Abstract
Background:
It is uncertain how often patients with autosomal dominant polycystic kidney disease (ADPKD) develop kidney stones.
Objective:
To review English-language studies reporting the incidence and prevalence of stones and stone interventions in adults with ADPKD.
Design:
Systematic review and meta-analysis.
Setting:
Any country of origin.
Patients:
Adult patients with ADPKD.
Measurements:
Incidence or prevalence of kidney stones and stone interventions.
Methods:
We reviewed 1812 citations from bibliographic databases, abstracted data from 49 eligible studies, and assessed methodological quality in duplicate. In some studies, the proportion of adults with ADPKD with the outcome were compared to adults without ADPKD; for these studies, prevalence risk ratios were calculated and pooled using a random effects model.
Results:
We identified 49 articles that met our review criteria. The methodological quality of many studies was limited (scores ranging from 2 to 14 out of 22, with a higher score indicating higher quality). No study clearly reported stone incidence, and in the cross-sectional studies, the definition of stones was often unclear. The prevalence of stones ranged from 3% to 59%, and a prevalence of stone interventions ranged from 1% to 8%; the average patient age at the time of assessment ranged from 26 to 61 years across the studies. Two studies reported a nonstatistically significant higher stone prevalence in patients with ADPKD compared to unaffected family members. Compared to unaffected family members, patients with ADPKD had a higher prevalence of kidney stones (6 cross-sectional studies; unadjusted prevalence ratio: 1.8; 95% confidence interval: 1.3 to 2.6; P = .0007; test for heterogeneity: I2 = 0%, P = .8).
Limitations:
Studies were limited to articles published in English.
Conclusions:
The prevalence of kidney stones and stone interventions in adults with ADPKD remains uncertain. Future studies of higher methodological quality are needed to better characterize the incidence and prevalence of kidney stones in patients with ADPKD.
Trial registration:
We did not register the protocol for this systematic review.
What was known before
It is uncertain how often patients with autosomal dominant polycystic kidney disease (ADPKD) develop kidney stones.
What this adds
This review summarized the results of 49 studies. The prevalence of kidney stones reported in the literature ranged between 3 and 59%, and the prevalence of stone intervention ranged from 1 to 8% in patients with ADPKD. The quality of published literature was poor, and no study clearly reported stone incidence in ADPKD. This review calls for better studies to be conducted in the future.
Introduction
Autosomal dominant polycystic kidney disease (ADPKD) is the most commonly inherited kidney disease and is characterized by focal cyst development in both kidneys. 1 In early stages of ADPKD, the cysts cause structural deformation to the kidney and damage adjacent nephrons, but overall kidney function is maintained by compensatory hyperfiltration of functioning nephrons.2,3 As the number and size of cysts increase progressively, more nephrons become damaged, and overall kidney function starts to decline. 4 By the age of 55 years, about half of the patients reach end-stage kidney disease (ESKD) and require kidney transplantation or dialysis to sustain life.5,6
End-stage kidney disease is not the only kidney manifestation of ADPKD. Previous studies suggest that kidney stones are more prevalent in patients with ADPKD compared to the general population; however, there remains uncertainty about the incidence and prevalence of kidney stone in patients with ADPKD.7-12 Kidney stones in patients with ADPKD are associated with significant morbidity. For example, stones are a significant determinant of pain and may accelerate disease progression to ESKD in patients with ADPKD.13,14
We conducted this systematic review to critically appraise and summarize studies which reported the incidence and prevalence of kidney stones and stone interventions in patients with ADPKD. This encompassed studies which also included patients without ADPKD as a comparator.
Methods
Design and Study Selection
We conducted this systematic review using a pre-specified protocol not previously published but detailed below and report this review according to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement (Supplementary Table S1). 15
The following studies met our eligibility criteria for review: (1) published English full-text articles and conference proceedings; (2) any study design (eg, cross-sectional or cohort study); (3) mean age of studied population 18 years or older; (4) study populations not solely restricted to patients with ESKD; (5) reported prevalence or incidence of stones; and (6) studies published any time after 1970 (the resolution of imaging modalities in older studies would be different from current ones). In some studies, patients without ADPKD were included as a comparator to patients with ADPKD, and in such cases, we abstracted information on both groups of patients.
Identifying Relevant Articles
We performed a comprehensive search of bibliographic databases from 1970 to February 2019 (MEDLINE, EMBASE, Web of Science, BIOSIS Preview, and CINAHL) to identify all relevant journal articles and conference proceedings (detailed in Supplementary Table S2). To identify further relevant articles, we also used the “cited by” function on Web of Science and Google Scholar and “related article” function on Google Scholar and “similar article” function on PubMed to identify other relevant articles. We also reviewed the reference lists of all relevant articles.
