Abstract
Background:
The frequency and outcomes of starting maintenance dialysis in the hospital as an inpatient in kidney transplant recipients with graft failure are poorly understood.
Objective:
To determine the frequency of inpatient dialysis starts in patients with kidney graft failure and examine whether dialysis start status (hospital inpatient vs outpatient setting) is associated with all-cause mortality and kidney re-transplantation.
Design:
Population-based cohort study.
Setting:
We used linked administrative healthcare databases from Ontario, Canada.
Patients:
We included 1164 patients with kidney graft failure from 1994 to 2016.
Measurements:
All-cause mortality and kidney re-transplantation.
Methods:
The cumulative incidence function was used to calculate the cumulative incidence of all-cause mortality and kidney re-transplantation, accounting for competing risks. Subdistribution hazard ratios from the Fine and Gray model were used to examine the relationship between inpatient dialysis starts (vs outpatient dialysis start [reference]) and the dependent variables (ie, mortality or re-transplant).
Results:
We included 1164 patients with kidney graft failure. More than half (55.8%) of patients with kidney graft failure, initiated dialysis as an inpatient. Compared with outpatient dialysis starters, inpatient dialysis starters had a significantly higher cumulative incidence of mortality and a significantly lower incidence of kidney re-transplantation (
Limitations:
Possibility of residual confounding and unable to determine inpatient dialysis starts that were unavoidable.
Conclusions:
In this study we identified that most patients with kidney graft failure had inpatient dialysis starts, which was associated with an increased risk of mortality. Further research is needed to better understand the reasons for an inpatient dialysis start in this patient population.
Introduction
Six percent of all Canadians who start maintenance dialysis do so because of kidney graft failure. 1 Kidney transplants have a median graft survival of 10 to 15 years from a deceased donor and 15 to 20 years from a living donor. 2 It is well established that patients with kidney graft failure have poor outcomes (eg, mortality).3-7 Kaplan et al 3 found an approximately 3-fold higher annual adjusted death rate in kidney transplant recipients after graft failure (9.4%) compared with before graft failure (2.8%).
There are a substantial number of studies on the effects of unplanned dialysis starts (commonly defined as dialysis initiation in hospital and/or with a central venous catheter) 8 in patients receiving kidney replacement therapy for the first time finding that unplanned dialysis starters experience worse outcomes (eg, increased risk of death and hospitalization) compared with planned dialysis starters.9,10 However, to our knowledge, no studies have considered whether the same is true for patients who start dialysis unplanned after kidney graft failure and few studies have quantified the frequency of unplanned dialysis starts in these patients.11-13 Therefore, we conducted this study to characterize the frequency of inpatient dialysis starts (ie, our study definition of an unplanned dialysis start) in patients with kidney graft failure and to examine whether dialysis start status (inpatient vs outpatient) is associated with all-cause mortality. We also examined the trends in inpatient dialysis starts over time and the association between dialysis start status and kidney re-transplantation or the number of days alive and out of the hospital (DAOH).
Methods
Design and Setting
We conducted a population-based cohort study using administrative healthcare databases held at ICES (Ontario, Canada). These data sets were linked using unique encoded identifiers and analyzed at ICES. The use of data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a Research Ethics Board. We followed the REporting of studies Conducted using Observational Routinely collected health Data guidelines for observational studies (the RECORD statement) (Supplementary Table S1). 14
Data Sources
To identify kidney transplant recipients and graft failure events we used the Canadian Organ Replacement Register (CORR). Vital status and demographic information were obtained from the Registered Persons Database. We used the Canadian Institute for Health Information Discharge Abstract Database (CIHI-DAD) to identify procedural and diagnostic codes associated with hospitalizations, whereas the CIHI Same Day Surgery database was used to identify day surgeries. The CIHI National Ambulatory Care Reporting System database contains information on emergency room visits. We used the Ontario Health Insurance Plan to identify physician submitted diagnostic and billing codes, whereas the ICES-derived Physician Database was used to identify nephrologists.
