Abstract
Haemophilus influenzae type b (Hib) is an important cause of meningitis and pneumonia in children. Despite the availability of Hib conjugate vaccine, many countries are still to implement it in their immunization schedule. Before introducing the vaccine in routine immunization programs, it is important to know not only the cumulative efficacy but also the efficacy of each vaccine dose. The primary objective of this review is to find whether two primary dose schedule of Hib vaccine is equally efficacious as the standard three primary dose schedule. A highly sensitive online search was run using the terms ‘Haemophilus Vaccines’ or ‘Haemophilus influenzae type b’ and ‘conjugate vaccine’, and Medline (Ovid), PubMed, Embase, CENTRAL and Scopus were explored for prospective randomized controlled studies. Data were extracted in a predesigned proforma and analyzed using RevMan software. Nine randomized studies were included in the analysis. Pooled vaccine efficacy using a fixed effects model against confirmed invasive Hib disease following the 3, 2 and 1 primary dose schedule were 82% [95% confidence interval (CI) 73-87], 79% (95% CI 54–90) and 65% (95% CI 23–84), respectively, and the overall efficacy was 80% (95% CI 72–85). To conclude, we found that Hib conjugate vaccine is highly efficacious and that the two dose regime is as good as the three dose regime. [The protocol was registered with PROSPERO (CRD42013004490)].
Keywords
Introduction
Haemophilus influenzae type b (Hib) is an important cause of both invasive (meningitis, pneumonia, epiglottitis, osteomyelitis, septicemia, arthritis and pericarditis) and noninvasive diseases in children [Kuhnert and Christensen, 2008]. Hib causes 90% of all the hospitalizations due to H. influenzae [Asensi et al. 1995]. Albeit that Hib may dwell in any age group, invasive diseases are prevalent in early childhood, appositely uncommon after five years of age. The true burden of Hib disease is not known, especially in low-income countries due to the lack of adequate laboratory testing facilities, increasing use of antibiotics prior to laboratory testing, lack of community-based studies and unpublished research data. Three million cases of serious diseases and child deaths ranging from 400,000 to 700,000 per year are caused by H. influenzae type b, as estimated by the World Health Organization [WHO, 1998].
Several Hib conjugate vaccines have been developed, which have been gradually introduced into routine immunization programs since the early 1990s. The number of countries using Hib conjugate vaccine increased from 89 in 2004 to 172 by the end of 2011.
Two generations of Hib vaccines are available. The first generation, polyribosolribitol phosphate (PRP) vaccines, failed to be efficacious in children aged less than 18 months, i.e. the age group with maximum disease burden [Kelly et al. 2004], due to the inability to generate memory B cells. The capsular polysaccharides were conjugated with protein carriers in an effort to ameliorate the immunogenicity of the T-cell independent immunogen. The second generation is the conjugate Hib vaccine: four basic types (PRP-T, PRP-D, HbOC and PRP-OMP) which differ in the carrier protein, their structure and length of the capsular polysaccharide, and PRP (Table 1) are available [Kelly et al. 2004]. The conjugate Hib vaccines differ in their immunogenicity because of different methods of coupling and choice of protein carrier [WHO, 2006]. They have the capacity to reduce the carrier state in vaccinated children and to reduce the pool of Hib infected children in the community, thereby providing herd immunity for unvaccinated children.
Characteristics of included studies.

DTP, diphtheria–tetanus–pertussis; HbOC, Haemophilus influenzae type b; IPV, inactivated polio virus; MMR, measles, mumps and rubella; OMPC, outer membrane protein complex; OPV, oral polio vaccine; PRP, polyribosolribitol phosphate.
After conjugate vaccines were introduced in the immunization schedules of high-income countries, there was a rapid decline in the burden of Hib diseases. However, Hib vaccines have yet to be introduced in many low-income countries as part of routine immunization programs. This may be due to paucity of available data and cost constraints on Hib disease [Kahn et al. 2014]. It is important to know not only the efficacy of Hib vaccine but also each dose efficacy; a cost-analysis should be made before introducing the vaccine in routine programs. Trials have shown that the seroprotection rate after two primary dose schedule may be equivalent to the three primary dose schedule [Campagne et al. 1998].
Objectives
The primary objective was to find out whether the two primary dose schedule of Hib vaccine is equally efficacious as the standard three primary dose schedule.
