Abstract
Objective:
Asymptomatic bacteriuria (ASB) during pregnancy is a risk factor for development of urinary tract infections and pyelonephritis, which can lead to maternal and foetal consequences. This study aimed to determine the prevalence of ASB during pregnancy, the most common causative pathogens and their resistance patterns.
Methods:
A retrospective analysis was performed using microbiology laboratory data from urine sample cultures from pregnant women collected at our University hospital over a 6-year period (2014–2019). Identification and susceptibility testing were performed using standard microbiology procedures based on British Society of Antimicrobial Chemotherapy and European Committee on Antimicrobial Susceptibility Testing.
Results:
From a total of 18,938 urine samples, 1522 (8.04%) were positive for bacteriuria, the most common isolates were Escherichia coli and Coliform (lactose fermenters) (n = 1171, 76.9%), followed by Enterococcus faecalis and other enterococci (n = 191, 12.5%). In 2019, the resistance of E. coli was 56.8%, 25.3% and 4.7% to amoxicillin, trimethoprim and gentamicin, respectively, with an increasing pattern of resistance to trimethoprim and gentamicin from 2014 to 2019. The resistance rates to nitrofurantoin were 1% and 5.5% for E. coli and Group B Streptococcus, respectively.
Conclusion:
Our study shows the trends of antimicrobial resistance in this vulnerable group and will help confirm treatment effectiveness and direct guideline recommendations locally and internationally.
Level of Evidence:
2b
Keywords
Introduction
Urinary tract infections (UTIs) and bacteriuria are common during pregnancy due to mechanical and hormonal changes. 1 Asymptomatic bacteriuria (ASB) is defined as a significant count of bacteria (>105 colony forming units (CFU)/mL) in the urine plus absence of clinical signs or symptoms.2,3
Approximately, 30% of pregnant women with untreated ASB develop acute pyelonephritis, which is associated with preterm labour, low birth weight and systemic maternal infection.3–5 Due to the potential consequences that untreated ASB can have, it is regularly screened for and treated in developed countries. Current National Institute for Health and Care Excellence guidelines recommend that nitrofurantoin should be prescribed as first line treatment followed by amoxicillin or cefalexin depending on antimicrobial sensitivities.6,7
The majority of bacteriuria in pregnancy is caused by Escherichia coli (63–85%), Klebsiella pneumoniae (8%) and rarer organisms such as Staphylococcus aureus, Group B Streptococcus and Pseudomonas spp. 1 Group B Streptococcus accounts for 2–10% of ASB positive cultures and is a known cause of neonatal and maternal infection if a membrane rupture occurs.8,9
The increasing rates of antimicrobial resistance (AMR), worldwide, means a rising fear of extended spectrum B lactamase (ESBL) and multidrug-resistant (MDR) organisms. 8 Antimicrobial stewardship (AMS) is one of the crucial elements of tackling AMR globally including the prescription of antibiotics for ASB.
Current data investigating the common pathogens and antimicrobial susceptibility patterns in ASB has been collected in non-pregnant populations or in other regions around the world. To our knowledge, this information is lacking in the United Kingdom, and therefore, we aimed to examine these features among pregnant women screened in antenatal clinics in the United Kingdom. By matching current antibiotic recommendations for treating ASB in pregnant women with the common pathogens and susceptibility patterns we aim to aid the reduction of inappropriate antibiotic prescription and thus slow the development of antimicrobial resistance.
Methods
This study used data collected the microbiology department in conjunction with the antenatal clinics of University Hospital Southampton NHS Foundation Trust. The bacteriuria screenings were done at the antenatal clinics using midstream urine (MSU) samples from pregnant women in their first 20 weeks of pregnancy as per NICE guidelines. 6 Follow up takes place at the Princess Anne Hospital Southampton which sees ~5,500 pregnant women each year. Identification and susceptibility testing were performed using standard microbiology procedures based on British Society of Antimicrobial Chemotherapy (BSAC) and/or European Committee on Antimicrobial Susceptibility Testing methodologies (EUCAST). 10 The microbiology lab extracted 6 years’ worth of data (January 2014–Decemeber 2019) which was condensed into an encrypted pseudonymised data set.
The data set was first filtered to exclude certain results that were not relevant to the analysis being done and to reduce the data set in order for a full analysis to be complete. Samples removed mainly included those that had no growth recorded as well as samples positive for Candida albicans. This filtering was done under guidance from a consultant clinical microbiologist and infection specialist as well as the microbiology lab specialist staff.
