Abstract

A 64-year-old Caucasian man was referred to the urology clinic with intermittent painless gross haematuria 16 years following renal transplantation. He was previously on haemodialysis for three years for end-stage renal failure due to lupus nephritis before undergoing deceased-donor renal transplantation at 48 years of age. His post-transplant recovery was unremarkable, with no evidence of rejection and normal renal function. His medications included the immunosuppression regimen of cyclosporin, mycophenolate mofetil and low-dose prednisolone.
His haematuria work-up included ultrasonography, which revealed a polypoid soft-tissue mass in the anterior wall of the urinary bladder with both endophytic and exophytic components (Figure 1). Subsequent computed tomography (CT) imaging demonstrated a 16 mm×12 mm smoothly marginated ovoid soft-tissue attenuation nodule in the mid-anterior urinary bladder (Figure 2). The transplanted kidney appeared normal, and both native kidneys demonstrated atrophic changes. There was no evidence of a patent urachus, urachal diverticulum or metastasis.
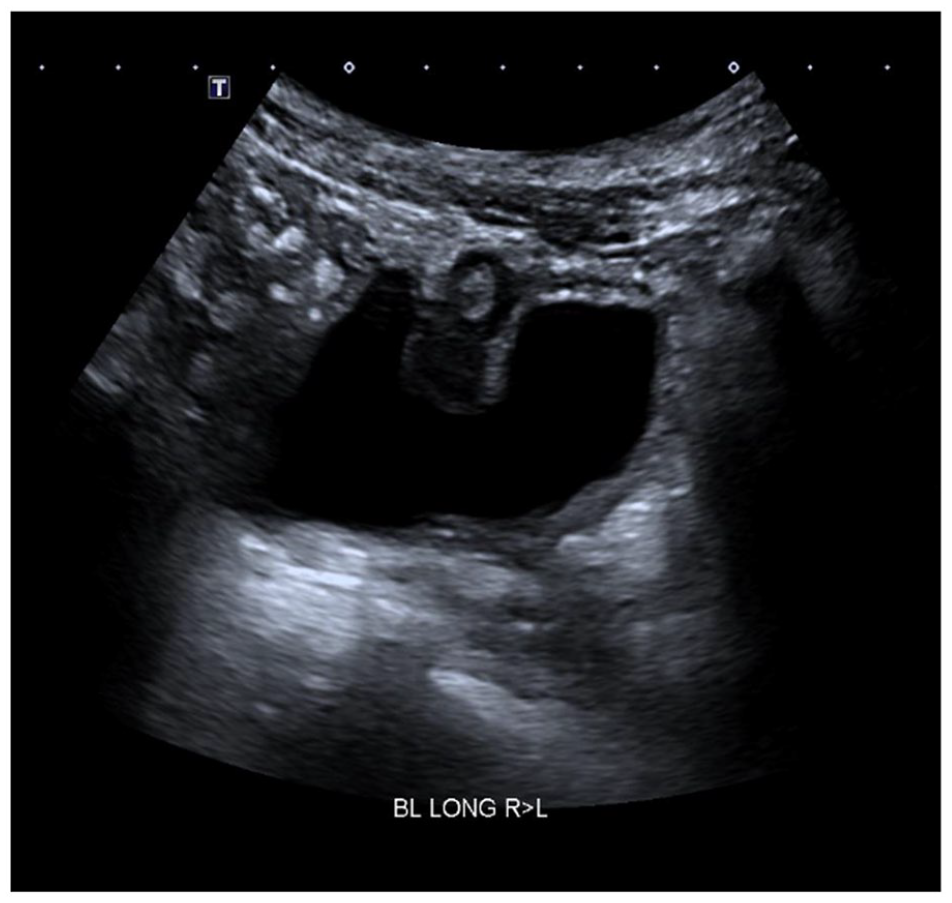
Ultrasound imaging of the bladder demonstrated a 20 mm polypoid soft-tissue mass in the anterior wall of the urinary bladder with both an endophytic and exophytic component.