Two reviewers (V.K. and G.G.) independently removed duplicates and rated the title and abstract of each citation as “relevant,” “possibly relevant” or “not relevant.” We then retrieved the full text of “relevant” and “possibly relevant” articles to assess study eligibility. The 2 reviewers resolved any disagreement through discussion and consensus.
Data Abstraction
Two reviewers (V.K. and G.G.) independently abstracted data from all included articles, recorded the data on the standardized abstraction form (Supplementary Table S3), and resolved any disagreements through discussion, or with the help of a third reviewer (D.M.N.). We collected data on study characteristics, patient characteristics, incidence or prevalence of stones, and stone characteristics. We abstracted the prevalence of stone intervention from the included studies that reported it.
We assessed the methodological quality of included studies using a modified Downs and Black checklist (Supplementary Table S4). We assigned all included studies a score between 0 and 22 based on our modified checklist with a higher score indicating a greater quality. 16
Data Analysis
We used a Fischer Exact test for studies with controls that did not statistically compare the prevalence of stones between patients with ADPKD and controls. We also calculated the prevalence ratio of kidney stones for each of the studies with controls using Cochrane Review Manager 5.3. We assessed for heterogeneity across all studies using the I2 test. I2 values below 25%, between 25% and 75%, and above 75% correspond to low, moderate, and high levels of heterogeneity, respectively. We conducted a meta-analysis to combine the results if I2 was less than 75%. We calculated the meta-analyzed prevalence ratio estimates for kidney stones using a random effects model and Cochrane Review Manager 5.3.
Results
Study Selection
A schematic diagram of the study selection process is presented in Figure 1. Our search yielded 1812 citations, and we identified 29 eligible articles that met our eligibility criteria. We identified an additional 20 eligible articles through our further search strategy described above, which resulted in a total of 49 eligible articles (a total of 9396 patients with ADPKD).7-12,14,17-58 The chance-corrected agreement between 2 independent reviewers for full-text eligibility was excellent (κ = 0.86).
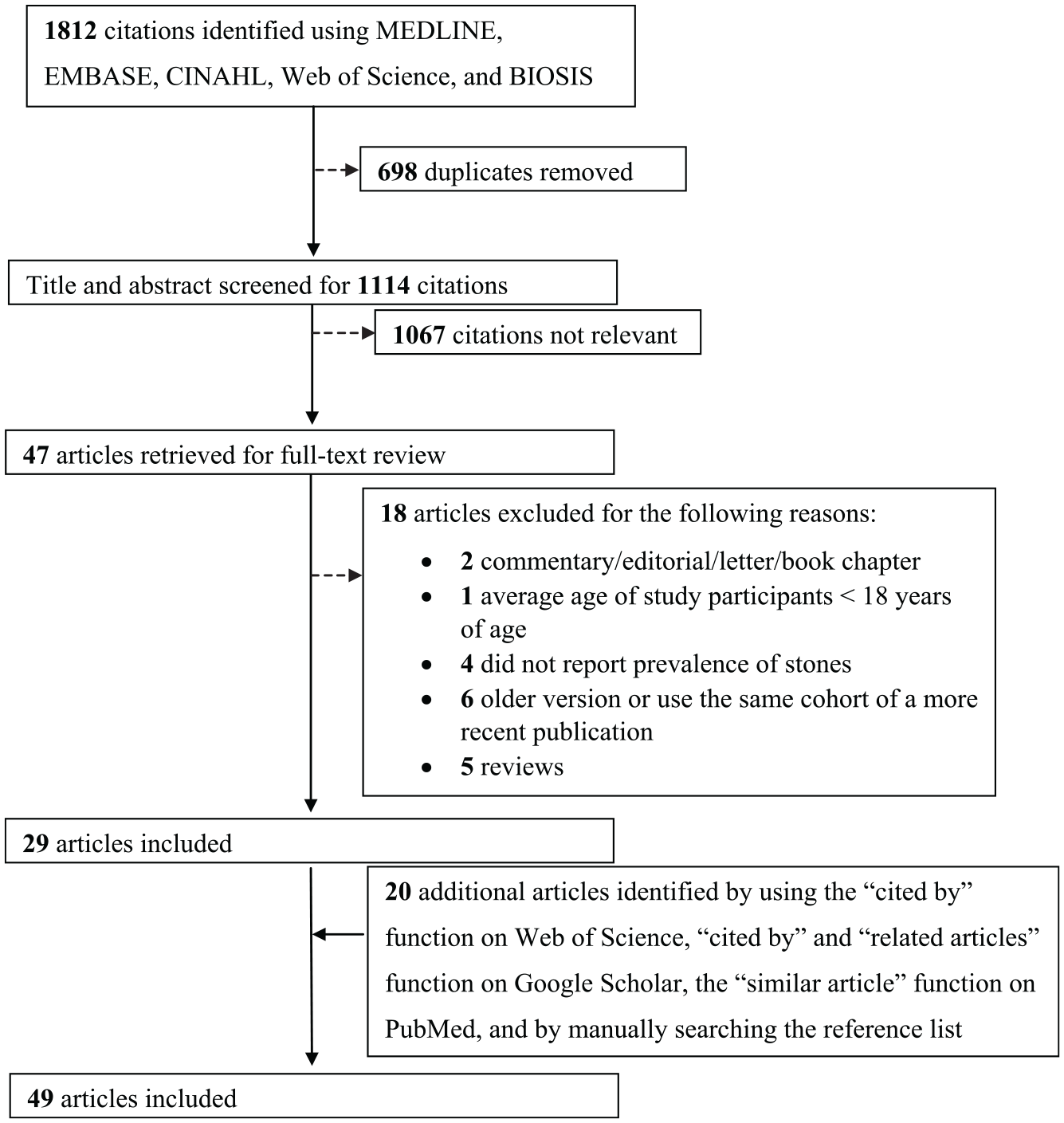
Study selection.