Methods
Search methods for identification of studies
A Medline (Ovid) search (till July 2014) was performed to identify clinical trial publications on the efficacy of Hib vaccination. The search strategies employed was to ‘explode’ the terms ‘Haemophilus Vaccines’ or ‘Haemophilus influenzae type b’ and ‘conjugate vaccine’. We used the similar search terms for other databases including PubMed, Embase (till July 2014) and the CENTRAL (Cochrane Central Register of Controlled Trials, 2014, issue 8). Two authors independently searched the database and scanned the references of retrieved articles to obtain articles not identified by Medline. Any discrepancy was resolved through discussion with the second author (M.S.). The protocol was registered with PROSPERO data base (CRD42013004490). For the search strategy, please refer to Supplementary file 1.
Inclusion criteria
Studies with prospectively and randomly/quasi-randomly allocated participants for comparing a commercially available Hib vaccine and a control to prevent invasive Hib disease among children less than 5 years old were considered eligible. We did not consider quality measure as a criterion for exclusion (so as to maximize the available data) nor did we add a language barrier. We excluded case control studies, burden studies of Hib carriage, reviews, immunogenicity studies, case reports and studies comparing the efficacy of two different Hib vaccines.
Data extraction and management
Two authors independently extracted data on a predesigned proforma which included the following details: study ID (author, date of publication), study setting, study population, type of intervention given and control used, number of Hib doses given, positive Hib cases after each dose, and serious adverse events if any. Any disagreement in the extracted data was resolved through discussion with the third author. The methodological quality was assessed using the Cochrane’s risk of bias tool [Higgins and Green, 2011].
Assessment of heterogeneity and sensitivity analysis
A quantitative analysis was performed using the intention-to-treat (ITT) principle. Meta-analysis was performed using a fixed-effect model. We used the Q test (chi-square statistics) with an alpha (α) of 0.1 to test heterogeneity among pooled estimates [Higgins and Green, 2011]. When there was statistically significant heterogeneity in outcomes across studies, we tried to explore the cause. A sensitivity analysis was performed by excluding studies with incomplete randomization, by excluding studies with cluster randomization, and by excluding studies with small effect size. The risk ratio (RR) with 95% confidence intervals (CIs) was calculated. Prespecified subgroup analyses were planned to compare the effectiveness according to the number of Hib doses given.
Data analysis
Data were analyzed using RevMan software (version 5.0.2). GRADEpro software developed by the Grading of Recommendations Assessment, Development and Evaluation (GRADE) working group was used to grade the evidence as low, moderate or high quality [Schünemann et al. 2011]. Vaccine efficacy (VE) was calculated using the formula: VE (%) = (1 – RR) × 100, where RR is the pooled risk ratio.
Results
Search results
The study flow is depicted in Figure 1. The search yielded 832 potentially eligible studies after elimination of the duplicates. Out of these, 230 studies did not report Hib, 131 reported about safety and/or immunogenicity, 190 reported about combination vaccines, 40 were review papers, 24 dealt with the impact of Hib in the postlicensure era, 123 reported about physiological aspects, and 83 reported other issues not relevant to the present review. Eleven studies were found meeting the inclusion criteria, of these, 2 studies were excluded. Finally, 9 studies (Table 1) were included in the analysis [Eskola et al. 1990; Ward et al. 1990; Black et al. 1991; Santosham et al. 1991; Vadheim et al. 1993; Booy et al. 1994; Lagos et al. 1996; Mulholland et al. 1997; Gessner et al. 2005].