Statistical analysis was performed in SPSS (version 26, IBM, Armonk, NY, USA) and graphs generated in R (Vienna, Austria) using the ‘ggplot’ package. Data were assumed to be non-normally distributed given the limited data available. Correlations were therefore tested with Spearman’s test and presented as spearman’s rho with an associated p-value. Chi-square tests were used to compare proportions of categorical variables, with Fisher’s Exact test used in the case of any counts dropping below n = 5. Significance was set at p < 0.05.
This study was conducted from October 5, 2020 to January 4, 2020 and approved by the UHS Ethics Board for Audits (ZAUD 6685) as well as the University Ethics Board (ERGO 55927).
Results
From January 2014 to Decemeber 2019, 18938 patient samples were examined by the University Hospital Southampton microbiology lab for ASB during the first trimester of pregnancy. Of these, 1522 (8%) had a positive urine culture which indicates the overall prevalence of ASB among this cohort. In addition, 372 samples showed yeast or Candida albicans, but were excluded from the remainder of the study due to relevance to the aims.
Bacteriuria was most prevalent in the 25–29 age group (n = 425, 27.92%), however, this group also had a large portion of the total records (n = 5638, 29.77%). The under 20 age group and the 20–24 age group displayed consistently high Chi-squared Goodness of Fit Test values (average of 5.21) indicating that these age groups have an increased portion of positive samples for ASB during pregnancy.
Prevalence of pathogens causing ASB in pregnancy
From the samples collected in the year 2019, majority of the culture results for positive bacteriuria were caused by E. coli and other lactose fermenting coliforms (77.58%) followed by Enterococcus faecalis and other enterococci (13.37%), Group B Streptococcus (4.64%), Morganella morganii, Citrobacter freundii and other non-lactose fermenting coliforms (NLF) (2.32%) (Figure 1). The remaining bacteria (Proteus spp., Pseudomonas spp., Staphylococcus aureus, Staphylococcus saprophyticus, Klebsiella pneumonaie and Acinetobacter spp.) all had very low prevalence with many of these accounting for less than 1% of the total positive samples from 2019.