Description of Included Studies
The characteristics of included studies are summarized in Table 1. The 49 eligible studies were published between 1977 and 2019, and the majority of the studies were conducted in Turkey (7 studies) followed by the United States (6 studies), Albania (5 studies), Brazil (3 studies), India (3 studies), Spain (3 studies), Canada (2 studies), Italy (2 studies), and Japan (2 studies). A single study was conducted in Bulgaria, China, Cyprus, Greece, Ireland, Korea, Pakistan, Philippines, Republic of Macedonia, Saudi Arabia, Senegal, Taiwan, Tunisia, and the United Kingdom, and one was a multinational study. The country where the study was conducted was unknown for one study. The number of centers participating in a study was unclear in 19 of 49 studies; of the remainder, 21 studies were single center and 9 were multicenter. Among the 49 included studies, 12 were cohort studies, 33 were cross-sectional studies, and the study design was unclear for 4 studies.
Study Characteristics.

Note. ADPKD = autosomal dominant polycystic kidney disease; NR = not reported; N/A = not applicable; U/S = ultrasound; CT = computed tomography; Fam Hx = family history; PKD = polycystic kidney disease.
A modified Downs and Black checklist was used to assess the methodological quality of each included study. The methods quality score ranged between 0 and 22 with higher scores indicating higher quality.
Data were abstracted and methodological quality was assessed for the portion of the multicomponent study that reported the prevalence of stones.
Patient Population
The sample size of patients with ADPKD ranged from 30 to 1139 (Table 2). The mean age of patients with ADPKD ranged from 26 to 61 years, 35% to 71% of the patients with ADPKD were male, up to 51% developed end-stage renal disease (ESRD), 5% to 88% were hypertensive, and 1% to 73% experienced at least one prior urinary tract infection (UTI; Table 2).
Patient Characteristics.
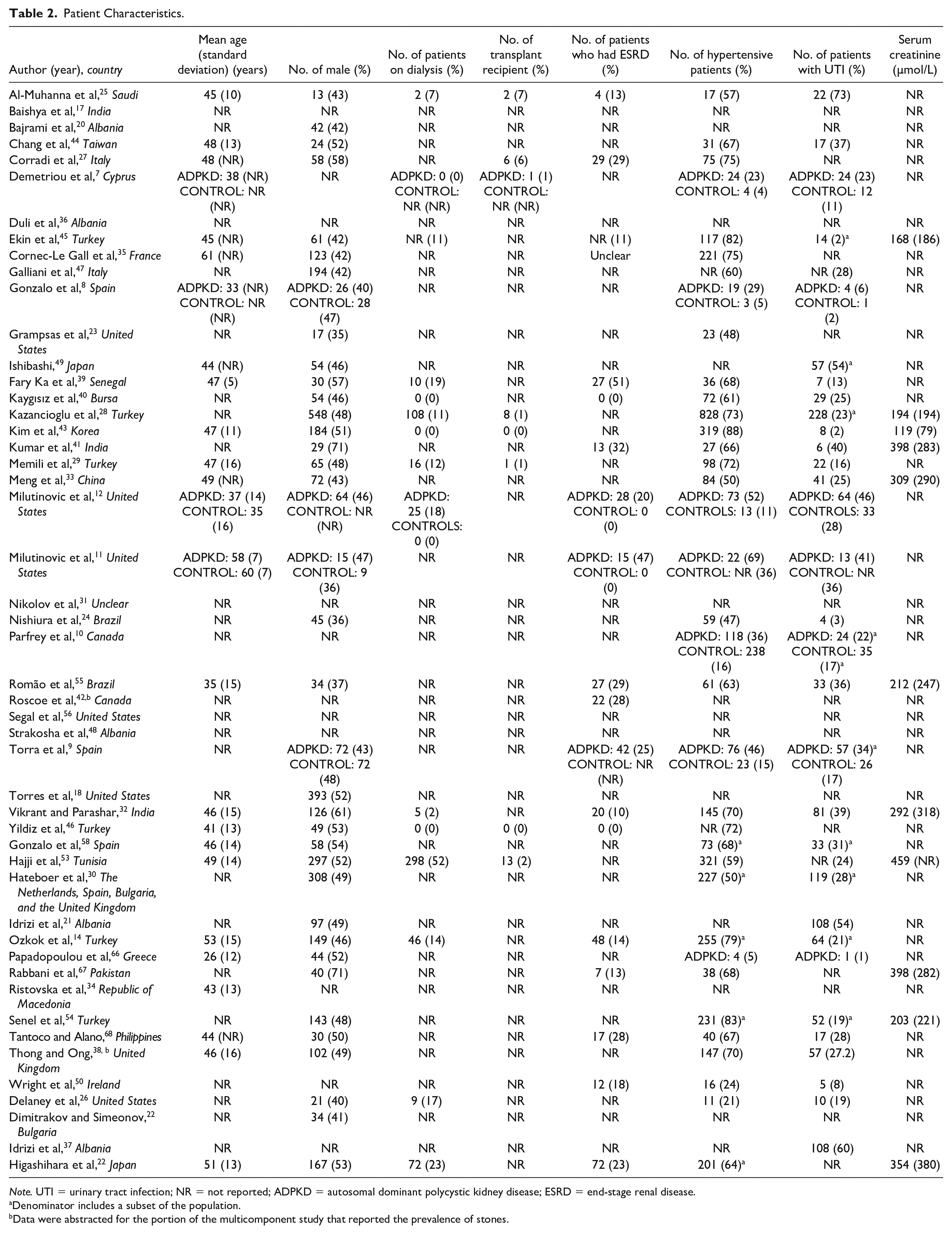
Note. UTI = urinary tract infection; NR = not reported; ADPKD = autosomal dominant polycystic kidney disease; ESRD = end-stage renal disease.
Denominator includes a subset of the population.
Data were abstracted for the portion of the multicomponent study that reported the prevalence of stones.
Six studies compared the prevalence of stones in patients with ADPKD to unaffected family members as controls.7-12 The mean age of controls ranged from 35 to 60 years, 36% to 48% of the controls were male, 4% to 36% were hypertensive, and 2% to 36% experienced a prior UTI (Table 2).
Quality Assessment of Studies
The methodological quality of the studies was limited as the methods quality score ranged from 2 to 14 out of 22 (where higher scores indicate higher methodological quality).
