Abstract
Heart failure is a complex disease with poor outcome. This complexity may prevent care providers from covering all aspects of care. This could not only be relevant for individual patient care, but also for care organisation. Disease management programmes applying a multidisciplinary approach are recommended to improve heart failure care. However, there is a scarcity of research considering how disease management programme perform, in what form they should be offered, and what care and support patients and care providers would benefit most. Therefore, the Improving kNowledge Transfer to Efficaciously Raise the level of Contemporary Treatment in Heart Failure (INTERACT-in-HF) study aims to explore the current processes of heart failure care and to identify factors that may facilitate and factors that may hamper heart failure care and guideline adherence. Within a cross-sectional mixed method design in three regions of the North-West part of Europe, patients (n = 88) and their care providers (n = 59) were interviewed. Prior to the in-depth interviews, patients were asked to complete three questionnaires: The Dutch Heart Failure Knowledge scale, The European Heart Failure Self-care Behaviour Scale and The global health status and social economic status. In parallel, retrospective data based on records from these (n = 88) and additional patients (n = 82) are reviewed. All interviews were audiotaped and transcribed verbatim for analysis.
Keywords
Introduction
Despite significant advances in therapy, heart failure (HF) is a highly prevalent chronic disorder with poor prognosis and high socio-economic impact.1–4 This impact is likely to further increase as the prevalence of HF in North-West European countries is expected to further increase from approximately 2% at present to 3% by 2025. 5
Because HF is a complex disease often accompanied by comorbidities, disease management programmes (DMPs) have been introduced.6–8 They include a multidisciplinary approach to manage patients with HF and encompass different components as highlighted in recent guidelines. 9 If implemented comprehensively, DMPs offer accessible and efficient provision of care, 10 which may result in benefits on both quality and cost-effectiveness of care.11–16 Therefore, guidelines 17 advice to organise HF care in multidisciplinary teams, including nurses, cardiologists and general practitioners (GPs).17,18 In addition to healthcare providers, patient involvement is an important part of DMPs.12,19,20 However, DMPs are not uniformly implemented in Europe. In part, this may be related to the fact that DMPs do not refer to one single, clearly defined programme, but to an overall concept of delivering care. Thus, care is typically adapted to local needs and national healthcare systems, and the interplay of structure, process and outcomes may vary significantly between different regions. 21 In clinical practice, it is largely unknown how care is organised. 22 In particular, the roles of care providers and inter-professional interactions are often indistinct17,18,23,24 and patient perspectives regarding optimal HF care have been hardly considered. 25 Moreover, care providers are sometimes not capable to consider all aspects of HF, given the complexity of the disease, 26 resulting in inadequate guideline adherence.26,27 Finally, it is unknown if the way how national healthcare is organised, may influence HF care and to what extent HF care differs between countries.
Therefore, the INTERACT-in-HF (Improving kNowledge Transfer to Efficaciously Raise the level of Contemporary Treatment in Heart Failure) study investigated current practice in chronic HF care from perspectives of both patients and care providers. We hypothesised that there is variation regarding all aspects mentioned below between individuals, but also between the participating regions and between patients and care providers. The aim of the study was to explore processes in HF care to identify factors that may facilitate and factors that may hamper HF care. For this purpose, a mixed methods study design was set up in three neighbouring regions in the North-West part of Europe. Particular attention was paid to organisational aspects of HF care including the patients’ perspective, the performance of DMPs if present, interaction between care providers and patients as well as interaction between care providers themselves and the needs and experiences of both. More specifically, we investigated
applied diagnostics and therapeutics as compared to current guidelines; reasons of not confirming to guidelines; stakeholders and their role in HF care from different perspectives; the patient flow, information transfer and communication between patients and care providers; referrals and reasons for admission; the patients’ and healthcare providers’ view on quality of HF care and factors that foster or hinder good HF care; the impact of national healthcare in the Netherlands, Belgium and Germany on HF care.
The present article presents the protocol of the INTERACT-in-HF study including some preliminary results to give a clear overview over the purpose of this trial, the methods used and the subjects included.