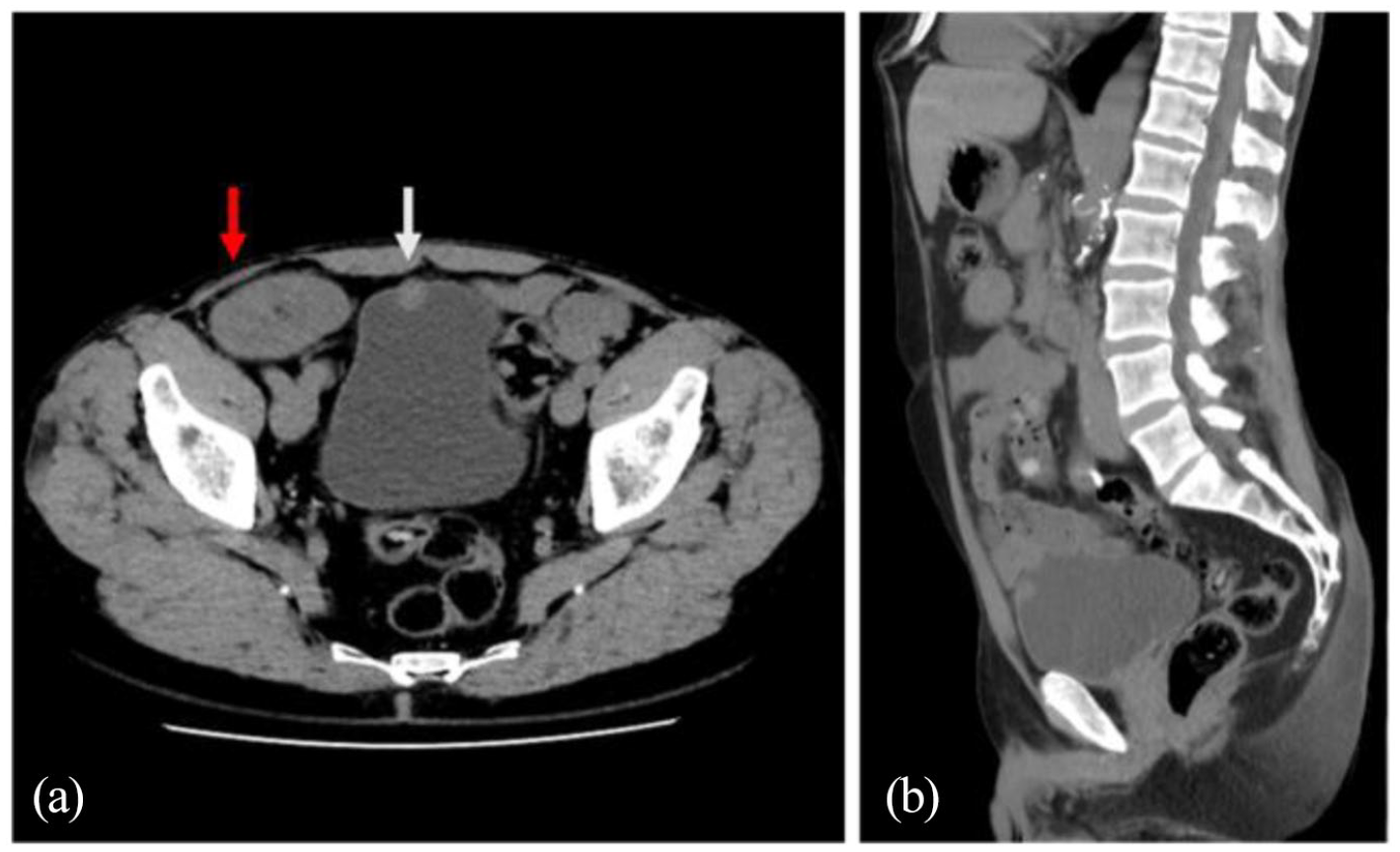
Axial (a) and sagittal (b) section computed tomography imaging of the abdomen and pelvis demonstrated a soft-tissue attenuation lesion in the anterior wall of the bladder (white arrows). The transplant kidney is seen in the right iliac fossa (red arrow).