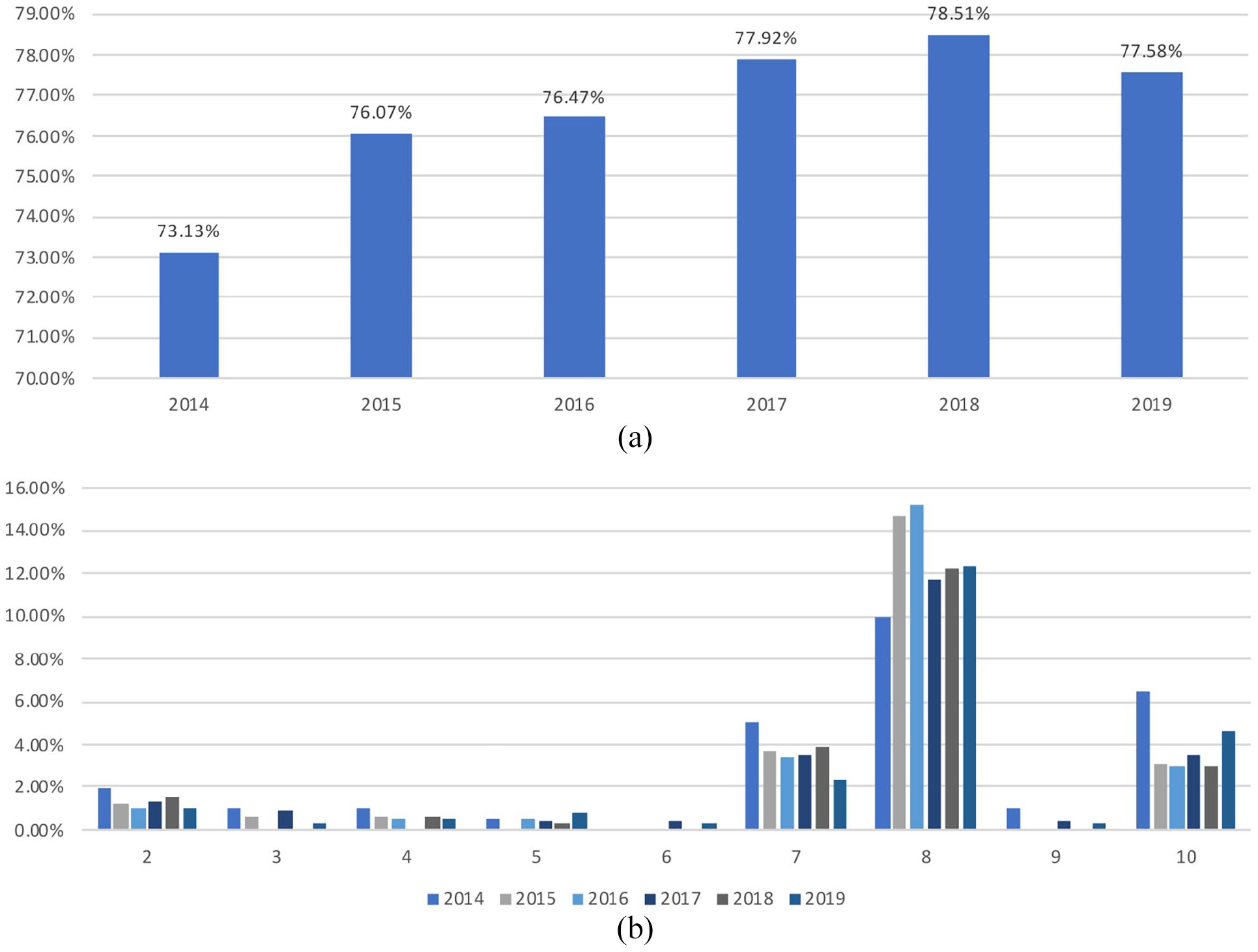
(a) Prevalence of E. coli and other lactose fermenting coliforms 2014–2019. (b) Prevalence of Bacteria Types Causing Bacteriuria in Pregnant Women-Excluding Group 1 (E. coli and other lactose fermenting coliforms).
In 2014, 73.13% of the total positive samples were caused by E. coli and other lactose fermenting coliforms and this remained consistent over the 6 years this data set covers. Although there appears to be a slight increase in proportion (from 73.13% in 2014 to 77.58% in 2019, a 4.45% increase), this was not statistically significant. Enterococcus faecalis and other enterococci, Acinetobacter lwoffi, Acinetobacter pittii and other Acinetobacter as well the Streptococcus Group B, Proteus spp., Pseudomonas spp., Staphylococcus aureus, Staphylococcus saprophyticus and Klebsiella pneumoniae showed no notable changes and a constant low prevalence over that time frame. However, Morganella morganii, Citrobacter freundii and other non-lactose fermenting coliform bacteria had a consistent number of samples (n = 10 in 2014 and n = 9 in 2019), this is noteworthy due to the fact that the total number of samples increased from 2014 to 2019, so the proportion of this group out of the total positive samples decreased (4.98% in 2014 to 2.32% in 2019, p ⩽ 0.05).
Resistance patterns
E. coli displays a significant resistance to amoxicillin (56.8% in 2019) and increasing resistance to trimethoprim (25.3%,8% increase from 2014 to 2019) and gentamicin (4.7%, 3.3% increase from 2014 to 2019). Other drugs such as ciprofloxacin (3.56% increase from 2014 to 2019) and co-amoxiclav (2.96% increase from 2014 to 2019) also show small increases in resistance over this time. However, since the resistance rates of these remain below 10%, they are not as clinically relevant as the resistance to amoxicillin and trimethoprim but may pose potential problems in the future (Figure 2).