Methods
Study design
An HF network of three North-West European regions, Maastricht (the Netherlands; approximately 1,22,461 inhabitants) Aachen (Germany; approximately 2,43,336 inhabitants) and Noorder-Kempen (Belgium; approximately 2,46,021 inhabitants), was set up. Within this network, data were collected from 88 HF patients in primary and secondary care and their care providers (i.e. cardiologists, GPs and (HF) nurses). A two-phase, cross-sectional, mixed methods design was used, encompassing both qualitative and quantitative methods (Figure 1).
Study timeline and mixed methods study design.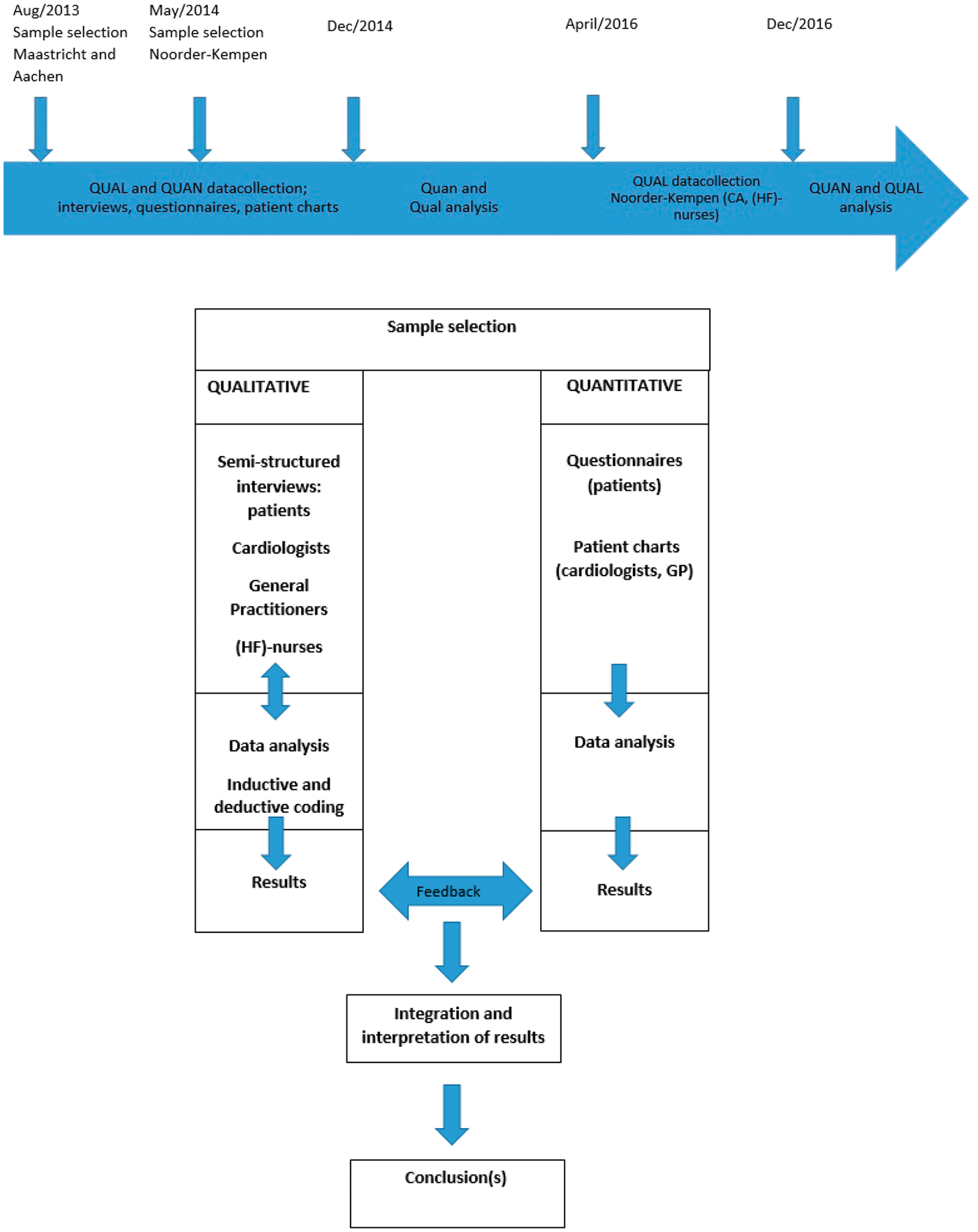
Application of different types of triangulation.

GPs: general practitioners; HF: heart failure; NWE: North West European.