The internal validity of studies’ results is affected by the definition of the exposure being investigated and the outcome of interest. Of the 49 studies, 29 specified the definition for ADPKD. Patients with ADPKD were identified using Ravine criteria in 6 studies, Ravine criteria or another additional criterion such as family history and liver cysts in 3 studies, Pei criteria in 3 studies, Pei criteria and an additional criterion in 2 studies, at least 5 cysts in each kidney in 3 studies, and other criteria in the remaining 13 studies; the definition for ADPKD was unclear or not reported in the remaining 19 studies. Ravine and Pei criteria to diagnose ADPKD are summarized in Supplementary Table S5 and Table S6, respectively.59,60 Some studies used a definition different from the most accepted diagnostic criteria at the time the study was published. For example, Ekin et al 45 and Kazancioglu et al 28 defined patients with at least 5 cysts in each kidney as patients with ADPKD, although Pei criteria were the most commonly used diagnostic criteria for ADPKD during the time period in which the studies were conducted.28,45
Thirty of the 49 studies described how they identified patients with stones, while the remaining 19 studies did not. Among the 30 studies that specified how the stones were detected, 3 studies relied on patient self-report of a history of stones, 14 solely relied on radiological evidence of stone, and 13 studies relied on combination of radiological evidence of stone and at least one other criterion (ie, stone passage and recovery, surgical removal of stone and self-report of stone). Among the 27 of the 30 studies that used radiological evidence of stones as one of their diagnostic criteria, 9 reviewed historic imaging, 10 reviewed recent imaging, and the nature of considered imaging was unclear in 8 studies. Eight of the 27 studies thoroughly described what they were looking for on the radiological image to identify stones. Among the 5 studies that reported asymptomatic stones, the percentage of patients ranged between 1% and 68%.17,18,21,37,48
The setting and source population from which the samples are recruited affects the study generalizability. For 21 of the studies, the setting or population from which the sample was recruited from was unclear or not reported. Patients were recruited from hospitals in 18 studies, outpatient clinics in 7 studies, solely from an inpatient setting in 1 study, an outpatient ADPKD speciality clinic in 1 study, and from both an inpatient and outpatient setting for 1 study. It is unclear if patients were recruited from an inpatient or outpatient setting for 20 studies and setting was not reported for one study.