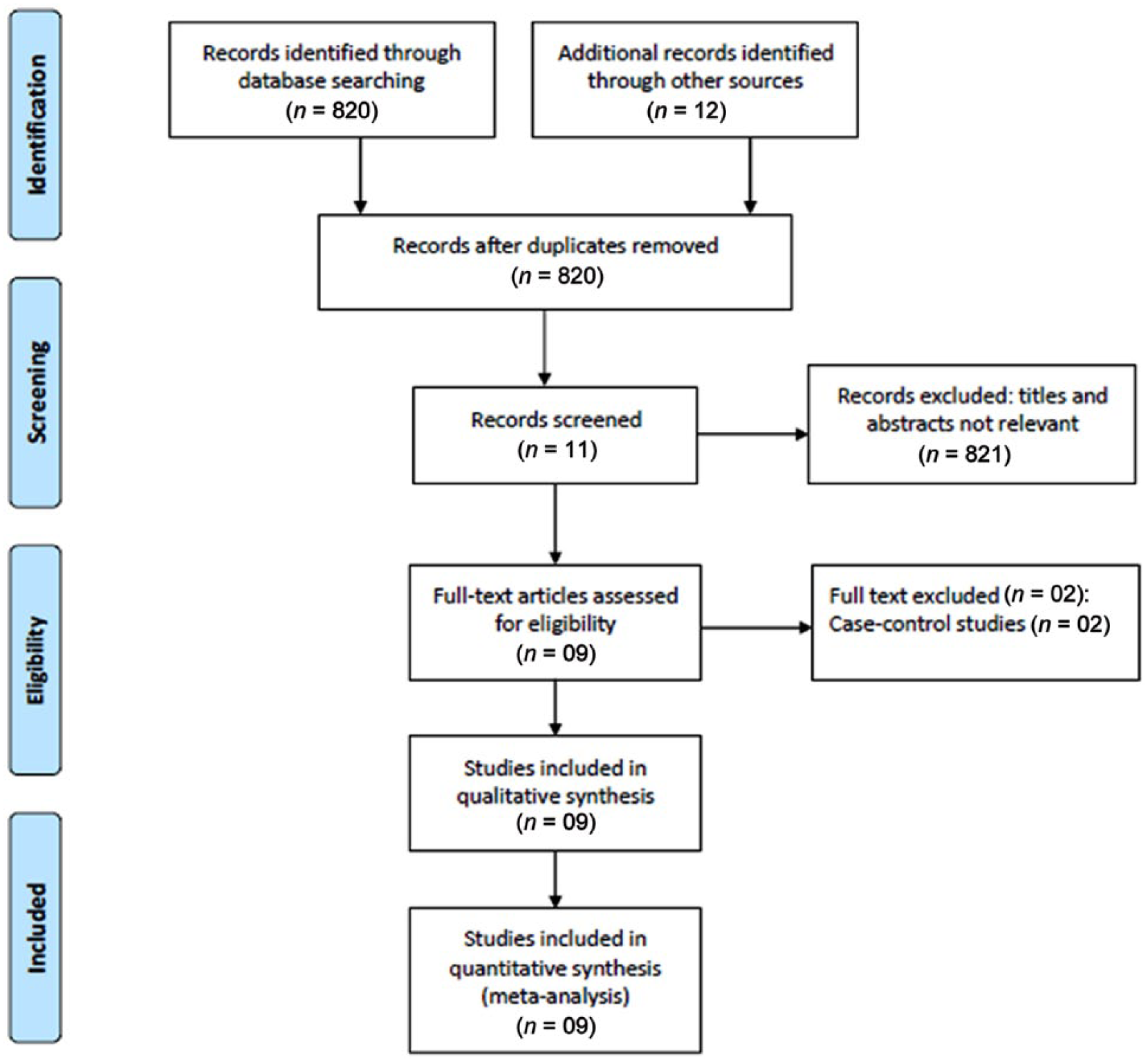
Study flow diagram (PRISMA flow chart).
Vaccine efficacy
The heterogeneity was ‘not significant’ among the studies reporting the efficacy of the three dose (I2 = 40%) and the one dose schedules (I2 = 28%), but was ‘significant’ among the studies reporting the efficacy of two dose schedule (I2 = 64%). Pooled vaccine efficacy following the primary three (overall protective efficacy), two and one dose schedules were 82% (RR 0.18; 95% CI 0.13–0.27), 79% (RR 0.21; 95% CI 0.10–0.47) and 65% (RR 0.35; 95% CI 0.16–0.77), respectively (Figure 2–4). The RRs of giving two primary doses compared with control and three primary doses compared with control were comparable and there was not much difference.

Forest plot showing single dose Hib conjugate vaccine efficacy against invasive Hib disease compared with placebo.

Forest plot showing two doses Hib conjugate vaccine efficacy against invasive Hib disease compared with placebo.

Forest plot showing three doses Hib conjugate vaccine efficacy against invasive Hib disease compared with placebo.
