Abstract

Introduction and background
Benign prostatic hyperplasia (BPH) is a prevalent condition. Transurethral resection of the prostate (TURP) is the gold-standard surgical treatment for patients with significant BPH-related lower urinary tract symptoms (LUTS) refractory to pharmacotherapy, recurrent urinary tract infections (UTI), development of bladder stones, refractory haematuria or recurrent acute urinary retention or in those who develop high-pressure chronic urinary retention. However, development of bladder neck contracture (BNC) after TURP is not uncommon. Treatment of post-TURP BNC is challenging, especially for those with recurrences following endoscopic treatments. We report and describe the use of intralesional injection of triamcinolone acetonide under cystoscopic guidance to treat a patient with multiple recurrent BNC after TURP successfully.
Presentation
A 57-year-old man was referred to our urology clinic for management of his recurrent post-TURP BNC. He first presented with voiding LUTS and underwent monopolar TURP six years ago in the private sector, when 6 g of adenoma was resected. His voiding LUTS improved initially after surgery. However, he later developed recurrent voiding LUTS. Cystoscopy demonstrated a patent prostatic channel, but a BNC was identified. The patient underwent repeated transurethral bladder neck incision (TUBNI) with or without concurrent TURP five times by different urologists over the subsequent years. He experienced short-term improvement of his symptoms after each procedure, but symptoms recurred within several months following treatment. He presented to our clinic with a bothersome dribbling urinary flow, associated with sensation of incomplete emptying and urinary frequency.
On urethrocystoscopy, he was found to have a blind-end bladder neck. A suprapubic catheter was inserted. Four weeks later, a staged rendezvous insertion of hydrophilic guidewire using a flexible cystoscope via the suprapubic tract was carried out, which was used to cannulate the bladder neck and exit through the penile meatus (Figure 1). A subsequent retrograde transurethral bladder neck incision was performed at the 12 o’clock position using an optical urethrotome (Figure 2), followed by a formal TUBNI using a Colling’s knife when the bladder neck was wide enough to pass instruments through endoscopically. Residual visible bladder neck scars were resected by electrocautery with loop transurethrally. An 18F indwelling urethral catheter was kept in situ for 10 days. Postoperatively, the patient had significant improvement of his symptoms. He had an excellent maximum flow rate (Qmax) of 34.9 mL/s on uroflowmetry assessment at six weeks.
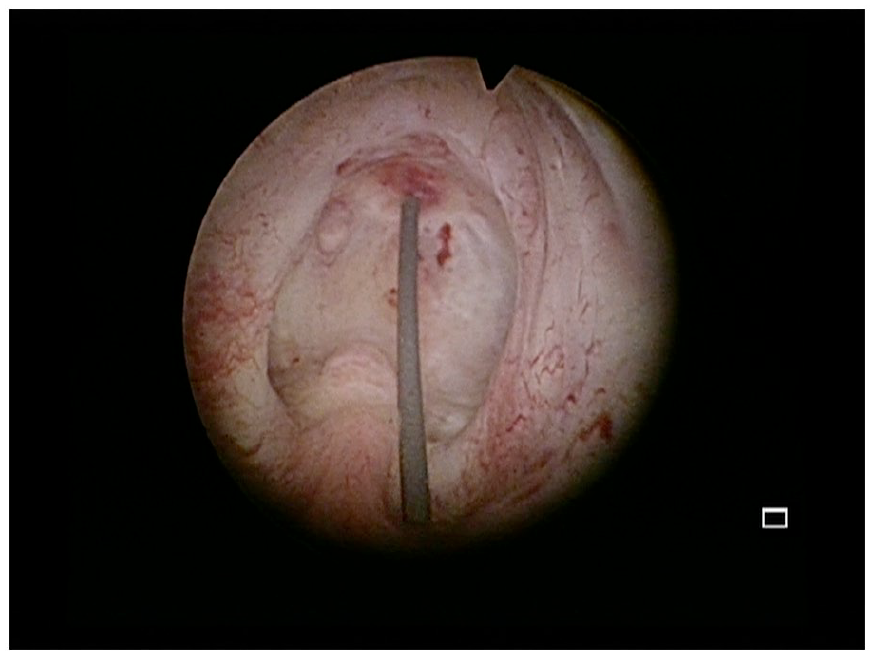
The patient presented with a very tight bladder neck contracture which only allowed a hydrophilic guidewire to pass through on presentation.