Six of the 49 studies compared the prevalence of stones in patients with ADPKD to controls, which were unaffected family members. All of these studies were cross-sectional. Only 2 of the 6 studies statistically compared the prevalence of stones in patients with ADPKD to controls. Both of these studies used univariate analyses and did not adjust for any confounders.
Prevalence and Characteristics of Stones and Prevalence of Stone Intervention
In patients with ADPKD, the prevalence of stones ranged between 3% and 59% (Table 3). Of those patients with stones, 2% to 47% underwent at least one stone intervention. Urinary tract infections and flank pain were the predominant precursor to diagnosis of stones in patients with ADPKD.17,21,24,37,40,48 In most patients, stones were solely located in the renal calyces.17,18 Most stones were composed of uric acid according to 6 studies7,18,20,21,37,48 and oxalate according to 2 studies (Table 4).22,26
Prevalence of Stones and Stone Intervention in Patients With ADPKD and Controls.
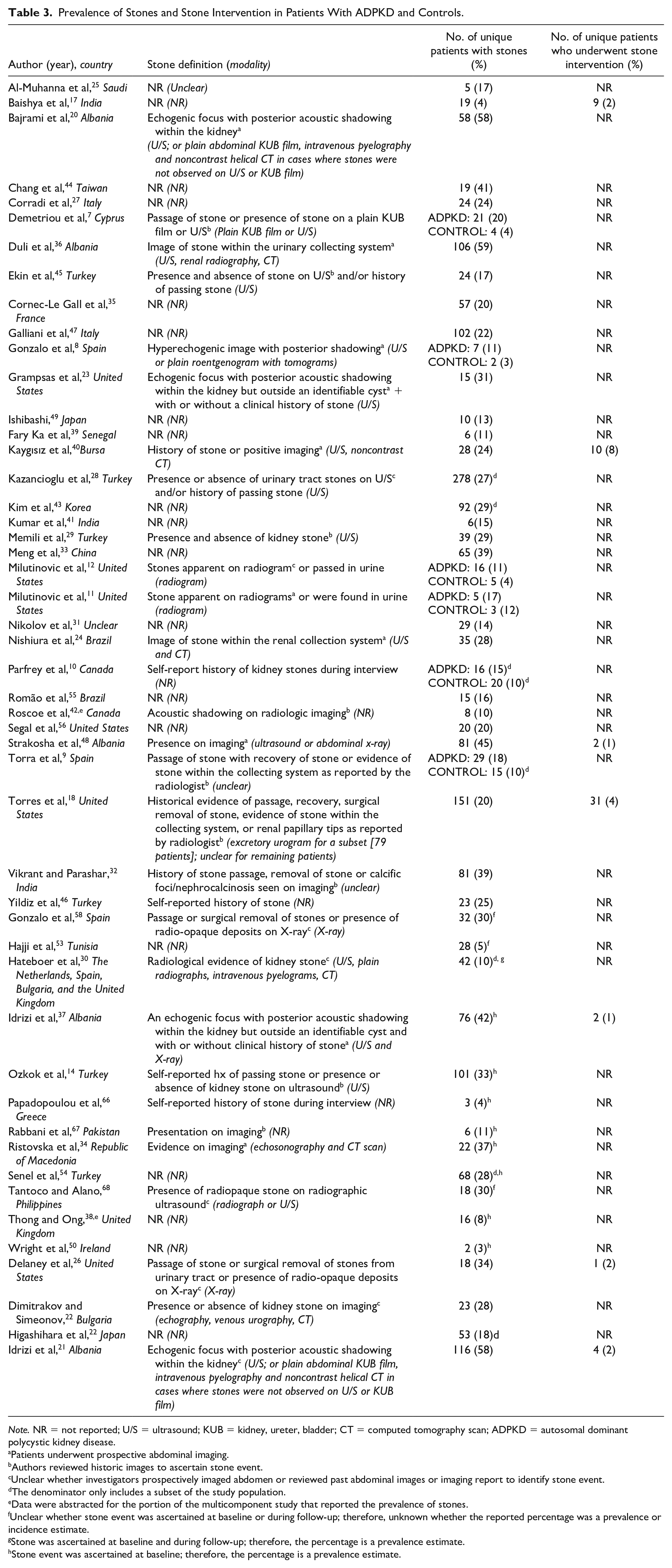
Note. NR = not reported; U/S = ultrasound; KUB = kidney, ureter, bladder; CT = computed tomography scan; ADPKD = autosomal dominant polycystic kidney disease.