A sensitivity analysis (fixed-effects model) was performed to determine the strength of the present study. The overall Hib efficacy using the fixed-effects model remained at 82% (RR 0.18; 95% CI 0.13–0.27) for the 8 included studies. Excluding the 2 cluster randomized studies [Lagos et al. 1996; Gessner et al. 2005] showed no change in the vaccine efficacy which was 79% (RR 0.21; 95% CI 0.14–0.32) for the three dose schedule and 79% (RR 0.21; 95% CI 0.19–0.46) for the two dose schedule. Similarly, the efficacy did not change much when we excluded the quasi randomized studies: 80% (RR 0.20; 95% CI 0.13–0.30) for the three dose schedule and 79% (RR 0.21; 95% CI 0.10–0.46) for the two dose schedule [Ward et al. 1990; Black et al. 1991].
Risk of bias in included studies
The details are provided in Figures 5 and 6.
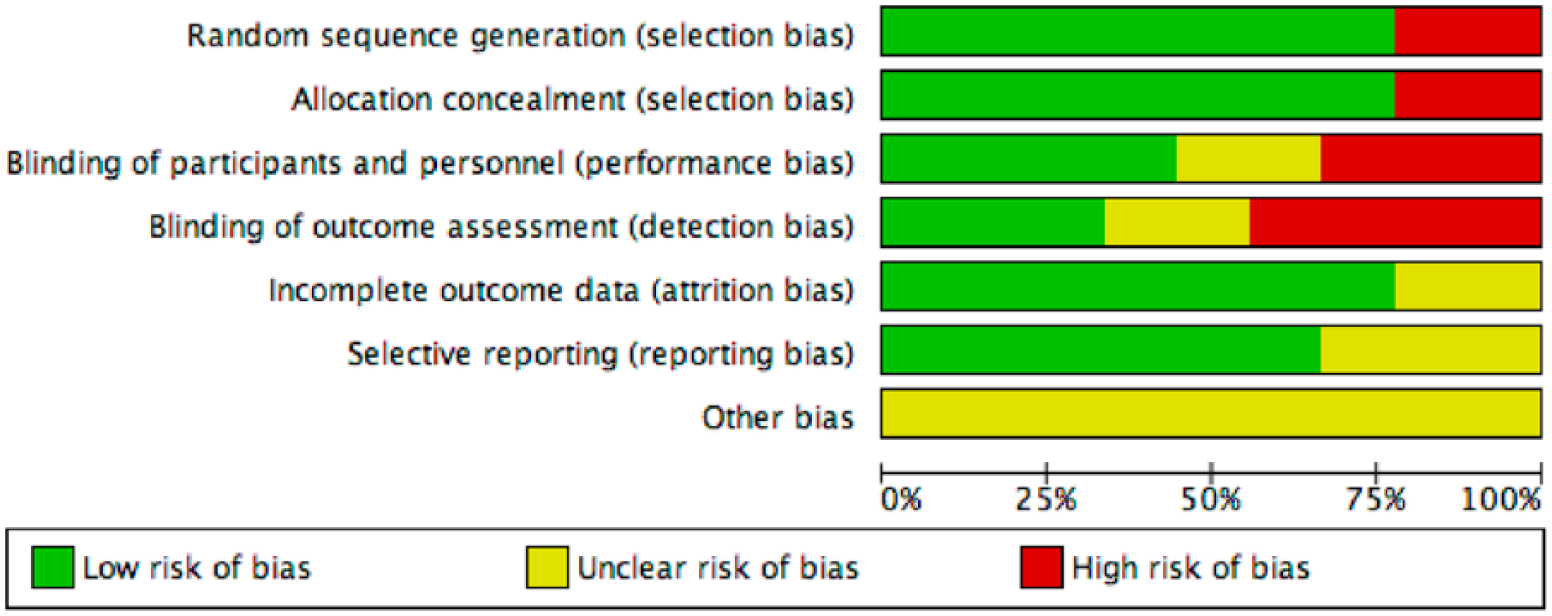
Graph showing risk of bias in the included studies.
