn Tiranathanagul, Somchai Eiam-ong and Paweena Susantitaphong in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We acknowledge all the staffs and HD nurses in every HD center, LEO Pharma C/O DKSH (Thailand) Limited, and Atlantic Pharmaceutical Co., Ltd. for providing us catheter locking solutions, heparin, and sodium bicarbonate, respectively.
Ethics Approval and Consent to Participate
Subjects have given their informed consent and that the study protocol has been approved by the Institute’s committee on human research.
Consent for Publication
All authors have provided consent for publication.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author’s Note
Manorom Poosoonthornsri is now affiliated with Clinical Pharmacy Services, Pharmaceutical Group, King Chulalongkorn Memorial Hospital, Bangkok, Thailand.
Author Contributions
Research idea and study design was contributed by WW, PK, and PS; data acquisition was contributed by WW, SL, PT, KC, and MP; data analysis/interpretation was contributed by WW, JP, PK, and PS; statistical analysis was contributed by JP; supervision or mentorship was provided by KT, KhT, PK, SE, PS. Each author contributed important intellectual content during manuscript drafting or revision, accepts personal accountability for the author’s own contributions, and agrees to ensure that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure form for Disclosure of Potential Conflicts of Interest and none were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by The Kidney Foundation of Thailand, Ratchadapiseksompotch Fund; Ratchadapiseksompotch Endorsement Fund (RA 63/080), Faculty of Medicine, Chulalongkorn University, Thailand, and the Research Unit for Metabolic Bone Disease in CKD patients, Faculty of Medicine, Chulalongkorn University (GRU 6201230005-1).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