Sample selection
Purposive sampling was used to include following stakeholders in HF care: patients, cardiologists, GPs and HF nurses. In order to avoid selection bias within the patient group, a selection of eligible patients was done by selecting the first, third, fifth, and seventh patient with HF out of the patient database of the participating cardiologists and GPs.
Based on the protocol of the study, less care providers than patients were included (Figure 2). When interviews revealed additional care providers relevant to HF care of individual patients they were added to the study sample.
Sample selection. (a) Maastricht, (b) Aachen and (c) Noorder-Kempen.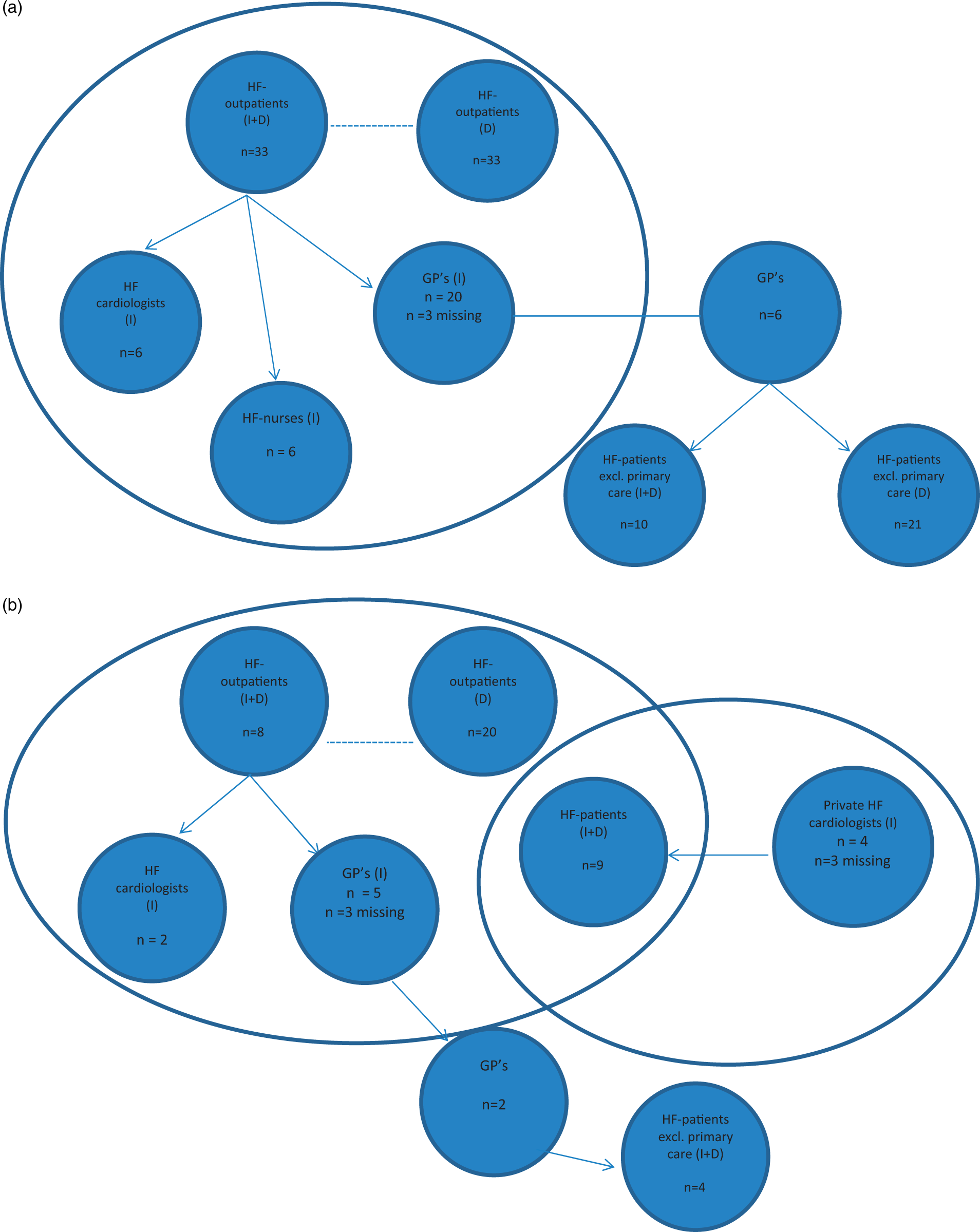
Maastricht and Aachen
Between August 2013 and December 2014, care providers from the outpatient HF clinics of both Maastricht University Medical Centre (The Netherlands) and University Hospital Aachen (Germany) were approached for participation. All agreed to participate and patients were selected as described above. These patients were asked for consent to participate and to allow interviews with their care providers. Subsequently, patients’ cardiologists, GPs and HF nurses were interviewed after providing consent for participation. At the HF centre in Aachen, no specialised HF nurses were employed. To include HF patients exclusively treated in primary care, participating GPs were asked to deliver a list of patients with the main diagnosis of HF that were treated only by them. From this list, the sample was selected as described (Figures 1 and 2(a) and (b)).