Study Population
Patients with kidney graft failure
We included patients who experienced kidney graft failure at least 6 months after their initial transplant between January 1, 1994 and March 31, 2016 (ie, both the kidney transplant and graft failure must have occurred during this time frame); a 6-month time frame was selected to ensure we were not including recipients who had primary nonfunction (ie, kidney never functioned) and recipients are managed slightly different in the first 6 months posttransplant with more frequent visits to a nephrologist compared with later follow-up. We excluded individuals who met the following criteria: age <18 years at the graft failure date, previous organ transplant recipient (including kidney), simultaneous multiorgan transplant (eg, kidney-pancreas transplant), primary nonfunction (ie, additional data check to exclude individuals whose kidney never functioned; defined as at least 3 codes for dialysis with at least 1 code appearing in the first 7 days, in the 8-90 days, and in the 91-150 days after the transplant date),15,16 graft failure date that preceded the re-transplant date (≤14 days) (ie, patients who had a pre-emptive re-transplant after graft failure), and no evidence of kidney replacement therapy in the ±30 days of the graft failure date (ie, data check to ensure the patient truly had graft failure). The cohort entry date (index date) was the date of graft failure.
Dialysis Start Status
We defined an inpatient dialysis start as the initiation of maintenance dialysis in the hospital as an inpatient. Specifically, we captured dialysis procedure/intervention codes occurring during a hospital admission in CIHI-DAD. We required the hospital admission date associated with the dialysis intervention code to occur within ±45 days of the graft failure date (including the graft failure date). We selected 45 days to ensure our definition of inpatient dialysis starts was sensitive (ie, we did not want to miss anyone with an inpatient dialysis start). Furthermore, this time frame allowed for potential inaccuracies in the capture of the graft failure date. The graft failure date is provided to CORR directly from the transplant centers but this date has not undergone formal validation. 17 Patients not captured as an inpatient dialysis start were included in the outpatient dialysis start group (ie, patient initiating maintenance dialysis in an outpatient setting).
Outcomes
Our primary outcome was all-cause mortality (Supplementary Table S2) which is accurately captured in our databases.18,19 As a secondary outcome, we assessed kidney re-transplantation, which we defined as a kidney transplant occurring after the graft failure date. In addition, we assessed DAOH in the first year after graft failure. Days alive and out of the hospital is a measure of healthcare utilization widely used in the cardiovascular literature and captures both morbidity and mortality.20-22 We calculated DAOH by subtracting the total days in hospital and total days dead from the potential total follow-up time (365 days). To calculate the total days in hospital we added the durations of each hospital stay, whereas for total days dead we calculated the number of days from the death date to the end of the 1-year follow-up. We defined the total potential follow-up time as the number of days from the index date (ie, graft failure date) until the last day of follow-up (ie, 1 year after the index date).
Statistical Analysis
We used standardized differences to describe differences between patients with and without an inpatient dialysis start; a meaningful difference between groups is presented as a difference ≥10%. These differences provide a measure of the difference between the 2 groups divided by the pooled standard deviation. 23
To account for competing risks, we used the cumulative incidence function to calculate the cumulative incidence of all-cause mortality and kidney re-transplantation. 24 Specifically, for mortality, kidney re-transplantation was considered a competing event and for kidney re-transplantation, mortality was a competing event. The cumulative incidence function keeps patients who experienced the competing risk event (ie, death or re-transplant) in the risk set using inverse probability weighting; therefore, patients who die or have a re-transplant contribute less to the risk set than those who did not and were still at risk of the event. It is recommended that the cumulative incidence function is used when the risk of the competing event is high which could potentially overestimate the incidence of the outcome.24,25 We used Gray’s test to determine whether there was a statistically significant difference in outcomes between inpatient vs outpatient dialysis starters.