Patients underwent prospective abdominal imaging.
Authors reviewed historic images to ascertain stone event.
Unclear whether investigators prospectively imaged abdomen or reviewed past abdominal images or imaging report to identify stone event.
The denominator only includes a subset of the study population.
Data were abstracted for the portion of the multicomponent study that reported the prevalence of stones.
Unclear whether stone event was ascertained at baseline or during follow-up; therefore, unknown whether the reported percentage was a prevalence or incidence estimate.
Stone was ascertained at baseline and during follow-up; therefore, the percentage is a prevalence estimate.
Stone event was ascertained at baseline; therefore, the percentage is a prevalence estimate.
Symptoms and Characteristics of Stones.
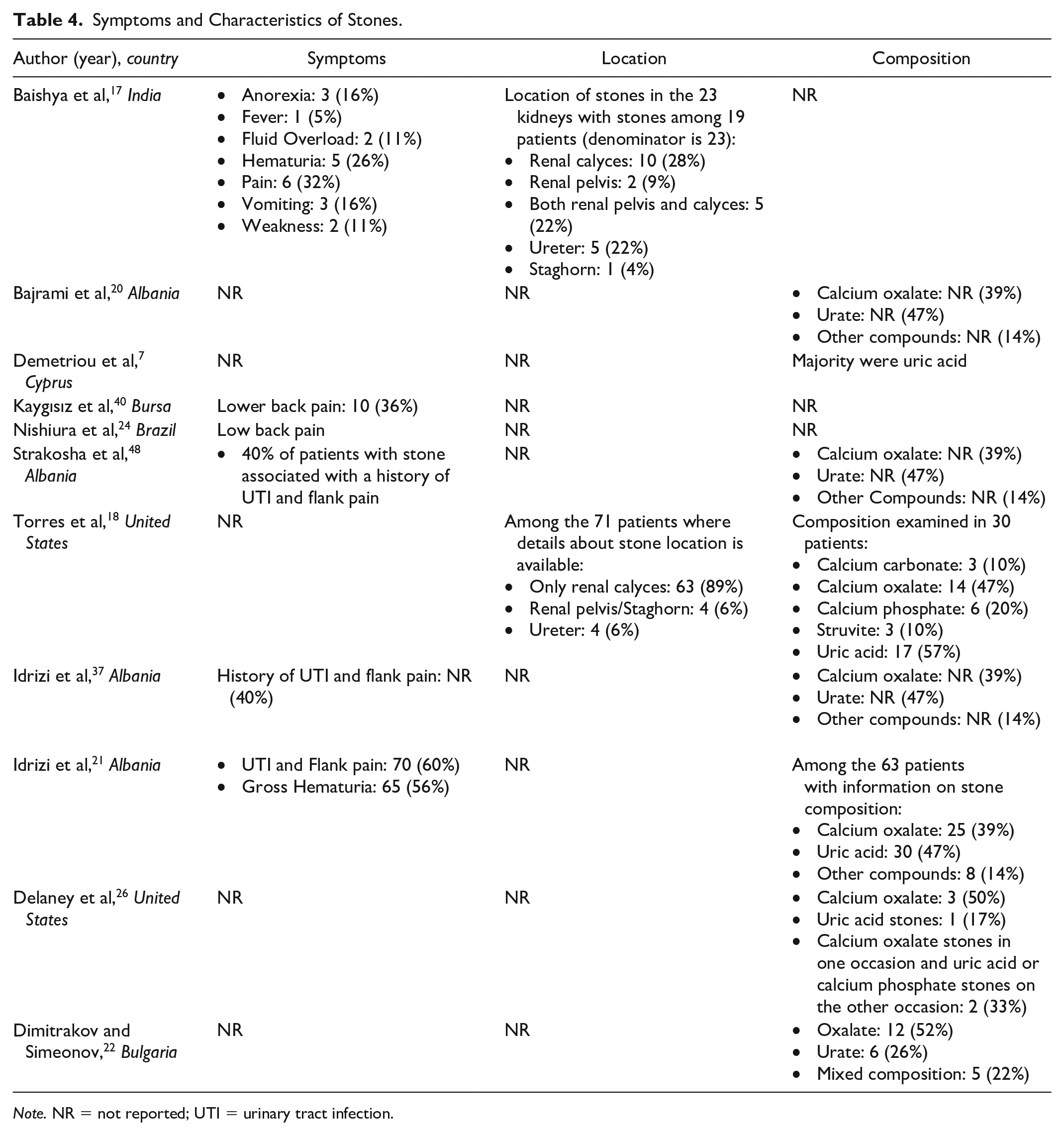
Note. NR = not reported; UTI = urinary tract infection.
