Abstract
Background:
Blood group incompatibility (ABOi) is the most common barrier to living donor kidney transplantation. Options for such recipients include kidney paired donation (KPD) or desensitization methodology to reduce blood antibody response.
Objective:
The objective of this study is to report on the first North America experience in ABOi living donor kidney transplantation using Glycosorb ABO immunoadsorption columns.
Design:
Retrospective observational cohort study.
Setting:
Renal transplant program at St. Michael’s Hospital, Unity Health Toronto, University of Toronto.
Patients:
Twenty-six ABOi living donor transplants from August 2011 through February 2020 were undertaken at our center.
Measurements:
Renal allograft and patient survival postdesensitization for ABOi living donor transplants and isohemagglutinin titer reduction.
Methods:
Preoperative immunosuppressive regimen consisted of a single dose of Rituximab 375 mg/m2 IV on day −28; tacrolimus, mycophenolic acid, and prednisone to start on day −7. Immunoadsorption treatments with Glycosorb A or B columns were performed on day −7 through day −1 based on anti-A or anti-B titers on Spectra Optia Apheresis System. Immunosuppression included basiliximab, solumedrol followed by oral prednisone, once-daily tacrolimus, and mycophenolic acid. The mean follow-up was 53 months (3-96 months).
Results:
A total of 26 individuals underwent an attempt at desensitization of whom 24 patients underwent immediate transplant. One patient had a rebound in titers and subsequently was transplanted from a blood group compatible living donor. A second patient had an unrelated medical issue and desensitization was discontinued. Five-year patient survival was 96% and death censored allograft survival was 92%. Posttransplant anti-A or anti-B titers were monitored daily for the first 7 days posttransplant and every 2 days from days 7 to 14. There were no acute rejections seen in this cohort of transplant recipients.
Limitations:
As our protocol was first initiated as proof of concept, a few recipients had low initial isohemagglutinin titers. This may have contributed to improved clinical outcomes.
Conclusions:
ABO column immunoadsorption with specific columns is a safe and effective method for ABOi living donor kidney transplantation, and an option when KPD is less than ideal.
Trial not registered as this was a retrospective cohort review.
Introduction
Despite the recent success of kidney paired donation (KPD) programs, there remains an ongoing need for direct living donation between blood group–incompatible donor/recipient pairs (ABOi). The desensitization methods for overcoming ABOi have evolved over the years, with many programs using plasma exchange (PLEX) and rituximab to replace splenectomy.1-3 In some jurisdictions such as in the United States, PLEX is the only available option for ABOi transplantation. While effective, there may be limits with PLEX to remove high levels of anti-blood group antibodies. 2 In addition, the use of plasma is not without inherent risk. The development of novel blood group–specific immunoadsorption (IA) columns has allowed for a more effective and rapid approach of desensitization for ABOi living renal transplantation. 4 In 2011, Health Canada approved the use of Glycosorb IA columns, Glycorex Transplantation AB Lund, Sweden, for ABOi kidney transplantation. The objective of this study is to report on the first and only Canadian/North American experience with these columns.
Materials and Methods
Patients
Twenty-six potential kidney transplant recipients with living donors underwent ABOi desensitization from August 2011 through February 2020. Although KPD was presented as an option to all patients, the ABOi desensitization protocol was offered as a first choice when KPD may have been a less suitable preference. These included (1) blood group AB donors, a cohort who are very difficult to match in a paired donation system; (2) blood group O recipients, who match less frequently in KPD; and (3) other donor/recipient pairs who did not want to wait for a match in KPD and could be transplanted within 30 days from the start of the protocol, in particular, 5 patients who were hoping to avoid dialysis. Inclusion criteria were recipients with incompatable blood groups against their donor, and the absence of any HLA donor-specific antibodies (DSA), and negative flow cross match. While all data were collected prospectively for clinical outcomes, the analysis of study was conducted as a retrospective review using our transplant electronic medical record and hospital database. The analysis of the retrospective study was approved by the research ethics board at St. Michael’s Hospital.