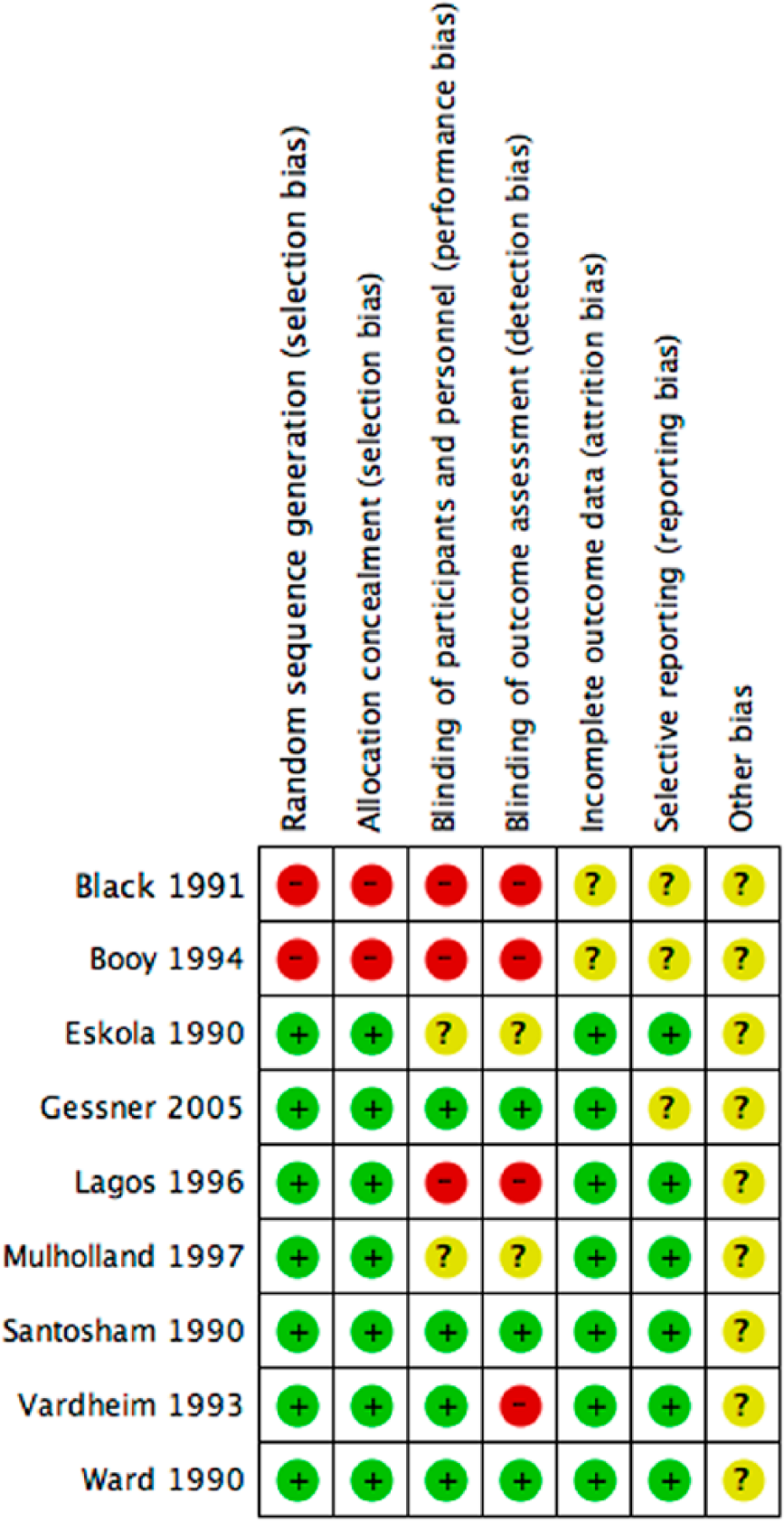
Summary of bias in the included studies.
Publication bias
To assess whether there was a bias in the meta-analysis, a funnel plot was constructed using the RR and 1/SE (standard error) values obtained from trials measuring outcome for the three primary dose schedule. In the absence of a publication bias, such a plot is expected to have a shape resembling an inverted funnel [Egger et al. 1997]. From the funnel plot generated, the possibility of bias in the analysis cannot be ruled out (Figure 7).