We present the subdistribution hazard ratios (SdHRs) from the Fine and Gray model to examine the relationship between inpatient dialysis starts (vs outpatient dialysis start [reference]) and the dependent variables (ie, mortality or re-transplant). We used Schoenfeld residuals to examine whether the proportional hazards assumption was met and examined our cumulative incidence function curves to determine any important departures from proportionality (none were noted). In our final model we adjusted for the following covariates which were selected based on clinical expertise and literature review: age at graft failure, sex, race, residence (rural vs urban), neighborhood income quintile, cause of end-stage kidney disease (ESKD), dialysis vintage (ie, time from dialysis initiation to kidney transplant), Charlson-Deyo comorbidity score, era of graft failure, era of transplant, delayed graft function, transplant vintage (ie, time from kidney transplant to graft failure), donor type (living vs deceased), number of hospitalizations in 6 months prior to graft failure, and days in hospital in 6 months prior to graft failure. To provide insight into the potential mechanism of the association between dialysis start status and mortality, in a post hoc analysis, we used the extended Cox model with a Heaviside function allowing us to obtain several cause-specific hazard ratios (HRs) over different time intervals. 26 To determine whether there was a statistically significant difference between individuals with an inpatient vs outpatient dialysis start in DAOH, we used the Wilcoxon-Mann-Whitney test. A logistic regression model was used to examine the association between the era of graft failure (1995-2004 [reference], 2005-2009, 2010-2013, 2014-2016) (independent variable) and inpatient dialysis starts (dependent variable).
There were some missing data for the following variables: income quintile (<1%), cause of ESKD (8.2%), and race (6.9%). For missing income quintile, we imputed the middle income category (ie, income quintile 3), whereas for cause of ESKD and race, we imputed the most common value, glomerulonephritis, and white race, respectively. We conducted all analyses in the Statistical Analysis System (SAS) version 9.4 (www.sas.com). We considered a 2-sided
Additional Analyses
To provide insight into the reasons for the hospitalization during which an inpatient dialysis start occurred, we examined the 5 most common diagnoses using the first 3 digits of the
Results
Baseline Characteristics
We included 1164 patients with kidney graft failure (see Figure S1 of Supplementary Appendix), of which 55.8% (n = 650) had an inpatient dialysis start and 44.2% (n = 514) had an outpatient dialysis start. Compared with outpatient dialysis starters, inpatient dialysis starters were significantly older (median 53 vs 49 years), more likely to be from the lowest income quintile (26.6 vs 18.1%), and more likely to have heart failure (34.0 vs 19.3%) (Table 1). The median (25th, 75th percentile) total follow-up was 3.3 years (1.6, 6.0 years).
Characteristics of Patients With Kidney Graft Failure Classified by Inpatient vs Outpatient Dialysis Start Status.

All baseline characteristics taken at the time of graft failure unless otherwise indicated.
Standardized differences were used to compare inpatient to outpatient dialysis starts. A meaningful difference between groups is presented as a difference ≥10%.
Quintiles of median neighborhood income.
Residing in an area with a population <10 000 represents a rural residence.
Measured prior to the initial kidney transplant.
Transplant vintage was defined as the time from the initial kidney transplant to the graft failure date [graft failure date - kidney transplant date].
Dialysis vintage was defined as the time from the dialysis initiation date (prior to the initial kidney transplant) to the kidney transplant date [kidney transplant date - dialysis initiation date]. Recipients of a pre-emptive transplant (ie, no dialysis prior to transplant) were given a dialysis vintage of 0.
Delayed graft function was defined as a dialysis code in the 1 to 7 days after the initial kidney transplant but no dialysis code in the 90 to 150 days after the kidney transplant date.
All comorbidities were assessed 5 years prior to the graft failure date.
Diabetes and hypertension defined as the presence of 2 Ontario Health Insurance Plan codes or 1 hospitalization with a diagnosis of diabetes or hypertension.
Kidney disease is a variable in the Charlson-Deyo index. Therefore, all kidney transplant recipients with a Charlso-Deyo index score of 0 were given a 2 and those with a score of 1 were given a 3.