Noorder-Kempen
Patients in Noorder-Kempen were initially selected at primary care level due to logistic reasons. GPs and members of the regional GP-association were approached for participation in this study. In order to have sufficient participation, the snowball sampling method was used. 33 Interviews with patients and GP’s were preformed between May and November 2014. Cardiologists and (HF) nurses have been interviewed between April and December 2016 (Figures 1 and 2(c)).
Inclusion and exclusion
Patients were included if they fulfilled the following criteria: HF diagnosis due to left ventricular dysfunction, irrespective of underlying diagnosis and left ventricular ejection fraction, age above 18 years and being capable to understand instructions. Except for heart transplantation and lack of informed consent, no exclusion criteria were applied.
Ethical considerations
The protocol was approved by the Ethics Committees of Maastricht University Medical Centre, the University of Antwerp and the University of Aachen. Participating patients and care providers provided written informed consent after receiving oral and written information. The study followed the principles according to the Declaration of Helsinki.
Measurements and data collection
Qualitative part: Interviews
Patients underwent a semi-structured interview, starting with the open question ‘How did your heart problems start?’ In response to this question, a number of upcoming issues in the patients’ narrative were discussed. Alongside emerging topics, a set of predefined topics and questions was addressed to all patients to address the objectives of this study (Appendix 1).
Care providers participated in semi-structured interviews about their experiences of HF care in general and in relation to their patient participating in the study. Interviews with care providers started with the open question ‘What does HF mean to you?’ The interview topics addressed understanding and knowledge of HF, use of guidelines, roles of care providers in HF care and communication (Appendix 2).
The topic list was developed after expert meetings with several HF-care stakeholders (patient organisation, GPs/GP networks, HF specialists, general cardiologists, internists, geriatrician, representatives of healthcare insurers and representatives of Dutch ministry of health). The topics were adjusted during the progress of the interviews. This so-called, iterative approach allows emerging themes and ideas to be included in subsequent interviews, but also altering the research methods and hypothesis as the study progresses. 33 Interviews were performed by trained research staff members (three male and six female) with different professional backgrounds. Interviews were performed by KB (F, 38 years, public health nurse, master in health sciences), RS (M, 40 years, Phsyiotherapist and Master of sience in Engenering Technology), MM (M, 50 years, Master of Sience in Engineering Technology and Post graduate in Relation and Communication Sciences), SB (F, 31 years, MD), ZS (M, 28 years, medical student), BON (F, 24 years, medical student), DR (F, 27 years, MD), CR (F; 25 years; MD student) and CH (F, 42 years, MD). CR and CH interviewed all German participants. CR is a native German speaker (German parents) living in the Netherlands since childhood.
Interviewers did not have a patient–care provider relationship and had no contact with the patient prior to the interview. In Maastricht and Aachen, most patient interviews were performed in the outpatient clinic. Due to mobility problems or patients’ preference, some interviews were performed at home. Because all Belgian patients were selected in primary care, interviews were conducted at home. In all participating countries, care providers were interviewed in their work environment. The average interview duration was 30 and 60 min for care providers and patients, respectively.
Dutch and Belgian interviews were coded separately. An attempt to merge the databases showed that there were too many codes and that it was impossible to merge because of the different ways of coding. Subsequently, all interviews were re-coded and codes were restrained from 6090 to 684 distributed over five levels. In case of disagreement, analysts deliberated until consensus was reached. Primarily the Dutch and Belgian codebooks were merged. After consensus about the German codes, all databases were merged.