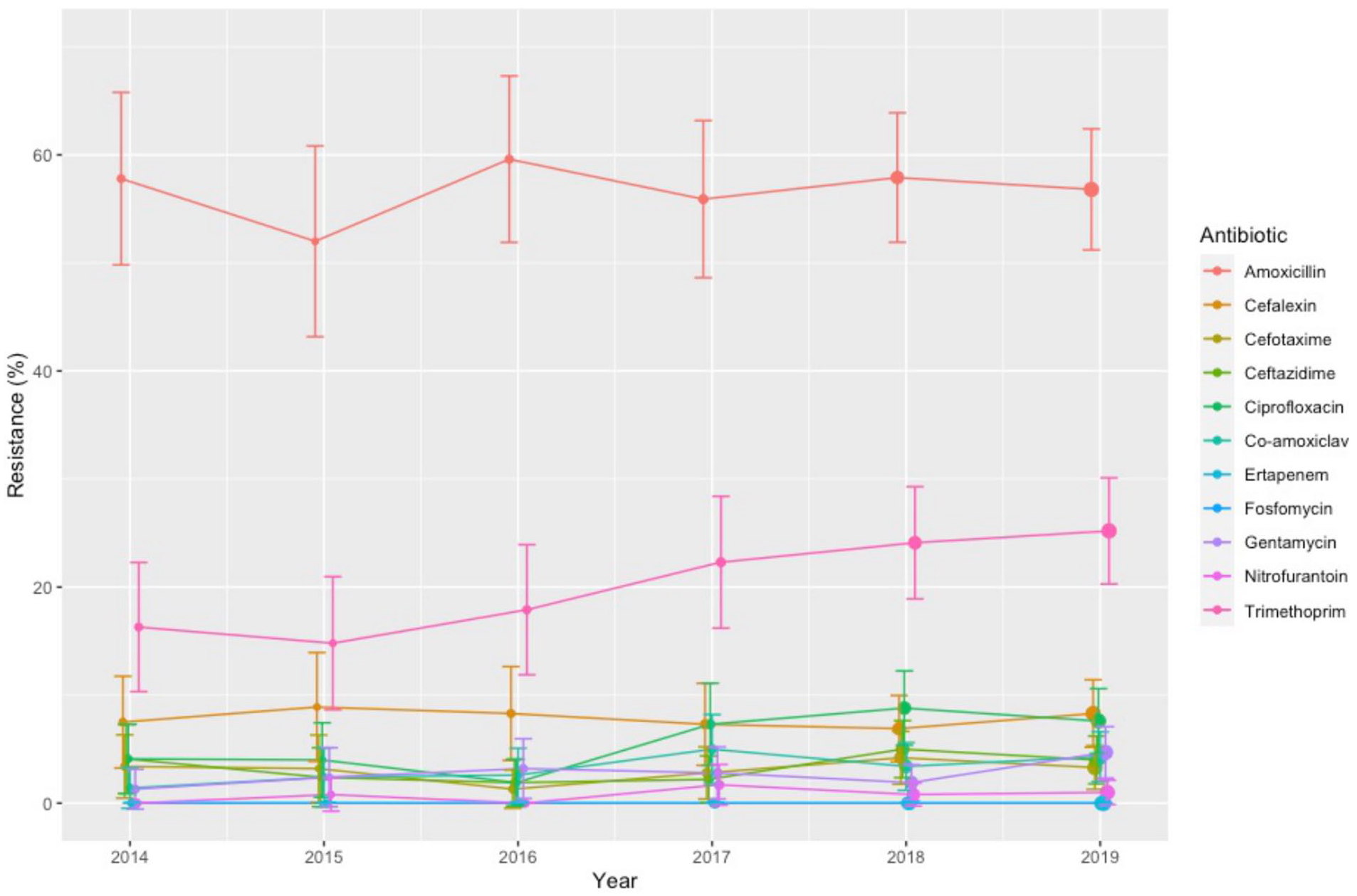
E. coli Coliform (lactose fermenters) antibiotic-resistance patterns from positive ASB samples in pregnant women 2014–2019.
As seen in Table 1, Enterococcus faecalis and other enterococci displayed an increasing resistance to trimethoprim (6.05% increase from 2016 to 2019) as well as some resistance to amoxicillin. Morganella morganii, Citrobacter freundii and other non-lactose fermenting coliforms also display resistance to amoxicillin (77.78% in 2019). The high-resistance rates of bacteria to amoxicillin suggests that amoxicillin should only be prescribed when there is a confirmed sensitivity.
Proportion of pos. samples for each bacteria resistant to named antibiotics from 2014 to 2019.