All-Cause Mortality
During follow-up, 283 (43.5 %) patients with an inpatient dialysis start died compared with 155 (30.2%) with an outpatient dialysis start. The cumulative incidence of all-cause mortality was significantly higher in inpatient dialysis starters compared with outpatient dialysis starters (
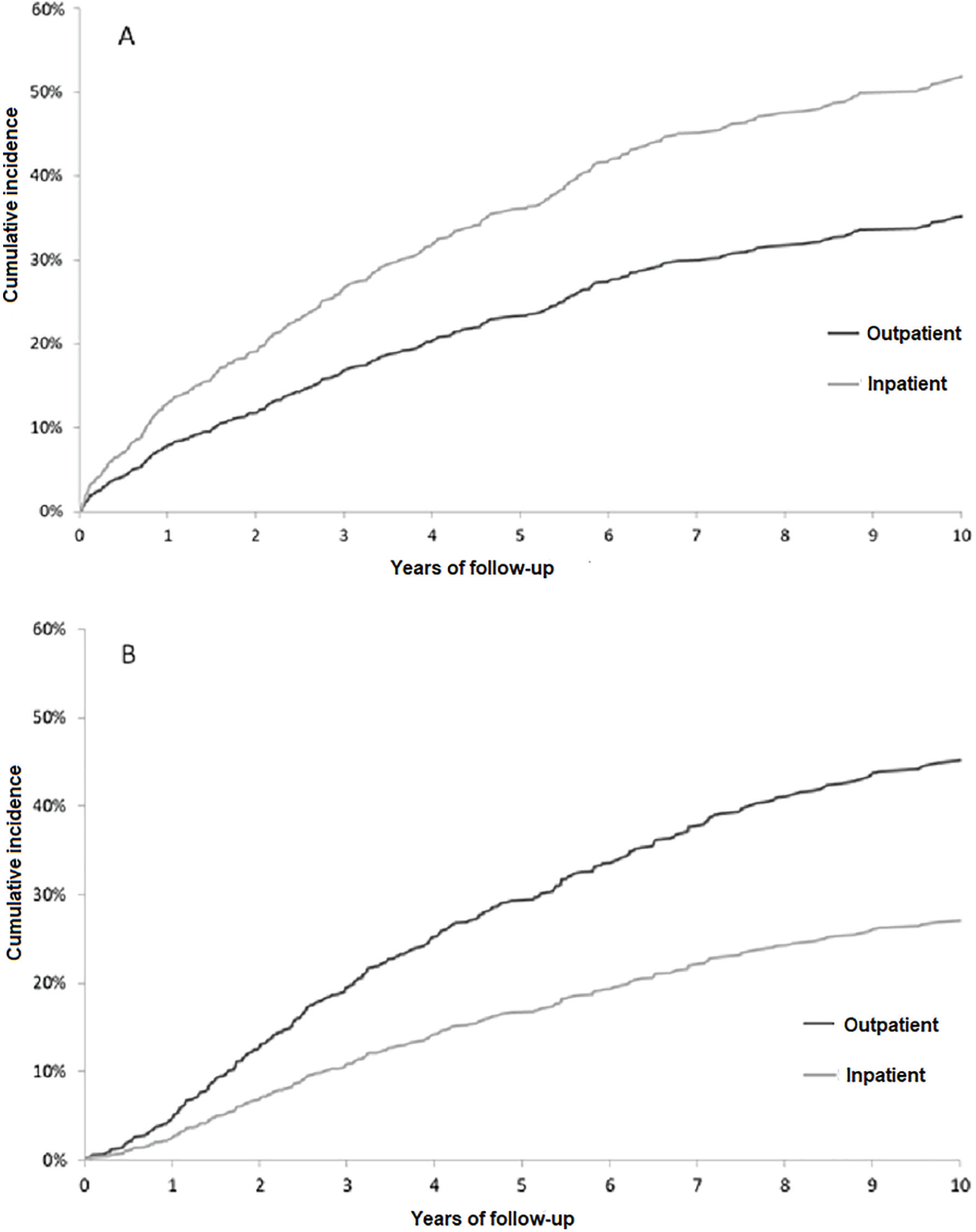
(A) Cumulative incidence of all-cause mortality, accounting for the competing risk of kidney re-transplant, in patients with kidney graft failure displayed by inpatient vs outpatient dialysis starts. (B) Cumulative incidence of kidney re-transplant, accounting for the competing risk of death, in patients with kidney graft failure displayed by inpatient vs outpatient dialysis starts.