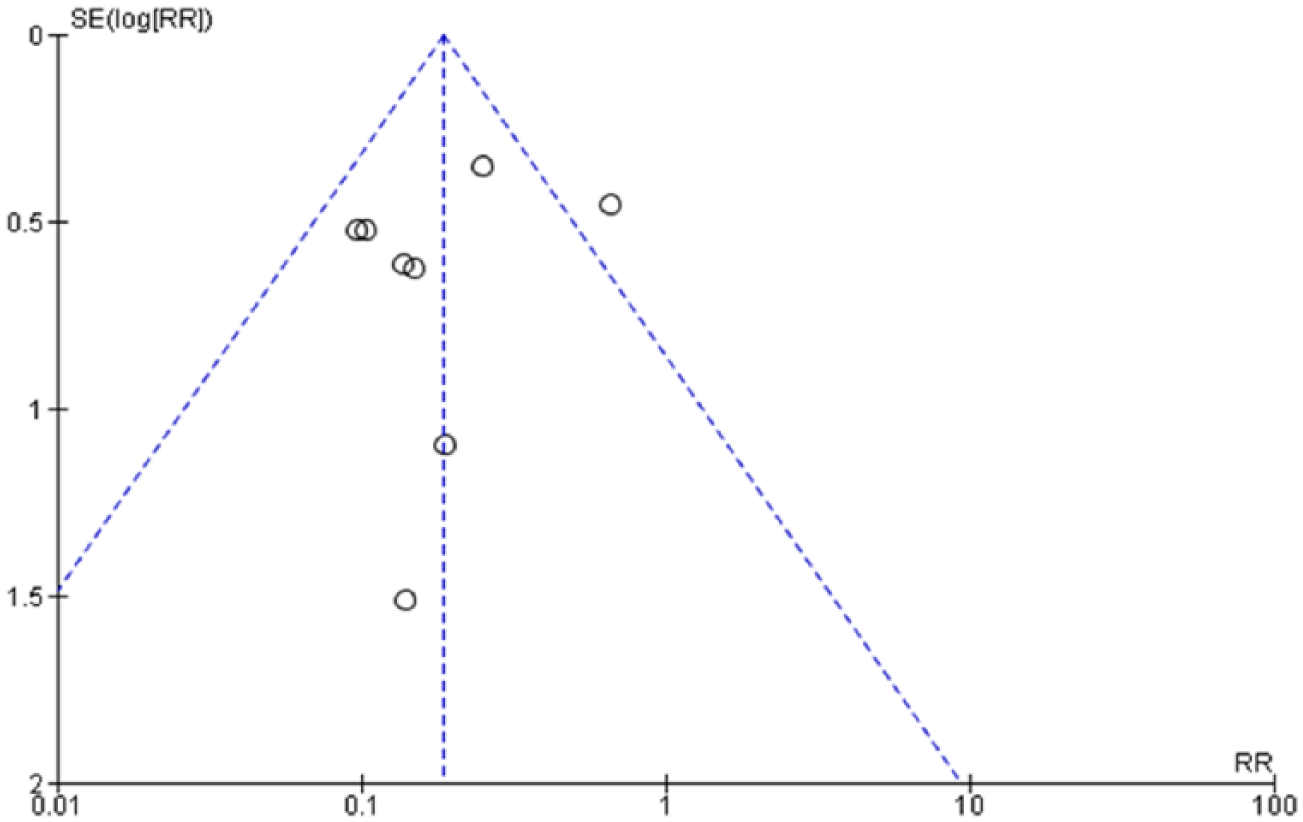
Funnel plot assessing bias in the analyses (generated from the trials assessing the efficacy of the three primary dose schedule).
Grade of evidence
The summary of findings table (Table 2) showed that the available evidence is of ‘moderate quality’ for the one dose and the two dose schedules, and of ‘low quality’ for overall and the three dose schedule. Quasi-randomization and the open label nature of trials downgraded the quality of evidence.
Summary of findings.