As shown in Figure 3, cystoscopic examination under general anaesthesia revealed a solitary 2 cm papillary tumour in the anterior bladder wall with a normal transplant and bilateral native ureteral orifices. Transurethral resection of the bladder tumour was performed. Histopathological examination revealed a mucin-producing adenocarcinoma infiltrating the lamina propria with suspicion for invasion of the detrusor muscle. Serum carcinoembryonic antigen (3 ng/mL, normal range <8 ng/mL) and prostate-specific antigen (0.43 ng/mL, normal range <4.0 ng/mL) were unremarkable. Colonoscopy revealed no abnormality.
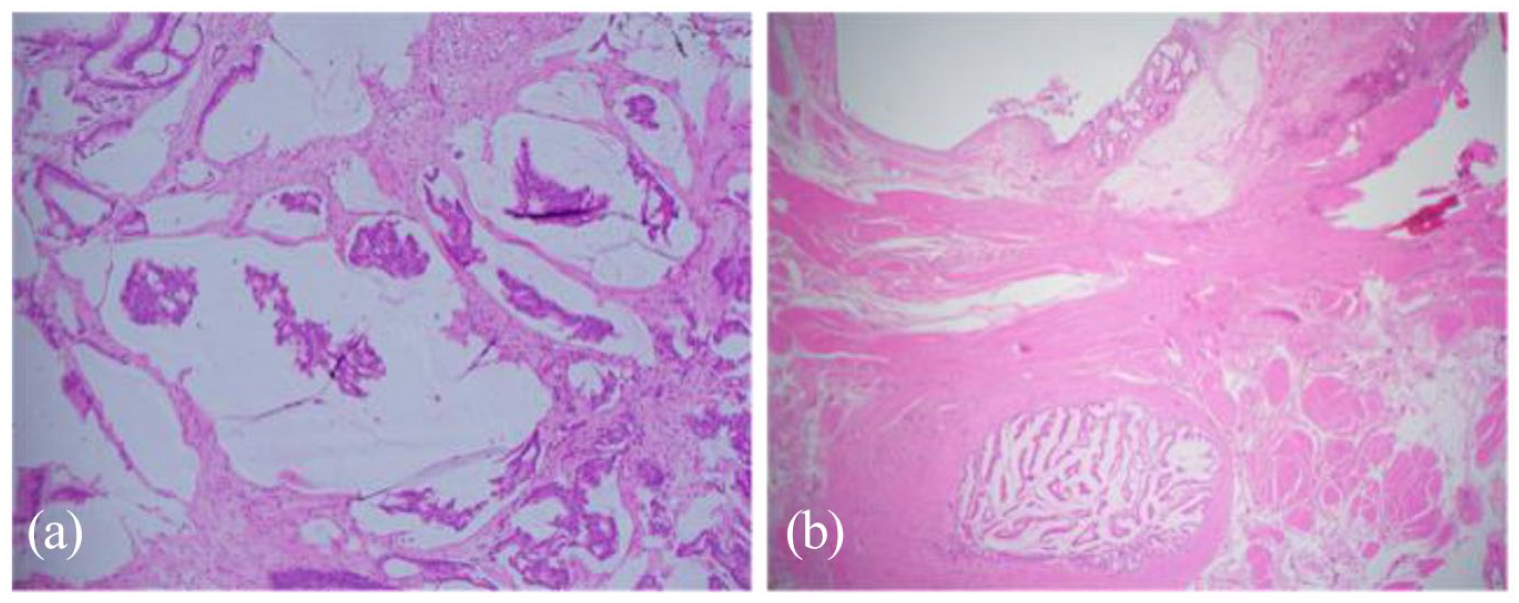
Microscopy examination demonstrating (a) mucinous adenocarcinoma on transurethral resection of the bladder tumour and (b) residual glandular tumour in region of the urachus with extravasated mucin on radical cystoprostatectomy.