All-Cause Mortality and Kidney Re-transplant for Patients With Kidney Graft Failure With an Inpatient vs Outpatient Dialysis Start.

Cumulative incidence of all-cause mortality, accounting for the competing risk of kidney re-transplantation.
Adjusted for age, sex, race, residence, income quintile, cause of ESKD, dialysis vintage, Charlson-Deyo index, era of graft failure, era of transplant, delayed graft function, transplant vintage, donor type, number of hospitalizations in 6 months prior to graft failure, and days in hospital in 6 months prior to graft failure.
Cumulative incidence of kidney re-transplant, accounting for the competing risk of death.
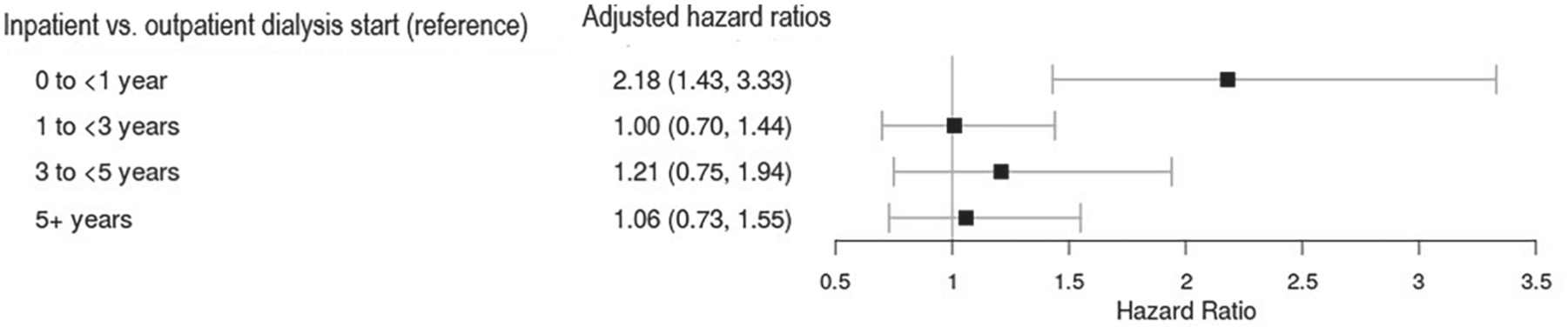
Adjusted hazard ratios for all-cause mortality in patients with kidney graft failure with an inpatient vs outpatient dialysis start.
Kidney Re-transplant
During follow-up, 138 (21.2%) patients with an inpatient dialysis start and 186 (36.2%) with an outpatient dialysis start had a kidney re-transplant. The cumulative incidence of a kidney re-transplant was significantly lower in inpatient dialysis starters compared with outpatient dialysis starters (
Additional Outcomes
The median (25th, 75th percentile) number of DAOH for patients with an inpatient dialysis start was 349 days (313, 359), whereas for outpatient dialysis starts, it was 361 days (352, 365) (
Odds Ratio of an Inpatient Dialysis Start in Patients With Kidney Graft Failure, Presented by Era of Graft Failure.