The prevalence of stones ranged from 3% to 12% in family members confirmed not to be affected with ADPKD (Table 3). None of the studies described the characteristics of stones in unaffected family members. All 6 studies that compared the prevalence of stones in patients with and without ADPKD reported stones were more prevalent in patients with ADPKD; however, 4 studies did not statistically analyze the prevalence of stones between the 2 groups, and the remaining 2 studies found no statistical difference. When we statistically compared the prevalence of stones in patients with ADPKD to unaffected family members in the 4 studies that did not conduct any statistical analyses, we found that only one out of the 4 studies found a significant difference. Meta-analysis of the calculated prevalence ratios across 6 cross-sectional studies show that patients with ADPKD had a higher prevalence of kidney stones compared to unaffected family members (unadjusted prevalence ratio: 1.8, 95% confidence interval: 1.3 to 2.6, P = .0007; test for heterogeneity: I2 = 0%, P = .8; Figure 2).

Calculated unadjusted prevalence ratio of stones in patients with autosomal dominant polycystic kidney disease compared to unaffected family members.
Six studies reported the prevalence of stone intervention in patients with ADPKD, which ranged between 1% and 8% (Table 3). None of the studies with controls reported the prevalence of stone intervention in unaffected family members.
Stone Incidence
No study clearly reported the incidence of kidney stones and the incidence of stone intervention in patients with ADPKD. Most cohort studies included in this review assessed kidney stones at cohort entry and not during follow-up. Whether the reported percentage was a prevalence or incidence estimate was unclear for 3 of the included cohort studies.
Discussion
Many popular educational materials and clinical practice guidelines state that kidney stones are common in patients with ADPKD, and its prevalence may be 5 to 10 times higher than the general population.61,62 This make clinical sense based on our knowledge of the pathophysiology of ADPKD; the kidney cysts in patients with ADPKD lead to urinary stasis which promotes stone formation. 23 Our review of the literature, however, indicates that the evidence to support these assertions is weak and illuminates several knowledge gaps about the clinical epidemiology of stones in ADPKD. No study has clearly reported the incidence of stones in ADPKD. Prevalence estimates in ADPKD varied widely ranging from 3% to 59% for kidney stones and from 1% to 8% for stone interventions. Urinary tract infections and flank pain were the predominant precursors to diagnosis of stones; however, UTI and flank pain are not specific to stones and are also manifestations of ADPKD independent of stones. It is likely that UTI and flank pain were associated with ADPKD itself rather than stone because most of the stones in ADPKD were located in the renal calyces where they would be less likely to be symptomatic. Uric acid stones are the most prevalent stone composition in patients with ADPKD. The wide-ranging prevalence estimates along with the discovery that no published studies clearly reported stone incidence confirm that how often patients with ADPKD develop kidney stones remains uncertain.
There are several reasons why prevalence estimates of stones varied drastically across studies. These include inconsistent stone definitions, different distributions of stone risk factors, potential recall bias in studies that relied on patient self-report to identify stone events, and relying on past imaging reports done for reasons other than stone identification. Self-report is particularly problematic because the symptoms of flank pain and hematuria are common with ADPKD in the absence of stone disease. Patients with ADPKD may be more likely to undergo renal imaging, which would lead to over-detection of potentially clinically insignificant stones which may also exist undetected in the general population. The variability in imaging modalities used across studies and even between patients in the same study may also explain the variable prevalence estimates across studies. For example, computed tomography (CT) is a more sensitive method of stone detection than ultrasound and would provide a more accurate estimate of stone prevalence.63,64 There are many in the current literature. Most of the studies published to date on stones in ADPKD were conducted in a single center and are of poor methodological quality. Additionally, only 6 studies compared the prevalence of stones in patients with ADPKD to controls.7-12 Among these 6 studies, only 2 statistically compared the prevalence of stones between the 2 groups,9,10 and none of these studies adjusted for confounders.7-12 Additionally, not all patients with ADPKD were hospitalized; as a result, prevalence estimates obtained from patients recruited from an inpatient setting must be generalized to the broader ADPKD population with caution. Similarly, the prevalence estimates obtained from patients recruited from an outpatient speciality clinic must also be generalized to the broader ADPKD population with caution due to increased surveillance. Also, only 8 of 49 of the included studies described the composition of stones in patients with ADPKD; none of the 8 studies compared the composition of stones in patients with ADPKD to patients without ADPKD.