Desentization Procedures
Following donor and recipient blood group determination, recipients’ isohemagglutinin titers were measured by manual tube method as follows: IgG titer by indirect antiglobulin test at 37°C and IgM titer by immediate spin at room temperature. Patients with isohemagglutinin titers exceeding 1:512 were not allowed to proceed with transplant under this protocol. The titer was used to determine the approximate number of the IA procedures required pretransplant. Titers were repeated prior to the first IA and then after every IA treatment. As per manufacturer’s recommendation, angiotensin-converting enzyme inhibitors were stopped prior to treatment. Immunoadsorption therapy was planned to begin at day −7 and number of treatments was based on the baseline titer. Either central venous catheter or an arteriovenous (AV) fistula/graft was used for access. The method for antibody removal employed antigen-specific IA using Glycosorb ABO columns. These low molecular carbohydrate columns including A or B blood group antigens linked to a sepharose matrix adsorb isohemagglutinins both effectively and specifically. 5 The columns were attached to a Spectra Optia Apheresis System and 2.5 plasma blood volumes were processed per session. All procedures were performed by the apheresis nurses. The need for further IA was based on the post-IA titers from a previous session. Transplant proceeded when the relevant isohemagglutinin titer was 1:4 or less. 6 Posttransplant IA was administered if either IgG or IgM isohemagglutinin titer exceeded 1:8 in week 1 and 1:16 in week 2, as per the protocol. 6
Immunosuppression
All patients received rituximab 375 mg/m2 at 30 days prior to the anticipated transplant. On day −7 patients began once-daily tacrolimus at 0.15 mg/kg, prednisone 0.5 mg/kg, and mycophenolic acid at a dose of 720 mg orally twice daily. For patients with anti-A, or anti-B pretransplant titers of 1:256 or greater, IViG 1 g/kg was given at day −1. At the time of transplant, patients received basiliximab induction 20 mg days 0 and 4, IV solumedrol 1 g/kg preoperatively and 0.5 g/kg twice daily for 48 hours postoperatively followed by an oral prednisone taper to 5 mg/d by 60 days, once-daily tacrolimus with C0 targets of 5 to 8 ng/mL, and mycophenolate acid 720 mg twice daily, reduced to 720 mg in a.m., and 360 mg in p.m. by month 3.
Statistical Analysis
This was a description of a cohort of patients. Outcome data were reported as means and standard deviations. Pearson correlation coefficient was used in correlating the number of treatments with predesensitization titers.
Results
Patient Demographics
Twenty-six patients underwent desensitization between August 2011 and February 2020. Their demographics are shown in Table 1. Twenty-four patients were transplanted. Most of the patients (84%) were male and the average age at transplant was 51 years. Of note 5 (20%) were preemptive transplant recipients. Seventeen of 24 recipients were blood group O, and 6 donors were blood group AB. Five patients were transplanted preemptively. The mean anti-A or anti-B IgG titers pretreatment was 1:64 (range = 1:4-1:512).