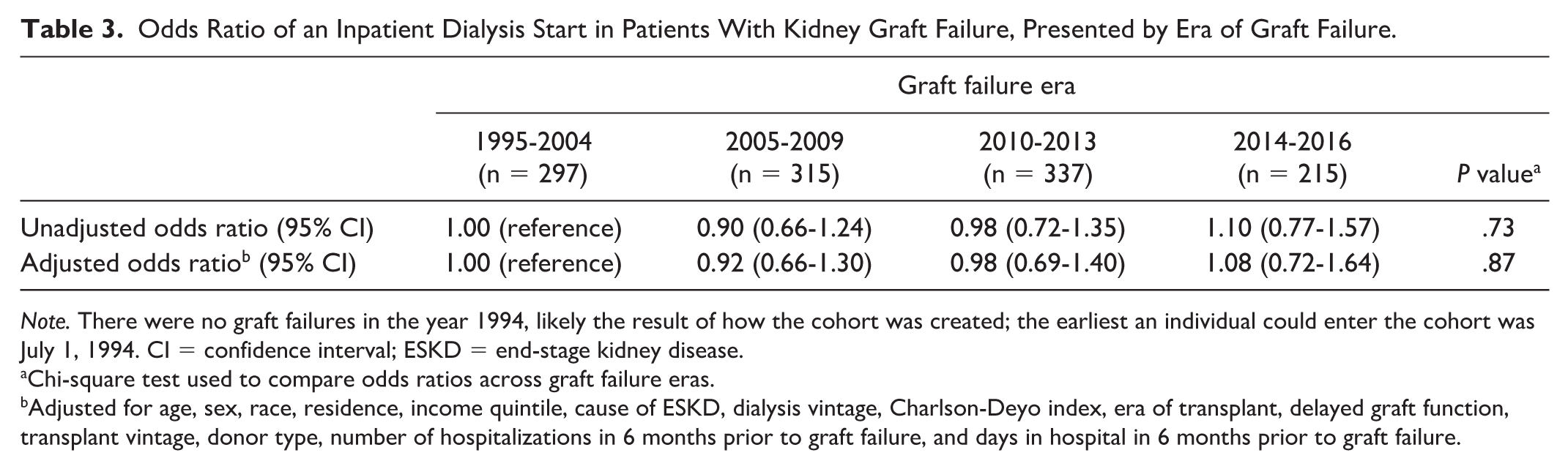
Chi-square test used to compare odds ratios across graft failure eras.
Adjusted for age, sex, race, residence, income quintile, cause of ESKD, dialysis vintage, Charlson-Deyo index, era of transplant, delayed graft function, transplant vintage, donor type, number of hospitalizations in 6 months prior to graft failure, and days in hospital in 6 months prior to graft failure.
Additional Analyses
When examining the most common reasons for hospital admissions associated with inpatient dialysis starts, we found that for patients with kidney graft failure after April 1, 2002, the 2 most common reasons were failure and rejection of transplanted organs and tissues (33.9%) and acute renal failure (12.5%) (Supplementary Table S3).
When restricting our analysis to inpatient dialysis starts that occurred within ±30 days of the graft failure date, we found 50.9% (n = 593) of patients with kidney graft failure initiated dialysis as an inpatient. Similar to our primary analysis, starting dialysis as an inpatient had a significantly higher cumulative incidence of all-cause mortality (
Discussion
In this study, we found that 56% of kidney transplant recipients with graft failure, initiated dialysis as an inpatient. These patients had worse outcomes, including a significantly higher cumulative incidence of all-cause mortality and fewer DAOH, while also having a lower cumulative incidence of kidney re-transplant compared with outpatient dialysis starters. These results highlight opportunities to improve the care of kidney transplant recipients leading up to graft failure.