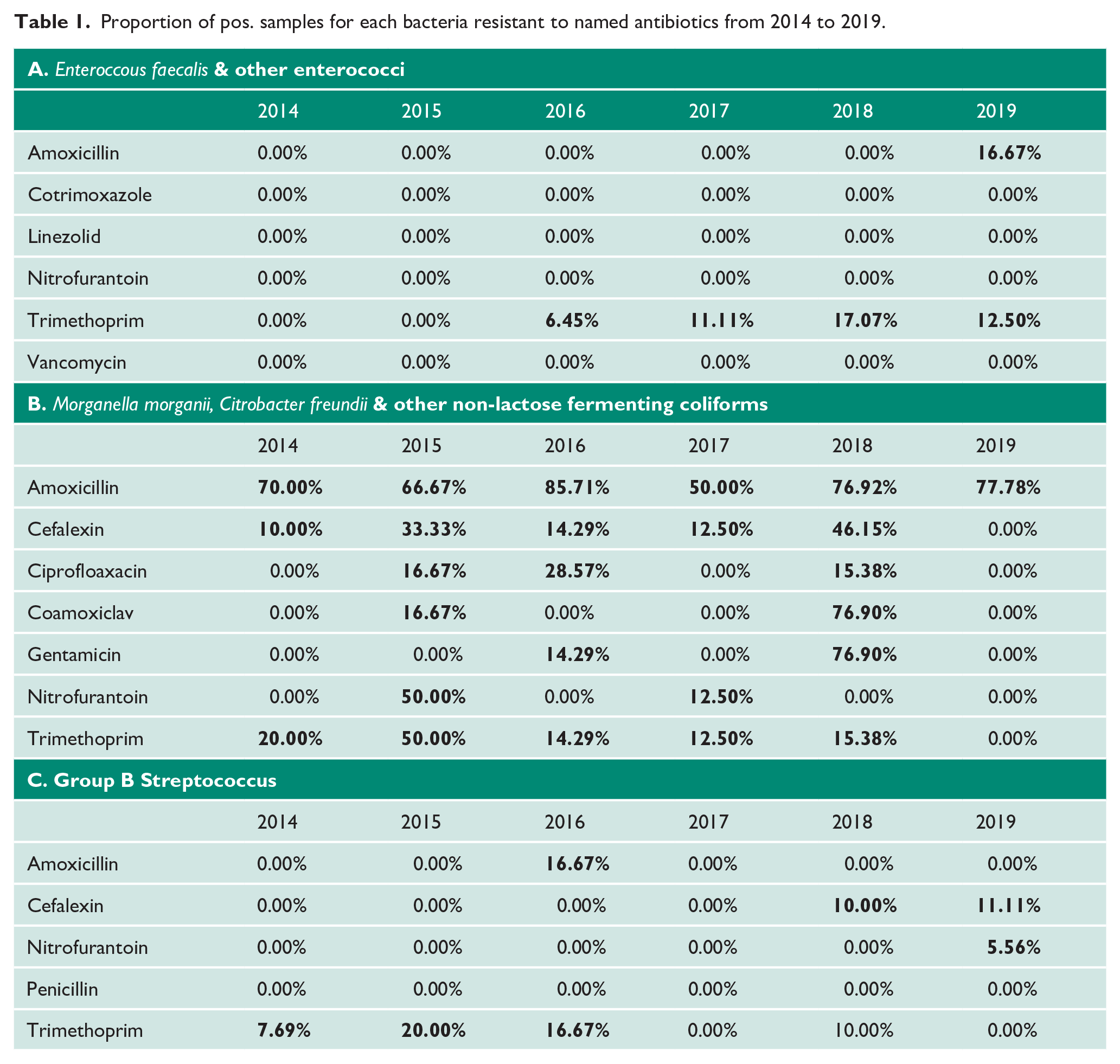
The recent resistance of Group B Streptococcus to nitrofurantoin may be of relevance since as shown in Table I, there are no instances of resistance to this drug before 2019. Finally, Klebsiella pneumoniae, as noted in Figure 1, has a very low prevalence but because of the severity of the infections it can cause it is important to note that there are a few instances of resistance to amoxicillin, cefalexin, cefotaxime and co-amoxiclav.
Patients with Multiple Positive Bacteriuria Samples
206 patients screened in the antenatal clinics from 2014 to 2019 had more than one sample positive for bacteriuria. Of these, 79.22% of patients who had multiple positive samples had all urine culture results which were all positive for E. coli or another lactose fermenting coliform. 106 patients experienced two positive samples of bacteriuria during one pregnancy with both of those samples being positive for E. coli or coliforms (lactose fermenting). Out of these, 31 (29.2%) of these patients had relapses of infection (defined as having an infection from the same organism 1–2 weeks after antimicrobial therapy, for the purpose of this study 4 weeks was used to consider scheduling dilemmas) and 75 (70.8%) patients either had a reinfection (same organism reinfecting the urinary tract) or a completely new infection 11 (Figure 3).