Baseline Demographics n = 26.
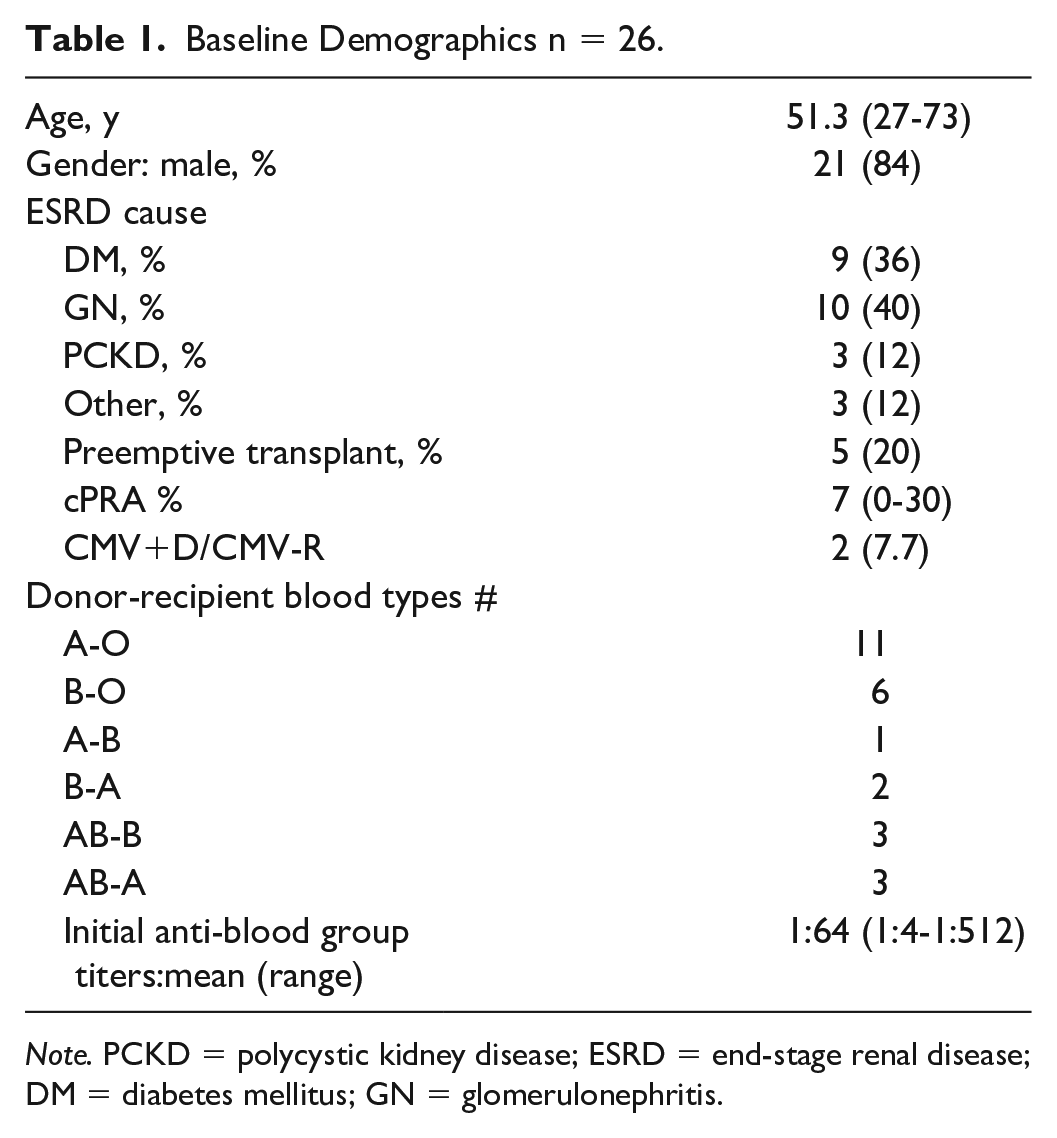
Note. PCKD = polycystic kidney disease; ESRD = end-stage renal disease; DM = diabetes mellitus; GN = glomerulonephritis.
Two of the 26 attempted IA recipients did not undergo immediate transplantation. One patient who underwent desensitization had a rebound in anti-A titers on the day of transplant, resulting in cancelation of the procedure. This patient ultimately found a blood group compatible donor and was later successfully transplanted. One patient underwent 2 IA procedures, but the transplant was postponed owing to an unrelated medical issue.
Patient and Allograft Survival
For the 24 transplanted recipients, patient survival was 96% at a mean of 53 months posttransplant follow-up, one patient died 3.5 years posttransplant with a functioning allograft from a cardiovascular event. Death censored graft survival was 91.3% (Table 2). There were 2 graft losses in 23 patients who remain alive. The first graft lost occurred secondary to renal vein thrombosis at the time of transplant. The second graft loss was at 4.5 years posttransplant in a patient who developed tuberculosis and required reduction in immunosuppression. The course was complicated by late acute rejection and BK nephritis following augmentation of immunosuppressive therapy. Otherwise, there were no rejections at any time point posttransplant in any other recipient.
Posttreatment, Posttransplant n = 24, Mean f/u 53 Months.

Glycosorb Treatments and A, B Titers
The mean anti-A, anti-B IgG titers pretransplant in this cohort was 1:64 (1:4-1:512). The mean number of Glycosorb treatments required was 2.66 (1.4), range 1 to 5. There was a direct correlation between pretreatment titers and treatments (Figure 1). Most patients were able to achieve titers between 1:1 and 1:2 at the time of transplant; however, a few recipients were transplanted with titers of 1:4. All patients were monitored with anti-A, B titers daily from days 0 to 7 and every second day from days 8 to 14. Two patients required a single treatment posttransplant for titers of 1:16 or greater, and one patient required 2 posttransplant treatments.

Correlation between anti-A, anti-B titers and number of treatments required.
There were no major adverse events related to IA. There were no cases of anaphylaxis. The most common complication was citrate toxicity manifested as mild paresthesia. which occurred in 2 patients. The risk of this complication was mitigated by monitoring ionized calcium and replacing calcium as needed.
Two patients were CMV mismatched (CMVD+/CMV−). They received oral valganciclovir prophylaxis for 6 months and remained free of CMV disease. A single patient who was CMV seropositive at the time of transplant developed CMV 4 months posttransplant, which readily resolved with treatment.