Few studies have quantified the frequency of inpatient dialysis starts in patients with kidney graft failure, with estimates ranging from 6 to 44%.11-13 For example, Evans et al 11 conducted a small study at 2 transplant centers (n = 179) in the United Kingdom and found that approximately 42% of patients with kidney graft failure started dialysis in the hospital as an acute admission (ie, not in an outpatient [planned] setting). One potential reason for the wide range in estimates across studies is the inconsistency in the definition of an unplanned dialysis start. 27
The large proportion of inpatient dialysis starts in this study may be surprising as kidney transplant recipients are followed regularly by a nephrologist. However, several studies have found that care in kidney transplant recipients is often not optimal with many clinical targets being unmet (eg, blood pressure, hemoglobin).28-30 The Dialysis Outcomes and Practice Patterns Study found patients on dialysis with kidney graft failure were less likely to have many of their clinical targets met (eg, parathyroid hormone levels) compared with dialysis patients on the kidney transplant waitlist who never had a transplant. 31 Possible explanations for why kidney transplant recipients with a failing graft may start dialysis as an inpatient include a focus on preserving the graft rather than on predialysis care, unwillingness by the patient and provider to accept that the transplant is failing, and limited studies on the optimal timing of dialysis initiation in this patient population.13,32 In our study, it is unlikely that inpatient dialysis starts were a result of patients not being followed frequently by a nephrologist as inpatient dialysis starters had a significantly higher number of median visits to a nephrologist in the year prior to graft failure vs outpatient dialysis starters (16 vs 12). Recognizing the potential suboptimal care of patients with failing kidney transplants, the British Transplant Society published guidelines for their management in 2014. 33 However, many of the recommendations were not supported by high-quality evidence. 33
One potential way to enhance the quality of care in kidney transplant recipients approaching graft failure is to improve the coordination of care, with a multidisciplinary team involving the nephrologist, other physicians, allied health workers, and the patient.9,34 Furthermore, care could be improved by implementing clinics dedicated to managing these patients, as suggested by the British Transplantation Society, although there is limited evidence to support this approach. 33 In Ontario, several transplant centers have clinics dedicated to the care of patients approaching graft failure, whereas other centers have patients managed through their chronic kidney disease clinics. Despite the initiation of these clinics, we found no decline in the incidence of inpatient dialysis starts over the 20-year study follow-up. Therefore, more research evaluating interventions to optimize care prior to dialysis initiation in patients with graft failure is needed. 35
It is important to note that regardless of the quality of management, a proportion of inpatient dialysis starts will be unavoidable due to sudden acute illness or an unpredictable rapid decline in renal function. In our study, we found that 22.6% of our inpatient dialysis starters had a dialysis start date that was captured in an intensive care unit setting which could suggest a sudden acute event resulting in an unavoidable start. Furthermore, we found that 12.5% of patients had acute kidney failure as their most common diagnosis for hospitalization associated with their inpatient dialysis start which could suggest an unavoidable start. However, we cannot definitively state these were unavoidable as this would require serial glomerular filtration rate measurements; our databases only had laboratory values for a small proportion of our cohort. Further research is needed to better understand the reasons for an inpatient dialysis start in this patient population.
There are several potential explanations for why patients with kidney graft failure who experience an inpatient dialysis start have higher mortality compared with outpatient dialysis starters. First, there could be unmeasured differences between the 2 groups (eg, medication, laboratory measures). Second, there could be differences in vascular access type, with arteriovenous (AV) access associated with superior survival compared with central venous catheters. 36 However, previous reports have found no difference in the proportion of patients with kidney graft failure with an AV access between inpatient and outpatient dialysis starters. 13 Third, in the general population patients who are admitted to the hospital have a significantly higher risk of death compared with patients with no admission; being admitted to the hospital could represent a greater overall burden of disease that we may not have been able to fully adjust for. 37 Fourth, patients who start dialysis as an inpatient may be sicker, being more likely to stop dialysis and receive conservative care. Future studies should examine conservative care and end-of-life care in patients with graft failure. Finally, patients with an inpatient dialysis start may be more likely to get an infection, with sepsis common in patients with graft failure. 38 However, it is important to note that when we presented HRs stratified by follow-up time, inpatient dialysis starters had a higher rate of all-cause mortality in the first year of follow-up, but at 1+ years, there was no significant difference between outpatient and inpatient dialysis starters. This potentially suggests that the mechanism of an inpatient dialysis start being associated with poorer outcomes is related to the immediate effects of the start itself (eg, inferior dialysis preparation). This could also suggest that inpatient dialysis starters who die in the first year have significant more medical complications and comorbidities which were not captured in our adjusted analysis.