Quantitative part: Questionnaires
Prior to the interviews, patients completed three questionnaires:
The Dutch Heart Failure Knowledge Scale (DHFKS), a 15-item, self-administered questionnaire (score 0–100), covers items concerning general HF knowledge, symptoms, symptom recognition and HF treatment.
34
The non-validated German version was used in Aachen.
35
The European Heart Failure Self-care Behaviour Scale (EHFScBS), a 12-item, self-administered questionnaire (score 0–100), addresses several items concerning self-care in HF behaviour. Every item scores on a Likert scale from 1 (completely agree) to 5 (completely disagree).
36
The global health status and social economic status (SES), a 20-item, self-developed, global, and socio-demographic questionnaire, was used to evaluate general understanding of HF. This questionnaire includes self-reported general health status, educational level, smoking status, physical activity status, employment, knowledge about HF and medication use.
Clinical characteristics
Data from patient charts were collected to determine patient characteristics. In addition, retrospective data of additional HF patients were collected to test if diagnostic and therapeutic processes of the interviewed patients were representative. The following clinical characteristics were extracted from patient charts: age, gender, cause of HF, other cardiovascular diseases, comorbidities, cardiovascular risk factors (diabetes, hypertension, hypercholesterolemia and smoking), healthcare utilisation within the last year, New York Heart Association class, weight and height, blood pressure, heart rate and diagnostics used: electrocardiogram (rhythm and QRS duration), echocardiography and laboratory findings. HF aetiology and comorbidities were only considered if registered in patient charts. Cardiac medication and changes over time were recorded. Medical charts were provided by the participating hospitals of Maastricht, Aachen and the GPs in Noorder-Kempen.
Analyses
Quantitative data are presented as frequencies (%), mean (SD) or median (IQR), as appropriate. The statistical program SPSS v 20.0 (IBM) was used for data analysis.
All interviews were audiotaped (Smartpen, Livescribe, Oakland, CA, USA), transcribed verbatim and analysed by the qualitative analysis software program NVivo 10. All interviews were coded independently by SB, LBD, KB and CR. An independent expert, MD, reviewed data extraction in NVivo. Areas upon which coding differed were reconsidered until consensus was achieved. 37
Results
Patient characteristics
Patient characteristics per region.
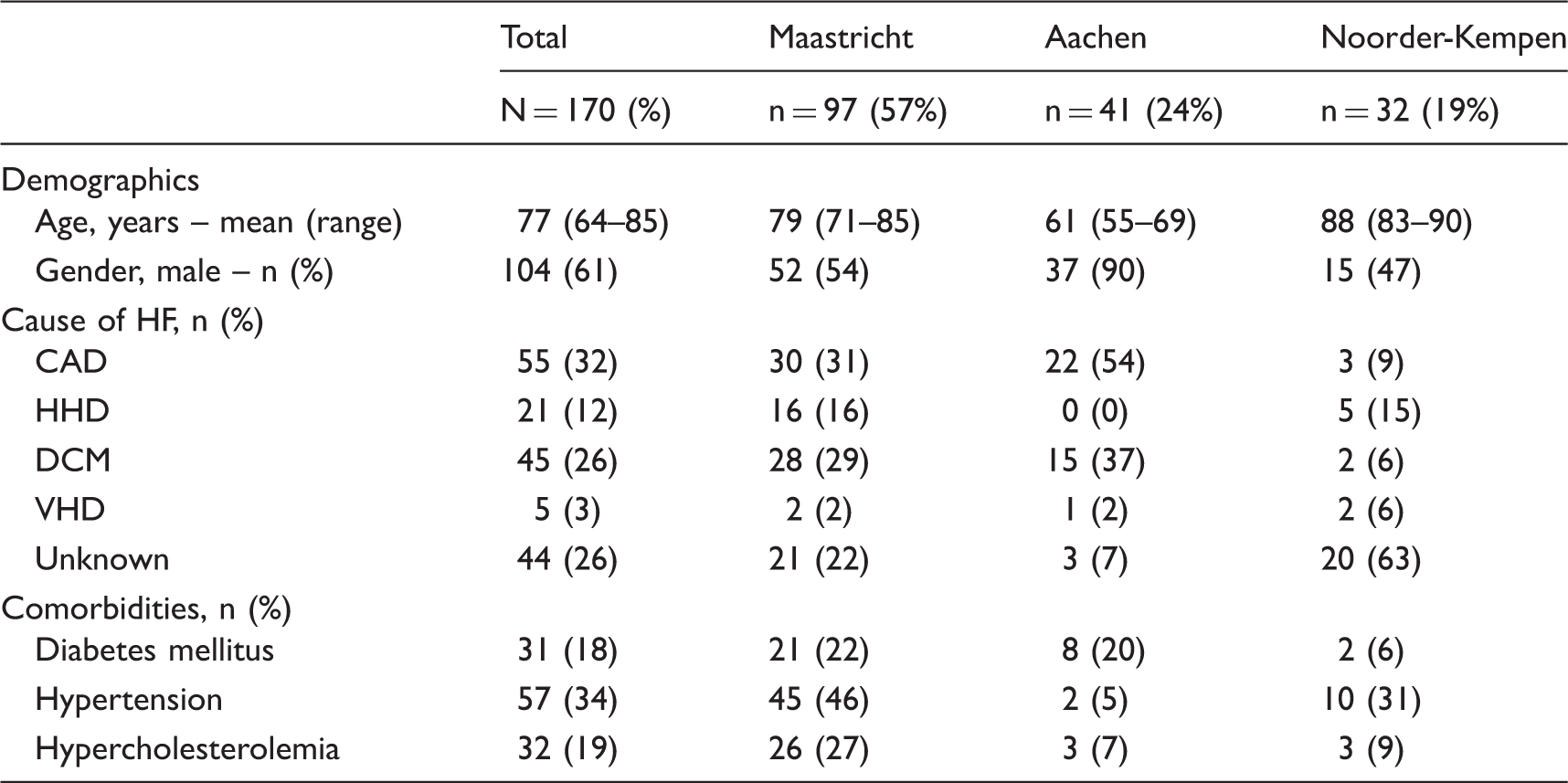
HF: heart failure; CAD: coronary artery disease; HHD: hypertensive heart disease; DCM: dilated cardiomyopathy; VHD: valvular heart disease.
Qualitative codebook
Framework of qualitative analyses.

Discussion