This review serves as a call to action for better research in this field. We recommend conducting large, multicenter studies that compare the risk of stones and risk of stone intervention between a representative population of ADPKD and controls to better characterize the magnitude of kidney stone and stone intervention risk in patients with ADPKD. We also recommend that such studies adjust for important confounders, such as hypertension, to better characterize the true association between ADPKD and kidney stones and stone intervention. Imaging tests are much more advanced, widespread, and frequent over time; this may lead to the possibility of detecting stones in ADPKD that may not be clinically relevant. Examining risk of kidney stone diagnosis and kidney stones that require intervention separately would provide insight into whether there is a potentially higher burden of asymptomatic stone that were detected incidentally on imaging. More reliable estimates of the magnitude of risk of stones and stone intervention would provide insight into clinical management practices and help patients with ADPKD and their physicians better prognosticate. If patients with ADPKD are truly at higher risk for kidney stones, then nephrologists may want to consider preventative measures for kidney stones. For example, if patients with ADPKD are at higher risk of kidney stones and hypocitraturia, then nephrologists may want to screen for hypocitraturia and treat patients with potassium citrate. Nephrologists may also want to consider treating large cysts that obstruct the urinary system and cause urinary stasis. Preventing stone formation would alleviate pain due to kidney stones and potentially slow down disease progression in patients with ADPKD. We also recommend comparing the composition of stones observed in patients with ADPKD compared to patients without ADPKD. New medications used in ADPKD, such as vasopressin receptor 2 antagonists, may alter the urine composition and change the types of renal stones that these patients get. Future ADPKD-specific risk factors, such as mutation type, of kidney stone studies may help identify patients at high risk for stones and provide further insight into the pathophysiology of kidney stones in patients with ADPKD.
Our study is the first to systematically review and summarize the prevalence of stones in patients with ADPKD. Unlike past narrative reviews, we used a comprehensive search strategy across 6 different databases, and 2 reviewers independently screened all citations retrieved from the search strategy to identify all relevant articles. We also conducted this review in accordance with an a priori protocol and published guidelines for systematic reviews. Two independent reviewers abstracted the data to minimize human error and bias.
There are some limitations inherent in our systematic review. First, we only included original journal articles and conference proceedings published in English. However, studies show that language-restricted meta-analysis does not lead to biased estimates. 65 Second, the definitions for ADPKD and stones varied across studies; therefore, the pooled estimate must be interpreted with caution.
Conclusions
Our systematic review highlights that there is poor consensus on the prevalence of stones in patients with ADPKD. A more methodologically robust study is needed to better characterize and understand the magnitude of risk of stones and stone intervention in patients with ADPKD. This information can help patients with ADPKD and physicians with their prognostication and might inform the use of interventions to reduce the risk of stones.
Supplemental Material
SupplementaryMaterials_PrevalenceOfStone_CJKHD – Supplemental material for Stone Prevalence in Autosomal Dominant Polycystic Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, SupplementaryMaterials_PrevalenceOfStone_CJKHD for Stone Prevalence in Autosomal Dominant Polycystic Kidney Disease: A Systematic Review and Meta-Analysis by Vinusha Kalatharan, Gary Grewal, Danielle M Nash, Blayne Welk, Sisira Sarma, York Pei and Amit X. Garg in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We thank John Costello for reviewing the search strategy developed by V.K.
Ethics Approval and Consent to Participate
Since this systematic review and meta-analysis did not involve human investigation, ethics approval was not required. Consent to participate was not required as our study did not rely on human subjects and reviewed the existing literature.
Consent for Publication
All authors have consented for publication.
Availability of Data and Materials
All data is presented in the original article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. York Pei served as an expert consultant on drug development (Otsuka, Pfizer, and Genzyme/Sanofi) related to autosomal dominant polycystic kidney disease. All other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ICES Kidney, Dialysis, and Transplantation Program provided funding for this study. Vinusha Kalatharan’s training was supported by the Canadian Institutes of Health Research Doctoral Scholarship and the Doctoral Scholarship from the KRESCENT Program (a national kidney research training partnership of the Kidney Foundation of Canada, the Canadian Society of Nephrology, and the Canadian Institutes of Health Research). Dr. Amit Garg was supported by the Dr. Adam Linton Chair in Kidney Health Analytics and a Clinician Investigator Award from the Canadian Institutes of Health Research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