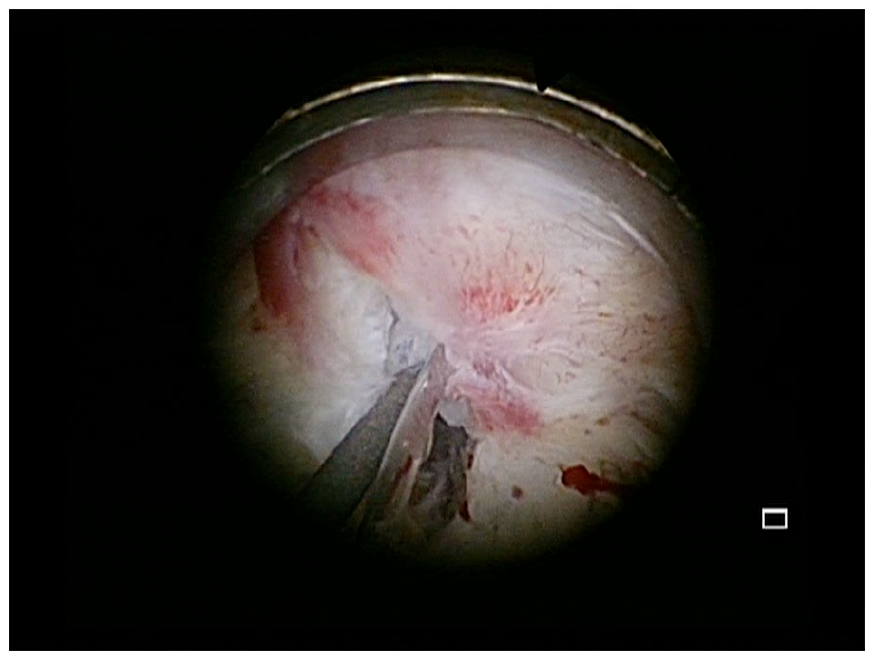
An optical urethrotome was used to incise the dense bladder neck scar and open up the channel.
Unfortunately, at three months postoperatively, the patient developed recurrence of LUTS. A repeat uroflowmetry assessment showed his Qmax decreased to 11.7 mL/s. Flexible urethrocystoscopy showed BNC recurrence, which was calibrated at 10F in size. Both options of transurethral cystoscopic bladder neck injection of triamicinolone acetonide and robotic bladder neck reconstruction with Y-V-plasty were offered to the patient. He opted for the former.
The procedure began with TUBNI using a Colling’s knife until scar tissues were incised to base of bladder neck at 3 and 9 o’clock positions. A total of 80 mg of triamcinolone acetonide mixed with 2 mL of 0.9% saline was injected at 3 and 9 o’clock positions of the bladder neck under cystoscopic guidance using a Williams cystoscopic injection needle (5.0F/35 cm; Cook Medical, Inc., Bloomington, IN). A 16F silicon urethral catheter was inserted at the end of the surgery, and the patient had a successful trial without catheter on day 5 postoperatively. The patient remained asymptomatic nine months following surgery, with a Qmax of 27.4 mL/s. A follow-up urethrocystoscopy demonstrated a patent bladder neck channel without any evidence of recurrent scarring or contracture (Figure 3).
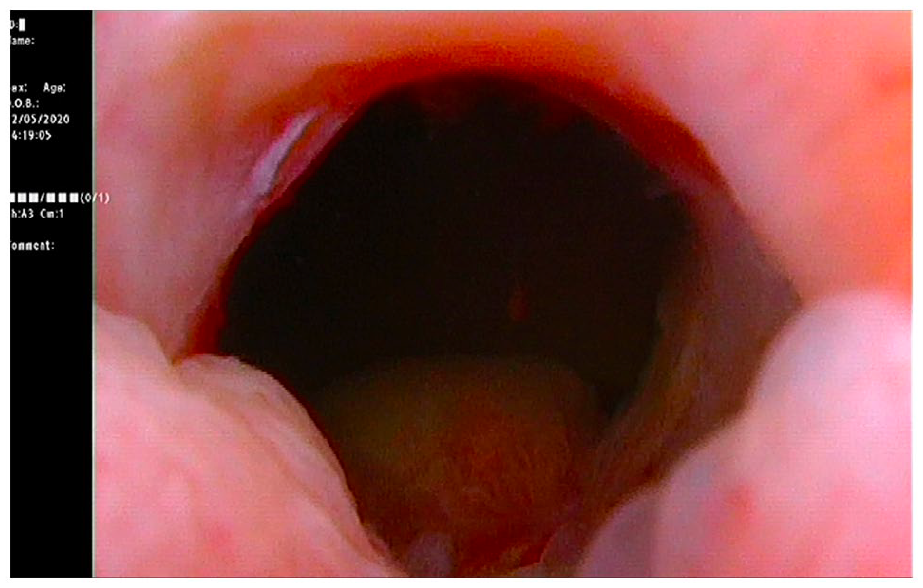
Urethroscopic view of the bladder neck six months following transurethral bladder neck incision and intralesional injection of triamcinolone at 3 and 6 o’clock. A patent bladder neck was observed, with no visible bladder neck scarring identified.