National and international guidelines are developed to define best practice in HF management. In order to achieve this, a multidisciplinary, patient-centred approach organised in DMP is suggested.15,17 Several initiatives have been initiated, yet implementation of DMP in Europe is still incomplete. 22 Although DMPs are structurally implemented in most centres in the Netherlands, this is not always the case in other European countries including Belgium and Germany. The INTERACT-in-HF study has been set up in three neighbouring North-West European regions because of these different, but comparable demographic features of included patients. Also, national organisation of healthcare differs between the participating regions.38,39 This allows us to get more insight into care processes, circumstances leading to successful implementation of guidelines and bottlenecks hindering optimal organisation of HF care.
This study included main stakeholders in the three regions in the North-West part of Europe. Underlying cause of HF was unknown in many patients suggesting differences in the diagnostic workup of patients in primary and secondary care, highlighting the need for a study such as this one. The attempt of displaying a comprehensive picture of HF management is crucial to understand current practice and its challenges. Several studies about HF-care organisation ignore the roles and responsibilities of different care providers possibly due to the fact that roles are not well defined in guidelines. This may, however, cause organisational and/or communicational uncertainties in daily practice.17,18,20,23,24,40–42
How to assess care process in chronic diseases?
Currently, there is no standardised approach to obtain a precise overview of the standard of HF care. 15 According to Bowling, 43 “the most persuasive evidence comes through a triangulation of measurement processes, as well as through minimising the error contained in each instrument.”43–45 Therefore, we used this mixed methods approach. This implies the use of multiple, complementary, measurement strategies to examine complex clinical problems.44,45 By using qualitative research, the study aims to gather an in-depth understanding of human behaviour and its governing reasons. More importantly, the qualitative method investigates the why and how of decision making, not just what, where and when. The combination with quantitative data improves validity as they provide information about the representativeness of patients included in the study.
During the initial coding process, we used an inductive approach to avoid relevant variables of interest remaining undetected. The most fundamental characteristic of an inductive approach is the holistic view. Thus, information provided in interviews was approached independently and without any prior knowledge of concepts, thus avoiding bias due to own experiences. Other characteristics of an inductive approach are purposive sampling and an iterative approach.45–49 Purposive sampling aims to include subjects with an additional value to answer the research question, 33 whereas the iterative approach is useful to investigate processes. The inductive approach generates validity because of its closeness to the truth.48,50 Finally, intercoder reliability is used to determine the coders’ consistency. Therefore, the approach used in this study will provide comprehensive insight into HF care in the three regions to test our hypothesis of significant variation in care between regions, and also between individuals.