He underwent a radical cystoprostatectomy, left pelvic lymph node dissection and ileal conduit urinary diversion through a 10 cm lower midline laparotomy incision. During cystoprostatectomy and lymph node dissection, particular care was taken when dissecting the right-sided bladder pedicles to protect the renal transplant. The position of the renal transplant in the right iliac fossa precluded right pelvic lymph node dissection. A 20 cm segment of ileum 15 cm proximal to the ileocaecal valve was harvested, and a side-to-side bowel anastomosis was performed. Uretero-ileal anastomoses were performed using the Wallace technique for the native ureters, and the transplant renal pelvis was spatulated and anastomosed to the side of the ileal conduit. Histopathological examination confirmed intestinal-type mucin-producing adenocarcinoma of the anterior bladder wall consistent with urachal adenocarcinoma with negative pathological nodes and surgical margins. There was a small focus of incidental localised Gleason score 3+3=6, Grade group 1 prostate adenocarcinoma. The postoperative function of the transplanted kidney remained stable, and follow-up ultrasonography did not reveal any hydronephrosis. Surveillance CT imaging at six months postoperatively did not reveal any evidence of recurrent or metastatic disease.
Renal transplant recipients have a two- to sevenfold increased risk of developing a secondary neoplasm compared to the general population, and transplant-associated malignancy is the leading cause of mortality among transplant recipients after 10 years of follow-up. 1 The majority of transplant-associated malignancies are skin cancer and lymphoma followed by urological cancers, of which most are renal-cell carcinoma.2,3 The urachus is a vestigial fibromuscular tract between the bladder and the umbilicus. Urachal adenocarcinoma accounts for 0.01% of all malignancies and for <1% of bladder cancers. 4 We report the fourth case of urachal adenocarcinoma following renal transplantation globally and the first case in Australia.4–6
The World Health Organization criteria for diagnosing urachal adenocarcinoma include (a) location of the tumour in the bladder dome and/or anterior wall, (b) epicentre of carcinoma in the bladder wall, (c) absence of widespread cystitis cystica and/or glandularis beyond the dome and anterior wall and (d) absence of a known primary tumour elsewhere. 7 Urachal adenocarcinoma is believed to evolve from intestinal metaplasia of the urachal remnant. It occurs most commonly in patients >50 years of age, and two-thirds of cases are in men. 8 Whilst gross haematuria is the most common clinical presentation, other symptoms may include urinary frequency, urinary urgency and abdominal pain. 8 CT imaging typically shows a soft-tissue attenuation mass at the dome of the bladder. Low-attenuation cystic components may be seen due areas of mucin production.
Five-year survival is approximately 40–50%, and adverse prognostic factors include locally advanced disease involving adjacent organs or abdominal wall, metastatic disease or residual disease after surgery.5,7 Surgery is the most commonly used treatment for non-metastatic urachal adenocarcinoma. 5 However, there is a lack of evidence-based guidelines regarding the best surgical approach because of the rarity of the condition, and both partial or radical cystectomy demonstrate similar oncological results.9,10 The use of uretero-ileal anastomoses for the native ureters and transplanted renal pelvis was preferred in the current case because of the risk of native ureteric obstruction following native ureteral ligation and the additional morbidity associated with bilateral native nephrectomy. While some authors recommend a bladder-sparing approach, the evidence for this is sparce, particularly in immunosuppressed patients. 5 Treatment by radiotherapy or chemotherapy could be considered in patients who have inoperable disease or positive margins, based on experience with bladder cancer in general. 7
Footnotes
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because it is a case report by the patient’s treating clinicians in our urology department.
Informed consent
Verbal and written informed consent was obtained from the patient for publication in a peer-reviewed journal.
Guarantor
A.A.
Contributorship
Each named author contributed substantially to the drafting of this manuscript. R.D. was responsible for the literature review and manuscript draft. S.C. and P.C. provided the pathology figures and their descriptions. J.M. was involved with manuscript revision, and A.A. provided supervision and critical revision of the article.