Discussion
The incidence of the BNC after TURP has been reported to be up to 9.6%. 1 Its pathophysiology has not been well understood, although it is believed to be aggravated by aggressive resection of the bladder neck, belligerent fulguration and hypertrophic scarring after TURP. 1 First-line treatments of post-TURP BNC are mainly endoscopic. Urethral stents have also been tried, but outcomes have been poor, with high re-intervention and complications rates. 2 Other endoscopic treatment options include urethral dilatation and endoscopic incision using cold-knife, electrocautery, laser, hot knife and loop resection. However, their outcomes have been poor, with many requiring multiple repeat surgical interventions, especially in patients with recurrent BNCs.
Open bladder neck reconstruction may be an option in experienced centre, but it is associated with a high morbidity rate. 2 Robotic Y-V plasty, whereby the BNC is incised anteriorly and a V-shaped bladder flap is advanced into the BNC in a Y-V plasty fashion, has been described with encouraging success rate a small series study, 3 but this is restricted to highly specialized units.
To avoid morbidities associated with BNC reconstructive surgery, endoscopic treatment utilizing intralesional injection of cytotoxic agents has been investigated. Redshaw et al. described a primary 58% (32/55) and overall 75% (41/55) success rate of TUBNI with intralesional injection of mitomycin C in a multi-centre retrospective review. 4 However, four (7%) patients developed serious adverse event,s including osteitis pubis, bladder floor necrosis and rectourethral fistula. Zhang et al. reported a 92.9% success rate of transurethral resection of bladder neck scar combined with intralesional triamcinolone injections in patients with highly recurrent post-TURP BNC. 5 No adverse events were reported. The injection of triamcinolone decreases fibroblast proliferation, increases collagen disintegration and suppresses inflammation. 5 Therefore, it reduces scar formation and is suitable for patients with recurrent BNCs. Zhang et al. suggested regular postoperative triamcinolone acetonide injections three times every four weeks to decrease the rate of recurrence further. Eltahawy et al. reported that triamcinolone injections after holmium laser incision can successfully treat 83% of 24 patients who had anastomotic stenosis after radical prostatectomy. 6 Although the pathophysiology may be different in anastomotic stenosis, the aim of triamcinolone injection remains the same: to reduce the recurrent scar formation after resection. No studies have described systemic absorption and toxicity of transurethrally injected triamcinolone. We suggest cautious use in patients with active tuberculosis, systemic fungal infections, active peptic ulcer disease, uncontrolled diabetes and severe hypertension until further studies can prove that the treatment does not affect these conditions.
We recommend not using urethral catheters with large diameters following treatment, as larger bore catheters in theory exert greater pressure on the bladder neck, reducing blood flow, and hinder re-epithelialisation, causing fibrosis and BNC recurrence. Silicone catheters should be preferred over latex catheters due to their more favourable biocompatibility and comparatively less antigenicity property.
Conclusion
We have described our technique of TUBNI with intralesional injection of triamcinolone to treat a patient with recurrent recalcitrant BNC successfully, who would have otherwise needed to undergo bladder neck reconstructive surgery or long-term intermittent self-dilatation with repeated endoscopic treatments.
Footnotes
Acknowledgements
None.
Conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The ethics committee of Queen Mary Hospital does not require ethical approval for publication of individual case reports.
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
Guarantor
W.L.
Contributorship
W.T.F. drafted the manuscript and coordinated the submission. W.L. performed the endoscopic procedure, reviewed the manuscript and provided important amendments. A.T.L.N. provided valuable idea in the concept of the treatment. J.H.L.T. reviewed the manuscript and approved the submission of the case report.