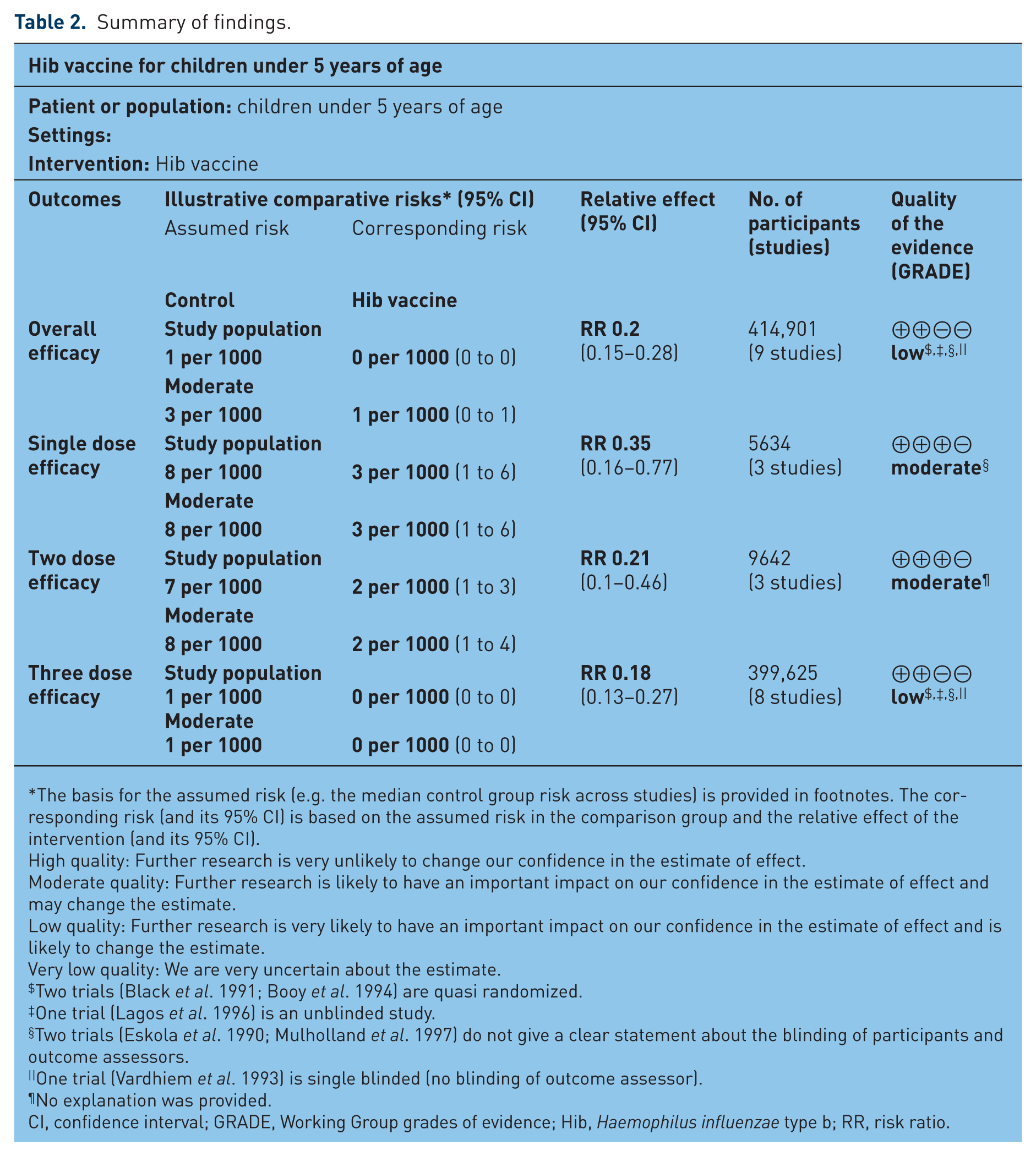
The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% CI) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
High quality: Further research is very unlikely to change our confidence in the estimate of effect.
Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate.
Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate.
Very low quality: We are very uncertain about the estimate.
Two trials (Black et al. 1991; Booy et al. 1994) are quasi randomized.
One trial (Lagos et al. 1996) is an unblinded study.
Two trials (Eskola et al. 1990; Mulholland et al. 1997) do not give a clear statement about the blinding of participants and outcome assessors.
One trial (Vardhiem et al. 1993) is single blinded (no blinding of outcome assessor).
No explanation was provided.
CI, confidence interval; GRADE, Working Group grades of evidence; Hib, Haemophilus influenzae type b; RR, risk ratio.