To the best of our knowledge, this is the first study, using a selection of participants within the purposive sample of patients comparing three different countries. This combined approach providing a large amount of data was used to achieve a representative description of HF care in the investigated regions. In order to maintain the benefits of the inductive approach and ensure reliability,48,50 the deductive codebook was based on the data gathered from the inductive approach. Deduction implies a focus on objective analysis of data and systemises qualitative research, thereby making it more reliable. The codebook was developed by three researchers with different professional backgrounds to eliminate selection of variables of interest, i.e. one MD and two nurses. One of the nurses was experienced in mixed methods studies, the MD had experience in HF and the other nurse in organisation of care. This allows objective description of important aspects of HF care in the different regions, which is a prerequisite to define properly hindering and supporting factors of good quality care. Altogether, this methodology is capable to provide a broad and reasonably unbiased insight into the organisation of HF care.
Strengths and limitations
The strength of this study is the inclusion of a broad study population with sufficient number of both patients and care providers in primary and secondary care.
Compared with other studies, triangulation of data collection and analysis is an advantage. Although interviews took place in three different European regions, all interviews were peer-reviewed by the same investigators. Furthermore, they were coded independently and results were merged after coding. To minimise bias by interviewers, predefined questionnaires were used by means of a semi-structured interview. To improve validity, the interview topic list was the same for all countries. Mastering of the German and Dutch language by the German interviewer prevented for bias due to translation. Thus, these measures may reduce the risk of bias significantly, whereas the combined use of inductive and deductive methodology improves reliability. Still, potential bias due to use of different interviewers, data analysts and languages cannot be completely excluded and needs to be considered when interpreting the results.
The patient-centred approach focused primarily on the patients’ perspective, which was similar in all three regions. This is important as care is often organised from the care providers’ perspective which may not always address the patients’ needs.
We acknowledge the limitation of retrospective quantitative data collection, but since we were interested in the least biased information obtained during interviews, we preferred the retrospective approach to collect quantitative data. Preliminary results demonstrate that the presence of comorbidities is not concordant to other evidence-based studies.51–53 This suggests that some comorbidities may be undetected. Still, this supports the need of in-depth studies on chronic diseases care such as this study.
Although guidelines state that DMP are necessary to address challenges of HF, the concrete content of DMP is not well defined and may vary significantly between individual centres. Therefore, results of this study might not be directly applicable to other centres using DMP. Also, we recognise the fact that the selected regions might not be representative for all regions of the participating countries, i.e. the Netherlands, Belgium and Germany, and more differences between regions and countries are likely regarding Europe as a whole. Still, our study will give important insight into differences regarding many aspects of HF care that are useful and applicable not only to the investigated regions.
Finally, patient recruitment in the three regions differed considerably, which resulted in differences between patient groups. In particular, patients were recruited from primary care only in Belgium due to logistic reasons, and mainly, tertiary care patients were included in Aachen. The potential impact of this difference cannot yet be determined but needs to be considered in further analyses.
Conclusion
This study will provide an in-depth insight into processes in chronic HF care, patients’ and care providers’ knowledge and preferences, the interaction and communication between stakeholders and the needs for improvements in HF care. It will also define bottlenecks that may hinder good clinical practice, and also aspects that support it. These results will help to understand the needs for providing well-organised, excellent HF care.
Footnotes
Authors’ contributions
HPBLR is the principle investigator and the project director for the INTERACT-in-HF study; he was, together with SSVW, responsible for the set-up of the study. SB, KB, JB, CK, SSvW and HPBLR contributed to the writing of this paper. All authors reviewed it critically for intellectual content and approved the final version submitted for publication.
Acknowledgements
We would like to thank Bao-Oanh Nguyen, Zjuul Seegers, Mark Mertens, Romy Sels and Desirée Rutten for performing semi-structured interviews and coding, Mireille HA Spanjers for her support in setting up the study, Mieke Derickx for teaching us the tricks of the trade concerning coding and transcribing and Carola Hausdorf for her support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