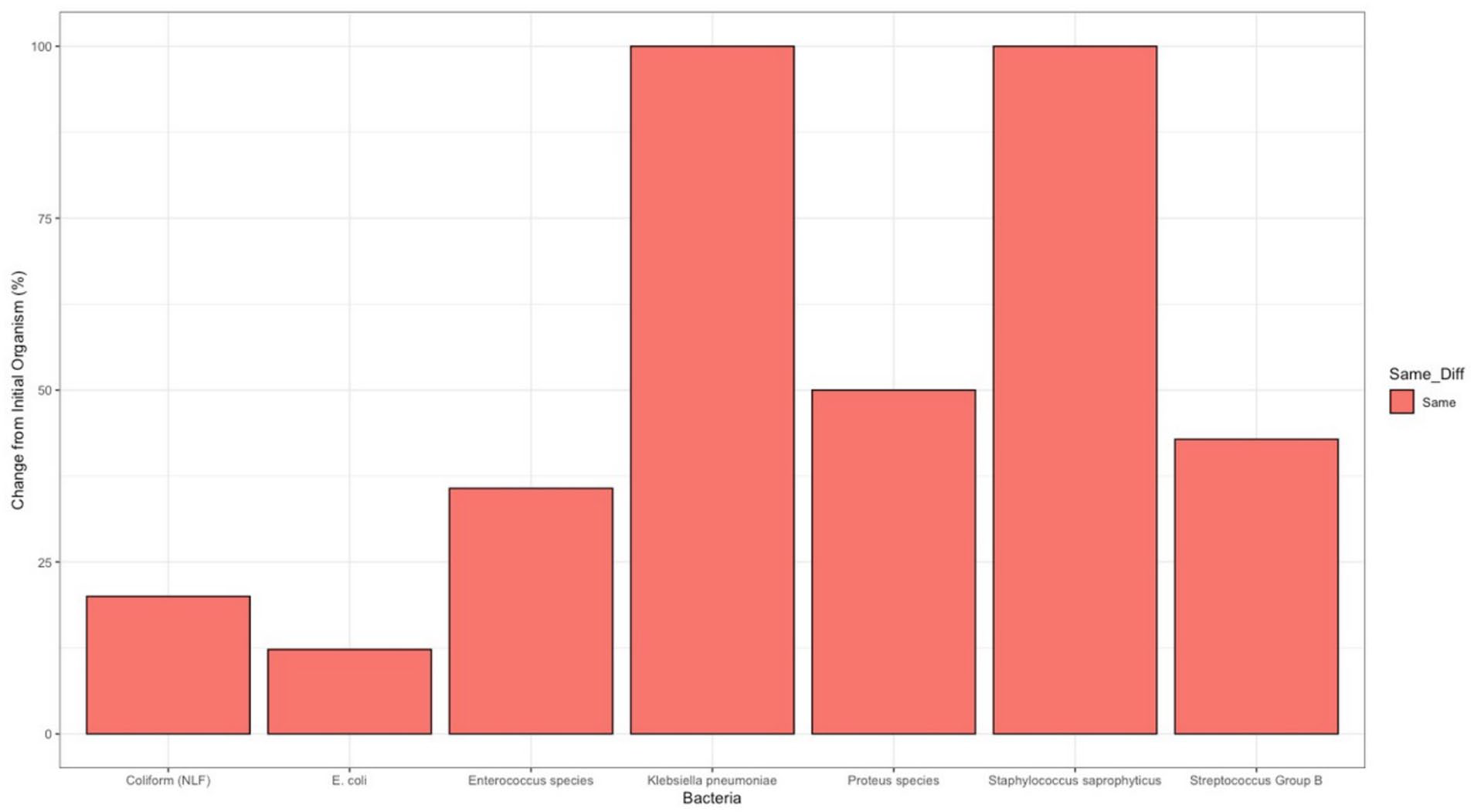
Proportion of repeat positive bacteriuria samples taken throughout a singular pregnancy in which the cultured bacteria differs from the initial screening sample.
Discussion
Main findings
To our knowledge, this is the largest study in the literature looking at prevalence and resistance patterns of ASB in pregnancy. We were able to reveal information about recent resistance patterns from the region and to confirm that the current regional guidelines still match these patterns.