Discussion
We studied the efficacy of Hib conjugate vaccine against invasive Hib diseases in children under 5 years of age. The Hib vaccine is 80% efficacious as showed by the pooled results in current meta-analysis. The sub-group analysis performed for various primary dose schedules showed that there is not much difference in the efficacy of two and three primary doses; two primary doses can prevent 79% of all invasive diseases (range: 54–90%) and three primary doses can prevent 82% of all invasive diseases (range: 73–87%).
The present review dealt with the efficacy of Hib conjugate vaccine only with regard to the invasive Hib diseases; it also dealt with the dose efficacy of Hib vaccine. These findings will be beneficial in helping low- and middle-income countries to decide on the inclusion of Hib conjugate vaccine in their national immunization programs, where cost plays a significant role in policy making. As Hib conjugate vaccine costs about 2.5 USD per dose, using a primary two dose schedule instead of a three dose schedule can be cost-effective [Campagne et al. 1998; Kumar et al. 2013].
Nine studies were included in the present review of which two were quasi randomized, one was open labelled, and one discussed Hib meningitis only. These study designs downgraded the quality of evidence (Figures 3 and 4). The heterogeneity was significant among trials reporting the efficacy of the two dose schedule, but not among one or three primary dose schedules. This can be due to the inclusion of quasi-randomized and cluster randomized studies. The sensitivity analysis conducted first excluding only quasi randomized studies [Black et al. 1991; Booy et al. 1994] and then excluding only cluster randomized studies [Lagos et al. 1996; Gessner et al. 2005] failed to explain the heterogeneity observed in the meta-analysis. The heterogeneity may also have arisen out of different timing of administration of vaccines doses (e.g. 2, 4, 6 months versus 3, 4, 6 months versus 6, 10, 14 weeks).
The study by Santosham et al. (1991) used a two dose schedule and found it to be highly efficacious. This study was devoid of any bias and thus generated good quality evidence. The other two studies [Ward et al. 1990; Mulholland et al. 1997] also found the two dose regime to be efficacious, but the range was too wide to be conclusive. However, eight studies favored the three dose regime. Two of these eight studies had wider CIs, but the overall pooled result favored them. Four of the included studies [Ward et al. 1990; Black et al. 1991; Vadheim et al. 1993; Lagos et al. 1996] had followed a 2, 4 and 6 months regime for primary doses. One study from Finland [Eskola et al. 1990] followed a 3, 4 and 6 months regime. Gessner and colleagues used a 6, 10 and 14 weeks regime which was closer to the 2, 4 and 6 months regime [Gessner et al. 2005]. The only study from the USA [Santosham et al. 1991] followed a 42–90 days and a 70–146 days schedule. However, WHO recommends 6, 10 and 14 weeks as the primary schedule.
The efficacy of Hib vaccine from the pooled data in the present meta-analysis is comparable with a previous meta-analysis conducted in 2006 that included 8 studies [84% (69–92%)] [Obonyo and Lau, 2006]. Another meta-analysis of 8 studies conducted in 2012 [Griffiths et al. 2012] showed the vaccine efficacy to be 93% and 92% for three and two primary doses, respectively, which was higher than the present finding. This meta-analysis excluded two studies by included in the present meta-analysis [Eskola et al. 1990; Ward et al. 1990] but included another study [Levine et al. 1999]. Similarly, another meta-analysis of observational studies [O’Loughlin et al. 2010] showed 95% (95% CI 82–99) efficacy after three doses and 92% (95% CI 81–99) efficacy following two doses. Recently, another meta-analysis of case-control studies showed vaccine efficacy of 55% (95% CI 2–80), 96% (95% CI 86–99) and 96% (95% CI 86–99) after one, two and three doses of Hib conjugate vaccine against Hib meningitis, respectively [Jackson et al. 2013]. However, in both of these meta-analysis [O’Loughlin et al. 2010; Jackson et al. 2013], the differences in the efficacies of two and three primary doses were not significant and the authors reached a similar conclusion to the present meta-analysis.
Conclusion
In the present meta-analysis, we found that Hib conjugate vaccine is highly efficacious and significantly reduces the risk of invasive Hib disease. The two dose regime is as good as the three dose regime. Randomized controlled trials with a large sample size and cost-effective analysis should be carried out in low to middle income countries to confer any policy decision.
Footnotes
Funding
The review was supported and funded by ICMR, New Delhi (grant number 5/7/592/11-RHN). The funding agency did not interfere with the review process or the results.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