Limitations of our study deserve mention. Residual confounding could have accounted for some of the differences in outcomes seen between patients. However, we adjusted for several clinical characteristics, accounting for many of the baseline differences between the 2 groups. Not all administrative database codes used in this study have been validated, including the graft failure date. Results may not be generalizable to other countries. Finally, we were not able to determine the proportion of inpatient dialysis starts that were unavoidable. If a high proportion of inpatient starts are unavoidable, there might be little that can be done to avoid these starts and their consequences, including mortality.
In conclusion, inpatient dialysis starts are common in patients with kidney graft failure. Inpatient dialysis starters have an increased risk of mortality and a decreased incidence of kidney re-transplantation compared with outpatient dialysis starters. These results serve as a call to action to better understand the causes of inpatient dialysis starts to guide the development and testing of interventions to reduce the burden and improve outcomes of inpatient dialysis starts in this patient population.
Supplemental Material
sj-pdf-1-cjk-10.1177_2054358120985376 – Supplemental material for Outcomes of an Inpatient Dialysis Start in Patients With Kidney Graft Failure: A Population-Based Multicentre Cohort Study
Supplemental material, sj-pdf-1-cjk-10.1177_2054358120985376 for Outcomes of an Inpatient Dialysis Start in Patients With Kidney Graft Failure: A Population-Based Multicentre Cohort Study by Kyla L. Naylor, Gregory A. Knoll, Eric McArthur, Amit X. Garg, Ngan N. Lam, Bonnie Field, Leah E. Getchell, Emma Hahn and S. Joseph Kim in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
G.A.K. is supported by the University of Ottawa Chair in Clinical Transplantation Research. Two patient partners were involved in setting the research questions, providing input on the selection of the outcome measures, commenting on the study results, and developing a dissemination plan. We thank our patient partners whose experiences with kidney transplantation and graft failure informed this research. This study was supported by the ICES Western site. ICES is funded by an annual grant from the Ontario Ministry of Health and Long- Term Care (MOHLTC). Core funding for ICES Western is provided by the Academic Medical Organization of Southwestern Ontario (AMOSO), the Schulich School of Medicine and Dentistry (SSMD), Western University, and the Lawson Health Research Institute (LHRI). The opinions, results, and conclusions are those of the authors and are independent from the funding sources. No endorsement by ICES, AMOSO, SSMD, LHRI, or the MOHLTC is intended or should be inferred. The research was conducted by members of the ICES Kidney, Dialysis and Transplantation team, at the ICES Western facility. Parts of this material are based on data and information compiled and provided by CIHI and MOHLTC. However, the analyses, conclusions, opinions, and statements expressed herein are those of the author, and not necessarily those of CIHI and MOHLTC.
Ethics Approval and Consent to Participate
ICES is a prescribed entity under section 45 of Ontario’s Personal Health Information Protection Act. Section 45 authorizes ICES to collect personal health information, without consent, for the purpose of analysis or compiling statistical information with respect to the management of, evaluation or monitoring of, the allocation of resources to or planning for all or part of the health system. Projects conducted under section 45, by definition, do not require review by a Research Ethics Board. This project was conducted under section 45, and approved by ICES’ Privacy and Compliance Office.
Consent for Publication
All authors consent to the publication of this study.
Availability of Data and Materials
The data set from this study is held securely in coded form at ICES. While data sharing agreements prohibit ICES from making the data set publicly available, access can be granted to those who meet prespecified criteria for confidential access, available at ![]() . The full data set creation plan and underlying analytic code are available from the authors upon request, understanding that the programs may rely upon coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
. The full data set creation plan and underlying analytic code are available from the authors upon request, understanding that the programs may rely upon coding templates or macros that are unique to ICES and are therefore either inaccessible or may require modification.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.J.K. and G.A.K. have received investigator-initiated research grants from Canadian Institutes of Health Research and Astellas Canada. A.X.G. received an investigator-initiated grant from Astellas for a Canadian Institutes of Health Research study in living kidney donors. The other authors declare no conflicts of interest. The results presented in this paper have not been published previously in whole or part.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