There has been some debate around the need of antibiotics in treating ASB in pregnancy but according to the current guidelines set by the European Association of Urology, evidence still suggests the need for antibiotics in the treatment of ASB in high-risk populations, such as pregnant women. This is to prevent the negative outcomes such as symptomatic urinary tract infections, low birth weight and preterm delivery. 12 As there is still a need to treat ASB in pregnant, the outcomes of this study are clinically relevant in providing further information about local resistance patterns and subsequently aiding the global movement against antimicrobial resistance.
There have been multiple studies conducted in other countries such as South Africa, Northern Ethiopia, Turkey and Egypt over the past few years investigating the prevalence of ASB, information on bacteria involved and antibiotic susceptibility patterns.2,3,9,13 All of these studies report E. coli as the most prevalent bacteria causing ASB in pregnant women. The Egyptian and the North Ethiopian studies also noted large resistance rates of E. coli to amoxicillin (100% in Northern Ethiopia).2,3 We noted much similarity in our study performed in the United Kingdom especially in regard to the resistance patterns of E. coli to amoxicillin. Although amoxicillin and co-amoxiclav (amoxicillin plus clavulanic acid) are not regularly used for treatment of ASB in pregnancy in the United Kingdom as current guidelines recommended the use of nitrofurantoin. However, based on the outcome from our study, we would recommend not using amoxicillin empirically but instead only in instances where a confirmed sensitivity of the organism to the antibiotic is established. It is also important to note that co-amoxiclav is not used as a result of findings from the ORACLE I study which determined that there is a risk of neonatal necrotizing enterocolitis when used for treatment of preterm rupture of the foetal membranes. 14
Although we found a very low prevalence of samples positive with Group B Streptococcus bacteria, it is important to note the recent resistance to nitrofurantoin in our study. In clinical practice, Group B Streptococcus infections in pregnant women are treated only when a patient in labour or the membranes have ruptured. In these instances, penicillin is typically used as a first line drug or a cephalosporin if the patient has a penicillin allergy. 15 The results from our study show no resistance of Group B Streptococcus to penicillin which confirms that this line of treatment should remain effective.
Strengths and limitations
The study has several limitations including being from a single centre, the inability to further examine the antibiotics prescribed for each patient and the related clinical outcomes. There were multiple patients who had multiple samples and it was left up to interpretation on whether there were issues with drug resistance or a new infection due to the lack of information. However, it still represents the largest data set in the United Kingdom for ASB in pregnancy and guides a potential follow-up study to further investigate patients who had multiple positive samples during a pregnancy and the causes behind this to gain more information about resistance patterns regarding bacteriuria in pregnant women.
Interpretation
The prevalence of bacteria and resistance patterns found in this study match what was expected especially in terms of the most common organisms causing ASB in pregnancy. The prevalence of E. coli in this study matches commonly accepted information that its prevalence causing UTIs is ~65–80%. 1 The resistance patterns found in this data matched other patterns from similar studies done in other regions of the world but provided a much-needed outlook into the resistance patterns in the South of England.2,3,9,13 There were some slight variations in resistance to specific drugs as previously mentioned but overall, the data showed no significant changes in resistance or prevalence of bacteria and supports the current guidelines for treating bacteriuria in pregnancy.
Conclusion
In this study, the prevalence of bacteria remains as expected and the resistance rates to first line treatment remains low. Antimicrobial resistance is a global issue and treatment of asymptomatic bacteriuria can be challenging during pregnancy. Our study shows the trends of AMR in this vulnerable group and will help confirm treatment effectiveness and direct guidelines recommendations locally and nationally. Similar studies should be conducted periodically to ensure that the guidelines for a sensitive population stay as accurate and effective as possible and to help reduce overprescribing.
Footnotes
Acknowledgements
The authors thank to the microbiology department particularly David Browning at the University Hospital Southampton NHS Foundation Trust, for providing the data to perform this study.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was conducted from October 5, 2020 to January 4, 2020 and approved by the UHS Ethics Board for Audits (ZAUD 6685) as well as the University Ethics Board (ERGO 55927).
Informed consent
Informed consent was not needed for this study as data used was pseudonymized data. Data can be obtained through specific request to the corresponding author.
Guarantor
BS and KS are joint senior authors.
EC is the corresponding author.
Contribution to Authorship
Study was organised by KS and BS in conjunction with the UHS microbiology department. EC analysed the data provided and interpreted the information with assistance with the statistics and figures from RG. The paper was then written by EC, KS, BS and SU all having input and different fields of study to contribute to the paper.