Discussion
Similar to a number of other jurisdictions, Canada has had a national KPD system since 2008. 7 All kidney transplant programs in the country partake in KPD which has facilitated transplants for donor/recipient pairs with blood group and HLA incompatibilities. Regardless, there remains a cohort of living donor pairs for whom paired donation is not the best option. Donors with blood group AB rarely match in a KPD program. In addition, blood group O recipients are disadvantaged compared with other blood groups. Some recipients wish to be transplanted preemptively. Finally, in Canada, unlike in American KPD programs, in most of the instances, donors travel to the intended recipient transplant center after a match, 7 instead of the donor kidneys being shipped, but some donors would prefer not to do so.
Glycosorb IA columns were approved for use by health Canada in 2010. However, at present, they have not been approved for use by the U.S. Food and Drug Administration (FDA) in the United States.
We sought to replicate work of Tydén et al who published their experience using these columns in 2005. 8 The potential benefits of specific IA versus other ABOi desensitization methods include specificity against blood group antibodies, a potential for reduction of higher levels of anti-blood group titers, 9 and a shorter duration of desensitization. However, these claims have never been subjected to the rigors of a randomized clinical trial and remain hypothesis generating.
A clear advantage, however, is the avoidance of exposure to blood products (albumin and/or plasma) and preservation of clotting factors (reducing the risk of bleeding) and all other immunoglobulins as IA specifically removes only anti-A and anti-B antibodies (avoiding further unnecessary immunosuppression). 10 This approach avoids the cost of blood products and any potential side effects relating to their use. Because during IA patient plasma is filtered and then returned to the patient (versus discarded in conventional PLEX), any circulating drugs are not removed. However, the specific IA columns come at a higher financial cost, although this has not uniformly been seen. 11 Although not explored in this study, a direct ABOi transplant obviates the costs asscociated with a kidney donor (and family member) having to travel to another city as is most often the case in the Canadian KPD system. 7
There have been reports of rebound of titers. 12 In our experience with 26 patients, there was a single individual in whom anti-A titers rebounded on more than one occasion with the IA columns ultimately resulting in the canceling of the transplant surgery. The reason for rebound in titers remains unclear at this time; however, anecdotally it may be more common with anti-A compared with anti-B blood group antibodies.
The Tyden protocol used IViG on day −1. Although we initially followed this, subsequently we modified the protocol to limit the use of IViG for those recipients with titers of 1:256 or greater. In addition, we discontinued the use of IVIG, as IVIG itself contains isohemagglutinins and may affect the isohemagglutinin titers. We also started out using a fixed protocol of posttransplant IA as per the original Swedish series but quickly switched to the German on-demand IA strategy. 6
At the time of writing, we are unaware of any other North American kidney transplant centers using the Glycosorb IA columns for ABOi. While used in Europe and Australia, the columns have not yet been approved for use by the FDA in the United States. Anecdotally, we have supplied columns to our local pediatric center for use in ABOi pediatric cardiac transplants.
Our experience with ABOi kidney transplant shows similar patient and allograft survival to blood group compatible living transplantation in our program. Five-year death censored allograft survival in ABOi with Glycosorb IA was 91.3%, compared with 92.3% in the blood group compatible cohort. There were no acute rejections in our patients. In the entire cohort, there was a single late rejection in a patient who required reduction in immunosuppression owing to tuberculosis and BK viremia.
While there have been a number of reported successful outcomes in ABOi renal transplantation, the experience using nonspecific desensitization methods has been variable. In some analysis, the outcomes of ABOi compared with compatible LD transplant are inferior with a negative bias to PLEX desensitization therapies being employed. 2 A meta-analysis by de Weerd could not demonstrate outcome differences regardless of desensitization technique. 13 However, Lo et al, after a mean of 26 months follow-up, demonstrated that overall graft survival was significantly worse after plasmapheresis compared with IA desensitization. 14
We as others have demonstrated that allograft accommodation occurs very early after transplantation, without the need for long-term monitoring. Of note, ABOi accommodation occurs despite the ongoing presence of recipient anti-A, anti-B antibodies, and the continued expression of A and B blood group antigens on the endothelium of vascularized organs. Various hypotheses for graft acceptance are beyond the scope of discussion but include alterations in pathways of apoptosis and alterations in cell transduction, in addition to a distinct pattern of gene expression, 15 quality of blood group antibody expression, 16 and C4d staining in peritubular capillaries without rejection. 17 A desired goal is to offer more transplants to obviate or avoid dialysis. In Canada, the national KPD system has had a strong impact on increasing the number of living donor transplants that were not previously possible. Desensitization using specific IA columns is yet another tool to help in achieving this outcome.
In summary, specific Glycosorb IA columns are efficacious for ABOi living kidney transplantation in the North American setting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
